Abstract
The critically endangered mangrove finch is now limited to one small population on the west coast of Isabela Island in the Galápagos, but 100 years ago multiple populations were found on the islands of Isabela and Fernandina. By accessing genetic datasets through museum sampling, we are able to put current levels of genetic diversity and hybridization with congenerics into a historical context for enhanced conservation. In this study, we compared neutral genetic diversity of the now extinct Fernandina population to historical and current diversity of the Isabela population using 14 microsatellite markers. We found that current genetic diversity of the last remnant population (~80–100 individuals) is far below levels 100 years ago, with only about half of the allelic diversity retained. Current genetic diversity is close to levels in the Fernandina population that went extinct by the 1970s. Bottleneck analysis did not show a strong signature of recent decline, and instead implies that this species may have consistently had low population sizes with wide fluctuations. Hybridization with congeneric woodpecker finches was found in the modern Isabela population, implying that some individuals within the few remaining breeding pairs are finding mates with woodpecker finches. Within the context of historical low population sizes and wide fluctuations, current conservation efforts may help the mangrove finch face current extinction threats and avoid the fate of the Fernandina population. However, this historically small lineage will likely continue to face challenges associated with small specialist species surrounded by a widely-distributed sister lineage producing viable hybrids.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Critically endangered species are vulnerable to a number of forces including habitat loss, habitat fragmentation, demographic instability, inbreeding, disease/parasites, introduced predators, hybridization with congenerics, and other factors which can tip the balance towards extinction (Purvis et al. 2000; Smith et al. 2006; O’Grady et al. 2006; Melbourne and Hastings 2008; Donlan and Wilcox 2008; Evans and Sheldon 2008; Méndez et al. 2014; Rivera-Parra et al. 2014). Accurately assessing the factors that may have lead to an initial decline and identifying ongoing processes that may threaten the future survival of these lineages is of great concern. However, determining the existence of a decline and linking potential causes is often hampered due to insufficient data on population conditions in the past.
Low genetic diversity in critically endangered species is of special concern to conservation biologists, as low effective population size may be the result of a recent bottleneck and/or linked to ongoing decline (Keller et al. 2001; Antao et al. 2011). Historical data from museum specimens collected during early human contact can provide insights into the long-term characteristics of species including distinguishing between recent declines, active declines, and long-term small effective population sizes. In addition, it can inform on range reductions and gene flow changes from historical levels (e.g., Hedrick and Waits 2005; Schwartz et al. 2007; Bristol et al. 2013; Spurgin et al. 2014; Dussex et al. 2015). Genetic datasets from now extinct population or species can also provide ecological and behavioral details of their decline, including identifying increased introgression with other species in the last known individuals of a dwindling population (e.g., Grant et al. 2005b).
In island bird populations, human activities such as culling or reduction of habitat have been definitively linked in some species to lower genetic diversity in extant populations compared to historical ones (e.g., Allentoft et al. 2014). In other cases, however, these datasets have shown that effective population size and genetic richness are often robust to even near-extinction levels of population decline (e.g., Dussex et al. 2015). Human activity has been directly implicated in the extinction of island birds across the globe (Case 1996; Blackburn et al. 2004), and recent and historical human activities are believed to be impacting many of the native species in the Galápagos Islands including many lineages of Darwin’s Finches (Mauchamp and Atkinson 2010; Watson et al. 2010; Dvorak et al. 2012; Parker and Whiteman 2012).
Darwin’s finches are a classic example of an adaptive radiation within the Galápagos Islands comprised of ~13–18 species with major groups corresponding to the ground finches (Geospiza), the tree finches (Camarhynchus), warbler finches (Certhidea), vegetarian finch (Platyspiza), a clade of sharp-beaked ground finches from Pinta, Fernandina and Santiago (currently known as Geospiza difficilis), and the Cocos Island finch (Pinaroloxias) (Farrington et al. 2014; Lamichhaney et al. 2015). Though many of the finch species have experienced negative conditions such as habitat reductions, invasive species interactions, and exposure to new diseases and parasites, there have yet to be any documented species extinctions within Darwin’s finches (Grant et al. 2005b; Watson et al. 2010; Dvorak et al. 2012; Levin et al. 2013). Local extinctions from individual islands have been documented, however (Grant et al. 2005b), as have many other species of vertebrates across the Galápagos (Steadman et al. 1991); highlighting the vulnerability of many lineages despite conservation protection efforts (Grant et al. 2005b).
Most Darwin’s finch species are classified by the IUCN red list as Least Concern, however in the Camarhynchus group 80% are classified with a threat status. Two tree finch species have been uplisted recently to Vulnerable in 2016, and another, the mangrove finch (Camarhynchus heliobates), has suffered extensive recent declines in its range and was listed as critically endangered in 2000 with only an estimated 80–100 birds remaining (IUCN 2016, population estimates from Charles Darwin Foundation Mangrove Finch Conservation Project 2016, Cunninghame et al. 2015). These mangrove finches have adapted to live in the scarce mangrove forests that occur in small patches on the coasts of some islands within the Galápagos Archipelago. As in all species within this radiation, breeding males hold small territories, build a dome-shaped nest, and sing to entice females to mate (mean territory size ~0.39 ha; Fessl et al. 2010b). In determining whether the presence of individuals indicates a breeding site or simply a foraging location, these “breeding behaviors” are used as indicators. Though mangrove finches may never have been abundant given the restricted occurrence of mangroves in Galápagos (Wium-Andersen and Hamann 1986), this species has recently been pushed to the edge of extinction due to egg predation by black rats and nestling mortality due to a blood sucking parasitic fly, Philornis downsi (juvenile mortality rate ~84 % without rat and parasite control; Fessl et al. 2010b). With the management of black rats beginning in 2008 (Fessl et al. 2010a), mortality caused by P.downsi is now the main factor driving the 70 % chick mortality (F. Cunninghame, unpublished).
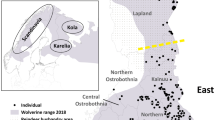
Two populations existed on Isabela until recently (Harris 1973; Grant and Grant 1997; Dvorak et al. 2004; Brumm et al. 2010). The small population at Cartago Bay (five singing males in 2008) was last seen in 2009 (two males) despite searches in 2011 and 2012 (Fessl et al. 2010b; Cunninghame et al. 2013; Young et al. 2013; Fig. 1). Now only the larger population in the northwestern part of the island persists. Mangrove finches formerly also occurred on the neighboring island of Fernandina (Fig. 1). The first and only record of a population of mangrove finches on Fernandina is from 1899, when 14 specimens were collected (Snodgrass and Heller 1904). Despite occasional sightings of non-singing individuals in recent years, the lack of territorial behavior (e.g. singing, nest building, approach towards playback) or breeding pairs implies that they are now locally extinct (Grant and Grant 1997; Dvorak et al. 2004).
Map of mangroves and mangrove finch breeding sites on Fernandina and Isabela Islands. Inset shows close-up of current breeding range of mangrove finches. All mangroves are highlighted (colored areas on the coasts of both islands). Red areas show the current mangrove forests utilized for breeding. The small yellow area and black star marks the now extinct breeding population at Bahia Cartago. Black areas represent mangroves that had breeding populations which are now extinct (before 1996). Green areas show mangroves with no known breeding. X’s mark single recent non-breeding records of mangrove finches
The final population of mangrove finches is restricted to mangrove forests on the western edge of Isabela Island concentrated in Playa Tortuga Negra and Caleta Black (Fig. 1). Their nearest phylogenetic sister species (woodpecker finch, Camarhynchus pallidus; Farrington et al. 2014; Lamichhaney et al. 2015) is also their nearest neighbor, with ranges overlapping at the edges of the mangrove forests (Fessl et al. 2010a). These species are exceptionally difficult to distinguish in the field except that the male mangrove finch often has a darkly speckled breast in maturity, while the woodpecker finch remains light greyish-olive and has a slightly larger beak (Fig. 2, Fessl et al. 2011).
The overlapping ranges and well-known examples of hybridization among many species of Darwin’s finches raise concerns about the integrity of the remnant mangrove finch population with respect to the woodpecker finches (Grant and Grant 1992; Grant et al. 2004; Grant et al. 2005a; Kleindorfer et al. 2014a). Hybridization has both positive and negative implications for severely threatened lineages, and should be evaluated in both an evolutionary and conservation context to understand the specific dynamics in each case. On the negative side, hybridization can erode the integrity of species targeted for conservation and ultimately lead to extinction of the rare species through outcrossing (Rhymer and Simberloff 1996; Wolf et al. 2001; Todesco et al. 2016). On the other hand, hybridization can also provide valuable genetic rescue to struggling populations, which may allow the transfer of adaptive traits between lineages (Hedrick 2009; Adams et al. 2011; Hedrick 2013).
In order to assess the current state of decline in mangrove finches in a historical perspective along with potential forces acting upon them including introgression with woodpecker finches, we surveyed genetic variation at fourteen autosomal microsatellite loci for modern field collections and historical museum collections (Fig. 1; Supplementary Table 1). Modern collections were from the remnant Isabela population and one individual from the recently extinct second Isabela population (Bahia Cartago, one specimen; Fig. 1). Historical collections from Isabela (near Vulcan Darwin, likely Playa Tortuga Negra) and Fernandina (Mangle Point and unknown eastern localities) were from the California Academy of Science Galápagos expedition (1905–1906) and Rollo Beck’s collection from a previous expedition (1899). We compared these specimens with modern (2006–2015) co-distributed woodpecker finches from Isabela and Fernandina to identify introgression and potential interspecies introgression.
Methods
Blood samples of mangrove finches were collected from the extant remnant population on Isabela throughout the region between Playa Tortuga Negra (PTN) and Caleta Black (CB) between 1999 and 2014 (N = 95), and from one eastern male (Bahia Cartago) in 2009 (Brumm et al. 2010) using mist nets (adults and juveniles) and nest sampling (eggs and nestlings collected for hand-rearing before returning to the wild as part of current intensive conservation management of the species). 56.5 % were males. Most birds were not known to be related except for 8 nestlings with one or more parent in the dataset (see below). All birds were blood sampled from the brachial vein with blood preserved on filter paper with EDTA. Historical specimens of mangrove finches from Isabela and Fernandina were collected in 1899 and 1906 by the California Academy of Sciences (Isabela N = 17, Fernandina N = 12). Tissue samples from museum specimens were obtained from toe pad shavings (approximately 3 × 2 mm).
Woodpecker finches from the same islands (Isabela and Fernandina) as well as a larger dataset of islands across the archipelago were surveyed to establish the possible interbreeding populations with mangrove finches. Woodpecker finches were also collected through both mist nests for adults and nest surveys for chicks. As these are not banded, the genetic dataset was checked to make sure that no individuals were sampled more than once. Woodpecker finches from Fernandina (N = 3) and Santiago (N = 6) were collected during the same time period. Woodpecker finches from San Cristóbal (N = 19), Isabela (N = 18), and Santa Cruz (N = 55), were collected between 2006 and 2015. As all other populations besides the Isabela and Fernandina populations were distantly related to the Isabela and Fernandina genetic cluster of woodpecker finches, we used just this smaller dataset for admixture analyses with mangrove finches (see below).
We included as many individuals as possible to represent the small extant population of mangrove finches on Isabela, thus suspected parents and siblings are also included in this dataset (8 mangrove finch nestlings with at least one parent in the dataset), as exclusion of these nestlings did not alter results (Table 2).
Museum specimens were processed following specialized protocols and modifications, including storage and processing in a room dedicated to ancient DNA work to avoid contamination (Petren et al. 2010; Farrington and Petren 2011; Parker et al. 2011). DNA was extracted from museum specimens using GeneClean Ancient DNA (MP Biomedicals) kits following the manufacturer protocol, with blank extractions included in every batch of samples processed.
Fourteen autosomal microsatellite markers that were optimized for ancient DNA specimens in Darwin’s finches (length <150 bp) were used to characterize both the modern and historical datasets (Farrington and Petren 2011). These markers were chosen for their ability to amplify across species from degraded museum specimens and because they are neutral (or “nearly neutral”) markers that can track population fluctuations without bias from selection (Petren et al. 2010). Samples from modern collections were processed following Petren et al. (2010). Genotypes of museum specimens were obtained from three independent replicate PCR runs following Farrington and Petren (2011) to reduce allelic dropout and genotyping error. PCR products were analyzed on an Applied Biosystems 3730xl DNA Analyzer at the Cornell University Life Sciences Core Laboratories Center and Max Planck Institute for Ornithology. Sample runs were genotyped in GENEMAPPER v. 3.7 (Applied Biosystems) without knowledge of population origin to limit scoring bias. Errors within the data (i.e. input errors, allelic dropout, stutter and null alleles) were assessed in MICRO-CHECKER v. 2.2 (Van Oosterhout et al. 2004). Due to sequencing at two facilities, we used large population datasets to ensure that peak scoring was consistent and sequenced one individual at both Cornell and Max Planck to check for machine inconsistencies.
In order to determine correct identification of mangrove finch individuals and identify any hybrid individuals, we completed assignment tests with the Bayesian clustering program STRUCTURE v. 2.3.4 (Pritchard et al. 2000). A number of combinations of individuals were assessed to determine the most relevant populations. We compared (1) all mangrove finches, (2) all woodpecker finches, and (3) all mangrove finches along with woodpecker finches from Isabela, Fernandina, Santa Cruz, Santiago, and San Cristobal. The genetic distance within woodpecker finches between the Isabela/Fernandina cluster (subspecies C.p. productus) and the rest of the woodpecker finches (C.p. pallidus and C.p. striatipecta) was sufficient to warrant removal of the non-codistributed woodpecker finch populations from analyses of admixture with the mangrove finches. We performed 10 runs at each value of the fixed parameter K (the number of clusters), from K = 1 to K = 6. Each run consisted of 500,000 replicates of the MCMC after a burn-in of 100,000. We used the admixture model without a population prior. All other parameters were set to default values. The program Structure Harvester v 0.6.92 (Earl and vonHoldt 2011) was used to visualize the STRUCTURE results and implement the Evanno et al. ad hoc method of K estimation, which detects the uppermost level of hierarchy (Evanno et al. 2005).
We used the program Ne-estimator v. 1.3 (Peel et al. 2004) to calculate population sizes of mangrove finch populations from microsatellites using the Linkage Disequilibrium (LD) method (Hill 1981). The principle of the LD method is that as Ne decreases, genetic drift generates nonrandom associations among alleles (i.e., increased LD). FSTAT v2.9.3.2 (Goudet 1995) was used for allele number and allelic richness estimation. All individuals identified as hybrids or possible hybrids were removed to assess the genetic diversity of the “pure” populations. The two populations with larger sample sizes (modern and historical Isabela) were randomly subsampled 100 times to 12 individuals to match the historical Fernandina population, and then averaged for a more even comparison of genetic diversity and sample sizes. Genepop v. 4.2 (Raymond and Rousset 1995) was used to calculate Fis for each population. Genetic diversity variables such as allele frequencies per locus, observed (Ho) and expected (He) heterozygosity, total number of alleles, the mean number of alleles, and the Garza-Williamson index were calculated in Arlequin v. 3.5.2.2 (Excoffier et al. 2005; Excoffier and Lischer 2010).
We tested each historical population for evidence of a prior population bottleneck based on deviations of allelic variation from mutation-drift equilibrium using the program BOTTLENECK v. 1.2.02 (Cornuet and Luikart 1996). The test was performed for the historical Isabela population, the historical Fernandina population, and the modern Isabela population using the two-phase model (TPM) of microsatellite evolution and a probability of 95 % for single-step mutations with variance of 12 (1000 replicates) as suggested by Piry et al. (1999). The Wilcoxon test was used to determine if significant excess or deficit of heterozygosity was detected based on observed number of alleles (H e > H eq < H d ). Excess heterozygosity would indicate a previous population bottleneck, while a heterozygosity deficit would indicate a population expansion. The modern Isabela mangrove finch population was also run without offspring of adults in the study to ensure that the full dataset was not biased due to increased relatedness.
Paternity was assessed through pedigree analysis for mangrove finch offspring in the modern Isabela dataset with one or more known potential parents. Potential parents were presumed correct unless they could be excluded genetically. Paternity was also checked in the historical Fernandina and Isabela datasets in Cervus v 3.0.7 (Kalinowski et al. 2007) to ensure that sampling bias did not impact estimates based on these small sample sizes.
Results
Locus summaries and parentage analysis
The historical Fernandina and Isabela populations of mangrove finches showed moderate levels of deviations from Hardy–Weinberg Equilibrium (HWE) and Linkage Disequilibrium (LD). These loci are not typically linked in Darwin’s finches (Petren et al. 1999; Petren et al. 2005). Fernandina had 16/91 pairwise LD comparisons that were significant at the 0.05 level, and loci gf1, gf4, gf6, gf7, and gf8 deviated from HWE. In the historical Isabela samples, 4/91 pairs showed disequilibrium, and loci gf1, gf4, gf5, gf7, gf10, and gf11 deviated from HWE at the 0.05 level. The modern Isabela population, with its much larger sample size, showed 13/68 significant pairwise LD comparisons, but no loci were out of HWE. All historical populations showed no potential parental/offspring relationships.
Structure analyses of species assignment and admixture
Bayesian clustering analysis with no prior species assignments identified K = 2 as the most likely number of genetic groups (Supplementary Fig. 1). However, the groupings assigned corresponded to the modern Isabela population of mangrove finches, and then split membership for all other populations. This pattern is likely an artifact of the strong membership assigned to the modern Isabela population of mangrove finches (due to inbreeding, Gao et al. 2007), thus K = 3 was explored. K = 3 assigned two clusters to the mangrove finches and one cluster to the woodpecker finches (Fig. 3).
Structure analysis results of all Isabela and Fernandina mangrove and woodpecker finches. Three genetically distinct groups: one lineage of woodpecker finch from Fernandina and Isabela (light gray), and two genetic clusters within the mangrove finches (black and dark gray). Mis-categorized individuals (isolated light gray bars with stars) in the modern mangrove finches are two juveniles cared for by a mangrove finch male and an un-banded and unidentified female. Introgressed individuals are indicated by a diamond
Six modern individuals that were morphologically identified as either mangrove or woodpecker finches showed <75 % membership to a single parental species (a cutoff implying at least one interspecific grandparent paring) (Fig. 3). These individuals were labeled “hybrids.” Two further individuals were identified as “possible hybrids” (85–88 % mangrove). All eight of these individuals (Fig. 3, indicated by diamonds) were removed from the dataset for analyses of genetic diversity and effective population size. No individuals from the historical samples showed introgression, but historical introgression cannot be ruled out due to the much smaller dataset. Two fledgling siblings identified as mangrove finches due to the presence of a mangrove finch male being the nest partner of an incubating female were confirmed as pure woodpecker finches, and not the offspring of the presumed father (PM078, PM080; Fig. 3, black stars).
A great deal of genetic diversity appears to have been lost between the historical mangrove finch populations and the current population, as two genetic clusters (admixed in the historical population) are assumed in the mangrove finches to accommodate the genetic uniformity of the extant population. The single Cartago individual (east coast of Isabela, Fig. 1) appears to be from a different genetic gene pool than the extant larger Isabela population with some alleles in common with the now extinct populations on both Isabela and Fernandina. This individual also had a different song (Brumm et al. 2010). With only one individual from this population, and small historical sample sizes, it is difficult to infer if the Cartago population was genetically more similar to the historical Isabela or Fernandina populations. Only one locus had an allele in common with the historical Fernandina population, while alleles at four other loci are shared with only historical and modern Isabela populations. Other loci in the Cartago individual do not match records in any other populations (gf11, allele 159; gf15, allele 123). The greater allelic affinity with Isabela populations implies that Cartago population was probably most closely related to the ones on the same island, despite the proximity of the Fernandina population.
Population genetic diversity and bottlenecks in the mangrove finch
Historical populations of mangrove finch from Isabela had higher genetic diversity than today, though the historical Fernandina population had similarly low levels of diversity compared to the modern Isabela population (Tables 1 and 2). Most strikingly, the historical Isabela population had a total of 100 alleles across microsatellites (from only 17 individuals), while the modern Isabela population has 56 alleles despite a dataset that was almost five times larger. The small historical Fernandina dataset (n = 12) had a similar number of alleles (n = 54). Even when the populations are subsampled to 12 individuals to match the smallest population, the modern Isabela population has approximately half of the alleles of the historical Isabela population (historical Isabela = 85.2, modern Isabela = 41.3). Mean allelic richness was also higher in the historical Isabela population (7.143, s.d. 3.110) than in the modern Isabela population (4.5, s.d. 2.355), though estimates overlap due to the size of the standard deviations. The historical Fernandina population (3.857, s.d. 1.406) was a close match to the modern Isabela population. The modern Isabela population had the lowest estimate of Fis (0.050) compared with 0.146 for historical Fernandina and 0.208 for historical Isabela. Estimates of effective population size between the historical and current Isabela populations (historical = 36.0 (22.9–72.2), modern = 21.5 (17.8–25.9)) are not significantly different, though both were significantly higher than the historical Fernandina population (1.2 (1.0–1.5)).
None of the populations conformed to expectations of a stable population size (Table 2). Detection of a recent bottleneck depended upon the estimation method. A recent bottleneck was predicted in all populations due to low M crit Garza-Williamson index values (below M < 0.68 as proposed by Garza and Williamson 2001). The historical Isabela population has the highest value (least likely to have had a historical bottleneck), while the historical Fernandina and modern Isabela populations were nearly identical. Contrary to this prediction, analyses in BOTTLENECK predicted no recent bottlenecks for any population (TPM model: historical Isabela p = 1.000, modern Isabela p = 0.974, historical Fernandina p = 1.000). The only changes inferred in these populations are recent expansions (TPM model: historical Isabela p = 0.000, modern Isabela p = 0.032, historical Fernandina = 0.000).
Discussion
Looking backwards from the current remnant population of mangrove finches on Isabela to the historical populations on both Fernandina and Isabela, it is clear that much has been lost in terms of population size and genetic richness for this endangered species. Both allelic richness and effective population size (Ne) were lower in the current Isabela population than in the historical Isabela population, though estimates from this last extant population are not as low as the now extinct Fernandina population. Effective population size in the current Isabela population was not as significantly affected by the loss in allelic richness as anticipated. This may be due to the fact that the population was predicted to be small before the presumed decline, and error bars were large for the relatively smaller historical dataset. Further studies including a larger historical dataset (if possible) and an expanded genomic dataset for greater precision in estimations will help to better evaluate the loss of genetic diversity, effective population size change, and severity of the extinction potential for this species. Eventual inclusion of functional markers which might document specific traits leading to extinction may also be beneficial in future studies to understand specific and general trends in declining lineages (Rollins et al. 2015).
Most strikingly, the estimate of effective population size in the historical Fernandina population was exceptionally low (1.2) compared with ~20–40 for the other populations. Both inbreeding and high variability in reproductive success between individuals are known causes of low Ne estimates, and these are both likely factors impacting the isolated Fernandina population. As only one breeding population was ever identified on Fernandina, inbreeding would be expected to be high. In contrast, populations on Isabela were historically distributed between multiple mangrove forests separated along multiple coasts of the island, which would have limited inbreeding. Variable reproductive success probably impacted all populations as Darwin’s finches are known for high variability in reproductive success for both traits under selection (Grant and Grant 2014). The small population size of the Fernandina population, however, would have made recovery from a low-success year particularly difficult.
The current and historical populations of mangrove finches all deviated from the expectations of a stable population, but the results were unclear as to whether recent bottlenecks or expansions had taken place (Table 2). Unless populations went through contractions and expansions before these surveys (a possibility from early sporadic human contact, see below), these conflicting results may relate to a small founding population of the species and periodic expansions and contractions. Ongoing size fluctuations may have left an ongoing signature of unstable population size. Alternatively, deviations from HWE could be an artifact of biases in collection if within-family relationships are over-represented in the dataset. In the historical samples, it is difficult to know if there were any sampling biases that could lead to the observed deviations. Collection notes are minimal, thus it is possible that historical collections might over-represent families within the larger population. No genetic parental/offspring relationships were inferred from paternity/maternity analyses, however, so if any familial biases are present we assume them to be minimal. In the current collection, the vast majority of banded individuals are included in this dataset, leading to a near-total species census. Thus in the current population, it is unlikely that signatures of a bottleneck or low effective population sizes are biased, because the sampling represents the true gene pool of the population.
The mangrove finches on Isabela Island are clearly in peril due to low population size, low reproductive success, and a severely restricted distribution (Dvorak et al. 2004; Fessl et al. 2010b; Cunninghame et al. 2013). Coupled with the recent local extinctions of additional populations on Fernandina and Isabela, the fate of the last remnant population is uncertain. Though genetic diversity and effective population size are not direct predictors of extinction probability on their own, they are strongly correlated with imperiled populations (Antao et al. 2011). As such, this last population should continue to be monitored for many aspects of fitness including census numbers, fledgling survivorship, response to conservation efforts, and genetic diversity and inbreeding. All of these factors combine to help determine the survival potential of the species. As our genetic estimates for the modern Isabela population are now similar to the genetic diversity of the now-extinct Fernandina population, this may be an indicator that this final population is also near extinction. Despite the documented recent declines, intensive conservation management may be able to provide a different future than the unmanaged Fernandina population.
Hybridization appears to be affecting the small mangrove finch population as a number of individuals are shown to be introgressed with woodpecker finches in the modern dataset. Though hybridization is often thought of as a detrimental process for small populations as they may be absorbed through introgression into a more widely distributed species, hybridization may also act as an evolutionarily constructive process for severely inbred populations (Anderson and Stebbins 1954; Arnold 2004; Hedrick 2013). Gene flow at certain loci may be beneficial in terms of increased genetic diversity and potentially increased habitat flexibility or resistance to parasites as seen in hybrids of other Galápagos tree finches (Grant et al. 2005a; Kleindorfer et al. 2014a).
Though no conclusive estimates of historical introgression can be made due to the smaller sample size of the historical populations and the missing historical populations of woodpeckers, the number of modern individuals with mixed ancestry speaks to a possible recent breakdown in reproductive isolation. One possible cause is that mangrove finches may have difficulty finding conspecific mates in their small population, or may have difficulty producing viable offspring with available mates due to inbreeding depression. Woodpecker finches inhabit the transition zone of habitat surrounding the mangrove forests, with some even living at the margin of the mangrove swamps themselves. Thus interspecific mates are readily available if pre-zygotic reproductive barriers are diminishing between the species.
Several hypotheses have been proposed to explain the decline of the mangrove finch including human causation (introduced predators, parasites, human disturbance), global shifts (global decline of mangrove habitat caused by geological shifts), and species-specific traits which lead to extinction vulnerability (naturally patchy distribution in mangrove habitat, correspondingly small population sizes, and the documented vulnerability of extreme habitat-specialists to extinction) (Grant and Grant 1997; Dvorak et al. 2004; Fessl et al. 2006; Fessl et al. 2010a). The protection granted to the Galápagos Islands as a National Park and concentrated conservation efforts from a variety of government and non-government agencies has helped reduce or eliminating many of the historical factors affecting species in Galápagos such as deforestation and habitat destruction as well as predation from pigs, goats, and introduced rats (Cruz et al. 2005; Walsh and Mena 2012). However, other risk factors including diseases and invasive species are on the rise as they are across all oceanic islands (Wikelski et al. 2004; Fessl et al. 2006, 2010b; Lee and Jetz 2011; Loehle and Eschenbach 2012; Levin et al. 2013; Kleindorfer et al. 2014b).
The continued low population size and genetic diversity of the mangrove finch suggests that forces involved in low population size predate most of the potential causes normally attributed to humans. Before 1920, few of the fifteen major Galápagos Islands were settled, and then only intermittently. Fernandina has never had a permanent human settlement. Isabela was settled in 1893, just prior to the museum collection expeditions incorporated in this study. To this day, settlement on Isabela is restricted to two towns in a small area in the southern part of this large (>4500 km2) island. Most areas of both islands have not been affected by introduced species until after 1900, especially the widely dispersed mangrove habitat (Dvorak et al. 2004). The highly invasive parasitic fly Philornis downsi was most likely introduced in the 1960s (Causton et al. 2006), and thus only contributes to recent declines instead of the historically low population sizes. The only likely human-mitigated stressor to the population before these historical samples were collected could be rat populations on Isabela, which are known to impact mangrove finch populations (Fessl et al. 2010b). Ship rats arrived on Isabela in 1830 (Harper and Carrion 2011) and might have arrived in mangrove finch habitat as early as this. As Fernandina has only endemic rice rats and no ship rats, yet this population went extinct first, it is unlikely that rats alone have influenced the species’ decline. One possible explanation for the extinction on Fernandina is that Fernandina is the most active volcanic islands in the archipelago, with a faster changing coastline than other islands, thus possibly making the mangrove forests equally unstable.
Information about the recent loss of the Cartago population on Isabela is largely lacking as it was only discovered in 1997. The population showed a gender skew lacking females in recent surveys and a more distant transition zone into woodpecker finch habitat, which would have prevented rescue from interspecies mating (Dvorak, pers. comm). This population would have been equally impacted by rats and Philornis (Fessl, unpublished) as the remnant Isabala population, though its long-term stability even without those pressures is unsure without longer-term records.
The remaining list of potential agents of decline include longer-term geological or climatic changes that affect mangrove habitat, natural or introduced pathogens, or other unknown causes. Independent of any human induced factors, the mangroves on Fernandina and Isabela have likely changed their distributions with volcanic activities/uplifts, and fluctuations in size may have a profound impact on dependent mangrove finch populations. Likewise, mangroves are declining globally due to human activity (Farnsworth and Ellison 1997; Alongi 2002; Alongi 2015), and lack of available habitat could slowly shrink mangrove finch populations. However, mangroves are known to undergo cycles of expansion and contraction according to changes in sea level, and thus may recover (Kendrick and Morse 1990). Regardless of the historical and ongoing causes of decline, introduced predators and a deadly nest parasite (Philornis downsi) are contributing to the current rapid decline of mangrove finches (Fessl et al. 2006, 2010a, b; Kleindorfer et al. 2014b).
We conclude that the decline of the mangrove finch is likely caused by a number of current and historical factors including an initially small founding stock and specialization to a narrowly distributed habitat that is declining and/or changing. This historically small lineage will likely continue to face challenges associated with small specialist species surrounded by a more widely-distributed sister lineage that produces viable hybrids despite current conservation efforts including rat control and intensive management through hand-rearing of chicks to circumvent the deadly Philornis larvae (Cunninghame et al. 2015). However, successful species rescue of nearly extinct bird species from very few individuals is possible (e.g., Chatham Island black robin, California condor, Norfolk Island green parrots; Butchart et al. 2006), and the extensive conservation management efforts underway for the mangrove finches on Isabela may be sufficient to save this species too from extinction.
References
Adams JR, Vucetich LM, Hedrick PW et al (2011) Genomic sweep and potential genetic rescue during limiting environmental conditions in an isolated wolf population. Proc R Soc B 278:3336–3344. doi:10.1098/rspb.2011.0261
Allentoft ME, Heller R, Oskam CL et al (2014) Extinct New Zealand megafauna were not in decline before human colonization. PNAS 111:4922–4927. doi:10.1073/pnas.1314972111
Alongi DM (2002) Present state and future of the world’s mangrove forests. Environ Conserv 29:331–349. doi:10.1017/S0376892902000231
Alongi DM (2015) The impact of climate change on mangrove forests. Curr Clim Change Rep 1:30–39. doi:10.1007/s40641-015-0002-x
Anderson E, Stebbins GL (1954) Hybridization as an evolutionary stimulus. Evolution 8:378–388. doi:10.2307/2405784
Antao T, Pérez-Figueroa A, Luikart G (2011) Early detection of population declines: high power of genetic monitoring using effective population size estimators. Evolut Appl 4:144–154. doi:10.1111/j.1752-4571.2010.00150.x
Arnold ML (2004) Transfer and origin of adaptations through natural hybridization: were Anderson and Stebbins right? Plant Cell 16:562–570. doi:10.1105/tpc.160370
Blackburn TM, Cassey P, Duncan RP et al (2004) Avian extinction and mammalian introductions on oceanic islands. Science 305:1955–1958. doi:10.1126/science.1101617
Bristol RM, Tucker R, Dawson DA et al (2013) Comparison of historical bottleneck effects and genetic consequences of re-introduction in a critically endangered island passerine. Mol Ecol 22:4644–4662. doi:10.1111/mec.12429
Brumm H, Farrington H, Petren K, Fessl B (2010) Evolutionary dead end in the Galápagos: divergence of sexual signals in the rarest of Darwin’s finches. PLoS One 5:e11191. doi:10.1371/journal.pone.0011191
Butchart SHM, Stattersfield AJ, Collar NJ (2006) How many bird extinctions have we prevented? Oryx 40:266–278. doi:10.1017/S0030605306000950
Case TJ (1996) Global patterns in the establishment and distribution of exotic birds. Biol Conserv 78:69–96. doi:10.1016/0006-3207(96)00019-5
Causton CE, Peck SB, Sinclair BJ et al (2006) Alien insects: threats and implications for conservation of Galápagos Islands. Ann Entomol Soc Am 99:121–143. doi:10.1603/0013-8746(2006)099
Cornuet JM, Luikart G (1996) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144:2001–2014
Cruz F, Josh Donlan C, Campbell K, Carrion V (2005) Conservation action in the Galàpagos: feral pig (Sus scrofa) eradication from Santiago Island. Biol Conserv 121:473–478. doi:10.1016/j.biocon.2004.05.018
Cunninghame F, Young H, Sevilla C, et al (2013) A trial translocation of the critically endangered mangrove finch: conservation management to prevent the extinction of Darwin’s rarest finch. Galapagos Report 2011–2012 GNPD, GCREG, CDF and GC Puerto Ayora, Galapagos, Ecuador 174–179
Cunninghame F, Switzer R, Parks B, et al (2015) Conserving the critically endangered mangrove finch: head-starting to increase population size. Galapagos Report 2013–2014 GNPD, GCREG, CDF and GC Puerto Ayora, Galapagos, Ecuador 151–157
Donlan CJ, Wilcox C (2008) Diversity, invasive species and extinctions in insular ecosystems. J Appl Ecol 45:1114–1123. doi:10.1111/j.1365-2664.2008.01482.x
Dussex N, Rawlence NJ, Robertson BC (2015) Ancient and contemporary DNA reveal a pre-human decline but no population bottleneck associated with recent human persecution in the Kea (Nestor notabilis). PLoS One 10:e0118522. doi:10.1371/journal.pone.0118522
Dvorak M, Vargas H, Fessl B, Tebbich S (2004) On the verge of extinction: a survey of the mangrove finch Cactospiza heliobates and its habitat on the Galápagos Islands. Oryx 38:171–179
Dvorak M, Fessl B, Nemeth E et al (2012) Distribution and abundance of Darwin’s finches and other land birds on Santa Cruz Island, Galápagos: evidence for declining populations. Oryx 46:78–86. doi:10.1017/S0030605311000597
Earl DA, vonHoldt BM (2011) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. doi:10.1007/s12686-011-9548-7
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. doi:10.1111/j.1365-294X.2005.02553.x
Evans SR, Sheldon BC (2008) Interspecific patterns of genetic diversity in birds: correlations with extinction risk. Conserv Biol 22:1016–1025. doi:10.1111/j.1523-1739.2008.00972.x
Excoffier L, Lischer HE (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567
Excoffier L, Laval G, Schneider S (2005) Arlequin (version 3.0): an integrated software package for population genetics data analysis. Evolutionary bioinformatics online 1:47
Farnsworth EJ, Ellison AM (1997) The global conservation status of mangroves. Ambio (Sweden) 26:328–334
Farrington HL, Petren K (2011) A century of genetic change and metapopulation dynamics in the Galápagos Warbler finches (Certhidea). Evolution 65:3148–3161. doi:10.1111/j.1558-5646.2011.01385.x
Farrington HL, Lawson LP, Clark CM, Petren K (2014) The evolutionary history of Darwin’s finches: speciation, gene flow, and introgression in a fragmented landscape. Evolution 68:2932–2944. doi:10.1111/evo.12484
Fessl B, Kleindorfer S, Tebbich S (2006) An experimental study on the effects of an introduced parasite in Darwin’s finches. Biol Conserv 127:55–61. doi:10.1016/j.biocon.2005.07.013
Fessl B, Loaiza AD, Tebbich S, Young HG (2010a) Feeding and nesting requirements of the critically endangered mangrove finch Camarhynchus heliobates. J Ornithol 152:453–460. doi:10.1007/s10336-010-0610-0
Fessl B, Young GH, Young RP et al (2010b) How to save the rarest Darwin’s finch from extinction: the mangrove finch on Isabela Island. Philos Trans R Soc of Lond B 365:1019–1030. doi:10.1098/rstb.2009.0288
Fessl B, Dvorak M, Vargas H, Young HG (2011) Recent conservation efforts and identification of the critically endangered mangrove Finche Camarhynchus heliobates in the Galapagos. Contiga 33:27–33
Garza JC, Williamson EG (2001) Detection of reduction in population size using data from microsatellite loci. Mol Ecol 10:305–318. doi:10.1046/j.1365-294X.2001.01190.x
Goudet J (1995) FSTAT (version 1.2): a computer program to calculate F-statistics. J Hered 86:485–486
Grant PR, Grant BR (1992) Hybridization of bird species. Science 256:193–197. doi:10.1126/science.256.5054.193
Grant PR, Grant BR (1997) The Rarest of Darwin’s finches. Conserv Biol 11:119–126. doi:10.1046/j.1523-1739.1997.95399.x
Grant PR, Grant BR (2014) 40 Years of Evolution: Darwin’s Finches on Daphne Major Island. Princeton University Press, Woodstock
Grant PR, Grant BR, Markert JA et al (2004) Convergent evolution of Darwin’s finches caused by introgressive hybridization and selection. Evolution 58:1588–1599
Grant PR, Grant BR, Petren K (2005a) Hybridization in the recent past. Am Nat 166:56–67
Grant PR, Grant BR, Petren K, Keller LF (2005b) Extinction behind our backs: the possible fate of one of the Darwin’s finch species on Isla Floreana, Galápagos. Biol Conserv 122:499–503
Harper GA, Carrion V (2011) Introduced rodents in the Galápagos: colonisation, removal and the future. In: Veitch CR, Clout MN, Towns DR (eds) Island invasives: eradication and management. IUCN, Gland, pp 63–66
Harris MP (1973) The Galápagos avifauna. Condor 75:265–278. doi:10.2307/1366166
Hedrick PW (2009) Evolutionary rescue in a changing world. Conserv Biol 9:996–1007. doi:10.1046/j.1523-1739.1995.9050988.x-i1
Hedrick PW (2013) Adaptive introgression in animals: examples and comparison to new mutation and standing variation as sources of adaptive variation. Mol Ecol 22:4606–4618. doi:10.1111/mec.12415
Hedrick P, Waits L (2005) Conservation genetics: what ancient DNA tells us. Heredity 94:463–464. doi:10.1038/sj.hdy.6800647
Hill WG (1981) Estimation of effective population size from data on linkage disequilibrium. Genet Res 38:209–216. doi:10.1017/S0016672300020553
IUCN (2016) The IUCN red list of threatened species. Version 2016–2
Kalinowski ST, Taper ML, Marshall TC (2007) Revising how the computer program cervus accommodates genotyping error increases success in paternity assignment. Mol Ecol 16:1099–1106. doi:10.1111/j.1365-294X.2007.03089.x
Keller LF, Jeffery KJ, Arcese P et al (2001) Immigration and the ephemerality of a natural population bottleneck: evidence from molecular markers. Proc R Soc Lond B 268:1387–1394. doi:10.1098/rspb.2001.1607
Kendrick GW, Morse K (1990) Evidence of recent mangrove decline from an archaeological site in Western Australia. Aust J Ecol 15:349–353. doi:10.1111/j.1442-9993.1990.tb01040.x
Kleindorfer S, O’Connor JA, Dudaniec RY et al (2014a) Species collapse via hybridization in Darwin’s tree finches. Am Nat 183:325–341. doi:10.1086/674899
Kleindorfer S, Peters KJ, Custance G et al (2014b) Changes in Philornis infestation behavior threaten Darwin’s finch survival. Curr Zool 60:542–550
Lamichhaney S, Berglund J, Almén MS et al (2015) Evolution of Darwin’s finches and their beaks revealed by genome sequencing. Nature 518:371–375. doi:10.1038/nature14181
Lee TM, Jetz W (2011) Unravelling the structure of species extinction risk for predictive conservation science. Pro R Soc Lond 278:1329–1338. doi:10.1098/rspb.2010.1877
Levin II, Zwiers P, Deem SL et al (2013) Multiple lineages of avian malaria parasites (Plasmodium) in the Galapagos Islands and evidence for arrival via migratory birds. Conserv Biol 27:1366–1377. doi:10.1111/cobi.12127
Loehle C, Eschenbach W (2012) Historical bird and terrestrial mammal extinction rates and causes. Divers Distrib 18:84–91. doi:10.1111/j.1472-4642.2011.00856.x
Mauchamp A, Atkinson R (2010) Rapid, recent and irreversible habitat loss: Scalesia forest on the Galapagos Islands. Galapagos Report 2009–2010 GNPD, GCREG, CDF and GC Puerto Ayora, Galapagos, Ecuador, 108-112
Melbourne BA, Hastings A (2008) Extinction risk depends strongly on factors contributing to stochasticity. Nature 454:100–103. doi:10.1038/nature06922
Méndez M, Vögeli M, Tella JL, Godoy JA (2014) Joint effects of population size and isolation on genetic erosion in fragmented populations: finding fragmentation thresholds for management. Evol Appl 7:506–518. doi:10.1111/eva.12154
O’Grady JJ, Brook BW, Reed DH et al (2006) Realistic levels of inbreeding depression strongly affect extinction risk in wild populations. Biol Cons 133:42–51. doi:10.1016/j.biocon.2006.05.016
Parker PG, Whiteman NK (2012) Evolution of pathogens and parasites on the Galápagos Islands. In: Wolff M, Gardener M (eds) The Role of science for conservation. Routledge, New York, pp 35–51
Parker PG, Buckles EL, Farrington H et al (2011) 110 Years of Avipoxvirus in the Galapagos Islands. PLoS One 6:e15989. doi:10.1371/journal.pone.0015989
Peel D, Ovenden JR, Peel SL (2004) NeEstimator: software for estimating effective population size, Version 1.3
Petren K, Grant BR, Grant PR (1999) A phylogeny of Darwin’s finches based on microsatellite DNA length variation. Proc R Soc Lond B 266:321–329
Petren K, Grant PR, Grant BR, Keller LF (2005) Comparative landscape genetics and the adaptive radiation of Darwin’s finches: the role of peripheral isolation. Mol Ecol 14:2943–2957. doi:10.1111/j.1365-294X.2005.02632.x
Petren K, Grant PR, Grant BR et al (2010) Multilocus genotypes from Charles Darwin’s finches: biodiversity lost since the voyage of the Beagle. Philos Trans R Soc Lond B 365:1009–1018. doi:10.1098/rstb.2009.0316
Piry S, Luikart G, Cornuet J-M (1999) Computer note. BOTTLENECK: a computer program for detecting recent reductions in the effective size using allele frequency data. J Hered 90:502–503. doi:10.1093/jhered/90.4.502
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Purvis A, Gittleman JL, Cowlishaw G, Mace GM (2000) Predicting extinction risk in declining species. Proc R Soc Lond B 267:1947–1952. doi:10.1098/rspb.2000.1234
Raymond M, Rousset F (1995) GENEPOP (Version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Rhymer JM, Simberloff D (1996) Extinction by hybridization and introgression. Annu Rev Ecol Syst 27:83–109
Rivera-Parra JL, Levin II, Parker PG (2014) Comparative ectoparasite loads of five seabird species in the Galapagos Islands. J Parasitol 100:569–577. doi:10.1645/12-141.1
Rollins LA, Whitehead MR, Woolnough AP et al (2015) Is there evidence of selection in the dopamine receptor D4 gene in Australian invasive starling populations? Curr Zool 61:505–519
Schwartz MK, Luikart G, Waples RS (2007) Genetic monitoring as a promising tool for conservation and management. Trends Ecol Evol 22:25–33. doi:10.1016/j.tree.2006.08.009
Smith KF, Sax DF, Lafferty KD (2006) Evidence for the role of infectious disease in species extinction and endangerment. Conserv Biol 20:1349–1357. doi:10.1111/j.1523-1739.2006.00524.x
Snodgrass RE, Heller E (1904) Papers from the Hopkins Stanford Galapagos expedition, 1898–1899. XVI. Birds. Proc Wash Acad Sci 5:231–372
Spurgin LG, Wright DJ, van der Velde M et al (2014) Museum DNA reveals the demographic history of the endangered Seychelles warbler. Evol Appl 7:1134–1143. doi:10.1111/eva.12191
Steadman DW, Stafford TW Jr, Donahue DJ, Jull AJT (1991) Chronology of holocene vertebrate extinction in the Galápagos Islands. Quatern Res 36:126–133. doi:10.1016/0033-5894(91)90021-V
Todesco M, Pascual MA, Owens GL et al (2016) Hybridization and extinction. Evol Appl. doi:10.1111/eva.12367
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) Micro-checker: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538. doi:10.1111/j.1471-8286.2004.00684.x
Walsh SJ, Mena CF (2012) Science and Conservation in the Galapagos Islands: Frameworks & Perspectives. Springer, New York
Watson J, Trueman M, Tufet M et al (2010) Mapping terrestrial anthropogenic degradation on the inhabited islands of the Galapagos Archipelago. Oryx 44:79–82. doi:10.1017/S0030605309990226
Wikelski M, Foufopoulos J, Vargas H, Snell H (2004) Galápagos birds and diseases: invasive pathogens as threats for island species. Ecol Soc 9:5–15
Wium-Andersen S, Hamann O (1986) Manglares de las Islas Galápagos. Inst Geogr Militar Rev Geogr 23:101–122
Wolf DE, Takebayashi N, Rieseberg LH (2001) Predicting the risk of extinction through hybridization. Conserv Biol 15:1039–1053. doi:10.1046/j.1523-1739.2001.0150041039.x
Young HG, Cunninghame F, Fessl B, Vargas FH (2013) Mangrove finch Camarhynchus heliobates an obligate mangrove specialist from the Galapagos Islands. In: Gleason G, Victor TR (eds) Mangrove ecosystems. Nova Science Publishers Inc, New York, pp 107–121
Acknowledgments
We thank the Galápagos National Park Directorate, the Charles Darwin Research Station, and the California Academy of Sciences. This research was supported by Darwin Initiative (15-005, EIDP0031, 162/12/018), Save our Species (2011A-023), Galápagos Conservancy (002-2015) and International Community Foundation with a grant awarded by The Leona M. and Harry B. Helmsley Charitable Trust (20160098), Galápagos Conservation Trust (CT14-171), the Mohammed Bin Zayed Species Conservation Fund (14259510), Durrell Wildlife Conservation Trust, the National Science Foundation (DEB-0317687), the Frankfurt Zoological Society (FZS 1224/97), Swiss Association of Friends of the Galápagos Islands, and the Max Planck Society. We thank Andrew Clack for assisting in ancient DNA techniques, Bart Kempenaers for providing materials and facility access at Max Planck for genotyping, and Peter and Rosemary Grant who initiated the work on the mangrove finch and have, over the years, helped and advised on the project. Research was carried out under IACUC protocol (12-04-09-01) and with approval for ethical research from Galápagos National Park. This publication is contribution number 2139 of the Charles Darwin Foundation for the Galápagos Islands.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lawson, L.P., Fessl, B., Hernán Vargas, F. et al. Slow motion extinction: inbreeding, introgression, and loss in the critically endangered mangrove finch (Camarhynchus heliobates). Conserv Genet 18, 159–170 (2017). https://doi.org/10.1007/s10592-016-0890-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-016-0890-x