Abstract
The nucleolus contains a lot of proteins unrelated to ribosome biogenesis. Some of these proteins shuttle between the nucleolus and the nucleoplasm regulating the cell cycle and stress response. The piRNA binding protein Piwi is involved in silencing of transposable elements (TEs) in the Drosophila gonads. Here we used cultured ovarian somatic cells (OSC) to characterize Piwi as a visitor to the nucleolus. Dynamic Piwi localization was shown to vary from its uniform distribution between the nucleoplasm and the nucleolus to pronounced nucleolar immobilization. We were intrigued by this localization behavior and revealed that nascent nucleolar transcripts recruit Piwi for nucleolar retention. Piwi eviction from the nucleolus was observed upon RNase treatment and after RNA polymerase (Pol) I inhibition, but not after Pol II inactivation. On the contrary, heat shock caused drastic Piwi redistribution from the nucleoplasm to the nucleolus, which occurred only in the presence of Pol I-mediated transcription. These results allow us to hypothesize that specific stress-induced transcripts made by Pol I promote the nucleolar sequestration of proteins in Drosophila, similar to previous observations in mammalian cells. We also found that in OSC, Piwi partially restricts expression of the rDNA copies containing R1 and R2 retrotransposon insertions especially upon heat shock-induced activation of these copies. Therefore, we suggest that Piwi intranuclear shuttling may have a functional role in ensuring a balance between silencing of rDNA-specific TEs under stress and the canonical Piwi function in non-nucleolar TE repression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The nucleolus is a membraneless intranuclear organelle involved not only in rDNA transcription and ribosome biogenesis, but also in regulating nucleoplasmic protein functions ensuring control of cell proliferation, stress response, and genome stability (Pederson and Tsai 2009; Lindstrom et al. 2018). Dynamic behavior of nucleolus components was demonstrated by FRAP experiments and proteomics approaches (Pederson 2011; Boulon et al. 2010 for review) and recent studies allow to consider the nucleolus as a dynamic droplet that is assembled by intranuclear liquid-liquid phase separation (Weber and Brangwynne 2015; Zhu and Brangwynne 2015), which may be driven by the presence of RNA molecules and modulated by RNA transcription (Berry et al. 2015). Much attention has been devoted to the study of the mobility of proteins within the nucleus, and nucleolar components have been shown to be engaged in complex movements due to nucleolar stress induced by transcription repression and other causes (Andersen et al. 2005; Holmberg Olausson et al. 2012). A lot of proteins that have no known roles in the biosynthesis of ribosomes are sequestered or released by the nucleolus in response to changes in cellular growth conditions or different stresses (Mekhail et al. 2005; Boulon et al. 2010). For some of these proteins including p53, nucleostemin, MDM2, Cdc14 phosphatase and others, the transient nucleolar localization was shown to be important to perform their cellular functions (Emmott and Hiscox 2009; Pederson and Tsai 2009). In particular, nucleolar detention was shown to be necessary for temporary inactivation of protein. The molecular mechanisms that determine dynamic nucleolar localization of nucleoplasmic proteins remain incompletely understood. The role of long noncoding RNAs originating from intergenic transcripts of the rDNA cluster in mammals in the capture and immobilization of nucleoplasmic proteins, including Hsp70, VHL, DNMT1, and POLD1, has been reported (Audas et al. 2012; Lam and Trinkle-Mulcahy 2015; Nemeth and Grummt 2018 for review). The formation of these intergenic spacer transcripts was shown to be induced by different stresses, such as heat shock and acidosis.
Among the proteins having no known role in the biosynthesis of ribosomes, but partially localized to the nucleolus is piRNA pathway protein Piwi (Mikhaleva et al. 2015). The Drosophila piRNA pathway operates mainly in the germline and somatic gonadal tissues and includes both cytoplasmic and nuclear piRNA-binding proteins. Piwi is a nuclear effector that is able to identify sequences of nuclear transcripts complementary to piRNAs, leading to the recruitment of protein complexes involved in chromatin modification and transcription repression. The main function of Piwi is silencing of transposable elements (TEs) (Czech and Hannon 2016; Iwasaki et al. 2015, for reviews); however, the biological role of piRNA-Piwi complexes has been extended to the regulation of expression of a number of proteins and participation in diverse cellular processes in development (Sarkar et al. 2017; Barckmann et al. 2015; Rojas-Rios et al. 2017).
Here we revealed Piwi shuttling between the nucleoplasm and the nucleolus, resembling the dynamic localization behavior of some other proteins detected in mammalian cell studies (Audas et al. 2012; Abdelmohsen and Gorospe 2012). Our results indicate that Piwi nucleolar retention is mediated by specific nucleolar transcription. Taking into account the well-known Piwi function in TE repression, we started to study the effect of Piwi accumulation in the nucleolus on the expression of rDNA copies containing retrotransposon insertions. Our observations suggest that the biological significance of Piwi nucleolar immobilization may be connected with the control of these TEs upon stress conditions.
Materials and methods
Drosophila stocks
Piwi-GFP line was provided by Alexey Aravin and Katalin Fejes Toth (Le Thomas et al. 2013). Flies were maintained at 25 °C on standard medium. The ovaries from 1- to 6-d-old flies were dissected for immunostaining.
Cell culture, RNAi, and treatment conditions
Ovarian somatic cells (OSCs) provided by M. Siomi were grown at 25 °C in Shields and Sang M3 insect medium (Sigma-Aldrich) supplemented with 10% heat inactivated fetal bovine serum (Gibco), 10% fly extract (http://biology.st-andrews.ac.uk/sites/flycell/flyextract.html), 10 μg/ml insulin (Sigma-Aldrich), 0.6 mg/ml glutathione (Sigma-Aldrich), 50 units/ml penicillin and 50 μg/ml streptomycin as described previously (Saito et al. 2009).
Actinomycin D (Act D) was used in the concentration of 1 μg/ml for 30 min or 1 h; however, additional experiments showed a similar effect on Piwi localization after 0.1 μg/ml Act D treatment for 15 min. Treatment with other inhibitors was carried out as follows:
9-Hydroxyellipticine (9НЕ) 5–10 μM 1 or 2 h; α-amanitin 20 μg/ml 2 h; 6-dichlorobenzimidazole 1-β-d-Ribofuranoside (DRB) 50 μM 4 h.
RNase A (Thermo Fisher Scientific) treatment of OSCs was done in the concentration of 0.05 mg/ml for 1 h. Acidosis was induced by 1 h incubation of growing cells in the same growth medium at pH 5.78. Oxidative stress was induced by diethyl maleate (2 mM 5 h). For heat shock, cells were incubated at 37 °C for 1 h.
To perform Piwi knockdown (Piwi KD) 40 pmol of siRNA duplex to piwi as well as to EGFP (control, Piwi non-KD) were nucleofected into 3 × 106 cells using the Cell Line 96-well Nucleofector Kit SF and program DG150 of the 96-well Shuttle Device (Lonza). Cells were harvested after 48 h incubation. The following siRNAs were used:
siEGFP-sense: GGCAAGCUGACCCUGAAGUTT
siEGFP-antisense: ACUUCAGGGUCAGCUUGCCTT
siPiwi-sense: GCUCCCAGGCGUGAAGGUGTT
siPiwi-antisense: CACCUUCACGCCUGGGAGCTT
The myc-Piwi plasmid kindly provided by M. Siomi (Saito et al. 2010) was transfected using FuGENE HD transfection reagent (#E2311, Promega) according to the manufacturer’s instructions.
Immunofluorescence
Immunostaining of OSC cells and ovaries was performed as described previously (Ilyin et al. 2017). The following primary antibodies were used: rabbit polyclonal anti-Piwi (1:500, provided by G. Hannon); mouse monoclonal anti-Piwi (serum, 1:5, provided by M. Siomi); rabbit polyclonal anti-Piwi (1:1000; Santa Cruz Biotechnology, sc-98,264); rabbit polyclonal anti-fibrillarin (1:1000, Abcam); mouse monoclonal anti-fibrillarin (1:300, Abcam, 4566); rabbit anti-lamin (1:500, provided by P. Fisher) (Osouda et al. 2005); guinea pig polyclonal anti-LBR (1:1000) (Wagner et al. 2004); guinea pig polyclonal anti-Udd (1:800, provided by M. Buszczak) (Zhang et al. 2014); and mouse monoclonal anti-c-MYC (1:100, DSHB, # 9E 10). Secondary antibodies (Invitrogen, Thermo Fisher Scientific) were the following: anti-rabbit IgG Alexa Fluor 488; anti-rabbit IgG Alexa Fluor 546; anti-rabbit IgG Alexa Fluor 633; anti-mouse IgG Alexa Fluor 488; anti-mouse IgG Alexa Fluor 633; and anti-guinea pig IgG Alexa Fluor 633. Confocal microscopy was done using LSM 510 META system (Zeiss).
EU (5-ethynyluridine) and H3-uridine incorporation assays
Nascent RNA in nucleoli was labeled with EU (#E10345, Life Technologies) added to the culture medium to a 1 mM final concentration for 1 h. Cells were fixed, permeabilized as described (Ilyin et al. 2017), and processed for EU label detection using the Click-iT™ reaction according to the manufacturer’s instructions. Click-iT reaction was carried out in a cocktail containing Alexa Fluor 647 azide, triethylammonium salt (#A10277, Invitrogen, Thermo Fisher Scientific) and Reaction Buffer Kit (#C10269, Invitrogen, Thermo Fisher Scientific) 30 min in the dark at room temperature. Then cells were washed in PBTX and processed for immunostaining. Quantification of EU signals was performed using ImageJ software. Thirty cell nucleoli per experimental condition (after heat shock; non-heat shocked control) were analyzed.
For the H3-uridine incorporation assay, [5,6-3H]Uridine was added to the culture medium to the final activity 1 MBq/ml for 1.5 h after 1 h of each inhibitor treatment.
RT-qPCR
Total RNA was extracted using Trizol reagent (Invitrogen, Thermo Fisher Scientific) and precipitated with 70% ethanol at − 20 °C. DNA was removed by precipitation with 3.3 M LiCl at − 20 °C and subsequent DNAse I (Ambion) treatment for 1 h at 37 °C according to the manufacturer’s instructions. 1 μg of RNA was used for the reverse transcription reaction with random primer and Mint reverse transcriptase (Evrogen). The resulting cDNAs were analyzed by RT-qPCR performed on DT96 real-time DNA amplifier (DNA-Technology) using SYBR Green chemistry (Applied Biosystems). The following primers were used for PCR:
R1s1 AATTCCCGAGCTGTGCTAGA and R1as1 GTCTCAAGGCACCTTTCAGC (for R1);
R2s1 TGCTCCCGAAACAACAAACCAC and R2as1 AACAATGACCACGCAGCCTC (for R2); R2rRs1 ACACACAGTGTTGGCAGACCTA and R2rRas1 CCTTGGCTGTGGTTTCGCT (for R2-28S co-transcript); Zam_for3 TCACATCCTTCCAGCAATCTTCAA and Zam_rev3 TATTACAGTTTCTGACATTATTTCTTCGTG (for Zam); MDG1_dir AACAGAAACGCCAGCAACAGC and MDG1_rev CGTTCCCATGTCCGTTGTGAT (for mdg1); and Rp49_up ATGACCATCCGCCCAGCATAC and Rp49_rev2 GCTTAGCATATCGATCCGACTGG (for rp49).
Results
Dynamic Piwi nucleolar retention depends on elevated temperature and Pol I-mediated transcription
Our previous studies of the Piwi in TE silencing allowed us to detect its inhomogeneous distribution in the nucleus of somatic and germ cells of Drosophila melanogaster ovaries and embryos (Mikhaleva et al. 2015). For further studies of environmental and intrinsic factors that may impact Piwi intranuclear localization, we used cultured ovarian somatic cells (OSCs) known to express Piwi (Saito et al. 2009). Piwi enrichment in OSC nucleoli was confirmed by its strong colocalization with a well-known nucleolar marker, fibrillarin (Fig. 1a, left panel), subunit of Pol I regulatory complex, Udd (Fig. 1b) (Zhang et al. 2014), and some other nucleolar proteins. The nucleolar Piwi enrichment was detected using different Piwi antibodies, as well as by Myc antibody staining of OSCs transfected with the Myc-Piwi construct (Fig. 1c). However, we revealed that Piwi localization sometimes varied from pronounced nucleolar immobilization (Fig. 1a, left panel) to a virtually uniform distribution between the nucleoplasm and the nucleolus (Fig. 1a, right panel). Causes of these variations are unclear and possibly related to the physiological state of the cells.
Piwi nucleolar localization in OSCs depends on Pol I–mediated transcription. a Pronounced Piwi (green) and fibrillarin (red) colocalization (left panel) or uniform Piwi distribution between the nucleoplasm and the nucleolus (right panel) under normal conditions. b Piwi colocalization with a subunit of Pol I regulatory complex, Udd (red). c Myc staining (green) of OSCs transfected with Myc-Piwi construct. d RNase A and actinomycin D (ActD) treatments cause Piwi eviction from the nucleolus (indicated by white arrows). e Cell treatment by Pol I-specific inhibitor 9HE, but not by Pol II inhibitor DRB leads to the loss of Piwi colocalization with fibrillarin. f 9HE treatment for 2 h drastically reduces the 5-ethynyl uridine (EU) incorporation (red) in the nucleoli demonstrating the inhibition of nucleolar transcription. Lamin (purple) indicates the nuclear envelope. Scale bars, 5 μm
We checked whether Piwi nucleolar localization is RNA-dependent. The nucleolar enrichment of Piwi completely vanished after cell treatment with RNase A leading to visualization of Piwi unstained nucleolar holes containing fibrillarin (Fig. 1d). OSC treatment with actinomycin D that inhibits all RNA polymerases (Bensaude 2011) or by the recently reported selective Pol I inhibitor, 9-hydroxyellipticin (thereafter 9НЕ) (Andrews et al. 2013), both caused Piwi eviction from the nucleolus (Fig. 1d, e). The effectiveness of the 9HE treatment for repression of nucleolar transcription in OSCs was demonstrated by 5-ethynyl uridine (EU) incorporation (Fig. 1f) and H3-uridine incorporation (Fig. S1) assays. At the same time Pol II inhibitors α-ammanitin (not shown) and DRB (Panse et al. 1999) exerted no effect on Piwi nucleolar localization (Fig. 1e). Thus, only Pol I but not Pol II–mediated transcription drives Piwi retention in the nucleolus.
The nucleolus plays an important role as a sensor and coordinator of the stress response. It is known that different cellular stresses affect nucleolar proteome content and drive protein release from the nucleolus or, by contrast, cause protein nucleolar sequestration (Andersen et al. 2005; Boulon et al. 2010). For instance, heat shock is a well-known physiological stimulus that induces nucleolar detention of Hsp70 and other proteins (Welch and Suhan 1985; Nollen et al. 2001; Audas et al. 2012). We evaluated the influence of some stress agents on Piwi accumulation in the nucleolus. Acidosis caused Piwi release from the nucleolus (Fig. 2a), whereas the oxidative stress agent diethyl maleate (2 mM 5 h) exerted no effect (not shown). Next, we revealed a strongly enhanced Piwi accumulation in the nucleolus accompanied by its leaving the nucleoplasm as a result of heat shock (37 °C, 1 h) (Fig. 2b). Importantly, the heat shock–induced Piwi movement to the nucleolus was prevented by 9HE inhibitor treatment revealing the dependence of this effect on Pol I-mediated transcription (Fig. 2c).
Cellular stress induced by heat shock, but not by acidosis, enhances Pol I–dependent accumulation of Piwi in the nucleolus despite the reduction of the total nucleolar transcription. a Acidosis causes Piwi (green) redistribution from the nucleolus marked by fibrillarin (red) to the nucleoplasm. b Heat shock (HS, 37 °C, 1 h) induces pronounced Piwi enrichment in the nucleolus accompanied by its leaving the nucleoplasm. Lamin B-receptor (LBR) staining (purple) marks the nuclear envelope. c Heat shock-induced Piwi detention in the nucleolus is prevented by repression of Pol I–mediated transcription. Cells were treated with 9HE inhibitor before or after heat shock (indicated as 9HE+HS and HS+9HE, respectively). In both cases, the pronounced nucleolar enrichment of Piwi was lost (indicated by arrows). Cells treated with 9HE before heat shock show also partial Piwi redistribution to the cytoplasm. d Heat shock (37 °C, 1 h) reduces but does not completely block nucleolar transcription, measured by the nucleolar EU incorporation assay. In the box plots, the median is represented by a thick horizontal line, the upper and lower hinges are 75th and 25th percentile, respectively, the whiskers indicate the highest and lowest values (n = 30; Student’s t test; p = 5.4 × 10−19). Scale bars, 5 μm
The repression of rDNA transcription caused by heat stress of mammalian cells and mediated by antisense transcription has been demonstrated (Ghoshal and Jacob 1996; Zhao et al. 2016; Zhao et al. 2018). In Drosophila, elevated temperature was shown to reduce rRNA synthesis (Bell et al. 1988) and a recent study revealed a strong but not complete decrease of Pol I densities on rDNA copies upon heat treatment in Drosophila cells (Raje et al. 2018). In concert with these results, we found about a threefold reduction of nucleolar transcription after 1 h of OSC heat shock (37 °C) using EU incorporation assay (Fig. 2d). The decrease of total nucleolar transcription due to the heat stress implies that the Piwi nucleolar sequestration may be provided not by ordinary rRNA transcription, but by a certain specific fraction of Pol I-produced RNAs. Our results represent the new evidence that Pol I-mediated transcription is an important factor in attracting nucleoplasmic proteins to the nucleolus, taking into account previously reported data on the role of stress-induced transcription of intergenic rDNA spacers (IGS) in protein detention in the nucleolus in mammalian cells (Audas et al. 2012; Wang et al. 2018). Of note, these IGS transcripts are produced by Pol I under stress conditions that repress normal rRNA transcription (Audas et al. 2012; Jacob et al. 2013). The nature of Pol I transcripts recruiting Piwi to the Drosophila nucleolus remains obscure.
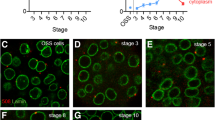
Next, we checked whether heat shock–dependent redistribution of Piwi from nucleoplasm to the nucleolus in cultured cells reflects the same stress response of ovarian cells in vivo. For this purpose, we studied the behavior of Piwi upon heat shock in ovaries of flies carrying the Piwi-GFP transgene. Drosophila ovaries are composed of ovarioles that contain progressively developing egg chambers. Each egg chamber includes a cluster of 16 germline cells (fifteen polyploid nurse cells and single oocyte) surrounded by a monolayer of somatic follicle cells. Normally, nucleoli of follicle cells are enriched in Piwi (Mikhaleva et al. 2015), whereas the degree of Piwi colocalization with the nucleolus in germ cells varies considerably (data not shown). Note that the nucleolus of a polyploid nurse cell is a branched structure that is differed from an ordinary round nucleolus (Dapples and King 1970). Elevated temperature caused pronounced Piwi-GFP detention in the nucleoli of both somatic and germline cells. In follicle cells, Piwi-GFP colocalization with fibrillarin was enhanced after heat shock (Fig. 3). In the nurse cells, Piwi-GFP strongly corresponded to the localization pattern of fibrillarin instead of uniformly distributed Piwi-GFP in unstressed nuclei (Fig. 3). Similar results were obtained by immunostaining of endogenous Piwi in ovaries (data not shown).
Heat shock induces Piwi accumulation in nucleoli of somatic and germline cells in ovaries. Egg chamber from control (upper panel) and heat shocked flies (HS, 37 °C, 2 h, lower panel). Without heat shock, Piwi-GFP is uniformly distributed in germline nurse cell (NC) nuclei, whereas some follicle cell (FC) nuclei show Piwi-GFP preference to colocalize with fibrillarin (arrows). Heat shock causes pronounced Piwi-GFP enrichment in both FC and NC nucleoli. Scale bars, 10 μm
Piwi partially restricts heat shock–induced activation of rDNA-specific retrotransposons R1 and R2
In Drosophila melanogaster, some rDNA units contain R1 and R2 non-LTR retrotransposons inserted into specific sites of the 28S rDNA sequence (Eickbush and Eickbush 2015). Importantly, R2 elements lacking their own promoters are transcribed only as a part of rRNA precursor transcripts and can then be excised by self-splicing (Eickbush and Eickbush 2010). Thus, transcription of R2 sequence is considered as a marker of the corresponding rDNA unit expression. Generally, rDNA genes with R1 and R2 insertions exhibit very low levels of transcription compared to normal uninserted rDNA genes (Jamrich and Miller 1984; Ye and Eickbush 2006; Eickbush and Eickbush 2015). This repression was suggested to be defined by formation of heterochromatin domains within rDNA cluster (Eickbush and Eickbush 2015). Also, the DNA-binding regulatory protein CTCF was shown to be involved in this process (Guerrero and Maggert 2011). Nevertheless, the mechanism of R1/R2 silencing remains underexplored.
It has been reported by the DiMario group that heat shock treatment of Drosophila larval and cultured S2 cells induces Pol I–dependent upregulation of R1 but not R2 element transcription (He et al. 2015; Raje et al. 2018). The piRNA pathway can potentially be involved in the control of retrotransposon-containing rDNA genes. We hypothesized that Piwi could function in the nucleolus as a regulator of the expression of inserted rDNA units during their abnormal activation due to the cellular stress. To address this question, we checked the effect of Piwi RNAi knockdown (Piwi-KD) on R1 and R2 transcript abundances upon heat shock. Quantitative RT-PCR revealed that a 1-h heat shock of the control OSC cells treated with siRNAs to GFP (hereafter, non-KD cells) resulted in 25-fold and fourfold increase of R1 and of R2 transcript levels, respectively (Fig. 4). We also observed a heat shock–induced tenfold upregulation of unprocessed transcripts encompassing the R2 3′-end and the downstream 28S rRNA sequence (R2-28S) (Fig. 4). Piwi KD without heat shock led to a non-significant increase in R1 expression and sevenfold increase in R2 transcript level compared to non-KD cells, whereas Piwi-KD cells upon heat shock exhibited significantly higher upregulation of R2 and R2-28S and especially R1 transcripts compared to both non-heat-shocked Piwi-KD and heat-shocked control cells (Fig. 4). To test whether this Piwi-regulated thermal stress-dependent activation is specific for the elements inserted into rDNA units, we checked the expression of non-nucleolar mdg1 and ZAM TEs, which are known to be strongly silenced by Piwi in ovarian somatic cells (Sienski et al. 2012; Rozhkov et al. 2013). These elements exhibited no or weak increase of transcript levels after heat shock in non-KD cells (Fig. 4). Heat shock upon Piwi KD did not lead to significant additional upregulation of mdg1 and ZAM compared to non-heat-shocked Piwi-KD cells (Fig. 4). Thus, our results show that Piwi specifically restricts heat shock-induced activation of rDNA genes containing retrotransposon insertions, especially of those with R1 elements. Our finding that the R2-containing rDNA copies are also susceptible to derepression due to heat shock (albeit to a lesser extent than R1), indicates that this effect reflects a general disruption of the mechanisms of inserted rDNA copy silencing due to nucleolar stress.
Piwi restricts heat shock–induced activation of rDNA-specific R1 and R2 retrotransposons. RT-qPCR values of transcript levels normalized to rp49 mRNA are indicated. Piwi-KD without heat shock upregulates R2 and R2-28S transcript levels, but does not significantly change R1 expression compared to control cells (ctrl). Heat shock of Piwi-KD cells (Piwi-KD HS) leads to a significantly higher increase of R2, unprocessed R2-28S co-transcripts, and especially R1 transcript levels, than heat shock of control cells (HS) or Piwi-KD without heat shock. Non-nucleolar retrotransposons mdg1 and ZAM are derepressed in Piwi-KD cells, but not in HS cells. In Piwi-KD HS cells, ZAM and mdg1 are upregulated non-significantly compared to non-heat-shocked Piwi-KD cells. Mean +/− s.d. are indicated. (Student’s t test; *p < 0.05, **p < 0.01, ns = not significant)
In summary, we suggest that under normal conditions the repression of inserted rDNA units, which transcription is several orders of magnitude below the level of normal pre-rRNA (Eickbush and Eickbush 2015), is determined by the mechanism likely unrelated to piRNA pathway; however, upon heat shock, this putative silencing mechanism becomes partially impaired. Although Piwi normally makes only a small contribution to the repression of the inserted rDNA units, during thermal stress Piwi, being amassed within the nucleolus, can substantially restrict activation of R1 and R2 elements.
Discussion
Here we demonstrated that Piwi localization in the nucleolus is dynamic and depends on the presence of Pol I–mediated transcription. This conclusion is based on the observation of Piwi eviction from the nucleolus in the presence of a specific inhibitor of Pol I transcription, 9HE. Piwi nucleolar accumulation in OSC cells seems to be transient, but factors driving this nucleolus-nucleoplasm shuttling are obscure and remain subject to further research. Nevertheless, our results suggest that the Piwi nucleolar sequestration upon stress depends on specific Pol I-produced transcripts, but not on general rRNA transcription, allowing us to draw parallels with a mechanism of IGS-mediated protein nucleolar detention in mammalian cells (Audas et al. 2012; Wang et al. 2018). It also cannot be excluded that R1/R2 transposon transcripts overexpressed upon heat shock may participate in nucleolar recruitment of Piwi. In addition, the possible role of chaperone machinery in stress-induced Piwi accumulation in the nucleolus can be assumed, taking into account the earlier reports indicating that chaperones are not only involved in stabilizing of nuclear proteins but can also drive them to the nucleolus during stress in mammalian cells (Nollen et al. 2001). Of note, functional interactions between Piwi and the Hsp90 chaperone were clearly demonstrated in Drosophila (Specchia et al. 2010; Gangaraju et al. 2011; Izumi et al. 2013).
Heterochromatinization of repeated rDNA copies is necessary to stabilize the rDNA cluster and prevent recombination, whereas upon stress rDNA transcription is usually repressed by additional chromatin modifications (Nemeth and Grummt 2018 for review). Epigenetic control of Drosophila rDNA genes remains underexplored compared to the mammalian rDNA clusters, where chromatin alterations are assisted by various long non-coding RNAs under both normal (Guetg et al. 2012; Xing et al. 2017; Mayer et al. 2006) and stress conditions (Bierhoff et al. 2014; Zhao et al. 2018). In particular, antisense long non-coding RNA mediates attenuation of murine pre-rRNA synthesis upon heat stress (Zhao et al. 2018). Some reports hint at the possibility of small RNA participation in the modulation of rDNA expression. For example, deeply conserved microRNA was shown to be hosted by internal transcribed spacer 1 region of Drosophila rDNA cluster (Chak et al. 2015). Human Argonaute 2 (Ago2) was found to be tethered to nascent rRNA through microRNA interactions (Atwood et al. 2016). rDNA-derived piRNAs and siRNAs which are probably formed as a result of rRNA cleavage and possibly have some functions, were found in different model organisms (Wei et al. 2013; García-López et al. 2015). Although the role of Piwi-piRNA pathway in transcriptional silencing of TEs and promotion of heterochromatin assembly has been thoroughly investigated in Drosophila (Czech and Hannon 2016; Iwasaki et al. 2015, for reviews), it remains unknown whether Piwi exerts any effect on the chromatin of the rDNA cluster during stress or acts posttranscriptionally in R1/R2 element silencing.
Our observation that upon heat shock Piwi accumulates in the nucleolus and concurrently restricts the activation of R1 and R2 elements indicates a possible functional role of Piwi intranuclear shuttling in the silencing of inserted rDNA copies. We propose that Piwi relocalization from nucleoplasm to the nucleolus may reflect a switch from its canonical role in non-nucleolar TE repression to specific nucleolar tasks. In general, this implies the existence of a finely tuned system that determines the balance between the nucleolar and nucleoplasmic localization of Piwi.
Abbreviations
- TEs:
-
Transposable elements
- OSC:
-
Ovarian somatic cells
- 9HE:
-
9-Hydroxyellipticine
- EU:
-
5-Ethynyl uridine
References
Abdelmohsen K, Gorospe M (2012) RNA-binding protein nucleolin in disease. RNA Biol 9:799–808. https://doi.org/10.4161/rna.19718
Andersen JS, Lam YW, Leung AK, Ong SE, Lyon CE, Lamond AI, Mann M (2005) Nucleolar proteome dynamics. Nature 433:77–83. https://doi.org/10.1038/nature03207
Andrews WJ, Panova T, Normand C, Gadal O, Tikhonova IG, Panov KI (2013) Old drug, new target: ellipticines selectively inhibit RNA polymerase I transcription. J Biol Chem 288:4567–4582. https://doi.org/10.1074/jbc.M112.411611
Atwood BL, Woolnough JL, Lefevre GM, Saint Just Ribeiro M, Felsenfeld G, Giles KE (2016) Human Argonaute 2 is tethered to ribosomal RNA through MicroRNA interactions. J Biol Chem 291:17919–17928. https://doi.org/10.1074/jbc.M116.725051
Audas TE, Jacob MD, Lee S (2012) Immobilization of proteins in the nucleolus by ribosomal intergenic spacer noncoding RNA. Mol Cell 45:147–157. https://doi.org/10.1016/j.molcel.2011.12.012
Barckmann B, Pierson S, Dufourt J, Papin C, Armenise C, Port F, Grentzinger T, Chambeyron S, Baronian G, Desvignes JP, Curk T, Simonelig M (2015) Aubergine iCLIP reveals piRNA-dependent decay of mRNAs involved in germ cell development in the early embryo. Cell Rep 12:1205–1216. https://doi.org/10.1016/j.celrep.2015.07.030
Bell J, Neilson L, Pelligrini M (1988) Effect of heat shock on ribosomal synthesis in Drosophila melanogaster. Mol Cell Biol 8(1):91–95. https://doi.org/10.1128/MCB.8.1.91
Bensaude O (2011) Inhibiting eukaryotic transcription: which compound to choose? How to evaluate its activity? Transcription 3:103–108. https://doi.org/10.4161/trns.2.3.16172
Berry J, Weber SC, Vaidya N, Haataja M, Brangwynne CP (2015) RNA transcription modulates phase transition-driven nuclear body assembly. Proc Natl Acad Sci U S A 112:E5237–E5245. https://doi.org/10.1073/pnas.1509317112
Bierhoff H, Dammert MA, Brocks D, Dambacher S, Schotta G, Grummt I (2014) Quiescence-induced LncRNAs trigger H4K20 trimethylation and transcriptional silencing. Mol Cell 54:675–682. https://doi.org/10.1016/j.molcel.2014.03.032
Boulon S, Westman BJ, Hutten S, Boisvert FM, Lamond AI (2010) The nucleolus under stress. Mol Cell 40:216–227. https://doi.org/10.1016/j.molcel.2010.09.024
Chak LL, Mohammed J, Lai EC, Tucker-Kellogg G, Okamura K (2015) A deeply conserved, noncanonical miRNA hosted by ribosomal DNA. RNA 21:375–384. https://doi.org/10.1261/rna.049098
Czech B, Hannon GJ (2016) One loop to rule them all: the ping-pong cycle and piRNA-guided silencing. Trends Biochem Sci 41:324–337. https://doi.org/10.1016/j.tibs.2015.12.008
Dapples CC, King RC (1970) The development of the nucleolus of the ovarian nurse cell of Drosophila melanogaster. Z Zellforsch Mikrosk Anat 103:34–47
Eickbush DG, Eickbush TH (2010) R2 retrotransposons encode a self-cleaving ribozyme for processing from an rRNA cotranscript. Mol Cell Biol 30:3142–3150. https://doi.org/10.1128/MCB.00300-10
Eickbush TH, Eickbush DG (2015) Integration, regulation, and long-term stability of R2 retrotransposons. Microbiol Spectrum 3: MDNA3–0011-2014 doi:https://doi.org/10.1128/microbiolspec.MDNA3-0011-2014
Emmott E, Hiscox JA (2009) Nucleolar targeting: the hub of the matter. EMBO Rep 10:231–238. https://doi.org/10.1038/embor.2009.14
Gangaraju VK, Yin H, Weiner MM, Wang J, Huang XA, Lin H (2011) Drosophila Piwi functions in Hsp90-mediated suppression of phenotypic variation. Nat Genet 43:153–158. https://doi.org/10.1038/ng.743
García-López J, Alonso L, Cárdenas DB, Artaza-Alvarez H, Hourcade Jde D, Martínez S, Brieño-Enríquez MA, Del Mazo J (2015) Diversity and functional convergence of small noncoding RNAs in male germ cell differentiation and fertilization. RNA 21:946–962. https://doi.org/10.1261/rna.048215.114
Ghoshal K, Jacob ST (1996) Heat shock selectively inhibits ribosomal RNA gene transcription and down-regulates E1BF/Ku in mouse lymphosarcoma cells. Biochem J 317:689–695. https://doi.org/10.1042/bj3170689
Guerrero PA, Maggert KA (2011) The CCCTC-binding factor (CTCF) of Drosophila contributes to the regulation of the ribosomal DNA and nucleolar stability. PLoS One 6:e16401. https://doi.org/10.1371/journal.pone.0016401
Guetg C, Scheifele F, Rosenthal F, Hottiger MO, Santoro R (2012) Inheritance of silent rDNA chromatin is mediated by PARP1 via noncoding RNA. Mol Cell 45:790–800. https://doi.org/10.1016/j.molcel.2012.01.024
He F, James A, Raje H, Ghaffari H, DiMario P (2015) Deletion of Drosophila Nopp140 induces subcellular ribosomopathies. Chromosoma 124:191–208. https://doi.org/10.1007/s00412-014-0490-9
Holmberg Olausson K, Nister M, Lindstrom MS (2012) p53-dependent and -independent nucleolar stress responses. Cells 1:774–798. https://doi.org/10.3390/cells1040774
Ilyin AA, Ryazansky SS, Doronin SA, Olenkina OM, Mikhaleva EA, Yakushev EY, Abramov YA, Belyakin SN, Ivankin AV, Pindyurin AV, Gvozdev VA, Klenov MS, Shevelyov YY (2017) Piwi interacts with chromatin at nuclear pores and promiscuously binds nuclear transcripts in Drosophila ovarian somatic cells. Nucleic Acids Res 45:7666–7680. https://doi.org/10.1093/nar/gkx355
Iwasaki YW, Siomi MC, Siomi H (2015) PIWI-interacting RNA: its biogenesis and functions. Annu Rev Biochem 84:405–433. https://doi.org/10.1146/annurev-biochem-060614-034258
Izumi N, Kawaoka S, Yasuhara S, Suzuki Y, Sugano S, Katsuma S, Tomari Y (2013) Hsp90 facilitates accurate loading of precursor piRNAs into PIWI proteins. RNA 19:896–901. https://doi.org/10.1261/rna.037200.112
Jacob MD, Audas TE, Uniacke J, Trinkle-Mulcahy L, Lee S (2013) Environmental cues induce a long noncoding RNA-dependent remodeling of the nucleolus. Mol Biol Cell 24:2943–2953. https://doi.org/10.1091/mbc.E13-04-0223
Jamrich M, Miller OLJ (1984) The rare transcripts of interrupted rRNA genes in Drosophila melanogaster are processed or degraded during synthesis. EMBO J 3(7):1541–1545
Lam YW, Trinkle-Mulcahy L (2015) New insights into nucleolar structure and function. F1000Prime Rep 7:48. https://doi.org/10.12703/P7-48
Le Thomas A, Rogers AK, Webster A, Marinov GK, Liao SE, Perkins EM, Hur JK, Aravin AA, Toth KF (2013) Piwi induces piRNA-guided transcriptional silencing and establishment of a repressive chromatin state. Genes Dev 27:390–399. https://doi.org/10.1101/gad.209841.112
Lindstrom MS, Jurada D, Bursac S, Orsolic I, Bartek J, Volarevic S (2018) Nucleolus as an emerging hub in maintenance of genome stability and cancer pathogenesis. Oncogene 37:2351–2366. https://doi.org/10.1038/s41388-017-0121-z
Mayer C, Schmitz KM, Li J, Grummt I, Santoro R (2006) Intergenic transcripts regulate the epigenetic state of rRNA genes. Mol Cell 22:351–361. https://doi.org/10.1016/j.molcel.2006.03.028
Mekhail K, Khacho M, Carrigan A, Hache RR, Gunaratnam L, Lee S (2005) Regulation of ubiquitin ligase dynamics by the nucleolus. J Cell Biol 170:733–744. https://doi.org/10.1083/jcb.200506030
Mikhaleva EA, Yakushev EY, Stolyarenko AD, Klenov MS, Rozovsky YM, Gvozdev VA (2015) PIWI protein as a nucleolus visitor in Drosophila melanogaster. Mol Biol (Mosk) 49:161–167. https://doi.org/10.1134/S0026893315010100
Nemeth A, Grummt I (2018) Dynamic regulation of nucleolar architecture. Curr Opin Cell Biol 52:105–111. https://doi.org/10.1016/j.ceb.2018.02.013
Nollen EA, Salomons FA, Brunsting JF, van der Want JJ, Sibon OC, Kampinga HH (2001) Dynamic changes in the localization of thermally unfolded nuclear proteins associated with chaperone-dependent protection. Proc Natl Acad Sci U S A 98:12038–12043. https://doi.org/10.1073/pnas.201112398
Osouda S, Nakamura Y, de Saint Phalle B, McConnell M, Horigome T, Sugiyama S, Fisher PA, Furukawa K (2005) Null mutants of Drosophila B-type lamin Dm(0) show aberrant tissue differentiation rather than obvious nuclear shape distortion or specific defects during cell proliferation. Dev Biol 284:219–232. https://doi.org/10.1016/j.ydbio.2005.05.022
Panse SL, Masson C, Heliot L, Chassery JM, Junera HR, Hernandez-Verdun D (1999) 3-D organization of ribosomal transcription units after DRB inhibition of RNA polymerase II transcription. J Cell Sci 112(Pt 13):2145–2154
Pederson T (2011) The nucleolus. Cold Spring Harb Perspect Biol 3:a000638. https://doi.org/10.1101/cshperspect.a000638
Pederson T, Tsai RY (2009) In search of nonribosomal nucleolar protein function and regulation. J Cell Biol 184:771–776. https://doi.org/10.1083/jcb.200812014
Raje HS, Lieux ME, DiMario PJ (2018) R1 retrotransposons in the nucleolar organizers of Drosophila melanogaster are transcribed by RNA polymerase I upon heat shock. Transcription 9:1–13. https://doi.org/10.1080/21541264.2018.1506682
Rojas-Rios P, Chartier A, Pierson S, Simonelig M (2017) Aubergine and piRNAs promote germline stem cell self-renewal by repressing the proto-oncogene Cbl. EMBO J 36:3194–3211. https://doi.org/10.15252/embj.201797259
Rozhkov NV, Hammell M, Hannon GJ (2013) Multiple roles for Piwi in silencing Drosophila transposons. Genes Dev 27:400–412. https://doi.org/10.1101/gad.209767.112
Saito K, Inagaki S, Mituyama T, Kawamura Y, Ono Y, Sakota E, Kotani H, Asai K, Siomi H, Siomi MC (2009) A regulatory circuit for piwi by the large Maf gene traffic jam in Drosophila. Nature 461:1296–1299. https://doi.org/10.1038/nature08501
Saito K, Ishizu H, Komai M, Kotani H, Kawamura Y, Nishida KM, Siomi H, Siomi MC (2010) Roles for the Yb body components Armitage and Yb in primary piRNA biogenesis in Drosophila. Genes Dev 24:2493–2498. https://doi.org/10.1101/gad.1989510
Sarkar A, Volff JN, Vaury C (2017) piRNAs and their diverse roles: a transposable element-driven tactic for gene regulation? FASEB J 31:436–446. https://doi.org/10.1096/fj.201600637RR
Sienski G, Donertas D, Brennecke J (2012) Transcriptional silencing of transposons by Piwi and maelstrom and its impact on chromatin state and gene expression. Cell 151:964–980. https://doi.org/10.1016/j.cell.2012.10.040
Specchia V, Piacentini L, Tritto P, Fanti L, D'Alessandro R, Palumbo G, Pimpinelli S, Bozzetti MP (2010) Hsp90 prevents phenotypic variation by suppressing the mutagenic activity of transposons. Nature 463:662–665. https://doi.org/10.1038/nature08739
Wagner N, Weber D, Seitz S, Krohne G (2004) The lamin B receptor of Drosophila melanogaster. J Cell Sci 117:2015–2028. https://doi.org/10.1242/jcs.01052
Wang M, Tao X, Jacob MD, Bennett CA, Ho JJD, Gonzalgo ML, Audas TE, Lee S (2018) Stress-induced low complexity RNA activates physiological amyloidogenesis. Cell Rep 24:1713–1721.e4. https://doi.org/10.1016/j.celrep.2018.07.040
Weber SC, Brangwynne CP (2015) Inverse size scaling of the nucleolus by a concentration-dependent phase transition. Curr Biol 25:641–646. https://doi.org/10.1016/j.cub.2015.01.012
Wei H, Zhou B, Zhang F, Tu Y, Hu Y, Zhang B, Zhai Q (2013) Profiling and identification of small rDNA-derived RNAs and their potential biological functions. PLoS One 8:e56842. https://doi.org/10.1371/journal.pone.0056842
Welch WJ, Suhan JP (1985) Morphological study of the mammalian stress response: characterization of changes in cytoplasmic organelles, cytoskeleton, and nucleoli, and appearance of intranuclear actin filaments in rat fibroblasts after heat-shock treatment. J Cell Biol 101:1198–1211. https://doi.org/10.1083/jcb.101.4.1198
Xing YH, Yao RW, Zhang Y, Guo CJ, Jiang S, Xu G, Dong R, Yang L, Chen LL (2017) SLERT regulates DDX21 rings associated with Pol I transcription. Cell 169:664–678.e16. https://doi.org/10.1016/j.cell.2017.04.011
Ye J, Eickbush TH (2006) Chromatin structure and transcription of the R1- and R2-inserted rRNA genes of Drosophila melanogaster. Mol Cell Biol 26:8781–8790. https://doi.org/10.1128/MCB.01409-06
Zhang Q, Shalaby NA, Buszczak M (2014) Changes in rRNA transcription influence proliferation and cell fate within a stem cell lineage. Science 343:298–301. https://doi.org/10.1126/science.1246384
Zhao Z, Dammert MA, Hoppe S, Bierhoff H, Grummt I (2016) Heat shock represses rRNA synthesis by inactivation of TIF-IA and lncRNA-dependent changes in nucleosome positioning. Nucleic Acids Res 44:8144–8152. https://doi.org/10.1093/nar/gkw496
Zhao Z, Senturk N, Song C, Grummt I (2018) lncRNA PAPAS tethered to the rDNA enhancer recruits hypophosphorylated CHD4/NuRD to repress rRNA synthesis at elevated temperatures. Genes Dev 32:836–848. https://doi.org/10.1101/gad.311688.118
Zhu L, Brangwynne CP (2015) Nuclear bodies: the emerging biophysics of nucleoplasmic phases. Curr Opin Cell Biol 34:23–30. https://doi.org/10.1016/j.ceb.2015.04.003
Acknowledgements
We are grateful to Mikiko Siomi for providing OSC line, myc-Piwi vector and help in the implementation of this work; Artem Ilyin for assistance in performing Piwi knockdowns; Elena Fefelova for helpful advice on Piwi-GFP; Igor Kireev for assistance with EU incorporation assay; Konstantin Panov for providing 9-hydroxyellipticine inhibitor, and Michael Buszczak for Udd antibody. The work was carried out with the use of the equipment of the common use center “Center of Cell and Gene Technology”, Institute of Molecular Genetics, RAS.
Authors’ contribution statement
EM, VG, and MK conceived and designed the study; EM, TL, and HI performed research; EA, VG, and MK analyzed data; and VG and MK wrote the paper.
Funding
The work was supported by the Russian Foundation for Basic Research (grant no. 18-54-50015 YaPh_a) and by the Presidium of the Russian Academy of Sciences program Molecular and Cell Biology for VAG.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Jennifer Gerton and Lev Porokhovnik
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig S1
H3-uridine incorporation in OCSs treated with different transcription inhibitors. Treatment with 9НЕ inhibitor of Pol I-mediated transcription causes more than 60% reduction of H3-uridine incorporation demonstrating a known significant contribution of rRNA to total RNA synthesis. Simultaneous 9HE and Pol II inhibitor DRB treatment reduces H3-uridine incorporation to the background level comparable with that in cells treated with actinomycin D (ActD), which inhibits both Pol I and Pol II. (PDF 46 kb)
Rights and permissions
About this article
Cite this article
Mikhaleva, E.A., Leinsoo, T.A., Ishizu, H. et al. The nucleolar transcriptome regulates Piwi shuttling between the nucleolus and the nucleoplasm. Chromosome Res 27, 141–152 (2019). https://doi.org/10.1007/s10577-018-9595-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-018-9595-y