Abstract
The metastasis is a multistep process in which a small proportion of cancer cells are detached from the colony to enter into blood cells for obtaining a new place for metastasis and proliferation. The metastasis and cell plasticity are considered major causes of cancer-related deaths since they improve the malignancy of cancer cells and provide poor prognosis for patients. Furthermore, enhancement in the aggressiveness of cancer cells has been related to the development of drug resistance. Metastasis of pancreatic cancer (PC) cells has been considered one of the major causes of death in patients and their undesirable prognosis. PC is among the most malignant tumors of the gastrointestinal tract and in addition to lifestyle, smoking, and other factors, genomic changes play a key role in its progression. The stimulation of EMT in PC cells occurs as a result of changes in molecular interaction, and in addition to increasing metastasis, EMT participates in the development of chemoresistance. The epithelial, mesenchymal, and acinar cell plasticity can occur and determines the progression of PC. The major molecular pathways including STAT3, PTEN, PI3K/Akt, and Wnt participate in regulating the metastasis of PC cells. The communication in tumor microenvironment can provide by exosomes in determining PC metastasis. The components of tumor microenvironment including macrophages, neutrophils, and cancer-associated fibroblasts can modulate PC progression and the response of cancer cells to chemotherapy.
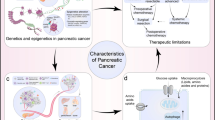
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
In Western countries, pancreatic cancer (PC) is suggested to be one of the leading causes (fourth in ranking) of death and by 2030, its mortality rate is suggested to enhance and become the second reason of death, just after lung cancer [1, 2]. In spite of advances in the field of medicine, PC is still among the major causes of death and it is considered as a lethal disease. The drawback of PC is a lack of symptoms in the early stages and major symptoms are revealed when the disease is at advanced stages [3]. There are epidemiological studies about diagnosed cases of PC that in 2018, 458,918 cases were diagnosed which caused 432,242 deaths among patients [4]. Although techniques for diagnosis and management of PC have reached a satisfactory level, the 5-year survival rate of PC is still 9% [4, 5]. The majority of PC cases is pancreatic ductal adenocarcinoma (PDAC) comprising 90% of cases. The risk factors for the development of PC include smoking, alcoholism, diabetes mellitus, obesity, aging, genetic and epigenetic alterations, family history, and others [4, 6]. The prognosis of PC patients is suggested to be low that is because of delays in diagnosis, rapid progression, and the ability of tumor cells in drug resistance development. The clinical symptoms lack in PC patients and therefore, there are challenges in the use of biomarkers including CA19-9 for the diagnosis of patients in terms of sensitivity and specificity [7, 8].
At the early stages of progression, surgical resection is used for the treatment of PC, and even after a successful operation, there is a high chance of recurrence [9]. Significant changes in genetic loci and remarkable mutations occur in PC to provide abnormal expression of tumor-suppressor and tumor-promoting factors, and dysregulation of important biological mechanisms including apoptosis, autophagy, drug resistance, malignancy, and metastasis [10]. The new therapeutics for PC are suggested to be focused on targeting KRAS and germline mutations since KRAS mutations are among the most common changes in the DNA of PC cells, and at the next rank, are germline changes [11, 12]. ATM, BRAF, BRCA1, BRCA2, CDKN2A, FGFR1, HER2, MET, MLH1, MSH2, MSH6, PALB2, PI3CA, PMS2, and PTEN are other genes that display abnormal changes in PC progression and they are responsible for drug resistance, malignant progression and other abnormal behaviour of PC cells [13]. More importantly, in 60–70% of PC cases, there are mutations that silence DKN2A, TP53, and SMAD4 [14, 15].
The significant advances in the field of biology have resulted in shedding more light on the understanding of genomic and epigenetic changes in the PC. SEMA3C has the ability to increase tumorigenesis in PC cells; Silencing SEMA3C diminishes cancer formation in vivo in the xenograft model. SEMA3C stimulates autophagy in increasing tumorigenesis, and silencing SEMA3C enhances sensitivity to KRAS or MEK1/2 [16]. The expression level of IPO7 enhances in PC; IPO7 escalates metastasis and prevents apoptosis. Silencing IPO7 impairs tumorigenesis and lung metastasis in vivo. IPO7 promotes ERBB2 expression in increasing tumorigenesis in PC [17]. The changes in the metabolism of PC cells can be provided by the dysregulation of molecular pathways; UBE2C has the ability to increase EGFR expression to induce PI3K/Akt axis in glycolysis stimulation [18]. COL10A1 binds to the DDR2 receptor on the surface of cells and after that, it stimulates MEK/ERK axis to mediate EMT in increasing liver metastasis of PC cells [19]. Furthermore, HOXA10 enhances NF-κB as a mechanism in enhancing colony formation and improving tumorigenesis in the xenograft model [20]. The epigenetic changes can also mediate carcinogenesis in PC; LINC01234 down-regulation can impair tumorigenesis in PC. LINC01234 sponges miR-513a-3p to elevate H6PD expression (Table 1) [21].
Owing to understanding the molecular dysregulation in increasing tumorigenesis in PC, the current review paper focuses on the role of molecular mechanisms in the metastasis of PC. The focus is on understanding the EMT mechanism and cell plasticity in PC cells. Moreover, the molecular profile of cancer metastasis is highlighted based on the most dysregulated mechanisms and pathways. The association between metastasis and drug resistance is provided. Finally, the bioinformatics analysis of the metastasis profile in PC is provided.
2 The concept of metastasis and related mechanisms
Metastasis is considered a factor in providing unfavourable prognosis and death of patients [36]. In metastasis, the cancer cells are detached from their primary site and disseminate into distal and surrounding organs. A few and small proportions of tumor cells have the ability to metastasis and after they metastasize into other tissues, they grow and proliferate [37, 38]. In the process of metastasis, plastic alterations occur in tumor cells and they may demonstrate features of epithelial-mesenchymal transition (EMT) [39, 40]. The metastasis of tumor cells is coordinated by genomic, transcriptome, and proteomic factors. The initial step for metastasis is to invade the surrounding tissues and cross over the endothelial barrier [41]. Metastasis is one of the features and hallmarks of tumor cells and the intrinsic and extrinsic factors in the tumor microenvironment (TME) participate in the invasion of cancer cells. After that, cancer cells have the capacity of entering blood vessels and they cross over the vascular endothelium called intravasation. The different factors and cell compartments participate in the metastasis and invasion [42,43,44]. Then, the cancer cells exit the blood vessels to accommodate in a new place, known as extravasation. At the primary site, the cancer cells proliferate to increase their population. The metastasis of cancer cells is coordinated by a number of molecular pathways in cancer. The tumor invasion can be inhibited by function of METTL3 acetylation [45]. The mitochondrial m5C can enhance metastasis and dissemination of tumor cells [46]. The cholesterol reprogramming and lymphangiogenesis can be accelerated by FASM to stimulate lymph node metastasis [47]. The transactivation of CDCA3 can be triggered by MYBL2 in escalating invasion of bladder tumor [48]. PRMT5 escalates arginine methylation of MTHFD1 to promote invasion and migration [49]. The inflammation increases metastasis of tumor cells and HECTD3 promotes IKKα activity to induce NF-κB axis [50]. The changes occurring in promoter of pathways can improve metastasis; EZH2 binds to promoter of KRT14 to escalate its expression in increasing peritoneal invasion [51]. Therefore, metastasis of tumor cells is coordinated by various molecular interactions (Fig. 1).
The process of metastasis in human cancer. The process of metastasis starts when a small proportion of cancer cells that have high migratory ability, are separated from other tumor cells in the colony to enter into the bloodstream known as intravasation. After circulation, it exists bloodstream, known as extravasation and after accommodation in new and secondary site, they proliferate and establish new colonies. The EMT induction enhances the invasion of tumor cells and upregulation of matrix metalloproteinase enzymes can result in the degradation of the basement membrane in increasing invasion of tumor cells
3 EMT in pancreatic cancer
3.1 Metastasis
EMT is a mechanism involved in cancer invasion that by changing the transformation of epithelial cells to mesenchymal cells in escalating metastasis and migration of tumor cells. The EMT involves changing the molecular levels along with phenotypic alterations. The N-cadherin and vimentin levels enhance, while E-cadherin levels reduce to increase the motility of cancer cells. The stimulation of EMT can enhance the invasion of tumor cells and it causes chemoresistance [52,53,54]. EMT has been considered as a potential mechanism in increasing metastasis of PC cells; the modulation of EMT in tumor cells can be provided by EMT-inducing transcription factors (EMT-TFs) that include Slug, Snail, ZEB proteins, and TGF-β. The decrease in glutamine level (depletion) can cause an increase in the invasion of PC cells that is because of Slug upregulation. In fact, loss of glutamine causes stimulation of ATF4 and ERK to enhance Slug expression in EMT induction and escalating invasion and metastasis of PC cells [55]. The upregulation of ZEB1 also participates in an increase in metastasis of PC cells through EMT induction. Musashi2 increases EGF expression to induce EMT and for this purpose, EGF promotes EGFR to increase ZEB1 expression, resulting in ERK/MAPK axis stimulation in escalating PC invasion through EMT induction [56]. The EGF-mediated EMT in PC cells is a potential subject and in addition to musashi2, calreticulin has the ability of EMT induction. The enhancement in the expression level of EGF is based on the function of calreticulin to promote integrin/EGFR and ERK/MAPK pathways in EMT induction [57]. Hence, ERK/MAPK axis is considered a factor in EMT induction and its suppression can prevent metastasis of PC. FAM172A suppresses ERK, vimentin, and MMP-9 levels, while it promotes E-cadherin and β-catenin levels to impair EMT-mediated invasion of PC cells [58]. However, only one mechanism has not been shown for the regulation of MAPK. GINS2 is able to induce ERK/MAPK axis in EMT stimulation and enhance PC invasion and metastasis [59]. Notably, the tumor cells choose a continuum of EMT states and the highest metastatic potential is observed when a late-hybrid EMT state occurs [60]. Another EMT-TF is SNAI1 which has the ability of EMT stimulation in accelerating PC invasion. The reason for focusing on EMT-TFs is that they are among the most common modulators of EMT induction and cancer metastasis, and if their related pathways are highlighted, the way for the development of new therapeutics is paved. MACC1 has been shown to positively interact with SNAIL in increasing fibronectin 1 (FN1) and trans-repression of CDH1 in improving PC invasion [61].
Owing to the critical function of EMT in increasing metastasis of PC cells, there has been an effort in EMT regulation. Emodin is an anti-cancer agent in reducing tumorigenesis in PC; emodin promotes miR-1271 expression as a way to down-regulate Twist1 in EMT inhibition and reduce the invasion of PC [62]. Betulinic acid administration is beneficial in reducing the metastasis of PC cells. Upon administration of betulinic acid, stimulation of AMPK occurs to mediate EMT and enhance the stemness of PC cells through upregulation of SOX2, Oct4, and Nanog [63]. When genomic changes occur in PC cells, the expression level of some of the factors alters. The upregulation of SMARCAD1 in PC results in the stimulation of Wnt/β-catenin to induce EMT in escalating PC invasion and metastasis [64]. Calpains2 has shown also potential in upregulation of the Wnt/β-catenin axis to trigger EMT in PC invasion and metastasis [65]. Hence, EMT is considered a mechanism involved in promoting PC invasion (Fig. 2).
A schematic representation of EMT. The mechanism of EMT is suggested to be vital for metastasis and drug resistance development in cancer cells. The tight junctions, adherens junctions, and desmosomes are responsible for keeping epithelial cells together so that during EMT, they are lost and mesenchymal cells with high motility are generated
3.2 Drug resistance association
The emergence of drug resistance in PC has been suggested to cause therapy failure in patients at the clinical level. With respect to the fact that enhancement in the aggressiveness potential of PC cells can escalate tumorigenesis and subsequent chemoresistance, there should be an association between metastasis and drug resistance in PC cells. The cisplatin resistance in PC cells can emanate from an increase in metastasis; Par-4 determines the response of PC cells to chemotherapy. Low expression levels of Par-4 in PC cells can develop cisplatin resistance through upregulation of PI3K/Akt to mediate EMT [66]. On the other hand, a reduction in the expression level of oncogenic factors can cause chemosensitivity. Low expression level of lncRNA NEAT1 in PC cells causes gemcitabine sensitivity. However, when the expression level of NEAT1 increases during tumorigenesis and progresses towards advanced stages, NEAT1 sponges miR-506-3p to escalate ZEB2 expression in EMT induction and enhancing metastasis, resulting in gemcitabine resistance [59]. Therefore, the function of EMT in the development of chemoresistance in PC is critical, and related molecular mechanisms should be highlighted. miR-3656 is capable of enhancing gemcitabine sensitivity in PC and for this purpose, it downregulates RHOF to suppress EMT-mediated gemcitabine resistance. However, the upregulation of TWIST1 can abrogate the function of miR-3656 in increasing drug sensitivity in PC cells [67]. As a result, therapeutics have been directed towards the regulation of EMT in increasing drug sensitivity. It has been reported that the application of oncolytic adenovirus co-expressing decorin and Wnt decoy receptor can participate in suppressing drug resistance in PC through ECM degradation and disruption of EMT [68]. Table 2 and Fig. 3 summarize the function of EMT in PC metastasis and chemoresistance.
4 Cell plasticity in pancreatic cancer
Although there are connections between EMT and cell plasticity in human cancers, it would be better to describe the cell plasticity in PC in a separate section to comprehensively discuss the related pathways regulating plasticity in cancer cells and its function in the modulation of invasion and aggressiveness. The KRAS mutation occurs in PC and this tumor-promoting factor is responsible for an increase in cell plasticity and tumor initiation through wild-type PI 3-kinase p110α [95]. The presence of stem cells can promote tumorigenesis in PC. The plasticity of PC stem cells can be escalated by the balance of MYC/PGC-1α balance. The down-regulation of MYC results in PGC-1α upregulation that participates in the oxidative metabolism of cancer stem cells that is abrogated in resistant populations [96]. The PC plasticity increases by CD44 upregulation. The PC cells with high levels of CD44 display cell plasticity, EMT induction, and low response to chemotherapy. The poor expression of CD44 enhances the chemosensitivity of PC cells [97]. In fact, the presence of epithelial plasticity has been shown to escalate PC invasion and metastasis [98]. However, the epithelial plasticity in PC is under the tight control of molecular interactions. The upregulation of TBK1 can occur by Axl to induce epithelial plasticity in PC [99]. After the identification of such pathways, their blockage can be performed. The application of warfarin can suppress Gas6-induced Axl upregulation to impair epithelial plasticity and invasion of PC cells [100]. Interestingly, Prrx1 has been related to an increase in mesenchymal plasticity of PC cells to enhance aggressiveness [101]. Therefore, both epithelial and mesenchymal plasticity are important factors in the determination of PC invasion and metastasis. In addition to epithelial and mesenchymal plasticity, acinar cell plasticity modulates the metastasis of PC cells. The acinar cell plasticity is facilitated by the function of IL-22 to stimulate PC invasion in mice, and suppression of STAT3 can abrogate this role [102]. Another aspect is the metabolic plasticity of PC cells that promotes G6PD expression in the development of erlotinib resistance in PC cells [103]. The PC plasticity can be accelerated upon loss of KDM6A that promotes activating A level to induce EMT and p-p38 in driving PDAC invasion [104]. Putting it all together, it appears that the process of cell plasticity in PC is highly controlled [105,106,107,108,109] and even changes in the tumor microenvironment can modulate cell plasticity [110]. Owing to the function of cell plasticity in determining PC invasion, studies have focused on targeting related pathways and mechanisms through the blockage to prevent the invasion of tumor cells [111]. Moreover, cell plasticity is a determining factor for the response of PC cells to immunotherapy [112].
5 Molecular profile of metastasis in pancreatic cancer
5.1 Non-coding RNAs
The microRNAs (miRNAs) are considered small and short non-coding RNAs with linear structures and a low number of nucleotides that are able to modulate important biological mechanisms in tumor cells, especially PC. The wealth of evidence has highlighted the function of miRNAs in the regulation of metastasis in PC. The invasion of PC cells can be significantly elevated by the function of miR-3613-5p which can be obtained through the reduction in CDK6 and also promotes lymph node metastasis [113]. Notably, miRNAs can exert two functions in PC cells that previous experiment highlighted the oncogenic function, while another study demonstrates that miR-1297 is capable of reducing MTDH expression to impair the invasion and migration of PC cells [114]. One of the most common and dysregulated factors in PC is KRAS in that its function is oncogenic and it displays high expression levels in PC cells and tissues. Interestingly, miR-216 has shown the ability in reducing KRAS expression to suppress metastasis of PC cells and to induce apoptosis [115]. The function of miRNAs in PC is versatile and in addition to metastasis, they can regulate proliferation, apoptosis, and metabolism. However, an interesting experiment has highlighted that when glycolysis-related enzymes are affected by miRNAs, it can affect the metastatic potential of PC cells. miR-489-3p reduces levels of PKM2 and LDHA as glycolytic enzymes to suppress glucose metabolism, growth, and metastasis of PC cells [116].
The long non-coding RNAs (lncRNAs) are an important member of non-coding RNAs lacking protein synthesis, similarity in structure to miRNAs (both of them are linear structures) and their length is more than 200 nts. The lncRNA location can be cytoplasm or nucleus, and they are potential regulators of molecular pathways, biological mechanisms, and tumorigenesis [117, 118]. The expression level of miRNAs in PC can be reduced by lncRNAs through sponging. LncRNA DANCR has the ability to sponge miR-33b to enhance MMP-16 expression in accelerating invasion and metastasis of PC cells [119]. The expression level of lncRNA AGAP2-AS1 can be increased by the function of RREB1 as an oncogenic factor to reduce levels of ANKRD1 and ANGPTL4 in promoting the growth and metastasis of PC cells [120]. The stability of mRNA can be increased by the function of lncRNA such as the role of lncRNA NEAT1 in enhancing stability of ELF3 at mRNA levels to promote PC invasion and metastasis [121]. The lung tissue is one of the targets of PC cells. When migration and invasion of PC cells enhance, they can metastasize into lung tissue. The lncRNA PLACT1 is able to recruit hnRNPA1 that binds to the IκBα promoter in reducing its expression, resulting in NF-κB upregulation in promoting PC metastasis [122].
The circular RNAs (circRNAs) are also a member of a large family of non-coding RNAs that can modulate tumorigenesis. The structure of circRNAs is different from other members of non-coding RNAs that they have circle structures and their stability is higher compared to linear structures. The circRNAs can function as modulators of metastasis in PC. Circ-001569 is considered a prognostic factor in PC and it can escalate lymph node metastasis and invasion of PC cells [123]. Similar to lncRNAs, the expression level of miRNAs can be regulated and sponged by circRNAs. The ability of circ-0000284 in the modulation of PC progression has been evaluated and it has been reported that circ-0000284 reduces miR-1179 expression through sponging to enhance RHPN2 expression in accelerating metastasis and angiogenesis in PC cells [124]. The EMT as an inducer of metastasis in PC cells can be modulated by circRNAs. The expression level of circ-STK39 escalates in PC cells and tissues, and it reduces miR-140-3p expression to enhance TRAM2 levels in EMT induction and facilitates tumorigenesis and invasion of PC cells [125]. Therefore, it can be concluded that metastasis of PC cells is regulated by a variety of molecular mechanisms that are considered downstream targets of non-coding RNAs (Table 3, Fig. 4).
5.2 Reactive oxygen species
The changes in levels of reactive oxygen species (ROS) have recently been emerged as a new aspect of regulating tumorigenesis that is because of the versatile function of such free radicals in modulating various biological mechanisms. The metastasis of PC cells is tightly regulated by levels of ROS. The function of Kras in PC is oncogenic and it promotes invasion and migration of tumor cells. Interestingly, Kras escalates levels of CCL15 to promote ROS and EMT levels. More importantly, ROS can stimulate EMT in enhancing the invasion of PC [156]. When the PC cells are exposed to the pressure of high glucose levels, it can cause enhancement in progression and mediate chemoresistance. High glucose levels are able to enhance ROS generation. NOX4 is required for increasing ROS generation to promote MMP-3 levels in increasing PC invasion and metastasis [157]. Moreover, the expression level of MMO-9 enhances to promote PC invasion. Palmitic acid has been shown to enhance invasion and metastasis of PC cells and to this end, it promotes TLR4 expression to induce ROS overproduction. Then, NF-κB axis induction occurs to enhance MMP-9 levels in escalating PC invasion and metastasis [158]. More importantly, when levels of ROS by antioxidants reduce, the ability of hypoxia in EMT induction and enhancing tumorigenesis in PC decreases [159].
5.3 Chromosomal instability
Although the role of chromosomal instability in the modulation of PC invasion has been ignored, it should be noted that the instability of chromosomes participates in the malignancy of tumor cells. Therefore, this aspect should be evaluated in terms of regulating PC invasion. The chromosomal instability can be accelerated by the function of APOBEC3A and it mediates co-deletions in DNA repair-related pathways to enhance the metastasis of PC cells [160].
5.4 STAT3
Another molecular mechanism participating in the regulation of PC metastasis is the STAT3 axis. The levels of STAT3 suppress by IL-37 in impairing metastasis of PC cells. However, the HIF-1α axis reduces IL-37 levels to induce the STAT3 axis in enhancing lymph node metastasis and invasion of visceral [161]. The expression level of STAT3 in PC is regulated by SOCS5 as an endogenous suppressor. Therefore, if the expression level of SOCS5 reduces, it causes upregulation of STAT3. miR-301a contributes to the downregulation of SOCS5 to induce JAK/STAT3 axis in escalating metastasis of PC cells [162]. Owing to the function of STAT3 in enhancing metastasis of PC cells, its down-regulation by RNAi can impair angiogenesis and progression [163]. Moreover, phosphorylation of STAT3 at Tyr705 can cause progression and metastasis of PC cells [164]. Hence, the small molecule inhibitors of STAT3 based on targeting these sites have been developed. It has been reported that WB436B is a small molecule that can suppress the phosphorylation of STAT3 at Tyr705 to impair the invasion of PC in vivo [165]. The silencing of STAT3 can disrupt the invasion of PC cells and reduces MMP-7 expression [166]. There is an association between STAT3 and cell plasticity. When the expression level of STAT3 enhances by NETO2 in PC cells, it causes EMT induction to promote invasion and metastasis [167]. An important inducer of STAT3 in addition to JAK2, is IL-6. The upregulation of STAT3 in PC can occur by IL-6 in promoting invasion of PC. HIC1 is capable of suppressing the IL-6/STAT3 axis to diminish levels of its downstream targets including c-Myc, VEGF, CyclinD1, MMP-2, and MMP-9 in suppressing metastasis of PC cells [168]. Therefore, STAT3 is a regulator of PC metastasis which is summarized in Table 4.
5.5 HIF-1α
Hypoxia is considered another factor in the progression of PC; the expression level of HIF-1α enhances as a response to hypoxia and by binding to promote lncRNA CF129, it diminishes CF129 expression to increase metastasis and invasion of PC cells through upregulation of FOXC2 [184]. The carcinogenesis in PC increases by the function of hypoxia. There is positive feedback and interaction between lncRNA ZEB1-AS1 and HIF-1α in which HIF-1α increases ZEB1-AS1 expression in promoting ZEB1 expression and escalating metastasis of PC cells [185]. Interestingly, the function of HIF-1α in the regulation of epigenetic factors can determine the invasion of PC cells. HIF-1α undergoes upregulation under hypoxia and it enhances miR-646 expression to down-regulate MIIP in enhancing tumorigenesis. MIIP is able to impair the deacetylase ability of histone deacetylase 6 (HDAC6) to induce acetylation and degradation of HIF-1α [186]. HIF-1α enhances miR-212 expression as a mechanism to develop lymph node metastasis and invasion of PC cells [187]. The progression of PC cells can occur as a response to the autocrine loop. HIF-1α promotes VEGF expression and then, VEGF mediates the interaction of NRP-2 and integrin-aβ to stimulate FAK/ERK axis in enhancing HIF-1α expression. Therefore, such a positive feedback loop enhances metastasis and progression of PC cells [188]. The involvement of HIF-1α in the regulation of PC invasion has been mentioned in both in vitro and in vivo, and when the stability of HIF-1α reduces, the progression of PC decreases [189]. The increase in acetylation of HIF-1α reduces its expression. However, when deacetylation of HIF-1α is mediated by lncRNA MTA2R, it causes accumulation of HIF-1α in enhancing carcinogenesis [190]. Notably, an increase in Snail expression can be stimulated by HIF-1α in enhancing tumorigenesis and invasion of PC cells [191]. Hence, the upregulation of HIF-1α can be considered a factor in enhancing the metastasis of PC cells.
5.6 PTEN/PI3K/Akt
Dysregulation of PTEN/PI3K/Akt axis has been well-documented in PC (Table 5) and it should be noted that PTEN has anti-cancer activity, while PI3K/Akt has oncogenic function. The expression level of PTEN reduces in PC patients and it causes lymph node metastasis of tumor cells. PTEN promotes FOXO3a expression to upregulate PLZF, resulting in VEGF down-regulation in preventing the progression and invasion of PC cells [192]. However, the expression level of PTEN reduces by miR-32-5p in increasing metastasis. LncRNA GAS5 promotes PTEN expression through miR-32-5p sponging in reducing invasion and metastasis of PC cells [193]. There are regions on PTEN that miRNAs can bind to them. miR-92a-3p diminishes PTEN expression to enhance the growth and invasion of PC cells [194]. GFRα2 has also the potential in promoting miR-17-5p expression to down-regulate PTEN in promoting the progression and perineural metastasis of PC cells [195]. On the other hand, the function of PI3K/Akt in PC is tumorigenic; interestingly, the invasion of PC stem cells can be increased by PI3K/Akt. BMI-1 has the ability of stimulation of PI3K/Akt to enhance metastasis [196]. The overexpression of GLP-1R has been suggested to prevent PC invasion and that is because of PI3K/Akt suppression by GLP-1R [197]. Although irradiation has been considered a major tool in PC therapy, this therapeutic tool can accelerate the invasion and metastasis of tumor cells. Upon exposure to carbon ion irradiation, nitric oxide obtains the ability to stimulate the PI3K/Akt axis to enhance the metastasis of PC cells [198].
5.7 Wnt/β-catenin
The abnormal expression of Wnt has been well-documented in human cancers, particularly PC. This aberrant expression has been shown to modulate hallmarks of PC that metastasis is among them. The acceleration in PC invasion can be performed by Wnt. The expression level of Wnt enhances by function WTAPP1 function to induce metastasis [207]. Both angiogenesis and metastasis increase by the function of Wnt in increasing carcinogenesis in PC [208]. The reduction in the expression level of Wnt disrupts the invasion and metastasis of PC cells. It has been reported that linc00261 sponges miR-552-5p to increase FOXO3 expression, leading to Wnt down-regulation to disrupt invasion [209]. Interestingly, upregulation in Wnt can cause EMT in increasing cancer metastasis, and CDH13 suppresses Wnt/β-catenin to prevent EMT-mediated invasion of PC cells [210]. The stroma of PC is dense and comprised of CAFs, immune cells, endothelial cells, and ECM [211, 212]. The extracellular collagen and matrix can be deposited by the function of CAFs and cause fibrous desmoplasia in enhancing tumorigenesis [213, 214]. Such desmoplastic reactions cause chemoresistance, immune evasion, carcinogenesis, and metastasis [215, 216]. The pancreatic stellate cells (PSCs) are precursors of CAFs in PC and they can be stimulated by PC cells to be activated from a quiescent phenotype into an active myofibroblast phenotype to generate carcinogenic ligands and proteins of ECM [217,218,219]. The growth factors and cytokines can be secreted by the surrounding stroma of PC to enhance tumorigenesis in PC. The PSCs are able to secrete Wnt and tenascin C ligands in the stimulation of β-catenin and YAP/TAZ pathways in enhancing progression and metastasis [220]. Owing to the function of Wnt in increasing tumorigenesis in human cancers, studies have focused on the modulation of Wnt in suppressing metastasis. When oridonin is utilized in the treatment of PC, it can inhibit Wnt/β-catenin in impairing EMT and reducing invasion and metastasis of tumor cells [221]. The expression level of Wnt increases by the function of FOXM1 in enhancing the invasion of PC cells, while Merlin suppresses FOXM1/Wnt axis in reducing metastasis [222]. The upregulation of GPX2 in PC cells can cause stimulation of the Wnt axis in the induction of EMT and enhance invasion and migration [223]. Therefore, Wnt is a regulator of PC metastasis, summarized in Table 6. Fig. 5 summarizes the role of molecular pathways in the regulation of PC metastasis.
6 Exosomes in pancreatic cancer metastasis
Exosomes are considered biocompatible nanostructures with a size of less than 100 nm that has been surrounded by bilayer membranes and can be loaded with bioactive components including proteins, lipids, nucleic acids, and RNA molecules [238]. The function of exosomes in recent years in tumorigenesis has been of importance in that their role in reducing/enhancing carcinogenesis can be related to the cargo that they carry [239]. The metastasis of PC cells is highly regulated by exosomes. The enrichment of circ-IARS can be found in exosomes and through modulation of endothelial monolayer permeability, they accelerate the invasion of PC cells [240]. The exosomes can be secreted by almost all the eukaryotic cells. The PC cells are able to secret exosomes in favour of promoting their invasion and generation of the liver pre-metastatic niche [241]. The exosomes derived from PC cells can affect molecular interactions in tumorigenesis. These exosomes escalate the expression level of TFAP2A, Bcl-2, and p-AKT/AKT, and down-regulate PTEN and BAX levels to prevent apoptosis in increasing tumorigenesis in PC and they also enhance invasion of tumor cells [27]. The application of exosomes as diagnostic factors is extremely increasing due to the capacity of their isolation from the serum of patients. The exosomes can be isolated from the serum of patients and such exosomes are considered as factors involved in EMT induction and escalating cancer metastasis [242]. Interestingly, prometastatic secretome trafficking can be facilitated by exosomes in enhancing the pulmonary invasion of PC cells [243]. Important actions can be performed by exosomes in the regulation of cancer invasion. The degradation and remodelling of ECM can be induced by exosomes at the primary site and they invade through the basement membrane. They can enter and exist in the vascular system, known as intravasation and extravasation, respectively [243]. The PC cells are not the only source for the secretion and isolation of exosomes in the regulation of tumor metastasis. There are exosomes in the ascites of PC patients that can mediate EndMT in increasing vascular permeability and promoting remote metastasis [244]. Even lymphangiogenesis is accelerated by a critical function of exosomes. The exosomes derived from PC cells can participate in the downregulation of ABHD11-AS1 to increase lymphangiogenesis [245]. Another source for exosomes is mesenchymal stem cells (MSCs) that delivery of miR-124 by such exosomes can impair metastasis and EMT in PC cells, while upregulation of EZH2 abrogates the function of such exosomes [246]. Hence, exosomes are critical regulators of PC metastasis summarized in Table 7.
7 Tumor microenvironment components and pancreatic cancer invasion
7.1 Macrophages and metastasis
The tumor microenvironment (TME) components participate in the regulation of tumorigenesis and macrophages as immune cells are among such components [260, 261]. The tumor-associated macrophages (TAMs) are capable of secretion of cytokines and inflammatory factors to provide optimal conditions for carcinogenesis [262, 263]. The macrophages can be found in two polarization types including M1 and M2 macrophages [264]. The identification of M1 macrophages is based on the expression of iNOS and can be developed in response to LPS or IFN-γ [265]. The M2 macrophages are anti-inflammatory factors in contrast to M1 polarized macrophages that are developed in response to IL-4 or IL-13, and they can escalate angiogenesis and matrix remodelling [266, 267]. The M1 polarized macrophages exert anti-cancer activity, while M2 macrophages enhance carcinogenesis [268]. The macrophages are able to regulate the metastasis potential of PC cells. Interestingly, The extracellular vesical Ezrin can be derived from PC cells and they stimulate M2 polarization of macrophages in enhancing metastasis of tumor cells [269]. The ability of macrophages is higher than their polarization status and based on their secretion, they can affect the progression and metastasis of PC cells. It has been reported that granulin can be secreted by macrophages and through triggering liver fibrosis, it promotes invasion and migration of PC cells [270]. Furthermore, M2-polarized macrophages are capable of secreting CCL20 to stimulate EMT in escalating invasion and metastasis of PC cells [271]. Interestingly, when PI3Kα is genetically suppressed, the number of CD206-positive macrophages reduces which impairs the invasion of PC cells [272], confirming the role of macrophages as potential regulators of metastasis of PC. In addition, pharmacological suppression of macrophages can disrupt the metastasis of PC cells [273]. Interestingly, the chemotherapy response of PC cells can be improved by the polarization of macrophages. UTMD is able to enhance the polarization of macrophages from M2 to M1, and they normalize vasculature in impairing invasion and metastasis of PC cells [274]. Therefore, the communications in the TME can modulate the polarization of macrophages in affecting PC invasion. The ANXA1 can be enriched in extracellular vesicles to stimulate M2 polarization of macrophages and then, such macrophages stimulate endothelial cells and fibroblasts in mediating angiogenesis and matrix degradation [275]. Therefore, macrophages are considered as regulators of metastasis of PC cells (Table 8).
7.2 Neutrophils and metastasis
In addition to macrophages, neutrophils play a significant role in the regulation of metastasis and progression of PC cells. The stimulation of NF-κB can occur by upregulation of SPRY1 to mediate CXCL12/CXCR4 axis in neutrophil and macrophage recruitment to promote tumorigenesis in PC [286]. Moreover, the neutrophil reprogramming upon loss of SETD2 causes immune escape in PC [287]. Therefore, tumor-associated neutrophils (TANs) are involved in the modulation of carcinogenesis. The neutrophils are able to suppress microbial attacks and prevent their spread through the production of reactive oxidants, stimulation of granular constituents, and neutrophil extracellular traps (NETs) [288]. There is a wealth of evidence that the presence of NETs can provide the function of TANs in increasing tumorigenesis [289]. Interestingly, the generation of NETs can be accelerated by the function of DDR1 in enhancing the invasion and migration of PC cells [290]. Therefore, if NETs are therapeutically targeted, the invasion potential of PC cells reduces. The HMGB1 derived from NETs is able to increase tumorigenesis. The extravasation can be increased by NETs in promoting liver metastasis. Notably, thrombomodulin is able to mediate degradation of HMGB1 to suppress NETs in impairing metastasis of PC cells [291]. More importantly, the DNA derived from NETs can stimulate pancreatic stellate cells in enhancing progression of PC cells [292]. Sometimes, the potential of NETs in increasing tumorigenesis and metastasis of PC is related to their impact on the other members of tumor microenvironment such as cancer-associated fibroblasts (CAFs). The CAF stimulation by NETs can increase metastasis of PC cells to liver tissue [293]. Moreover, when PC cells are exposed to chemotherapy, the infiltration of neutrophils enhances that by stimulation of Gas6/AXL axis, it causes increase in metastasis of PC cells [294]. The ability of TANs in secretion of cytokines including TNF-α and TGF-β1 can escalate the liver metastasis of PC cells [295].
7.3 Cancer-associated fibroblasts and metastasis
Another member of TME is cancer-associated fibroblasts (CAFs) that their function in increasing the invasion of PC cells has been well-documented. The stimulation of CAFs can occur by ACLP to mediate fibrosis in TME. ACLP enhances levels of MMP-1 and MMP-3 in CAFs to enhance PC invasion [296]. Moreover, by upregulation of PERK, CAFs have the ability to obtain endothelial cell-like phenotype to stimulate angiogenesis in promoting tumorigenesis in PC [297]. The upregulation of FAK in CAFs can improve the metastatic potential of PC cells. The suppression of FAK impairs metastasis and invasion of CAFs and diminishes the amount and level of ECM by CAFs in reducing PC invasion [298]. In fact, the crosstalk between CAFs and tumor cells in TME can determine the aggressiveness and metastasis potential of PC cells [299]. The changes in the shape of CAFs in TME can affect the metastasis of PC cells. The TGF-β exposure promotes stiffness, elongation, spread, lamellipodia generation, and spheroid invasion of CAFs that are induced by the function of Rac, RhoA, and ROCK upon exposure to TGF-β [300]. Furthermore, an increase in the generation of invadopodia in CAFs by paladin can increase the metastasis of PC cells [301]. The metabolic changes including upregulation of LDH and PKM2 mediated by CAFs can escalate the metastatic potential of PC cells [302]. Hence, if CAFs are therapeutically targeted, the metastatic potential of PC cells reduces. Curcumin is able to suppress CAFs in EMT inhibition and reduce the metastasis of PC cells [303]. Exposure to chemotherapy can also induce the activation of CAFs. When exposure to nab-paclitaxel occurs, it causes overexpression of CCL26 in CAFs to stimulate the PI3K/Akt/mTOR axis in enhancing metastasis of PC cells [304]. Therefore, the shape of CAFs, their involvement in the regulation of metabolism, EMT modulation, and secretion of different factors can participate in the regulation of metastasis in PC (Fig. 6).
8 Therapeutic modulation of metastasis in pancreatic cancer
The therapeutic compounds have been extensively utilized for disrupting the invasion and metastasis of PC cells. Owing to the function of metastasis in increasing aggressiveness and mediating chemoresistance in PC, therapeutic modulation of metastasis can participate in tumor therapy. The modulation of citrate metabolism in PC cells by berberine can suppress the biosynthesis of fatty acids impairing invasion and metastasis [305]. One of the targets in which PC cells can metastasize is lung tissue. The potential of berberine in suppressing PC invasion can be related to the downregulation of TGFBR1 in vitro and in vivo [306]. A variety of pathways and mechanisms participate in an increase in metastasis and progression of PC cells. Upon down-regulation of HDAC, (-)-epigallocatechin 3-gallate (EGCG) stimulates RKIP to enhance histone H3 expression. Then, down-regulation of Snail, NF-κB, MMP-2, and MMP-9 occurs to reduce the invasion of PC cells [307]. The dysregulation of NF-κB can participate in EMT induction and enhancement in the progression of PC cells. However, curcumin as anti-cancer drugs, suppresses the NF-κB axis through down-regulation of IL-6/ERK to suppress EMT and metastasis of PC cells [78]. The potential of curcumin in reducing invasion can be accelerated by loading in nanostructures. The chitosan/PEG-blended PLGA nanostructures can be loaded with curcumin in suppressing metastasis and inducing apoptosis in PC cells [308].
The synergistic regulation of PC metastasis can be provided by a combination of pharmacological compounds. A mixture of quercetin and resveratrol can be utilized to suppress EMT through N-cadherin down-regulation [309]. The modulation of EMT in PC can be performed by regulation of NF-κB. Resveratrol is able to reduce levels of PI3K/Akt in suppression of NF-κB to inhibit EMT in reducing PC invasion and metastasis [310]. Quercetin is also a pharmacological and therapeutic compound in the regulation of EMT in PC therapy. The administration of quercetin can suppress EMT in PC cells through the downregulation of STAT3 to impair metastasis [311]. The down-regulation of SHH and TGF-β/Smad axis can be occurred by quercetin in suppressing metastasis of PC cells [312]. Sulforaphane as another anti-cancer compound has the ability to reduce vimentin, Twist-1, and ZEB1 levels to impair EMT-mediated metastasis of PC cells [313]. The growth factors such as bFGF participate in increasing the invasion of PC cells, while quercetin-3-O-glucoside is able to impair the metastasis of PC cells mediated by growth factors [314]. When there is a loss of SMAD4, the metastasis of PC cells enhances due to overexpression of HNF4G in that this axis is suppressed by the function of metformin [315]. Upon down-regulation of ADAM8 by propofol, reductions in levels of MMP-2, MMP-9, ERK1/2, and integrin β1 occur to impair metastasis and progression of PC cells [316]. Therefore, EMT, MMPs, and other factors involved in the metastasis of PC cells are modulated by therapeutic compounds in reducing tumorigenesis and aggressiveness.
9 Clinical insight and implications
The metastasis of PC and the related therapeutics have been exploited in clinical for the treatment of patients. A phase I/II study has shown the potential of Parenteral Ascorbate at high concentrations for the reduction in proliferation and metastasis of PC, while its toxicity is partial [317]. Although gemcitabine is a good choice in the treatment of patients, the application of paclitaxel through the intraperitoneal route has been shown in phase I/II study to be beneficial in peritoneal metastasis of PC with good tolerability of the drug [318]. However, the problem related to the clinical studies is the low number of participants [319], and therefore, for having a good insight towards the clinical application of drugs in minimizing metastasis, it is suggested to enhance the number of patients. Regardless of treatment, the whole-body FDG-PET is suggested to be beneficial in the detection of metastasis in PC patients [320]. Furthermore, when pharmacoangiographic computed tomography is performed, angiotensin-II can be utilized for the identification of liver metastasis in PC [321]. At the clinical level, when surgical resection of PC is performed, there is a chance of liver metastasis, and therefore, intra-arterial adjuvant chemotherapy is suggested for its prevention [322]. The vaccination has been developed as a new way for the treatment of metastasis in PC and vaccination with HLA-A2 restricted peptide derived from the universal tumor antigen survival could result in a decrease in metastasis of PC cells to the liver [323].
10 Conclusion and remarks
The ability of PC cells in metastasis has been a problem at both pre-clinical and clinical levels. The gene analysis displays that dysregulation of molecular pathways and mechanisms significantly participates in the progression of PC cells. The invasion of PC cells does not rely on a single pathway or mechanism, and it has been reported that a variety and complicated molecular mechanisms participate in this mechanism. The aim of the current review was not to only focus on the mechanism of PC cell invasion but also to provide a concept that how metastasis of PC cells enhances and what is the association between this metastasis and chemoresistance development. In the first concept, cell plasticity (mesenchymal and epithelial plasticity) and EMT determine the metastasis of PC cells. The enhancement of PC invasion is mediated by EMT induction and based on the pieces of evidence, EMT-mediated enhancement in aggression of PC cells can stimulate drug resistance. The non-coding RNA transcripts, STAT3, Wnt/β-catenin, and PTEN/PI3K/Akt, are among the most common mechanisms participating in PC progression and metastasis. Therefore, novel therapeutic mechanisms can participate in targeting such pathways for preventing the invasion of PC cells. However, the metastasis of PC cells is more than single participation of pathways and the factors involved in cell-cell communication such as exosomes and the components of TME including macrophages, neutrophils, and CAFs can determine the metastasis of PC cells. The therapeutic modulation of PC metastasis with natural products, small molecules, and nanostructures has been performed to impair tumorigenesis. However, it appears that current studies lack providing a complicated and rational connection between metastasis and drug resistance in PC. In fact, it is obvious that EMT and an increase in metastasis can mediate chemoresistance. However, most of the studies have focused on only understanding the metastasis-related pathways without focusing on therapy response. Therefore, complicated and comprehensive studies in the future are required to reveal and highlight the function of molecular pathways in the regulation of metastasis-related chemoresistance in PC.
There are several perspectives for future papers. For instance, in the case of EMT and metastasis of PC cells, the main emphasis has been on its regulation by upstream mediators, and among all the factors, non-coding RNA transcripts have been critically investigated. However, researchers should focus on the discovery of new proteins related to EMT that have not been investigated before. In the case of EMT, it has been clearly demonstrated that EMT has a close relationship with chemoresistance, but in other cases, the association with drug resistance has not been completely shown and therefore, studies should perform better and more detailed experiments in understanding the role of metastasis in development of chemoresistance in PC. For instance, the TME has various cells and components, critically regulates tumorigenesis and metastasis of PC cells and although the studies have shown the contribution of TME components in enhancement in progression and invasion, more focus should be directed on TME component-mediated metastasis and drug resistance in PC cells.
Data Availability
This is a review article and no data has been used.
References
Zeng, S., et al. (2019). Chemoresistance in Pancreatic Cancer. International Journal of Molecular Sciences, 20(18), 4504.
Rahib, L., et al. (2014). Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Research, 74(11), 2913–2921.
Kamisawa, T., et al. (2016). Pancreatic cancer. Lancet, 388(10039), 73–85.
Rawla, P., Sunkara, T., & Gaduputi, V. (2019). Epidemiology of pancreatic cancer: Global trends, etiology and risk factors. World journal of oncology, 10(1), 10–27.
Siegel, R. L., Miller, K. D., & Jemal, A. (2018). Cancer statistics, 2018. CA: a Cancer Journal for Clinicians, 68(1), 7–30.
Kleeff, J., et al. (2016). Pancreatic cancer. Nature reviews Disease primers, 2, 16022.
Kaur, S., et al. (2017). A combination of MUC5AC and CA19-9 improves the diagnosis of pancreatic cancer: A multicenter study. The American Journal of Gastroenterology, 112(1), 172–183.
Kaur, S., et al. (2012). Early diagnosis of pancreatic cancer: challenges and new developments. Biomarkers in Medicine, 6(5), 597–612.
Papaefthymiou, A., Doukatas, A., & Galanopoulos, M. (2022). Pancreatic cancer and oligonucleotide therapy: Exploring novel therapeutic options and targeting chemoresistance. Clinics and Research in Hepatology and Gastroenterology, 46(5), 101911.
Kamimura, K., Yokoo, T., & Terai, S. (2018). Gene therapy for pancreatic diseases: current status. International journal of molecular sciences, 19(11), 3415.
Hu, C., et al. (2018). Association between inherited germline mutations in cancer predisposition genes and risk of pancreatic cancer. Jama, 319(23), 2401–2409.
Di Magliano, M. P., & Logsdon, C. D. J. G. (2013). Roles for KRAS in pancreatic tumor development and progression. Gastroenterology, 144(6), 1220–1229.
Gleeson, F. C., et al. (2016). Targeted next generation sequencing of endoscopic ultrasound acquired cytology from ampullary and pancreatic adenocarcinoma has the potential to aid patient stratification for optimal therapy selection. Oncotarget, 7(34), 54526.
Wang, S., et al. (2020). Tumor microenvironment in chemoresistance, metastasis and immunotherapy of pancreatic cancer. American Journal of Cancer Research, 10(7), 1937–1953.
Jones, S., et al. (2008). Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science, 321(5897), 1801–1806.
Zhang, D., et al. (2022). SEMA3C supports pancreatic cancer progression by regulating the autophagy process and tumor immune microenvironment. Frontiers in Oncology, 12, 890154.
Li, M., et al. (2022). IPO7 promotes pancreatic cancer progression via regulating ERBB pathway. Clinics (São Paulo, Brazil), 77, 100044.
Cao, J. Z., et al. (2022). UBE2C promotes the progression of pancreatic cancer and glycolytic activity via EGFR stabilization-mediated PI3K-Akt pathway activation. Journal of gastrointestinal oncology, 13(3), 1444–1453.
Wen, Z., et al. (2022). COL10A1-DDR2 axis promotes the progression of pancreatic cancer by regulating MEK/ERK signal transduction. Frontiers in Oncology, 12, 1049345.
Li, J., et al. (2022). HOXA10 promote pancreatic cancer progression via directly activating canonical NF-κB signaling pathway. Carcinogenesis, 43(8), 787–796.
Song, Y., Gao, Z., & Zheng, C. (2022). Silencing LINC01234 represses pancreatic cancer progression by inhibiting the malignant phenotypes of pancreatic cancer cells. Immunobiology, 227(6), 152295.
Shi, J., et al. (2022). JNK inhibitor IX restrains pancreatic cancer through p53 and p21. Frontiers in Oncology, 12, 1006131.
Nimmakayala, R. K., et al. (2022). PAF1 cooperates with YAP1 in metaplastic ducts to promote pancreatic cancer. Cell Death & Disease, 13(10), 839.
Park, S. J., et al. (2023). Enhanced glutaminolysis drives hypoxia-induced chemoresistance in pancreatic cancer. Cancer Research, 83(5), 735–752.
Liu, H., et al. (2023). Pancreatic stellate cells exploit Wnt/β-catenin/TCF7-mediated glutamine metabolism to promote pancreatic cancer cells growth. Cancer Letters, 555, 216040.
Huang, C. K., et al. (2023). ENO1 promotes immunosuppression and tumor growth in pancreatic cancer. Clinical & Translational Oncology, 25(7), 2250–2264.
Wu, J. (2022). Pancreatic cancer-derived exosomes promote the proliferation, invasion, and metastasis of pancreatic cancer by the miR-3960/TFAP2A Axis. Journal of Oncology, 2022, 3590326.
Zhang, W., et al. (2022). Thiostrepton induces ferroptosis in pancreatic cancer cells through STAT3/GPX4 signalling. Cell Death & Disease, 13(7), 630.
Jacoberger-Foissac, C., et al. (2023). CD73 Inhibits cGAS-STING and cooperates with CD39 to promote pancreatic cancer. Cancer Immunology Research, 11(1), 56–71.
Simmler, P., et al. (2022). SF3B1 facilitates HIF1-signaling and promotes malignancy in pancreatic cancer. Cell Reports, 40(8), 111266.
Lee, M. S., et al. (2023). Ornithine aminotransferase supports polyamine synthesis in pancreatic cancer. Nature, 616(7956), 339–347.
Lefler, J. E., et al. (2022). STAT3 in tumor fibroblasts promotes an immunosuppressive microenvironment in pancreatic cancer. Life Science Alliance, 5(11), 1.
Su, H., et al. (2022). Collagenolysis-dependent DDR1 signalling dictates pancreatic cancer outcome. Nature, 610(7931), 366–372.
Rademaker, G., et al. (2022). Myoferlin targeting triggers mitophagy and primes ferroptosis in pancreatic cancer cells. Redox Biology, 53, 102324.
Lan, L., et al. (2022). GREM1 is required to maintain cellular heterogeneity in pancreatic cancer. Nature, 607(7917), 163–168.
Wang, S., et al. (2022). RNA-binding proteins and cancer metastasis. In Seminars in Cancer Biology. Elsevier.
Fidler, I. J. (2003). The pathogenesis of cancer metastasis: The'seed and soil'hypothesis revisited. Nature reviews cancer, 3(6), 453–458.
Chambers, A. F., Groom, A. C., & MacDonald, I. C. J. N. R. C. (2002). Dissemination and growth of cancer cells in metastatic sites. Nature Reviews Cancer, 2(8), 563–572.
Polyak, K., & Weinberg, R. A. J. N. R. C. (2009). Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nature Reviews Cancer, 9(4), 265–273.
Bakir, B., et al. (2020). EMT, MET, plasticity, and tumor metastasis. Trends in cell biology, 30(10), 764–776.
Yang, Y., & Cao, Y. (2022). The impact of VEGF on cancer metastasis and systemic disease. In Seminars in Cancer Biology. Elsevier.
Friedl, P., & Alexander, S. J. C. (2011). Cancer invasion and the microenvironment: Plasticity and reciprocity. Cell, 147(5), 992–1009.
Sun, X., et al. (2022). Inflammatory cell-derived CXCL3 promotes pancreatic cancer metastasis through a novel myofibroblast-hijacked cancer escape mechanism. Gut, 71(1), 129–147.
Yang, Y., et al. (2016). The PDGF-BB-SOX7 axis-modulated IL-33 in pericytes and stromal cells promotes metastasis through tumour-associated macrophages. Nature communications, 7(1), 11385.
Li, Y., et al. (2022). METTL3 acetylation impedes cancer metastasis via fine-tuning its nuclear and cytosolic functions. Nature Communications, 13(1), 6350.
Delaunay, S., et al. (2022). Mitochondrial RNA modifications shape metabolic plasticity in metastasis. Nature, 607(7919), 593–603.
Du, Q., et al. (2022). FASN promotes lymph node metastasis in cervical cancer via cholesterol reprogramming and lymphangiogenesis. Cell Death & Disease, 13(5), 488.
Liu, W., et al. (2022). MYBL2 promotes proliferation and metastasis of bladder cancer through transactivation of CDCA3. Oncogene, 41(41), 4606–4617.
Meng, Q., et al. (2022). Arginine methylation of MTHFD1 by PRMT5 enhances anoikis resistance and cancer metastasis. Oncogene, 41(32), 3912–3924.
Li, F., et al. (2022). Targeting HECTD3-IKKα axis inhibits inflammation-related metastasis. Signal Transduction and Targeted Therapy, 7(1), 264.
Verma, A., et al. (2022). EZH2-H3K27me3 mediated KRT14 upregulation promotes TNBC peritoneal metastasis. Nature Communications, 13(1), 7344.
Mirzaei, S., et al. (2023). Exosome-mediated miR-200a delivery into TGF-β-treated AGS cells abolished epithelial-mesenchymal transition with normalization of ZEB1, vimentin and Snail1 expression. Environmental Research, 231(Part 1), 116115.
Ashrafizadeh, M., et al. (2020). Association of the epithelial–mesenchymal transition (EMT) with cisplatin resistance. International journal of molecular sciences, 21(11), 4002.
Ashrafizadeh, M., et al. (2021). New insight towards development of paclitaxel and docetaxel resistance in cancer cells: EMT as a novel molecular mechanism and therapeutic possibilities. Biomedicine & Pharmacotherapy, 141, 111824.
Recouvreux, M. V., et al. (2020). Glutamine depletion regulates Slug to promote EMT and metastasis in pancreatic cancer. The Journal of Experimental Medicine, 217(9), 1.
Sheng, W., et al. (2020). Musashi2 promotes EGF-induced EMT in pancreatic cancer via ZEB1-ERK/MAPK signaling. Journal of Experimental & Clinical Cancer Research, 39(1), 16.
Sheng, W., et al. (2017). Calreticulin promotes EGF-induced EMT in pancreatic cancer cells via Integrin/EGFR-ERK/MAPK signaling pathway. Cell Death & Disease, 8(10), e3147.
Chen, Y., et al. (2020). FAM172A inhibits EMT in pancreatic cancer via ERK-MAPK signaling. Biology open, 9(2), bio048462.
Fu, X., et al. (2021). Downregulation of NEAT1 sensitizes gemcitabine-resistant pancreatic cancer cells to gemcitabine through modulation of the miR-506-3p/ZEB2/EMT axis. American Journal of Cancer Research, 11(8), 3841–3856.
Simeonov, K. P., et al. (2021). Single-cell lineage tracing of metastatic cancer reveals selection of hybrid EMT states. Cancer Cell, 39(8), 1150–1162.e9.
Zhang, X., et al. (2022). MACC1 promotes pancreatic cancer metastasis by interacting with the EMT regulator SNAI1. Cell Death & Disease, 13(11), 923.
Li, N., et al. (2018). Emodin inhibits pancreatic cancer EMT and invasion by up-regulating microRNA-1271. Molecular Medicine Reports, 18(3), 3366–3374.
Sun, L., et al. (2019). Betulinic acid inhibits stemness and EMT of pancreatic cancer cells via activation of AMPK signaling. International Journal of Oncology, 54(1), 98–110.
Liu, F., et al. (2019). SMARCAD1 promotes pancreatic cancer cell growth and metastasis through Wnt/β-catenin-mediated EMT. International Journal of Biological Sciences, 15(3), 636–646.
Peng, X., et al. (2022). Calpain2 upregulation regulates EMT-mediated pancreatic cancer metastasis via the Wnt/β-catenin Signaling Pathway. Frontiers of medicine (Lausanne), 9, 783592.
Tan, J., et al. (2014). Par-4 downregulation confers cisplatin resistance in pancreatic cancer cells via PI3K/Akt pathway-dependent EMT. Toxicology Letters, 224(1), 7–15.
Yang, R. M., et al. (2017). miR-3656 expression enhances the chemosensitivity of pancreatic cancer to gemcitabine through modulation of the RHOF/EMT axis. Cell Death & Disease, 8(10), e3129.
Li, Y., et al. (2019). Oncolytic Ad co-expressing decorin and Wnt decoy receptor overcomes chemoresistance of desmoplastic tumor through degradation of ECM and inhibition of EMT. Cancer Letters, 459, 15–29.
Okada, Y., et al. (2021). LAMC2 promotes cancer progression and gemcitabine resistance through modulation of EMT and ATP-binding cassette transporters in pancreatic ductal adenocarcinoma. Carcinogenesis, 42(4), 546–556.
An, N., & Zheng, B. (2020). MiR-203a-3p inhibits pancreatic cancer cell proliferation, EMT, and apoptosis by regulating SLUG. Technology in Cancer Research & Treatment, 19, 1533033819898729.
Chen, L., et al. (2022). ATP11A promotes EMT by regulating Numb PRR(L) in pancreatic cancer cells. PeerJ, 10, e13172.
Yu, S., et al. (2021). Circ_0092367 Inhibits EMT and gemcitabine resistance in pancreatic cancer via regulating the miR-1206/ESRP1 axis. Genes (Basel), 12(11), 1701.
Zhao, Y., et al. (2021). Targeted intervention of eIF4A1 inhibits EMT and metastasis of pancreatic cancer cells via c-MYC/miR-9 signaling. Cancer Cell International, 21(1), 670.
Hu, W., et al. (2019). IQGAP1 promotes pancreatic cancer progression and epithelial-mesenchymal transition (EMT) through Wnt/β-catenin signaling. Scientific Reports, 9(1), 7539.
Wang, W., et al. (2018). E-cadherin is downregulated by microenvironmental changes in pancreatic cancer and induces EMT. Oncology Reports, 40(3), 1641–1649.
Garg, R., et al. (2022). Targeting FTO suppresses pancreatic carcinogenesis via regulating stem cell maintenance and EMT pathway. Cancers (Basel), 14(23), 5919.
Santoro, R., et al. (2018). MEKK3 Sustains EMT and Stemness in Pancreatic Cancer by Regulating YAP and TAZ Transcriptional Activity. Anticancer Research, 38(4), 1937–1946.
Li, W., et al. (2020). Curcumin inhibits pancreatic cancer cell invasion and EMT by interfering with tumor-stromal crosstalk under hypoxic conditions via the IL-6/ERK/NF-κB axis. Oncology Reports, 44(1), 382–392.
Sheng, W., et al. (2020). Calreticulin promotes EMT in pancreatic cancer via mediating Ca(2+) dependent acute and chronic endoplasmic reticulum stress. Journal of Experimental & Clinical Cancer Research, 39(1), 209.
Yan, T., et al. (2017). Astaxanthin inhibits gemcitabine-resistant human pancreatic cancer progression through EMT inhibition and gemcitabine resensitization. Oncology Letters, 14(5), 5400–5408.
Weadick, B., et al. (2021). EMT-induced gemcitabine resistance in pancreatic cancer involves the functional loss of equilibrative nucleoside transporter 1. Molecular Cancer Therapeutics, 20(2), 410–422.
Zhu, S., et al. (2017). ASIC1 and ASIC3 contribute to acidity-induced EMT of pancreatic cancer through activating Ca(2+)/RhoA pathway. Cell Death & Disease, 8(5), e2806.
Shi, C., et al. (2021). NUDCD1 knockdown inhibits the proliferation, migration, and invasion of pancreatic cancer via the EMT process. Aging (Albany NY), 13(14), 18298–18309.
Zhang, Z., et al. (2017). miR-135b-5p Promotes migration, invasion and EMT of pancreatic cancer cells by targeting NR3C2. Biomedicine & Pharmacotherapy, 96, 1341–1348.
Rumman, M., et al. (2016). HS-173, a novel PI3K inhibitor suppresses EMT and metastasis in pancreatic cancer. Oncotarget, 7(47), 78029–78047.
Shen, J., et al. (2019). LncRNA XIST promotes pancreatic cancer migration, invasion and EMT by sponging miR-429 to modulate ZEB1 expression. The International Journal of Biochemistry & Cell Biology, 113, 17–26.
Zhang, R., et al. (2021). Silencing of circRNA circ_0001666 represses EMT in pancreatic cancer through upregulating miR-1251 and downregulating SOX4. Frontiers in Molecular Biosciences, 8, 684866.
Zhang, Y., et al. (2019). Upregulation of LASP2 inhibits pancreatic cancer cell migration and invasion through suppressing TGF-β-induced EMT. Journal of Cellular Biochemistry, 120(8), 13651–13657.
Bhutia, Y. D., et al. (2020). Chronic exposure to excess iron promotes EMT and cancer via p53 loss in pancreatic cancer. Asian Journal of Pharmaceutical Sciences, 15(2), 237–251.
Li, P., et al. (2020). FHL3 promotes pancreatic cancer invasion and metastasis through preventing the ubiquitination degradation of EMT associated transcription factors. Aging (Albany NY), 12(1), 53–69.
Zhang, L., et al. (2016). CCL21/CCR7 axis contributed to CD133+ pancreatic cancer stem-like cell metastasis via EMT and Erk/NF-κB pathway. PLoS One, 11(8), e0158529.
Zhao, H., et al. (2017). Up-regulation of glycolysis promotes the stemness and EMT phenotypes in gemcitabine-resistant pancreatic cancer cells. Journal of Cellular and Molecular Medicine, 21(9), 2055–2067.
Martinelli, P., et al. (2017). GATA6 regulates EMT and tumour dissemination, and is a marker of response to adjuvant chemotherapy in pancreatic cancer. Gut, 66(9), 1665–1676.
Gao, C., et al. (2021). Yap1-2 Isoform Is the Primary Mediator in TGF-β1 Induced EMT in Pancreatic Cancer. Frontiers in Oncology, 11, 649290.
Baer, R., et al. (2014). Pancreatic cell plasticity and cancer initiation induced by oncogenic Kras is completely dependent on wild-type PI 3-kinase p110α. Genes & Development, 28(23), 2621–2635.
Sancho, P., et al. (2015). MYC/PGC-1α balance determines the metabolic phenotype and plasticity of pancreatic cancer stem cells. Cell Metabolism, 22(4), 590–605.
Zhao, S., et al. (2016). CD44 expression level and isoform contributes to pancreatic cancer cell plasticity, invasiveness, and response to therapy. Clinical Cancer Research, 22(22), 5592–5604.
Suzuki, K., et al. (2017). Metadherin promotes metastasis by supporting putative cancer stem cell properties and epithelial plasticity in pancreatic cancer. Oncotarget, 8(39), 66098–66111.
Cruz, V. H., et al. (2019). Axl-mediated activation of TBK1 drives epithelial plasticity in pancreatic cancer. JCI Insight, 5(9), e126117.
Kirane, A., et al. (2015). Warfarin blocks Gas6-mediated Axl activation required for pancreatic cancer epithelial plasticity and metastasis. Cancer Research, 75(18), 3699–3705.
Feldmann, K., et al. (2021). Mesenchymal Plasticity Regulated by Prrx1 Drives Aggressive Pancreatic Cancer Biology. Gastroenterology, 160(1), 346–361.e24.
Perusina Lanfranca, M., et al. (2020). Interleukin 22 signaling regulates acinar cell plasticity to promote pancreatic tumor development in mice. Gastroenterology, 158(5), 1417–1432.e11.
Sharma, N., et al. (2020). Metabolic plasticity imparts erlotinib-resistance in pancreatic cancer by upregulating glucose-6-phosphate dehydrogenase. Cancer and Metabolism, 8, 19.
Yi, Z., et al. (2022). KDM6A Regulates Cell Plasticity and Pancreatic Cancer Progression by Noncanonical Activin Pathway. Cellular and Molecular Gastroenterology and Hepatology, 13(2), 643–667.
Singh, S. K., et al. (2015). Antithetical NFATc1-Sox2 and p53-miR200 signaling networks govern pancreatic cancer cell plasticity. The EMBO Journal, 34(4), 517–530.
Liu, M., et al. (2021). Zinc-Dependent regulation of ZEB1 and YAP1 coactivation promotes epithelial-mesenchymal transition plasticity and metastasis in pancreatic cancer. Gastroenterology, 160(5), 1771–1783.e1.
Krebs, A. M., et al. (2017). The EMT-activator Zeb1 is a key factor for cell plasticity and promotes metastasis in pancreatic cancer. Nature Cell Biology, 19(5), 518–529.
Kloesch, B., et al. (2022). A GATA6-centred gene regulatory network involving HNFs and ΔNp63 controls plasticity and immune escape in pancreatic cancer. Gut, 71(4), 766–777.
Nishino, H., et al. (2017). Grainyhead-like 2 (GRHL2) regulates epithelial plasticity in pancreatic cancer progression. Cancer Medicine, 6(11), 2686–2696.
Raghavan, S., et al. (2021). Microenvironment drives cell state, plasticity, and drug response in pancreatic cancer. Cell, 184(25), 6119–6137.e26.
Ireland, L., et al. (2020). Blockade of Stromal Gas6 Alters Cancer Cell Plasticity, Activates NK Cells, and Inhibits Pancreatic Cancer Metastasis. Frontiers in Immunology, 11, 297.
Datta, J., et al. (2022). Combined MEK and STAT3 inhibition uncovers stromal plasticity by enriching for cancer-associated fibroblasts with mesenchymal stem cell-like features to overcome immunotherapy resistance in pancreatic cancer. Gastroenterology, 163(6), 1593–1612.
Cao, R., et al. (2020). miR-3613-5p enhances the metastasis of pancreatic cancer by targeting CDK6. Cell Cycle, 19(22), 3086–3095.
Chen, Z., et al. (2018). MiR-1297 suppresses pancreatic cancer cell proliferation and metastasis by targeting MTDH. Molecular and Cellular Probes, 40, 19–26.
Wu, X., et al. (2018). MiR-216b inhibits pancreatic cancer cell progression and promotes apoptosis by down-regulating KRAS. Archives of Medical Science, 14(6), 1321–1332.
Zhang, D., et al. (2021). MiR-489-3p reduced pancreatic cancer proliferation and metastasis by targeting PKM2 and LDHA involving glycolysis. Frontiers in Oncology, 11, 651535.
Ashrafizadeh, M., et al. (2023). Noncoding RNAs as regulators of STAT3 pathway in gastrointestinal cancers: Roles in cancer progression and therapeutic response. Medicinal Research Reviews. https://doi.org/10.1002/med.21950
Mirzaei, S., et al. (2022). Molecular landscape of LncRNAs in prostate cancer: A focus on pathways and therapeutic targets for intervention. Journal of Experimental & Clinical Cancer Research, 41(1), 214.
Luo, Y., et al. (2020). LncRNA DANCR promotes proliferation and metastasis in pancreatic cancer by regulating miRNA-33b. FEBS Open Bio, 10(1), 18–27.
Hui, B., et al. (2019). RREB1-induced upregulation of the lncRNA AGAP2-AS1 regulates the proliferation and migration of pancreatic cancer partly through suppressing ANKRD1 and ANGPTL4. Cell Death & Disease, 10(3), 207.
Feng, Y., et al. (2020). LncRNA NEAT1 facilitates pancreatic cancer growth and metastasis through stabilizing ELF3 mRNA. American Journal of Cancer Research, 10(1), 237–248.
Ren, X., et al. (2020). lncRNA-PLACT1 sustains activation of NF-κB pathway through a positive feedback loop with IκBα/E2F1 axis in pancreatic cancer. Molecular Cancer, 19(1), 35.
Shen, X., et al. (2021). Identification of Circ_001569 as a Potential biomarker in the diagnosis and prognosis of pancreatic cancer. Technology in Cancer Research & Treatment, 20, 1533033820983302.
Zhang, J., et al. (2023). Circ_0000284 upregulates RHPN2 to facilitate pancreatic cancer proliferation, metastasis, and angiogenesis through sponging miR-1179. Journal of Biochemical and Molecular Toxicology, 37(3), e23274.
Li, C., et al. (2023). Downregulation of circ-STK39 suppresses pancreatic cancer progression by sponging mir-140-3p and regulating TRAM2-mediated epithelial-mesenchymal transition. Apoptosis, 28, 1024–1034.
Li, M., et al. (2020). miR-193a-5p promotes pancreatic cancer cell metastasis through SRSF6-mediated alternative splicing of OGDHL and ECM1. American Journal of Cancer Research, 10(1), 38–59.
Mody, H. R., et al. (2017). miR-202 Diminishes TGFβ Receptors and Attenuates TGFβ1-Induced EMT in Pancreatic Cancer. Molecular Cancer Research, 15(8), 1029–1039.
Jiang, J., et al. (2015). Reduction of miR-29c enhances pancreatic cancer cell migration and stem cell-like phenotype. Oncotarget, 6(5), 2767–2778.
Wu, X., et al. (2016). MiR-200a suppresses the proliferation and metastasis in pancreatic ductal adenocarcinoma through downregulation of DEK Gene. Translational Oncology, 9(1), 25–31.
Hu, Y., et al. (2012). miR-143 inhibits the metastasis of pancreatic cancer and an associated signaling pathway. Tumour Biology, 33(6), 1863–1870.
Chen, Y., et al. (2019). MicroRNA hsa-miR-623 directly suppresses MMP1 and attenuates IL-8-induced metastasis in pancreatic cancer. International Journal of Oncology, 55(1), 142–156.
Ma, L., et al. (2019). The miR-141/neuropilin-1 axis is associated with the clinicopathology and contributes to the growth and metastasis of pancreatic cancer. Cancer Cell International, 19, 248.
Wang, L., Mu, N., & Qu, N. (2021). Methylation of the miR-29b-3p promoter contributes to angiogenesis, invasion, and migration in pancreatic cancer. Oncology Reports, 45(1), 65–72.
Ma, C., et al. (2014). miR-212 promotes pancreatic cancer cell growth and invasion by targeting the hedgehog signaling pathway receptor patched-1. Journal of Experimental & Clinical Cancer Research, 33(1), 54.
Xue, L., et al. (2020). miR-539 suppresses the proliferation, migration, invasion and epithelial mesenchymal transition of pancreatic cancer cells through targeting SP1. International Journal of Molecular Medicine, 45(6), 1771–1782.
Hiramoto, H., et al. (2017). miR-509-5p and miR-1243 increase the sensitivity to gemcitabine by inhibiting epithelial-mesenchymal transition in pancreatic cancer. Scientific Reports, 7(1), 4002.
Deng, S. J., et al. (2018). Hypoxia-induced LncRNA-BX111 promotes metastasis and progression of pancreatic cancer through regulating ZEB1 transcription. Oncogene, 37(44), 5811–5828.
Cheng, C., et al. (2022). Positive feedback regulation of lncRNA TPT1-AS1 and ITGB3 promotes cell growth and metastasis in pancreatic cancer. Cancer Science, 113(9), 2986–3001.
Lou, S., et al. (2019). Downregulation of lncRNA AFAP1-AS1 by oridonin inhibits the epithelial-to-mesenchymal transition and proliferation of pancreatic cancer cells. Acta Biochimica et Biophysica Sinica Shanghai, 51(8), 814–825.
Cui, X. P., et al. (2019). LncRNA TP73-AS1 sponges miR-141-3p to promote the migration and invasion of pancreatic cancer cells through the up-regulation of BDH2. Bioscience Reports, 39(3), BSR20181937.
Liu, W., et al. (2020). A novel lncRNA PTTG3P/miR-132/212-3p/FoxM1 feedback loop facilitates tumorigenesis and metastasis of pancreatic cancer. Cell death discovery, 6(1), 136.
An, Y., et al. (2018). LncRNA DLX6-AS1 promoted cancer cell proliferation and invasion by attenuating the endogenous function of miR-181b in pancreatic cancer. Cancer Cell International, 18, 143.
Liu, B., et al. (2018). lncRNA GAS5 reverses EMT and tumor stem cell-mediated gemcitabine resistance and metastasis by targeting miR-221/SOCS3 in pancreatic cancer. Molecular Therapy--Nucleic Acids, 13, 472–482.
Gu, L., et al. (2017). lncRNA MEG3 had anti-cancer effects to suppress pancreatic cancer activity. Biomedicine & Pharmacotherapy, 89, 1269–1276.
Liu, S., et al. (2019). LncRNA SNHG16 promotes tumor growth of pancreatic cancer by targeting miR-218-5p. Biomedicine & Pharmacotherapy, 114, 108862.
Deng, P. C., et al. (2019). LncRNA SNHG14 potentiates pancreatic cancer progression via modulation of annexin A2 expression by acting as a competing endogenous RNA for miR-613. Journal of Cellular and Molecular Medicine, 23(11), 7222–7232.
Huang, S., et al. (2021). LncRNA PWAR6 regulates proliferation and migration by epigenetically silencing YAP1 in tumorigenesis of pancreatic ductal adenocarcinoma. Journal of Cellular and Molecular Medicine, 25(9), 4275–4286.
Chen, S., et al. (2020). LncRNA STXBP5-AS1 suppresses stem cell-like properties of pancreatic cancer by epigenetically inhibiting neighboring androglobin gene expression. Clinical Epigenetics, 12(1), 168.
Cai, J., et al. (2021). Curcumin attenuates lncRNA H19-induced epithelial-mesenchymal transition in tamoxifen-resistant breast cancer cells. Molecular Medicine Reports, 23(1), 1.
Zhou, M., et al. (2022). LncRNA FAM83H-AS1 promotes the malignant progression of pancreatic ductal adenocarcinoma by stabilizing FAM83H mRNA to protect β-catenin from degradation. Journal of Experimental & Clinical Cancer Research, 41(1), 288.
Cai, H., et al. (2017). LncRNA HOTAIR acts a competing endogenous RNA to control the expression of notch3 via sponging miR-613 in pancreatic cancer. Oncotarget, 8(20), 32905–32917.
Miao, H., et al. (2021). LncRNA TP73-AS1 enhances the malignant properties of pancreatic ductal adenocarcinoma by increasing MMP14 expression through miRNA -200a sponging. Journal of Cellular and Molecular Medicine, 25(7), 3654–3664.
Zhang, X., et al. (2020). Circ_0075829 facilitates the progression of pancreatic carcinoma by sponging miR-1287-5p and activating LAMTOR3 signalling. Journal of Cellular and Molecular Medicine, 24(24), 14596–14607.
Xing, C., et al. (2019). Circular RNA ADAM9 facilitates the malignant behaviours of pancreatic cancer by sponging miR-217 and upregulating PRSS3 expression. Artificial Cells, Nanomedicine and Biotechnology, 47(1), 3920–3928.
Wang, K. Q., et al. (2022). Circular RNA fibroblast growth factor receptor 1 promotes pancreatic cancer progression by targeting microRNA-532-3p/PIK3CB Axis. Pancreas, 51(8), 930–942.
Messex, J. K., et al. (2022). Oncogenic Kras-mediated cytokine CCL15 regulates pancreatic cancer cell migration and invasion through ROS. Cancers (Basel), 14(9), 2153.
Deng, J., et al. (2022). High Glucose promotes pancreatic ductal adenocarcinoma gemcitabine resistance and invasion through modulating ROS/MMP-3 signaling pathway. Oxidative Medicine and Cellular Longevity, 2022, 3243647.
Binker-Cosen, M. J., et al. (2017). Palmitic acid increases invasiveness of pancreatic cancer cells AsPC-1 through TLR4/ROS/NF-κB/MMP-9 signaling pathway. Biochemical and Biophysical Research Communications, 484(1), 152–158.
Shimojo, Y., et al. (2013). Attenuation of reactive oxygen species by antioxidants suppresses hypoxia-induced epithelial-mesenchymal transition and metastasis of pancreatic cancer cells. Clinical & Experimental Metastasis, 30(2), 143–154.
Wörmann, S. M., et al. (2021). APOBEC3A drives deaminase domain-independent chromosomal instability to promote pancreatic cancer metastasis. Nature Cancer, 2(12), 1338–1356.
Zhao, T., et al. (2020). IL-37/ STAT3/ HIF-1α negative feedback signaling drives gemcitabine resistance in pancreatic cancer. Theranostics, 10(9), 4088–4100.
Hu, H., et al. (2020). MicroRNA-301a promotes pancreatic cancer invasion and metastasis through the JAK/STAT3 signaling pathway by targeting SOCS5. Carcinogenesis, 41(4), 502–514.
Huang, C., et al. (2011). STAT3-targeting RNA interference inhibits pancreatic cancer angiogenesis in vitro and in vivo. International Journal of Oncology, 38(6), 1637–1644.
Fofaria, N. M., & Srivastava, S. K. (2015). STAT3 induces anoikis resistance, promotes cell invasion and metastatic potential in pancreatic cancer cells. Carcinogenesis, 36(1), 142–150.
Chen, H., et al. (2023). Selectively Targeting STAT3 Using a Small Molecule Inhibitor is a Potential Therapeutic Strategy for Pancreatic Cancer. Clinical Cancer Research, 29(4), 815–830.
Li, H., et al. (2011). STAT3 knockdown reduces pancreatic cancer cell invasiveness and matrix metalloproteinase-7 expression in nude mice. PLoS One, 6(10), e25941.
Li, Y., Zhang, Y., & Liu, J. (2019). NETO2 promotes pancreatic cancer cell proliferation, invasion and migration via activation of the STAT3 signaling pathway. Cancer Management and Research, 11, 5147–5156.
Hu, B., et al. (2016). HIC1 attenuates invasion and metastasis by inhibiting the IL-6/STAT3 signalling pathway in human pancreatic cancer. Cancer Letters, 376(2), 387–398.
Zuo, C., et al. (2018). Celecoxib suppresses proliferation and metastasis of pancreatic cancer cells by down-regulating STAT3 / NF-kB and L1CAM activities. Pancreatology, 18(3), 328–333.
Lian, J., et al. (2020). Ubiquitin specific peptidase 5 enhances STAT3 signaling and promotes migration and invasion in Pancreatic Cancer. Journal of Cancer, 11(23), 6802–6811.
Chen, J., et al. (2016). Interleukin-32α inactivates JAK2/STAT3 signaling and reverses interleukin-6-induced epithelial-mesenchymal transition, invasion, and metastasis in pancreatic cancer cells. Oncotargets and Therapy, 9, 4225–4237.
Zhang, Z., et al. (2019). BRM transcriptionally regulates miR-302a-3p to target SOCS5/STAT3 signaling axis to potentiate pancreatic cancer metastasis. Cancer Letters, 449, 215–225.
Chang, Y. T., et al. (2023). Pancreatic cancer-derived small extracellular vesical ezrin activates fibroblasts to exacerbate cancer metastasis through STAT3 and YAP-1 signaling pathways. Molecular Oncology. https://doi.org/10.1002/1878-0261.13442
Liu, L., et al. (2019). Circular RNA ciRS-7 promotes the proliferation and metastasis of pancreatic cancer by regulating miR-7-mediated EGFR/STAT3 signaling pathway. Hepatobiliary & Pancreatic Diseases International, 18(6), 580–586.
Guo, Y., et al. (2021). The anti-dysenteric drug fraxetin enhances anti-tumor efficacy of gemcitabine and suppresses pancreatic cancer development by antagonizing STAT3 activation. Aging (Albany NY), 13(14), 18545–18563.
Al-Ismaeel, Q., et al. (2019). ZEB1 and IL-6/11-STAT3 signalling cooperate to define invasive potential of pancreatic cancer cells via differential regulation of the expression of S100 proteins. British Journal of Cancer, 121(1), 65–75.
Su, K., et al. (2023). Solasodine suppresses the metastasis of gastric cancer through claudin-2 via the AMPK/STAT3/NF-κB pathway. Chemico-Biological Interactions, 379, 110520.
Huang, C., et al. (2013). Regulation of miR-155 affects pancreatic cancer cell invasiveness and migration by modulating the STAT3 signaling pathway through SOCS1. Oncology Reports, 30(3), 1223–1230.
Zhao, G., et al. (2013). MiR-130b is a prognostic marker and inhibits cell proliferation and invasion in pancreatic cancer through targeting STAT3. PLoS One, 8(9), e73803.
Wei, D., et al. (2003). Stat3 activation regulates the expression of vascular endothelial growth factor and human pancreatic cancer angiogenesis and metastasis. Oncogene, 22(3), 319–329.
Zhao, S., et al. (2008). Inhibition of STAT3 Tyr705 phosphorylation by Smad4 suppresses transforming growth factor beta-mediated invasion and metastasis in pancreatic cancer cells. Cancer Research, 68(11), 4221–4228.
Sasaki, N., et al. (2022). Gp130-Mediated STAT3 Activation contributes to the aggressiveness of pancreatic cancer through H19 long non-coding RNA expression. Cancers (Basel), 14(9), 2055.
Wu, Y. S., et al. (2017). Paracrine IL-6 signaling mediates the effects of pancreatic stellate cells on epithelial-mesenchymal transition via Stat3/Nrf2 pathway in pancreatic cancer cells. Biochimica et Biophysica Acta - General Subjects, 1861(2), 296–306.
Liu, M., et al. (2019). Hypoxia-induced feedback of HIF-1α and lncRNA-CF129 contributes to pancreatic cancer progression through stabilization of p53 protein. Theranostics, 9(16), 4795–4810.
Jin, Y., et al. (2021). Positive Reciprocal Feedback of lncRNA ZEB1-AS1 and HIF-1α contributes to hypoxia-promoted tumorigenesis and metastasis of pancreatic cancer. Frontiers in Oncology, 11, 761979.
Niu, Y., et al. (2018). MiRNA-646-mediated reciprocal repression between HIF-1α and MIIP contributes to tumorigenesis of pancreatic cancer. Oncogene, 37(13), 1743–1758.
Yue, H., Liu, L., & Song, Z. (2019). miR-212 regulated by HIF-1α promotes the progression of pancreatic cancer. Experimental and Therapeutic Medicine, 17(3), 2359–2365.
Wang, L., et al. (2021). N2E4, a Monoclonal antibody targeting neuropilin-2, inhibits tumor growth and metastasis in pancreatic ductal adenocarcinoma via suppressing FAK/Erk/HIF-1α Signaling. Frontiers in Oncology, 11, 657008.
Zhang, D., et al. (2016). β2-adrenogenic signaling regulates NNK-induced pancreatic cancer progression via upregulation of HIF-1α. Oncotarget, 7(14), 17760–17772.
Zeng, Z., et al. (2019). LncRNA-MTA2TR functions as a promoter in pancreatic cancer via driving deacetylation-dependent accumulation of HIF-1α. Theranostics, 9(18), 5298–5314.
Zhu, G. H., et al. (2013). Hypoxia-induced snail expression through transcriptional regulation by HIF-1α in pancreatic cancer cells. Digestive Diseases and Sciences, 58(12), 3503–3515.
Zhang, Q., et al. (2020). Expression of the PTEN/FOXO3a/PLZF signalling pathway in pancreatic cancer and its significance in tumourigenesis and progression. Investigational New Drugs, 38(2), 321–328.
Gao, Z. Q., et al. (2017). Long non-coding RNA GAS5 suppresses pancreatic cancer metastasis through modulating miR-32-5p/PTEN axis. Cell & Bioscience, 7, 66.
Liu, Y., et al. (2020). Role of miR-92a-3p/PTEN axis in regulation of pancreatic cancer cell proliferation and metastasis. Zhong Nan Da Xue Xue Bao. Yi Xue Ban, 45(3), 280–289.
Gu, J., et al. (2016). GFRα2 prompts cell growth and chemoresistance through down-regulating tumor suppressor gene PTEN via Mir-17-5p in pancreatic cancer. Cancer Letters, 380(2), 434–441.
Wang, M. C., et al. (2016). Polycomb complex protein BMI-1 promotes invasion and metastasis of pancreatic cancer stem cells by activating PI3K/AKT signaling, an ex vivo, in vitro, and in vivo study. Oncotarget, 7(8), 9586–9599.
Zhao, H., et al. (2014). Activation of glucagon-like peptide-1 receptor inhibits tumourigenicity and metastasis of human pancreatic cancer cells via PI3K/Akt pathway. Diabetes, Obesity and Metabolism, 16(9), 850–860.
Fujita, M., et al. (2014). Nitric oxide increases the invasion of pancreatic cancer cells via activation of the PI3K-AKT and RhoA pathways after carbon ion irradiation. FEBS Letters, 588(17), 3240–3250.
Li, H., et al. (2014). Effect of PTEN and KAI1 gene overexpression on the proliferation, metastasis and radiosensitivity of ASPC-1 pancreatic cancer cells under hypoxic conditions. Molecular Medicine Reports, 10(4), 1973–1977.
Ma, J., et al. (2010). IGF-1 mediates PTEN suppression and enhances cell invasion and proliferation via activation of the IGF-1/PI3K/Akt signaling pathway in pancreatic cancer cells. The Journal of Surgical Research, 160(1), 90–101.
Zhang, H., et al. (2019). MiR-132 promotes the proliferation, invasion and migration of human pancreatic carcinoma by inhibition of the tumor suppressor gene PTEN. Progress in Biophysics and Molecular Biology, 148, 65–72.
Zhang, H., et al. (2019). Long non-coding RNA CASC2 upregulates PTEN to suppress pancreatic carcinoma cell metastasis by downregulating miR-21. Cancer Cell International, 19, 18.
Wu, Y., et al. (2019). ITGA6 and RPSA synergistically promote pancreatic cancer invasion and metastasis via PI3K and MAPK signaling pathways. Experimental Cell Research, 379(1), 30–47.
Zhu, J. H., et al. (2020). MiR-139-5p/SLC7A11 inhibits the proliferation, invasion and metastasis of pancreatic carcinoma via PI3K/Akt signaling pathway. Biochimica et Biophysica Acta - Molecular Basis of Disease, 1866(6), 165747.
Li, W., et al. (2018). Curcumin inhibits superoxide dismutase-induced epithelial-to-mesenchymal transition via the PI3K/Akt/NF-κB pathway in pancreatic cancer cells. International Journal of Oncology, 52(5), 1593–1602.
Liu, X., et al. (2018). Phosphoglycerate Mutase 1 (PGAM1) Promotes pancreatic ductal adenocarcinoma (PDAC) Metastasis by acting as a novel downstream target of the PI3K/Akt/mTOR Pathway. Oncology Research, 26(7), 1123–1131.
Deng, J., et al. (2021). N(6) -methyladenosine-mediated upregulation of WTAPP1 promotes WTAP translation and Wnt signaling to facilitate pancreatic cancer progression. Cancer Research, 81(20), 5268–5283.
Gokturk, F., Erkoc-Kaya, D., & Arikoglu, H. (2021). Juglone can inhibit angiogenesis and metastasis in pancreatic cancer cells by targeting Wnt/β-catenin signaling. Bratislavské Lekárske Listy, 122(2), 132–137.
Chen, T., et al. (2020). Linc00261 inhibits metastasis and the WNT signaling pathway of pancreatic cancer by regulating a miR-552-5p/FOXO3 axis. Oncology Reports, 43(3), 930–942.
Xu, D., et al. (2020). Cadherin 13 Inhibits Pancreatic Cancer Progression and Epithelial-mesenchymal Transition by Wnt/β-Catenin Signaling. Journal of Cancer, 11(8), 2101–2112.
Nielsen, M. F., Mortensen, M. B., & Detlefsen, S. (2016). Key players in pancreatic cancer-stroma interaction: Cancer-associated fibroblasts, endothelial and inflammatory cells. World Journal of Gastroenterology, 22(9), 2678–2700.
Hosein, A. N., Brekken, R. A., & Maitra, A. (2020). Pancreatic cancer stroma: an update on therapeutic targeting strategies. Nature Reviews. Gastroenterology & Hepatology, 17(8), 487–505.
Veenstra, V. L., et al. (2018). ADAM12 is a circulating marker for stromal activation in pancreatic cancer and predicts response to chemotherapy. Oncogenesis, 7(11), 87.
Cannon, A., et al. (2018). Desmoplasia in pancreatic ductal adenocarcinoma: insight into pathological function and therapeutic potential. Genes & Cancer, 9(3-4), 78–86.
Schnittert, J., et al. (2019). Integrin α11 in pancreatic stellate cells regulates tumor stroma interaction in pancreatic cancer. The FASEB Journal, 33(5), 6609–6621.
Goehrig, D., et al. (2019). Stromal protein βig-h3 reprogrammes tumour microenvironment in pancreatic cancer. Gut, 68(4), 693–707.
Awaji, M., & Singh, R. K. (2019). Cancer-associated fibroblasts' functional heterogeneity in pancreatic ductal adenocarcinoma. Cancers (Basel), 11(3), 290.
Sun, Q., et al. (2018). The impact of cancer-associated fibroblasts on major hallmarks of pancreatic cancer. Theranostics, 8(18), 5072–5087.
Kobayashi, H., et al. (2019). Cancer-associated fibroblasts in gastrointestinal cancer. Nature Reviews. Gastroenterology & Hepatology, 16(5), 282–295.
Geleta, B., et al. (2022). Targeting Wnt/tenascin C-mediated cross talk between pancreatic cancer cells and stellate cells via activation of the metastasis suppressor NDRG1. The Journal of Biological Chemistry, 298(3), 101608.
Liu, Q. Q., et al. (2016). Oridonin inhibits pancreatic cancer cell migration and epithelial-mesenchymal transition by suppressing Wnt/β-catenin signaling pathway. Cancer Cell International, 16, 57.
Quan, M., et al. (2015). Merlin/NF2 suppresses pancreatic tumor growth and metastasis by attenuating the FOXM1-mediated Wnt/β-Catenin signaling. Cancer Research, 75(22), 4778–4789.
Li, F., Dai, L., & Niu, J. (2020). GPX2 silencing relieves epithelial-mesenchymal transition, invasion, and metastasis in pancreatic cancer by downregulating Wnt pathway. Journal of Cellular Physiology, 235(11), 7780–7790.
Garg, B., et al. (2017). Modulation of post-translational modifications in β-catenin and LRP6 inhibits Wnt signaling pathway in pancreatic cancer. Cancer Letters, 388, 64–72.
Jiang, H., et al. (2014). Activation of the Wnt pathway through Wnt2 promotes metastasis in pancreatic cancer. American Journal of Cancer Research, 4(5), 537–544.
Zhou, P., et al. (2019). NMIIA promotes tumor growth and metastasis by activating the Wnt/β-catenin signaling pathway and EMT in pancreatic cancer. Oncogene, 38(27), 5500–5515.
Wang, L., et al. (2009). Oncogenic function of ATDC in pancreatic cancer through Wnt pathway activation and beta-catenin stabilization. Cancer Cell, 15(3), 207–219.
Sun, Y., et al. (2019). Restoration of miRNA-148a in pancreatic cancer reduces invasion and metastasis by inhibiting the Wnt/β-catenin signaling pathway via downregulating maternally expressed gene-3. Experimental and Therapeutic Medicine, 17(1), 639–648.
Zhang, Q., et al. (2017). Hypoxia-inducible factor-2α promotes tumor progression and has crosstalk with Wnt/β-catenin signaling in pancreatic cancer. Molecular Cancer, 16(1), 119.
Weng, Y. C., et al. (2019). Long non-coding RNA LINC01133 silencing exerts antioncogenic effect in pancreatic cancer through the methylation of DKK1 promoter and the activation of Wnt signaling pathway. Cancer Biology & Therapy, 20(3), 368–380.
Wu, X., et al. (2019). LncRNA BANCR promotes pancreatic cancer tumorigenesis via modulating MiR-195-5p/Wnt/β-catenin signaling pathway. Technology in Cancer Research & Treatment, 18, 1533033819887962.
Yang, Y., Bai, Y. S., & Wang, Q. (2017). CDGSH iron sulfur domain 2 activates proliferation and EMT of pancreatic cancer cells via Wnt/β-catenin pathway and has prognostic value in human pancreatic cancer. Oncology Research, 25(4), 605–615.
Kim, J. H., et al. (2022). Vitamin C suppresses pancreatic carcinogenesis through the inhibition of both glucose metabolism and Wnt signaling. International Journal of Molecular Sciences, 23(20), 12249.
Zhan, T., et al. (2020). miR-455-3p functions as a tumor suppressor by restraining Wnt/β-catenin signaling via TAZ in pancreatic cancer. Cancer Management and Research, 12, 1483–1492.
Yang, J., et al. (2019). Long noncoding RNA DLX6-AS1 promotes tumorigenesis by modulating miR-497-5p/FZD4/FZD6/Wnt/β-catenin pathway in pancreatic cancer. Cancer Management and Research, 11, 4209–4221.
Wang, Z., et al. (2008). Blockade of SDF-1/CXCR4 signalling inhibits pancreatic cancer progression in vitro via inactivation of canonical Wnt pathway. British Journal of Cancer, 99(10), 1695–1703.
Douchi, D., et al. (2015). Silencing of LRRFIP1 reverses the epithelial-mesenchymal transition via inhibition of the Wnt/β-catenin signaling pathway. Cancer Letters, 365(1), 132–140.
Ashrafizadeh, M., et al. (2022). Exosomes as promising nanostructures in diabetes mellitus: From insulin sensitivity to ameliorating diabetic complications. International Journal of Nanomedicine, 17, 1229–1253.
Paskeh, M. D. A., et al. (2022). Emerging role of exosomes in cancer progression and tumor microenvironment remodeling. Journal of Hematology & Oncology, 15(1), 1–39.
Li, J., et al. (2018). Circular RNA IARS (circ-IARS) secreted by pancreatic cancer cells and located within exosomes regulates endothelial monolayer permeability to promote tumor metastasis. Journal of Experimental & Clinical Cancer Research, 37(1), 177.
Yu, Z., et al. (2017). Pancreatic cancer-derived exosomes promote tumor metastasis and liver pre-metastatic niche formation. Oncotarget, 8(38), 63461–63483.
Tang, P., et al. (2019). Serum derived exosomes from pancreatic cancer patients promoted metastasis: An iTRAQ-based proteomic analysis. Oncotargets and Therapy, 12, 9329–9339.
Ogawa, K., et al. (2020). Prometastatic secretome trafficking via exosomes initiates pancreatic cancer pulmonary metastasis. Cancer Letters, 481, 63–75.
Kimoto, A., et al. (2023). Exosomes in ascites from patients with human pancreatic cancer enhance remote metastasis partially through endothelial-mesenchymal transition. Pancreatology, 23(4), 377–388.
Zhou, X., et al. (2022). Pancreatic cancer cell-derived exosomes promote lymphangiogenesis by downregulating ABHD11-AS1 expression. Cancers (Basel), 14(19), 4612.
Xu, Y., et al. (2020). Anticancer effects of miR-124 delivered by BM-MSC derived exosomes on cell proliferation, epithelial mesenchymal transition, and chemotherapy sensitivity of pancreatic cancer cells. Aging (Albany NY), 12(19), 19660–19676.
Wei, Q., et al. (2020). EphA2-enriched exosomes promote cell migration and are a potential diagnostic serum marker in pancreatic cancer. Molecular Medicine Reports, 22(4), 2941–2947.
Zhang, Y. F., et al. (2019). Pancreatic cancer-derived exosomes promoted pancreatic stellate cells recruitment by pancreatic cancer. Journal of Cancer, 10(18), 4397–4407.
Nakayama, F., et al. (2022). Pancreatic cancer cell-derived exosomes induce epithelial-mesenchymal transition in human pancreatic cancer cells themselves partially via transforming growth factor β1. Medical Molecular Morphology, 55(3), 227–235.
Wang, X., et al. (2018). Hypoxic tumor-derived exosomal miR-301a mediates M2 macrophage polarization via PTEN/PI3Kγ to promote pancreatic cancer metastasis. Cancer Research, 78(16), 4586–4598.
Chen, K., et al. (2022). Hypoxic pancreatic cancer derived exosomal miR-30b-5p promotes tumor angiogenesis by inhibiting GJA1 expression. International Journal of Biological Sciences, 18(3), 1220–1237.
Li, M., et al. (2022). Exosomal miR-485-3p derived from pancreatic ductal epithelial cells inhibits pancreatic cancer metastasis through targeting PAK1. Chinese Medical Journal, 135(19), 2326–2337.
Cao, W., et al. (2021). Hypoxic pancreatic stellate cell-derived exosomal mirnas promote proliferation and invasion of pancreatic cancer through the PTEN/AKT pathway. Aging (Albany NY), 13(5), 7120–7132.
Li, Z., et al. (2018). Tumor-secreted exosomal miR-222 promotes tumor progression via regulating P27 expression and re-localization in pancreatic cancer. Cellular Physiology and Biochemistry, 51(2), 610–629.
Sun, Z., et al. (2021). Exosomal linc-ROR mediates crosstalk between cancer cells and adipocytes to promote tumor growth in pancreatic cancer. Molecular Therapy--Nucleic Acids, 26, 253–268.
Wu, M., et al. (2020). Role of exosomal microRNA-125b-5p in conferring the metastatic phenotype among pancreatic cancer cells with different potential of metastasis. Life Sciences, 255, 117857.
Xie, Z., et al. (2022). Exosome-delivered CD44v6/C1QBP complex drives pancreatic cancer liver metastasis by promoting fibrotic liver microenvironment. Gut, 71(3), 568–579.
He, Z., et al. (2022). Exosome-derived FGD5-AS1 promotes tumor-associated macrophage M2 polarization-mediated pancreatic cancer cell proliferation and metastasis. Cancer Letters, 548, 215751.
Costa-Silva, B., et al. (2015). Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nature Cell Biology, 17(6), 816–826.
Williams, C. B., Yeh, E. S., & Soloff, A. C. (2016). Tumor-associated macrophages: unwitting accomplices in breast cancer malignancy. NPJ Breast Cancer, 2, 15025.
Ye, H., et al. (2018). Tumor-associated macrophages promote progression and the Warburg effect via CCL18/NF-kB/VCAM-1 pathway in pancreatic ductal adenocarcinoma. Cell Death & Disease, 9(5), 453.
Meng, F., et al. (2015). CCL18 promotes epithelial-mesenchymal transition, invasion and migration of pancreatic cancer cells in pancreatic ductal adenocarcinoma. International Journal of Oncology, 46(3), 1109–1120.
Quail, D. F., & Joyce, J. A. (2013). Microenvironmental regulation of tumor progression and metastasis. Nature Medicine, 19(11), 1423–1437.
Chávez-Galán, L., et al. (2015). Much More than M1 and M2 Macrophages, There are also CD169(+) and TCR(+) Macrophages. Frontiers in Immunology, 6, 263.
Tarique, A. A., et al. (2015). Phenotypic, functional, and plasticity features of classical and alternatively activated human macrophages. American Journal of Respiratory Cell and Molecular Biology, 53(5), 676–688.
Helm, O., et al. (2014). M1 and M2: there is no "good" and "bad"-How macrophages promote malignancy-associated features in tumorigenesis. Oncoimmunology, 3(7), e946818.
Stöger, J. L., et al. (2012). Distribution of macrophage polarization markers in human atherosclerosis. Atherosclerosis, 225(2), 461–468.
Genard, G., Lucas, S., & Michiels, C. (2017). reprogramming of tumor-associated macrophages with anticancer therapies: Radiotherapy versus chemo- and immunotherapies. Frontiers in Immunology, 8, 828.
Chang, Y. T., et al. (2020). Pancreatic cancer-derived small extracellular vesical Ezrin regulates macrophage polarization and promotes metastasis. American Journal of Cancer Research, 10(1), 12–37.
Nielsen, S. R., et al. (2016). Macrophage-secreted granulin supports pancreatic cancer metastasis by inducing liver fibrosis. Nature Cell Biology, 18(5), 549–560.
Liu, B., et al. (2016). Tumor-associated macrophage-derived CCL20 enhances the growth and metastasis of pancreatic cancer. Acta Biochimica et Biophysica Sinica Shanghai, 48(12), 1067–1074.
Thibault, B., et al. (2021). Pancreatic cancer intrinsic PI3Kα activity accelerates metastasis and rewires macrophage component. EMBO Molecular Medicine, 13(7), e13502.
Griesmann, H., et al. (2017). Pharmacological macrophage inhibition decreases metastasis formation in a genetic model of pancreatic cancer. Gut, 66(7), 1278–1285.
Lin, L., et al. (2023). UTMD inhibits pancreatic cancer growth and metastasis by inducing macrophage polarization and vessel normalization. Biomedicine & Pharmacotherapy, 160, 114322.
Novizio, N., et al. (2021). ANXA1 contained in EVs Regulates macrophage polarization in tumor microenvironment and promotes pancreatic cancer progression and metastasis. International Journal of Molecular Sciences, 22(20), 11018.
Song, Y., et al. (2023). Circ_0018909 knockdown inhibits the development of pancreatic cancer via the miR-545-3p/FASN axis and reduces macrophage polarization to M2. Journal of Biochemical and Molecular Toxicology, 37(4), e23293.
Kurahara, H., et al. (2013). M2-polarized tumor-associated macrophage infiltration of regional lymph nodes is associated with nodal lymphangiogenesis and occult nodal involvement in pN0 pancreatic cancer. Pancreas, 42(1), 155–159.
Ma, X., et al. (2016). The pancreatic cancer secreted REG4 promotes macrophage polarization to M2 through EGFR/AKT/CREB pathway. Oncology Reports, 35(1), 189–196.
Geng, Y., et al. (2021). A notch-dependent inflammatory feedback circuit between macrophages and cancer cells regulates pancreatic cancer metastasis. Cancer Research, 81(1), 64–76.
Wu, J., et al. (2022). TNFSF9 promotes metastasis of pancreatic cancer by regulating M2 polarization of macrophages through Src/FAK/p-Akt/IL-1β signaling. International Immunopharmacology, 102, 108429.
Chen, Q., et al. (2019). Tumour cell-derived debris and IgG synergistically promote metastasis of pancreatic cancer by inducing inflammation via tumour-associated macrophages. British Journal of Cancer, 121(9), 786–795.
Gu, H., et al. (2022). NLRP3 activation in tumor-associated macrophages enhances lung metastasis of pancreatic ductal adenocarcinoma. Translational Lung Cancer Research, 11(5), 858–868.
Menen, R. S., et al. (2012). Tumor-educated macrophages promote tumor growth and peritoneal metastasis in an orthotopic nude mouse model of human pancreatic cancer. In Vivo, 26(4), 565–569.
Penny, H. L., et al. (2016). Warburg metabolism in tumor-conditioned macrophages promotes metastasis in human pancreatic ductal adenocarcinoma. Oncoimmunology, 5(8), e1191731.
Liu, C. Y., et al. (2013). M2-polarized tumor-associated macrophages promoted epithelial-mesenchymal transition in pancreatic cancer cells, partially through TLR4/IL-10 signaling pathway. Laboratory Investigation, 93(7), 844–854.
Shi, T., Li, X., Zheng, J., et al. (2023). Increased SPRY1 expression activates NF-κB signaling and promotes pancreatic cancer progression by recruiting neutrophils and macrophages through CXCL12-CXCR4 axis. Cellular Oncology. https://doi.org/10.1007/s13402-023-00791-z
Niu, N., et al. (2023). Tumor Cell-Intrinsic SETD2 Deficiency reprograms neutrophils to foster immune escape in pancreatic tumorigenesis. Advanced Science(Weinh), 10(2), e2202937.
Papayannopoulos, V. (2018). Neutrophil extracellular traps in immunity and disease. Nature Reviews. Immunology, 18(2), 134–147.
Cedervall, J., & Olsson, A. K. (2016). Immunity gone astray - NETs in cancer. Trends Cancer, 2(11), 633–634.
Deng, J., et al. (2021). DDR1-induced neutrophil extracellular traps drive pancreatic cancer metastasis. JCI Insight, 6(17), e146133.
Kajioka, H., et al. (2021). Targeting neutrophil extracellular traps with thrombomodulin prevents pancreatic cancer metastasis. Cancer Letters, 497, 1–13.
Miller-Ocuin, J. L., et al. (2019). DNA released from neutrophil extracellular traps (NETs) activates pancreatic stellate cells and enhances pancreatic tumor growth. Oncoimmunology, 8(9), e1605822.
Takesue, S., et al. (2020). Neutrophil extracellular traps promote liver micrometastasis in pancreatic ductal adenocarcinoma via the activation of cancer-associated fibroblasts. International Journal of Oncology, 56(2), 596–605.
Bellomo, G., et al. (2022). Chemotherapy-induced infiltration of neutrophils promotes pancreatic cancer metastasis via Gas6/AXL signalling axis. Gut, 71(11), 2284–2299.
Lianyuan, T., et al. (2020). Tumor associated neutrophils promote the metastasis of pancreatic ductal adenocarcinoma. Cancer Biology & Therapy, 21(10), 937–945.
Li, Y. X., et al. (2022). ACLP promotes activation of cancer-associated fibroblasts and tumor metastasis via ACLP-PPARγ-ACLP feedback loop in pancreatic cancer. Cancer Letters, 544, 215802.
Cai, W., et al. (2021). PERK-eIF2α-ERK1/2 axis drives mesenchymal-endothelial transition of cancer-associated fibroblasts in pancreatic cancer. Cancer Letters, 515, 86–95.
Zaghdoudi, S., et al. (2020). FAK activity in cancer-associated fibroblasts is a prognostic marker and a druggable key metastatic player in pancreatic cancer. EMBO Molecular Medicine, 12(11), e12010.
Chu, X., Yang, Y., & Tian, X. (2022). Crosstalk between Pancreatic Cancer Cells and Cancer-Associated Fibroblasts in the Tumor Microenvironment Mediated by Exosomal MicroRNAs. International Journal of Molecular Sciences, 23(17), 9512.
Stylianou, A., Gkretsi, V., & Stylianopoulos, T. (2018). Transforming growth factor-β modulates pancreatic cancer associated fibroblasts cell shape, stiffness and invasion. Biochimica et Biophysica Acta - General Subjects, 1862(7), 1537–1546.
Goicoechea, S. M., et al. (2014). Palladin promotes invasion of pancreatic cancer cells by enhancing invadopodia formation in cancer-associated fibroblasts. Oncogene, 33(10), 1265–1273.
Shan, T., et al. (2017). Cancer-associated fibroblasts enhance pancreatic cancer cell invasion by remodeling the metabolic conversion mechanism. Oncology Reports, 37(4), 1971–1979.
Wang, Q., et al. (2017). Curcumin suppresses epithelial-to-mesenchymal transition and metastasis of pancreatic cancer cells by inhibiting cancer-associated fibroblasts. American Journal of Cancer Research, 7(1), 125–133.
Chen, X., et al. (2021). CCL26 is upregulated by nab-paclitaxel in pancreatic cancer-associated fibroblasts and promotes PDAC invasiveness through activation of the PI3K/AKT/mTOR pathway. Acta Biochimica et Biophysica Sinica Shanghai, 53(5), 612–619.
Liu, J., et al. (2020). Cell metabolomics reveals berberine-inhibited pancreatic cancer cell viability and metastasis by regulating citrate metabolism. Journal of Proteome Research, 19(9), 3825–3836.
Tian, W., et al. (2022). Berberine suppresses lung metastasis of cancer via inhibiting endothelial transforming growth factor beta receptor 1. Frontiers in Pharmacology, 13, 917827.
Kim, S. O., & Kim, M. R. (2013). (-)-Epigallocatechin 3-gallate inhibits invasion by inducing the expression of Raf kinase inhibitor protein in AsPC-1 human pancreatic adenocarcinoma cells through the modulation of histone deacetylase activity. International Journal of Oncology, 42(1), 349–358.
Arya, G., Das, M., & Sahoo, S. K. (2018). Evaluation of curcumin loaded chitosan/PEG blended PLGA nanoparticles for effective treatment of pancreatic cancer. Biomedicine & Pharmacotherapy, 102, 555–566.
Hoca, M., et al. (2020). The effect of resveratrol and quercetin on epithelial-mesenchymal transition in pancreatic cancer stem cell. Nutrition and Cancer, 72(7), 1231–1242.
Li, W., et al. (2013). Resveratrol inhibits the epithelial-mesenchymal transition of pancreatic cancer cells via suppression of the PI-3K/Akt/NF-κB pathway. Current Medicinal Chemistry, 20(33), 4185–4194.
Yu, D., et al. (2017). Quercetin inhibits epithelial-mesenchymal transition, decreases invasiveness and metastasis, and reverses IL-6 induced epithelial-mesenchymal transition, expression of MMP by inhibiting STAT3 signaling in pancreatic cancer cells. Oncotargets and Therapy, 10, 4719–4729.
Guo, Y., et al. (2021). Quercetin suppresses pancreatic ductal adenocarcinoma progression via inhibition of SHH and TGF-β/Smad signaling pathways. Cell Biology and Toxicology, 37(3), 479–496.
Srivastava, R. K., et al. (2011). Sulforaphane synergizes with quercetin to inhibit self-renewal capacity of pancreatic cancer stem cells. Frontiers in Bioscience (Elite Edition), 3(2), 515–528.
Lee, J., et al. (2016). Quercetin-3-O-glucoside suppresses pancreatic cancer cell migration induced by tumor-deteriorated growth factors in vitro. Oncology Reports, 35(4), 2473–2479.
Wang, C., et al. (2021). Metformin inhibits pancreatic cancer metastasis caused by SMAD4 deficiency and consequent HNF4G upregulation. Protein & Cell, 12(2), 128–144.
Yu, X., et al. (2020). Propofol affects the growth and metastasis of pancreatic cancer via ADAM8. Pharmacological Reports, 72(2), 418–426.
Polireddy, K., et al. (2017). High Dose Parenteral Ascorbate Inhibited Pancreatic Cancer Growth and Metastasis: Mechanisms and a Phase I/IIa study. Scientific Reports, 7(1), 17188.
Yamada, S., et al. (2020). Phase I/II study of adding intraperitoneal paclitaxel in patients with pancreatic cancer and peritoneal metastasis. The British Journal of Surgery, 107(13), 1811–1817.
Takahara, N., et al. (2021). A phase I study of intraperitoneal paclitaxel combined with gemcitabine plus nab-paclitaxel for pancreatic cancer with peritoneal metastasis. Investigational New Drugs, 39(1), 175–181.
Nishiyama, Y., et al. (2005). Contribution of whole body FDG-PET to the detection of distant metastasis in pancreatic cancer. Annals of Nuclear Medicine, 19(6), 491–497.
Ishikawa, T., et al. (2007). Angiotensin-II administration is useful for the detection of liver metastasis from pancreatic cancer during pharmacoangiographic computed tomography. World Journal of Gastroenterology, 13(22), 3080–3083.
Hayashibe, A., et al. (2007). Clinical results on intra-arterial adjuvant chemotherapy for prevention of liver metastasis following curative resection of pancreatic cancer. Annals of Surgical Oncology, 14(1), 190–194.
Wobser, M., et al. (2006). Complete remission of liver metastasis of pancreatic cancer under vaccination with a HLA-A2 restricted peptide derived from the universal tumor antigen survivin. Cancer Immunology, Immunotherapy, 55(10), 1294–1298.
Acknowledgements
The biorender software (Biorender.com) was used to prepare the figures for this paper.
Author information
Authors and Affiliations
Contributions
Zhenli Guo, Milad Ashrafizadeh, Wei Zhang, and Rongjun Zou wrote the first draft of the manuscript. Gautam Sethi and Xianbin Zhang developed the concept, collected papers, and performed English Editing.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Informed consent
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, Z., Ashrafizadeh, M., Zhang, W. et al. Molecular profile of metastasis, cell plasticity and EMT in pancreatic cancer: a pre-clinical connection to aggressiveness and drug resistance. Cancer Metastasis Rev 43, 29–53 (2024). https://doi.org/10.1007/s10555-023-10125-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-023-10125-y