Abstract
Thoracic aortic disease, including thoracic aortic aneurysm (TAA), is frequently seen in patients with bicuspid aortic valve (BAV). We hypothesized that BAV morphotype would be associated with aortic aneurysm phenotypes but that other patient variables would significantly modify this relationship. 829 patients between 18 and 90 years with BAV and available raw imaging of the aortic valve and the ascending aorta to its mid-portion prior to aortic valve and aortic surgery were examined. The sinuses of Valsalva and proximal ascending aorta were measured from 2-dimensional co-planar echocardiographic images. We observed strong associations between patient habitus and raw and normalized dimensions of the aortic root and ascending aorta. Patients with R–L morphotype presented at an older age with larger aortic root but similar ascending aortic dimensions. After accounting for patient morphometric characteristics and severity of aortic valve disease, patients with R–L valve morphotype were marginally more likely to have an aortic root aneurysm (86% vs. 78%; P = 0.043), defined as aortic root dimension Z score ≥3. We observed only small differences in aortic dimensions between BAV morphotypes, that are eclipsed by variation in patient habitus. We interpret these findings to mean that BAV patients will not likely benefit from therapies based on aortic valve morphotype. Rather, we propose that all BAV patients should undergo longitudinal follow-up, independent of valve morphotype. Guidelines for aortic surgery based upon dimensions alone may be improved by considering patient characteristics such as age, body size and other characteristics.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Bicuspid aortic valve (BAV) is the most common congenital cardiac valve abnormality, with an incidence of 0.6–1.5% in the general population and is seen more frequent in males and Caucasians [1]. Thoracic aortic disease, including thoracic aortic aneurysm (TAA), is more frequently seen in patients with BAV than the general population [1, 2]. Prior literature has variably reported association between BAV morphotype has been associated with TAA phenotype; notably, right–left (R–L) coronary cusp fusion is associated with aortic root aneurysm and right–non-coronary (R–NC) cusp fusion has similarly been associated with ascending aortic aneurysm [3].
The etiology of these reported associations is not yet apparent, but they may be of genetic or biomechanical causes [4]. There is a common embryological origin of the aortic valve and ascending aorta, developing from neural crest cells early in embryogenesis [5]. It might be reasonable to believe that a genetic defect specifically altering the function and development of neural crest cells could result in defects of both the aortic valve and the media of the ascending aorta. Additionally, there is evidence for increased shear stress upon the aortic root and ascending aorta caused by abnormal flow direction through the bicuspid aortic valve [6], even in the absence of aortic leaflet thickening, calcification and stenosis [7]. Differences in direction of flow in the ascending aorta have been observed between R–NC and R–L morphotypes of BAV, thus formulating a mechanistic hypothesis for differences in aortic dimensions between BAV morphotypes [8].
We hypothesized that BAV morphotype would be associated with aortic aneurysm phenotype in an BAV cohort, but that other patient variables would significantly modify this relationship and have important clinical implications to care of the BAV patient. To examine this hypothesis, we used a cohort of BAV patients predominantly ascertained by presentation for cardiac surgery.
Methods
Patients
After local Institutional Review Board approval, patients with BAV and with available raw imaging of the aortic valve and the ascending aorta to its mid-portion prior to aortic valve and aortic surgery were identified from administrative records of Partners Healthcare (Boston, MA). For patients with an ICD-9 code 746.4, we extracted imaging, operative and discharge records and performed text searches for bicuspid aortic valve. We excluded patients with unavailable, inadequate or disputed imaging recorded prior to any reported cardiac surgery, patients with tricuspid aortic valves on arbitrated imaging, syndromic aortic disease or prior coarctation repair, a history of rheumatic or endocarditic valvular heart disease, patients with bicuspid NC–L fusion because of the rarity of this morphotype, and those under 18 or over 90 years of age, yielding 829 patients for analysis.
Age, height, weight, the severity of aortic stenosis and incompetence, and other clinical variables were harvested from clinical and surgical reports closest to, but not >180 days prior to, the date of aortic or aortic valve surgery. If a patient did not have aortic surgery the information was taken from the most recent clinical reports and imaging available.
Valve classification
Valve morphotype was classified as L–R (fusion between left and right leaflets), R–NC (fusion between right and non-coronary leaflets) (Fig. 1), or not bicuspid (tricuspid valves with no leaflet fusion) [3]. BAV was established by visualization of the aortic valve when seen in an axial view of the most recent transesophageal or transthoracic echocardiogram prior to surgery. Surgical notes and previous echocardiograms were used, when available, to confirm BAV diagnosis only. Identification of raphe was inconsistent among the cohort so this parameter was not included. All echocardiograms were performed and analyzed by trained echocardiography technicians and interpreted by a board-certified cardiologist. Two hundred and fifty randomly chosen patients and patients with indeterminate imaging were arbitrated twice to determine accuracy of the initial echocardiography reporting. Aortic measurements of 68 patients where the dimension differed by more than 3 mm were re-arbitrated by a third individual. Patients were discarded from analysis if imaging remained indeterminate even after third arbitration. Only patients in which in both valve morphotype and aortic measurements were confidently identified were included in analysis. The severity of aortic stenosis and incompetence was determined by echocardiographic criteria [9, 10] or if unavailable, by cardiac catheterization.
Sievers’ classification [11] of the surgical view of the bicuspid aortic valve. Type 0 has no visible raphe and can occur as a vertical or horizontal oriented commissure. Type 1 is the most common form of BAV and can occur as fusion of two of any three of the commissures, but most commonly a left–right (L–R) fusion or right–non-coronary (R–N) fusion. Type 2 is a reported bi-commissural fusion, which can be difficult to differentiate from Type 1 in echocardiographic imaging and during surgical inspection of the severely calcific aortic valve
Aortic dilation classification
The sinuses of Valsalva and proximal ascending aorta were measured from 2-dimensional images from inner edge to inner edge in the parasternal long-axis view at end-diastole for TTE images and from the mid-esophageal long axis for TEE images (Fig. 2) [12]. Because we observed strong univariate associations between older age, male gender, taller height, heavier weight and greater body mass index with increased dimensions of the aortic root and ascending aorta in patients with BAV, we performed additional analyses using normalized aortic dimensions to Z scores [13].
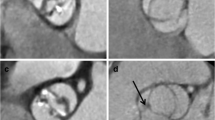
Three trans-esophageal images of the bicuspid aortic valve. The upper panel shows a long axis view of the left ventricular outflow tract, the bicuspid aortic valve and the dilated ascending aorta. The middle panel shows a short axis view of a type I bicuspid aortic valve during diastole. Only in systole (lower panel) is it apparent that the valve is bicuspid with complete fusion of the left and right coronary cusps
Z scores of aortic dimension were calculated using separate formulae for aortic root and tubular ascending aorta, and population norms derived by Campens et al. that take into account age, gender and body surface area [13]. The aorta was defined as dilated when Z score ≥1.96, and aneurysmal when Z score ≥3 [14]. Aortic phenotype was classified as root (maximal dimension at the sinuses of Valsalva), or ascending (maximum dimension at the mid-ascending aorta) dilation, both root and ascending aortic dilation, or being without dilation [15]. We did not use aortic shape classifications [16, 17] to describe the joint dimensions of the aortic root or ascending aorta as they lack concordance [15] and are potentially prone to observer variation.
Statistical analysis
Normally distributed continuous variables were summarized as mean and standard deviation and comparisons between groups were made by means of Student’s t test. Categorical variables were summarized with percentages and inter-group differences analyzed using Chi square statistics. Multivariate linear and logistic regression models were developed to find independent predictors of aortic dimensions and categorical aortic dilatation (Z score >1.96 and 3.0). Multivariable regression models were developed by forward stepwise modeling criteria of F probability <0.05 for entry and >0.1 for removal, while forcing age, gender, BSA, valve morphotype and the presence of aortic stenosis and incompetence into the model. All analyses were performed using SAS or JMP (SAS Institute, Cary, NC, version 9.1) with a two-tailed significance level set at 0.05.
Sensitivity analysis was performed to compare the relative importance of BAV morphotype to patient habitus upon aortic dimensions. This was performed by comparing the relative effect of valve morphotype to the 20th to 80th percentiles of age (41–66 years), BSA (1.77–2.15 m2) and gender (female-male) that we observed in developed multivariable models of aortic dimension, while holding other covariates constant, in this cohort.
Results
Cohort characteristics
From the source population of 2007 BAV patients we identified images for 927 patients where the valve fusion could be definitively identified on available imaging. Of these, 829 patients had sufficient imaging to measure aortic dimensions. The characteristics of the cohort are described in Table 1. Of note, the cohort was relatively young (53 ± 14 years) and predominantly had R–L valve morphotype. Patient characteristics between the two morphotypes were similar, however those with R–L morphotype were older and had a higher incidence of hyperlipidemia. Moderate or greater aortic stenosis or aortic incompetence was observed in 20 and 12% of patients, respectively. No patient had an unrepaired coarctation of the aorta. The majority of patients (56%) had surgery for either aortic valve repair or replacement, or surgery of the aortic root, ascending aorta or the aortic arch. The remainder had not yet had surgery. There was no difference in the frequency of surgery between the two aortic morphotypes.
Predictors of aortic dilatation
Patients with R–L morphotype presented at an older age with larger aortic root (Table 1). There were strong univariate associations between older age, male gender, taller height, heavier weight and greater body mass index with increased dimensions of the aortic root and ascending aorta in patients with BAV. The dimension of the ascending aorta at presentation did not differ between BAV morphotypes, even after adjustment for age, gender and BSA to a Z score. However, mean aortic root dimensions differed by 2 mm between BAV morphotype at the time of presentation and remained significantly different after translation to Z score. After adjustment for age, gender, BSA and the presence of moderate or severe aortic valve disease, R–L BAV morphotype was associated with small (<1 mm) increase in mean aortic root size, but not with mean ascending aorta size at presentation (Table 2). Patients with moderate or severe aortic stenosis had a slightly smaller aortic root, but similar ascending aortic dimensions while patients with moderate or severe aortic incompetence had slightly larger aortic root, but similar ascending aortic, dimensions (Table 2).
Patients with a dilated aortic root, defined as aortic root dimension Z score ≥1.96 independent of ascending aortic dimension, were younger, more likely to be male, have lower BSAs, have a decreased frequency of moderate or greater aortic stenosis, and increased frequency of moderate or greater aortic incompetence (Table 3). After accounting for these variables, there was a modest association of aortic root Z score with R–L valve morphotype (84% vs. 78%; P = 0.019). Even after normalization of aortic dimension to Z-score using an algorithm that makes use of age, gender and BSA [13], we observed persistence of age, gender and BSA in multivariable models of aortic dimensions (Tables 3, 4), implying that the utilized Z-score transformation does not fully account for these factors.
Patients with aortic root aneurysm, defined as aortic root dimension Z score ≥3, were similarly younger, more likely to be male, have lower BSAs, have a decreased frequency of moderate or greater aortic stenosis, and increased frequency of moderate or greater aortic incompetence (Table 3). After accounting for these variables, there was a marginal association of aortic root Z score with R–L valve morphotype (86% vs. 78%; P = 0.043).
Patients with ascending aortic dilatation, defined as ascending aortic dimension Z score ≥1.96 regardless of aortic root dimensions, were younger, but had similar frequency of other clinical variables (Table 4). Patients with ascending aortic aneurysm, defined as ascending aortic dimension Z score ≥3, were younger and more likely to be female. After accounting for these variables, there was a no association of the ascending aorta Z score ≥1.96 (77% vs. 80%; P = 0.56), or Z score ≥3 (77% vs. 80%; P = 0.41) with R–L valve morphotype (Table 4).
Sensitivity analysis examining the relative importance of BAV morphotype to age, gender and BSA using the multivariable models shown in Table 2. In the aortic root, the presence of male gender, an increase in age of 25 years, an increase in BSA of 0.38 m2, aortic stenosis, and statin use all had a greater effect upon aortic root dimension than bicuspid valve morphotype. In the ascending aorta, all measured variables had a greater effect upon aortic root dimension than bicuspid valve morphotype.
Discussion
The current study aimed to identify association between BAV morphotype and aortic dimensions and dilation. In doing so, we hoped to provide potential further insight into the cause of aortic dilation in patients with BAV and to identify patient parameters associated with aortic dilation. As previously described in other cohorts [3, 16, 18–23], we observed BAV patients with R–L phenotype have marginally greater aortic root dimensions than patients with R–NC morphotype, and may therefore be at greater risk of requiring surgery [24]. However, prior studies have not been in agreement with these findings [25–29]. Unlike previous cohorts, this study differed from prior studies by inclusion of older patients who had not yet undergone surgery, thus perhaps representing a cohort more representative of an overall referral population than previously described surgical cohorts. Similarly, studies making use of both CT and MRI were also unable to find an association between BAV morphotype and specific regional aortic dilatation, validating our findings across different imaging modalities [25, 30]. Thus we argue that controversy still remains on this issue and that using different parameters, namely age, gender, and BSA, to predict aortic outcomes may be more clinically useful than using BAV morphology.
Ascending aortic dimensions and risk of aortic dilation did not differ between the two morphotypes, in contrast to, and agreement with previous observations. Furthermore, the majority of individuals with a BAV are not aware of their aortic valve anatomy until they have symptoms of aortic valve disease or aortic aneurysm. Thus the observations we, and other studies, have made regarding associations between aortic valve morphotype and aortic dimensions may predominantly reflect the development of symptomatic disease rather than the rate of aortic dilation associated with aortic valve morphotype.
Previous studies have observed differences in aortic dimensions and rate of aortic dilation [31] between BAV morphotypes and suggested that BAV morphotype may be of use in risk stratification of BAV patients. However, we observed small differences in aortic dimensions between morphotypes in our study, that most likely do not have a major influence on incidence of aortic complications or determining timing of aortic surgery in BAV patients.
Wide inter-individual variability in aortic growth rates have been observed [27, 32] that are likely of greater clinical significance than a population averages for a specific BAV morphotype. Thus, we propose that longitudinal follow-up of all individuals with BAV will have greater utility, rather than increased intensity of follow-up of any particular BAV morphotype.
There is a lack of consensus on whether and how to adjust aortic dimensions for age, gender and body size when making clinical decisions. We observed strong univariate associations between older age, male gender, taller height, heavier weight, greater body mass index and surface area with increased dimensions of the aortic root and ascending aorta in patients with BAV, independent of BAV morphotype. These observations strongly support accounting for the patient’s body habitus when defining normal and abnormal aortic dimensions.
Currently, expert Guidelines use absolute aortic dimensions without normalization for patient characteristics and do not account for the site of the maximal aortic dimension [33]. Our observations that age, gender and BSA modify the association between BAV morphotype and aortic root dimensions can be interpreted as indicating that use of aortic root Z scores may be advantageous when assessing the dilated aortic root. We did not observe the same effect for the ascending aorta. Based on these findings, we suggest that Guidelines make use of morphometric parameters such as BSA, age and gender in determining a cutoff value for surgery. We observed persistence of these three variables in multivariate analyses of aortic dimensions, demonstrating that the methods of normalization of aortic dimensions to Z score are not optimized [13] and that large-scale studies to improve Z score normalization methods are required. Similarly, we believe that Guidelines should take into account the site of aortic dimensions, thus allowing for normal differences in aortic dimension between the aortic sinuses and the tubular ascending aorta.
Limitations
The current study is a retrospective study without longitudinal follow-up. The rate of aortic expansion has important prognostic significance that was not examined in this population. The majority of patients examined in this study were imaged prior to planned aortic valve or aortic surgery and thus do not represent the full spectrum of the BAV population. Some studies have classified BAV into five or more different categories based on the presence (or absence of raphes), however, because many of our images were from just prior to surgery, severe calcification very often made it difficult for us to determine the presence of a raphe. Surgical descriptions for the presence of a raphe were not provided in a consistent manner. Thus we elected to not include stratification based on the presence of a raphe. We were comfortable with the surgical description of valve orientation as this is usually discernable and consistent and thus we categorized the valve morphotype in a way we believed to be more clinically beneficial. These limitations potentially make the time and cause of presentation an important cause of observed differences in aortic size. In addition, we are unable to provide information in this cohort of the value of blood pressure control or other drug therapies upon aortic dimensions or rate of growth. Two issues that we cannot resolve in this study are a lack of standardization of imaging dimensions between pediatric and adult patients and the differences in measurement of the aortic dimensions between different imaging modalities. For the majority of our patients, only echocardiograms were available so we were unable to verify our techniques through the use of other imaging techniques. As a result, this study only focuses on the use of measurements derived from echocardiograms in determining z-scores and we are unable to make any assessment on analysis of z-scores based on measurements from other imaging studies. The same goes for determining BAV morphotype.
Conclusions
The current study shows only small differences in aortic dimensions between BAV morphotypes, that are eclipsed by patient characteristics such as age, gender, height and weight. We interpret these findings to mean that BAV patients will not likely benefit from risk stratification or therapies based on aortic valve morphotype. Rather, we propose that all BAV patients should undergo longitudinal follow-up, independent of valve morphotype. Guidelines for aortic surgery based upon dimensions alone may be improved by considering patient characteristics such as age, body size and other characteristics.
Abbreviations
- IE:
-
Infective endocarditis
- BAV:
-
Bicuspid aortic valve
- TAV:
-
Tricuspid aortic valve
References
Michelena HI, Prakash SK, Della Corte A, Bissell MM, Anavekar N, Mathieu P, Bosse Y, Limongelli G, Bossone E, Benson DW, Lancellotti P, Isselbacher EM, Enriquez-Sarano M, Sundt TM 3rd, Pibarot P, Evangelista A, Milewicz DM, Body SC, Investigators BA (2014) Bicuspid aortic valve: identifying knowledge gaps and rising to the challenge from the International bicuspid aortic valve consortium (BAVCon). Circulation 129(25):2691–2704. doi:10.1161/CIRCULATIONAHA.113.007851
Nkomo VT, Enriquez-Sarano M, Ammash NM, Melton LJ 3rd, Bailey KR, Desjardins V, Horn RA, Tajik AJ (2003) Bicuspid aortic valve associated with aortic dilatation: a community-based study. Arterioscler Thromb Vasc Biol 23(2):351–356
Della Corte A, Bancone C, Dialetto G, Covino FE, Manduca S, D’Oria V, Petrone G, De Feo M, Nappi G (2014) Towards an individualized approach to bicuspid aortopathy: different valve types have unique determinants of aortic dilatation. Eur J Cardiothorac Surg 45(4):e118–124, (discussion e124). doi:10.1093/ejcts/ezt601
Prakash SK, Bosse Y, Muehlschlegel JD, Michelena HI, Limongelli G, Della Corte A, Pluchinotta FR, Russo MG, Evangelista A, Benson DW, Body SC, Milewicz DM, Investigators B (2014) A roadmap to investigate the genetic basis of bicuspid aortic valve and its complications: insights from the International BAVCon (Bicuspid Aortic Valve Consortium). J Am Coll Cardiol 64(8):832–839. doi:10.1016/j.jacc.2014.04.073
Martin P, Kloesel B, Norris R, Lindsay M, Milan D, Body S (2015) Embryonic development of the bicuspid aortic valve. J Cardiovasc Dev Dis 2(4):248–272
Conti CA, Della Corte A, Votta E, Del Viscovo L, Bancone C, De Santo LS, Redaelli A (2010) Biomechanical implications of the congenital bicuspid aortic valve: a finite element study of aortic root function from in vivo data. J Thorac Cardiovasc Surg 140(4):890–896. doi:10.1016/j.jtcvs.2010.01.016
Bissell MM, Hess AT, Biasiolli L, Glaze SJ, Loudon M, Pitcher A, Davis A, Prendergast B, Markl M, Barker AJ, Neubauer S, Myerson SG (2014) Response to letter regarding article, “Aortic dilation in bicuspid aortic valve disease: flow pattern is a major contributor and differs with valve fusion type”. Circ Cardiovasc Imaging 7(1):214. doi:10.1161/CIRCIMAGING.113.001497
Bissell MM, Hess AT, Biasiolli L, Glaze SJ, Loudon M, Pitcher A, Davis A, Prendergast B, Markl M, Barker AJ, Neubauer S, Myerson SG (2013) Aortic dilation in bicuspid aortic valve disease: flow pattern is a major contributor and differs with valve fusion type. Circ Cardiovasc Imaging 6(4):499–507. doi:10.1161/CIRCIMAGING.113.000528
Baumgartner H, Hung J, Bermejo J, Chambers JB, Evangelista A, Griffin BP, Iung B, Otto CM, Pellikka PA, Quinones M, American Society of E, European Association of E (2009) Echocardiographic assessment of valve stenosis: EAE/ASE recommendations for clinical practice. J Am Soc Echocardiogr 22(1):1–23, (quiz 101–102). doi:10.1016/j.echo.2008.11.029
Zoghbi WA, Enriquez-Sarano M, Foster E, Grayburn PA, Kraft CD, Levine RA, Nihoyannopoulos P, Otto CM, Quinones MA, Rakowski H, Stewart WJ, Waggoner A, Weissman NJ, American Society of E (2003) Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J Am Soc Echocardiogr 16(7):777–802. doi:10.1016/S0894-7317(03)00335-3
Sievers HH, Schmidtke C (2007) A classification system for the bicuspid aortic valve from 304 surgical specimens. J Thorac Cardiovasc Surg 133(5):1226–1233. doi:10.1016/j.jtcvs.2007.01.039
Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, Picard MH, Roman MJ, Seward J, Shanewise JS, Solomon SD, Spencer KT, Sutton MS, Stewart WJ, Chamber Quantification Writing G, American Society of Echocardiography’s G, Standards C, European Association of E (2005) Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr 18(12):1440–1463. doi:10.1016/j.echo.2005.10.005
Campens L, Demulier L, De Groote K, Vandekerckhove K, De Wolf D, Roman MJ, Devereux RB, De Paepe A, De Backer J (2014) Reference values for echocardiographic assessment of the diameter of the aortic root and ascending aorta spanning all age categories. Am J Cardiol 114 (6):914–920. doi:10.1016/j.amjcard.2014.06.024
Legget ME, Unger TA, O’Sullivan CK, Zwink TR, Bennett RL, Byers PH, Otto CM (1996) Aortic root complications in Marfan’s syndrome: identification of a lower risk group. Heart 75(4):389–395
Della Corte A, Bancone C, Dialetto G, Covino FE, Manduca S, Montibello MV, De Feo M, Buonocore M, Nappi G (2014) The ascending aorta with bicuspid aortic valve: a phenotypic classification with potential prognostic significance. Eur J Cardiothorac Surg 46(2):240–247, (discussion 247). doi:10.1093/ejcts/ezt621
Schaefer BM, Lewin MB, Stout KK, Gill E, Prueitt A, Byers PH, Otto CM (2008) The bicuspid aortic valve: an integrated phenotypic classification of leaflet morphology and aortic root shape. Heart 94(12):1634–1638. doi:10.1136/hrt.2007.132092
Park CB, Greason KL, Suri RM, Michelena HI, Schaff HV, Sundt TM 3rd (2011) Fate of nonreplaced sinuses of Valsalva in bicuspid aortic valve disease. J Thorac Cardiovasc Surg 142(2):278–284. doi:10.1016/j.jtcvs.2010.08.055
Khoo C, Cheung C, Jue J (2013) Patterns of aortic dilatation in bicuspid aortic valve-associated aortopathy. J Am Soc Echocardiogr 26(6):600–605. doi:10.1016/j.echo.2013.02.017
Kang JW, Song HG, Yang DH, Baek S, Kim DH, Song JM, Kang DH, Lim TH, Song JK (2013) Association between bicuspid aortic valve phenotype and patterns of valvular dysfunction and bicuspid aortopathy: comprehensive evaluation using MDCT and echocardiography. JACC Cardiovasc Imaging 6(2):150–161. doi:10.1016/j.jcmg.2012.11.007
Fernandes S, Khairy P, Graham DA, Colan SD, Galvin TC, Sanders SP, Singh MN, Bhatt A, Lacro RV (2012) Bicuspid aortic valve and associated aortic dilation in the young. Heart 98(13):1014–1019. doi:10.1136/heartjnl-2012-301773
Jassal DS, Bhagirath KM, Tam JW, Sochowski RA, Dumesnil JG, Giannoccaro PJ, Jue J, Pandey AS, Joyner CD, Teo KK, Chan KL (2010) Association of Bicuspid aortic valve morphology and aortic root dimensions: a substudy of the aortic stenosis progression observation measuring effects of rosuvastatin (ASTRONOMER) study. Echocardiography 27(2):174–179. doi:10.1111/j.1540-8175.2009.00993.x
Miskowiec DL, Lipiec P, Kasprzak JD (2016) Bicuspid aortic valve morphology and its association with aortic diameter: an echocardiographic study. Kardiol Pol 74(2):151–158. doi:10.5603/KP.a2015.0134
Ruzmetov M, Shah JJ, Fortuna RS, Welke KF (2015) The association between aortic valve leaflet morphology and patterns of aortic dilation in patients with bicuspid aortic valves. Ann Thorac Surg 99(6):2101–2107, (discussion 2107–2108). doi:10.1016/j.athoracsur.2015.02.036
Prakash SK, Bosse Y, Muehlschlegel JD, Michelena HI, Limongelli G, Della Corte A, Pluchinotta FR, Russo MG, Evangelista A, Benson DW, Body SC, Milewicz DM, Investigators BA (2014) A roadmap to investigate the genetic basis of bicuspid aortic valve and its complications: insights from the International BAVCon (Bicuspid Aortic Valve Consortium). J Am Coll Cardiol 64(8):832–839. doi:10.1016/j.jacc.2014.04.073
Buchner S, Hulsmann M, Poschenrieder F, Hamer OW, Fellner C, Kobuch R, Feuerbach S, Riegger GA, Djavidani B, Luchner A, Debl K (2010) Variable phenotypes of bicuspid aortic valve disease: classification by cardiovascular magnetic resonance. Heart 96(15):1233–1240. doi:10.1136/hrt.2009.186254
Thanassoulis G, Yip JW, Filion K, Jamorski M, Webb G, Siu SC, Therrien J (2008) Retrospective study to identify predictors of the presence and rapid progression of aortic dilatation in patients with bicuspid aortic valves. Nat Clini Pract Cardiovasc Med 5(12):821–828. doi:10.1038/ncpcardio1369
Detaint D, Michelena HI, Nkomo VT, Vahanian A, Jondeau G, Sarano ME (2014) Aortic dilatation patterns and rates in adults with bicuspid aortic valves: a comparative study with Marfan syndrome and degenerative aortopathy. Heart 100(2):126–134. doi:10.1136/heartjnl-2013-304920
Kinoshita T, Naito S, Suzuki T, Asai T (2016) Valve phenotype and risk factors of aortic dilatation after aortic valve replacement in Japanese patients with bicuspid aortic valve. Circul J 80(6):1356–1361. doi:10.1253/circj.CJ-15-1378
Novaro GM, Tiong IY, Pearce GL, Grimm RA, Smedira N, Griffin BP (2003) Features and predictors of ascending aortic dilatation in association with a congenital bicuspid aortic valve. Am J Cardiol 92(1):99–101
Fazel SS, Mallidi HR, Lee RS, Sheehan MP, Liang D, Fleischman D, Herfkens R, Mitchell RS, Miller DC (2008) The aortopathy of bicuspid aortic valve disease has distinctive patterns and usually involves the transverse aortic arch. J Thorac Cardiovasc Surg 135(4):901–907. doi:10.1016/j.jtcvs.2008.01.022
Page M, Mongeon FP, Stevens LM, Souliere V, Khairy P, El-Hamamsy I (2014) Aortic dilation rates in patients with biscuspid aortic valve: correlations with cusp fusion phenotype. J Heart Valve Dis 23(4):450–457
Charitos EI, Stierle U, Petersen M, Mohamed SA, Hanke T, Schmidtke C, Klotz S, Sievers HH (2014) The fate of the bicuspid valve aortopathy after aortic valve replacement. Eur J Cardiothorac Surg 45(5):e128–e135. doi:10.1093/ejcts/ezt666
Accf/Aha/Aats/Acr/Asa/Sca/Scai/Sir/Sts/Svm Guidelines For The Diagnosis Management Of Patients With Thoracic Aortic Disease Representative M, Hiratzka LF, Creager MA, Isselbacher EM, Svensson LG, Members* AAGftMoPWVHDR, Nishimura RA, Bonow RO, Guyton RA, Sundt TM 3rd, ACC/AHA Task Force M, Halperin JL, Levine GN, Anderson JL, Albert NM, Al-Khatib SM, Birtcher KK, Bozkurt B, Brindis RG, Cigarroa JE, Curtis LH, Fleisher LA, Gentile F, Gidding S, Hlatky MA, Ikonomidis J, Joglar J, Kovacs RJ, Ohman EM, Pressler SJ, Sellke FW, Shen WK, Wijeysundera DN (2016) Surgery for aortic dilatation in patients with bicuspid aortic valves: a statement of clarification from the American college of cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 133(7):680–686. doi:10.1161/CIR.0000000000000331
Funding
This work was funded by NIH Grants R01HL114823 (SCB) and R01HL118266 (JDM).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The Institutional Review Board determined that individual patient consent was not required.
Appendix 1: List of BAVCon sites
Appendix 1: List of BAVCon sites
U Penn | Rita Milewski | milewskr@uphs.upenn.edu |
Uruguay | Victor Dayan | Victor_dayan@hotmail.com |
GenTAC | Barbara L. Kroner | byk@rti.org |
Harvard | Simon Body | sbody@partners.org |
Laval | Yohan Bossé | Yohan.Bosse@criucpq.ulaval.ca |
Mayo | Hector I. Michelena | michelena.hector@mayo.edu |
Michigan | Bo Yang | boya@med.umich.edu |
Monaldi | Giuseppe Limongelli | limongelligiuseppe@libero.it |
Oxford | Malenka Bissell | malenka.bissell@cardiov.ox.ac.uk |
Salerno (Naples) | Eduardo Bossone | ebossone@hotmail.com |
San Donato (Milan) | Francesca Pluchinotta | francesca.pluchinotta@grupposandonato.it |
Second University of Naples | Alessandro Della Corte | aledellacorte@libero.it |
Texas | Dianna M. Milewicz | Dianna.M.Milewicz@uth.tmc.edu |
Tufts | Gordon Huggins | ghuggins@tuftsmedicalcenter.org |
Vall d’Hebron (Barcelona) | Arturo Evangelista | arturevangelistamasip@gmail.com |
Vanderbilt | Joshua C Denny | josh.denny@vanderbilt.edu |
Rights and permissions
About this article
Cite this article
Habchi, K.M., Ashikhmina, E., Vieira, V.M. et al. Association between bicuspid aortic valve morphotype and regional dilatation of the aortic root and trunk. Int J Cardiovasc Imaging 33, 341–349 (2017). https://doi.org/10.1007/s10554-016-1016-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-016-1016-8