Abstract
Purpose
Anthracyclines are frequently used in adjuvant treatment for early-stage breast cancer (ESBC). The purpose of this study was to evaluate cardiotoxic effects in the first five years after treatment with different anthracycline-based regimens.
Methods
CCTG MA.21 (NCT000142) was a phase III trial in ESBC that compared cyclophosphamide (75 mg/m2) orally for 14 days, epirubicin (60 mg/m2) and fluorouracil, IV days one and eight (CEF) for six cycles; dose-dense epirubicin (120 mg/m2) and cyclophosphamide, IV every 2 weeks for six cycles with concurrent G-CSF then paclitaxel every 2 weeks for four cycles (ddEC/T); doxorubicin (60 mg/m2) and cyclophosphamide (600 mg/m2) every 3 weeks for four cycles then four cycles q3 weekly paclitaxel (175 mg/m2) (AC/T). Endpoints: LVEF decline; LV function changes (heart failure), or Grade 3–4 cardiac ischemia/infarction. A competing risk analysis was performed with endpoints of cardiotoxicity or recurrence in first 5 years after completion of chemotherapy.
Results
2104 women were randomized. Compliance with cardiac LVEF assessments was 70% at 5 years in all arms. The 5-year cumulative risks of any cardiac event for CEF, ddECT, and AC/T were 22.3% (95%CI 18.9 to 25.7), 14.2% (95%CI 11.0 to 17.3), and 8.1% (95%CI 5.8 to 10.4), respectively, p < 0.0001. At 5 years, women in the ddEC/T and AC/T group had significantly lower risk of cardiotoxicity than those given CEF (HR 0.599 and 0.371, respectively). Most events were asymptomatic drop in LVEF.
Conclusions
Asymptomatic changes in LVEF accounted for most of the cardiotoxicity. The majority of cardiac events occurred in year one although occurrence of cardiotoxicity over time highlights the need for improved risk stratification to guide cardiac surveillance strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mortality rates from breast cancer have declined over the past 30 years as a result of early detection and advances in adjuvant systemic therapy [1,2,3]. It is estimated that by 2020, there will be over 18 million cancer survivors in the United States alone, including 3.5 million breast cancer survivors [4]. This increase in survivorship, however, can come at a cost—cancer treatment is associated with both short- and long-term toxicity including cardiotoxicity [5,6,7].
Anthracyclines (doxorubicin, epirubicin), which are commonly used to treat a variety of cancers, including breast, Hodgkin and non-Hodgkin lymphoma, and sarcoma, are associated with a dose-dependent progressive cardiac dysfunction that manifests clinically as heart failure [6]. The risk of doxorubicin-induced heart failure increases with the cumulative dose of anthracycline: 3–5% at 400 mg/m2, 7–26% at 550 mg/m2, and 18–48% at 700 mg/m2 [11, 14] and can occur either acutely (within hours of treatment) or years to decades after drug exposure [13]. Similarly, there is a correlation between the dose of epirubicin and cumulative incidence of heart failure, but the dose response curve is shifted to the right compared to doxorubicin [11, 15].
With the increase in the number of cancer survivors and the recognition of cancer therapy-associated cardiotoxicity, protecting and maintaining long-term cardiovascular health has become increasingly important. Few clinical trials have reported on cardiovascular outcomes in women with early-stage breast cancer outside of those studying trastuzumab-based therapy [16,17,18,19]. A meta-analysis of 55 clinical trials studied patients randomized to anthracycline-based regimens versus non-anthracycline-containing regimens [12]. Although this meta-analysis included patients with a variety of malignancies (breast ovarian, lymphoma, myeloma, and sarcoma), most were patients with advanced or metastatic breast cancer. Those receiving anthracyclines were at greater risk of experiencing clinical cardiotoxicity (heart failure or a decline in left ventricular ejection fraction (LVEF) requiring the discontinuation of treatment) compared to those treated with non-anthracycline-based regimens (OR 5.43 (95%CI 2.34, 12.62) [12]. Trials included in this meta-analysis had varying lengths of follow-up and included both early (during and within one year after treatment) and late (at least one year after completing treatment) cardiotoxicity. Another meta-analysis examining incidence and predictors of anthracycline-associated cardiotoxicity included 49,017 cancer patients of whom 23,764 had breast cancer and 22,815 were treated with anthracycline-based chemotherapy. Clinical cardiotoxicity (symptomatic heart disease) was reported in 6% (95%CI 3% to 9%), and subclinical cardiotoxicity (asymptomatic systolic dysfunction) in 18% (95%CI 12% to 24%) with a median follow-up of 9 years [13].
While the risk of cardiotoxicity has been clearly established with conventional dosing of anthracyclines with anti-HER2 therapy, there is less data on the risk of cardiotoxicity associated with dose-dense (every 2 weeks) anthracycline-based regimens in women with non-trastuzumab-treated early-stage breast cancer. CALGB 9741 reported superior clinical efficacy (DFS HR = 0.74) with dose-dense versus traditional anthracycline-containing chemotherapy (every 3 weeks) in women with early-stage HER2 – breast cancer, at a median follow-up of 36 months. Low rates of ‘late’ cardiotoxicity requiring treatment were reported in all arms with slightly higher rates in the q3 weekly anthracycline dosing schedule (2% vs 1%); however, the definition of cardiotoxicity and frequency and modality of cardiac monitoring were not described [20].
The Canadian Cancer Trials Group (CCTG) MA.21 (NCT00014222) was a large-phase III trial comparing three different anthracycline-based chemotherapy regimens in node-positive or high-risk node-negative early-stage breast cancer [21]. The definition of cardiotoxicity and frequency and type of cardiac monitoring were pre-specified in the protocol. This paper represents an analysis of cardiotoxicity as pre-defined in this study and carried out up to 5 years after completion of adjuvant therapy.
Methods
Patient population
MA.21 was a multicentre phase III randomized control trial conducted in Canada and the United States that enrolled patients between December 2000 and May 2005. Women were eligible to participate if they were 60 years old or younger, premenopausal or early postmenopausal, with early-stage breast cancer (stage I–IIIB) and had undergone curative breast cancer surgery with axillary lymph node dissection. Prior chemotherapy, hormonal therapy, immunotherapy, or radiotherapy for breast cancer was not permitted. Women were excluded from this study if they had a left ventricular ejection fraction (LVEF) at or below the lower limit of normal in their local laboratory by multigated acquisition scan (MUGA) or echocardiogram, arrhythmias requiring ongoing treatment, heart failure, or documented coronary artery disease. The study was led by the CCTG and approved by participating center institutional research ethics boards.
Treatment
Patients were randomized to one of three adjuvant anthracycline-based chemotherapy regimens containing either epirubicin (E) or doxorubicin (A). Arm A (CEF) received six 28-day cycles of cyclophosphamide (75 mg/m2 orally, days 1–14), epirubicin (60 mg/m2 IV, days 1 and 8), 5-fluorouracil (500 mg/m2 IV, days 1 and 8). Arm B (ddEC/T) received six 14-day cycles of epirubicin (120 mg/m2 IV, day 1), and cyclophosphamide (830 mg/m2 IV, day 1) with concurrent G-CSF, followed by four 21-day cycles of paclitaxel (175 mg/m2 IV). Arm C (AC/T) received four 21-day cycles of doxorubicin (60 mg/m2 IV, day 1) and cyclophosphamide (600 mg/m2 IV, day 1) followed by four 21-day cycles of paclitaxel (175 mg/m2 IV). All patients were required to start chemotherapy within 48 h of randomization. All patients who had breast conserving surgery (BCS) received breast irradiation, but use of other locoregional radiotherapy was administered according to local institutional practice. Patients with estrogen receptor (ER)-positive breast cancer received tamoxifen after completion of chemotherapy. In 2002, the protocol was amended to permit aromatase inhibitor therapy in postmenopausal women and in 2005 trastuzumab was permitted for patients with HER2-positive disease.
Per-protocol cardiac monitoring included baseline LVEF measurement with either MUGA or echocardiogram before commencing, and at the end of chemotherapy and annual testing for 5 years after the end of chemotherapy. Patients were to be followed by the same method of evaluation throughout the study and MUGA scans were the recommended modality.
Cardiac endpoints
For the purpose of this analysis, the cardiac endpoints included were the following: LVEF decline of at least 10% and an absolute value of less than 50% either by MUGA or ECHO, or Grade 3–4 cardiac LV function changes (heart failure, HF) as reported using the National Cancer Institute Common Toxicity Criteria (NCICTC) Version 2.0, or Grade 3–4 cardiac ischemia/infarction as reported using the NCICCTC criteria. All patients were evaluated clinically on the first day of each chemotherapy cycle.
Statistical analysis
The statistical analysis was done as a time to cardiotoxicity event competing risk analysis [18]. The initial time point was the date of randomization. A cardiotoxicity event was defined as the first occurrence of one of the cardiac endpoints as defined above. The competing event was recurrence of breast cancer. For example, if a patient developed recurrence before the occurrence of any cardiac event, then she was censored as cardiotoxicity event free by the competing event at the time of recurrence. If a patient did not have a recurrence at 5 years and no cardiac event at 5 years, then she was censored as cardiovascular event free and competing event free at 5 years. If a patient did not have 5 years of LVEF measurements but was recurrence free at 5 years and had no evidence of HF or MI, she was considered as having no cardiac events at the latest date of LVEF measurement. Cumulative incidences curves were fitted using Gray’s method [22].
A Cox proportional hazards model was used to perform a multivariable analysis of the cause-specific function of the competing risks for cardiotoxicity [23]. All analyses were conducted with SAS software, version 9.3 (SAS Institute, Cary, NC).
Results
Two thousand one hundred and four (2,104) patients with early-stage breast cancer were randomized to one of three treatment arms: Arm A—CEF (n = 701); Arm B—ddEC/T (n = 701); or Arm C—AC/T (n = 702) between May 2000 and December 2005. Baseline patient characteristics and clinical outcomes of this study have previously been reported [21]. Recurrence-free survival was inferior in patients randomized to the AC/T arm (85.0%) compared to CEF (90.1%) and to ddEC/T (89.5%). Baseline patient cardiovascular risk factors are reported in Table 1. Risk factors were well balanced between treatment arms: the majority of patients were less than 50 years of age (60%) with a body mass index < 30 (67%). Cardiovascular risk factors were infrequent across all treatment arms: diabetes (4–5%); hypertension (2%), hypercholesterolemia (1%), and history of cardiovascular disease (1–2%). Information on smoking history and family history of cardiovascular disease was not collected.
The most commonly used modality for left ventricular assessment was MUGA scan (80%). During the follow-up period, patients either experienced a cardiac event, experienced a recurrence, died, were lost to follow-up, or remained event free. The disposition of patients is presented over the 5-year follow-up in Table 2, a modified consort diagram presented in tabular form. Table 2 demonstrates the events that occurred in patients during annual periods and how many patients entered the next interval. By year 5, 70% of remaining patients (404/577) in the CEF arm, 69% (406/588) in EC/T, and 69.5% (382/550) in AC/T had completed all per-protocol cardiac testing. The maximum cumulative dose of anthracyclines patients could receive was 720 mg/m2 epirubicin in Arm A and B, and 240 mg/m2 doxorubicin in Arm C. The distribution of cumulative anthracycline dose mg/m2 by treatment arm is shown in Table 3.
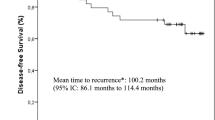
The number of patients experiencing a cardiac event in each treatment arm and the type of event is shown in Table 4. We found that CEF was associated with a significantly higher risk of developing cardiotoxicity than either of the other two regimens. Figure 1 shows the cumulative incidence of time to cardiotoxicity event (with recurrence as a competing event) for each treatment arm. The 5-year cumulative risks of first event of any of LVEF decline, HF, or MI for CEF, ddECT and ACT were 22.3% (95%CI 18.9 to 25.7), 14.2% (95%CI 11.0 to 17.3), and 8.1% (95%CI 5.8 to 10.4), respectively. Gray test shows there is a significant difference in cumulative incidence functions among these three groups (p < 0.0001). The hazard ratio for each year is shown in Table 2. Most of the cardiac events were asymptomatic drops in LVEF (Table 4). In the multivariable analysis, BMI over 30 and treatment (CEF versus ddEC/T) were significantly associated with increased risk of cardiotoxicity in the first 5 years after completion of treatment (Table 5). In addition, there was a trend for an association between age and risk of cardiotoxicity.
Discussion
In 2010, we published the results of MA.21, a phase III clinical trial comparing the efficacy of three different anthracycline-based chemotherapy regimens—CEF, ddEC/T, and AC/T [21]. That study showed that AC/T was significantly inferior to either epirubicin-based regimen based on 3-year recurrence-free survival rates. During chemotherapy treatment itself, rates of grade three or more heart failure were similar between the three treatment arms, 0.4% with CEF, 0.3% each with ddEC/T or AC/T. In this manuscript, we report the cardiac outcomes of patients in the first five years after chemotherapy.
The patients who received CEF had a statistically significantly increased risk of cardiotoxicity, compared to either of the other two regimens. This difference is seen despite the fact that both CEF and ddEC/T regimens had a similar cumulative dose of epirubicin: median dose – 682 mg/m2 for CEF and 712 mg/m2 for ddEC/T, suggesting that the dose intensity of chemotherapy, not just cumulative dose, has an impact on risk of cardiac dysfunction.
In all measured outcomes, the majority of events occurred within the first year after chemotherapy; however, a small number of patients continued to experience cardiac complications in subsequent years. It should be noted that the majority of events consisted of falls in LVEF and that the incidence of heart failure and MI was much lower (Table 4). In this study, we conducted a competing risk analysis comparing risk of experiencing a cardiac event to risk of breast cancer recurrence prior to a cardiac event (see Table 2). At year 5, twenty-two patients experienced a cardiac event in the CEF arm while four patients experienced a breast cancer recurrence. This is in contrast to the ddEC/T and AC/T arms in which there were 14 and two patients with cardiac events and eight and two patients with breast recurrences prior to a cardiac event at year five, respectively. These findings highlight the importance of considering not only the risk of breast cancer recurrence when selecting a cancer therapy, but also the risk of competing events, such as cardiotoxicity, which might lead to increased morbidity and/or mortality. Almost 12% of patients with normal LVEF at the time of completing anthracycline-based chemotherapy have been shown to develop left ventricular dysfunction in subsequent years [24]. The use of CEF resulted in the best recurrence-free survival (90.1%) of all treatment arms; however, this regimen has fallen out of favor due to treatment-related toxicities (e.g., febrile neutropenia), including risk of cardiotoxicity. Although current clinical practice has evolved to prescribing dose-dense AC/taxol, the results of our study suggest less risk of cardiotoxicity with this schedule of anthracycline administration (e.g., CEF vs dd EC/T arm). Future studies evaluating novel breast cancer regimens (e.g., immunotherapy) should take into consideration not only the risk of breast cancer recurrence and mortality but competing risks, such as cardiovascular risk factors and disease, when evaluating the incremental benefit of a new cancer treatment.
The incorporation of taxanes (paclitaxel, docetaxel) in the treatment of early breast cancer has led to a reduction in anthracycline exposure, with modern regimens typically consisting of three or four cycles (e.g., FEC/D, ddAC/T) compared to six cycles of anthracyclines (e.g., CEF, ddEC/T) or non-anthracycline-containing regimens (TC × 4–6) [25]. While a direct comparison of the efficacy of ddEC/T and ddAC/T has not occurred, the safety profile of dose-dense regimens has not been associated with an increase in cardiotoxicity [20, 26]. Trastuzumab-based regimens are standard of care for the 15–20% of women with a breast cancer diagnosis who overexpress HER2 and are associated with an increased risk of cardiotoxicity, mainly manifesting as asymptomatic drops in LVEF [27]. Only a small number of patients enrolled in MA.21 received trastuzumab: 29 (4.1%) in the CEF arm, 27 (3.9%) in the ddEC/T arm, and 26 (3.7%) in the AC/T arm, so it is difficult to precisely understand the confounding effect, if any, that this could have played. Given the current popularity of ddAC/T in clinical practice, understanding the ongoing risk of cardiotoxicity in the first 5 years after treatment is important to inform both patients and providers caring for breast cancer survivors.
This study had several limitations. Women included in the original trial were young with few cardiovascular risk factors and thus perhaps not representative of the patient population now being treated in the real-world setting. Over 60% of study patients were under age 50. In contrast, the median age of breast cancer patients at diagnosis in Ontario, Canada, is 60 [28]. When we consider the effects of breast cancer therapy in an older real-world breast cancer population, the risk of cardiotoxicity with high-dose anthracycline-based regimens may be higher than we report here. Two of the treatment regimens (CEF, ddEC/T) which included high-dose anthracycline (> 600 mg/m2) are not commonly used in clinical practice today. These regimens resulted in the highest incidence of cardiac events in this study. It should be noted, however, that patients treated with AC/T, considered a low-dose anthracycline (total 240 mg/m2) regimen, still experienced cardiac events out to 5 years after initiation of chemotherapy.
Cardiac endpoints in this study were defined based on changes in LVEF (decline of at least 10% and an absolute value of less than 50%) and the National Cancer Institute Common Toxicity Criteria (NCI CTCAE) version 2.0. Definitions of cardiovascular adverse events have evolved over time (NCIC CTCAE version 5.0), making it challenging to interpret findings across oncology clinical trials. The majority of cardiac events in our study were asymptomatic drops in LVEF and therefore we would not expect to see significant changes in our conclusions based on the current NCIC CTCAE version 5.0.
Although our data, demonstrating a low rate of cardiac events out to five years after completion of chemotherapy, are reassuring, clinicians should remain vigilant in monitoring and assessing cardiac function in breast cancer survivors, particularly in older breast cancer patients with co-existing cardiac risk factors.
The American Society of Clinical Oncology (ASCO) currently recommends cardiac assessment between 6 and 12 months after completion of cancer-directed therapy in asymptomatic patients considered to be at increased risk of cardiac dysfunction [29]. In patients treated with lower-dose anthracycline-based regimens (doxorubicin < 250 mg/m2), close surveillance for cardiac dysfunction is needed particularly in individuals > 60 years of age with ≥ 2 cardiac risk factors including smoking, hypertension, dyslipidemia, obesity during or after treatment [29]. The European Society of Medical Oncology (ESMO) recently published consensus recommendations on management of cardiac disease in cancer patients throughout oncological treatment, suggesting that in asymptomatic patients, it would be reasonable to consider cardiac imaging 2 years post-treatment and periodically thereafter [30]. Our findings support these recommendations. In our study, young women with normal baseline heart function and few co-morbidities treated with anthracycline-based chemotherapy continued to experience cardiac events 5 years after completion of cancer therapy suggesting the increased risk of left ventricular dysfunction is lifelong for these individuals. Cardiac surveillance recommendations have generally been based on expert opinion as there is limited long-term data on the cardiovascular consequences of exposure to potentially cardiotoxic cancer therapy in adult cancer survivors. Prospective cohort studies are needed to determine the best cardiovascular surveillance strategies (e.g., type of cardiovascular imaging, timing, cardiac biomarkers) in adult cancer patients exposed to potentially cardiotoxic cancer therapy, to guide health care providers in cancer survivorship care.
In this study of 5-year cardiac safety outcomes in patients from the MA.21 clinical trial comparing three different anthracycline-based chemotherapy regimens, we found that dose-dense EC/T and AC/T had significantly less treatment-related cardiotoxicity in the first 5 years after beginning chemotherapy compared to CEF. It should be noted that the vast majority were asymptomatic drops in LVEF, rather than clinical events. Dose-dense anthracycline-based chemotherapy continues to play a significant role in the treatment of early-stage breast cancer. When combined with the original efficacy results, the ddEC/T regimen had the best combination of efficacy and cardiac safety in a relatively young population of breast cancer patients who had few cardiac risk factors. The ddECT used in MA.21 had other significant toxicities, including anemia, that may render it less suitable for standard use. Nonetheless, the illustration that the dose-dense application of anthracycline at the same cumulative dose is no more cardiotoxic, and equally effective, should be explored in other datasets. This is particularly so in view of the recently EBCTCG meta-analysis suggesting their superiority in efficacy [26]
Cardiovascular disease is a significant cause of morbidity and mortality in breast cancer survivors, likely a consequence of previous cancer therapies (chemotherapy, targeted agents, radiation), cardiovascular risk factors/co-morbidities and aging. Our increased understanding of the interplay between cardiovascular and cancer risk factors, cancer treatments, and aging should lead to increased vigilance in the identification and management of cardiovascular disease in this patient population.
References
Howlader N, Ries LAG, Mariotto AB et al (2010) Improved estimates of cancer-specific survival rates from population-based data. J Natl Cancer Inst 102(20):1584–1598
Jemal A, Ward E, Hao Y et al (2005) Trends in the leading causes of death in the United States, 1970–2002. JAMA 294(10):1255–1259
Jemal A, Ward E, Thun M (2010) Declining death rates reflect progress against cancer. PLoS ONE 5(3):e9584
Miller KD, Siegel RL, Lin CC et al (2016) Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 66(4):271–289
Canadian Cancer Society’s Advisory Committee on Cancer Statistics. Canadian Cancer Statistics 2017. Toronto, ON: Canadian Cancer Society; 2017 available at www.cancer.ca/Canadian-Cancer-Statistics-2017-EN.pdf August 16, 2017
Bodai BI, Tuso P (2015) Breast cancer survivorship: a comprehensive review of long-term medical issues and lifestyle recommendations. Perm J 19(2):48–79
Runowicz CD, Leach CR, Henry L et al (2016) American Cancer socieity/American Society of Clinical oncology breast cancer survivorship care guideline. J Clin Oncol 34(6):611–635
Curigliano G, Cardinale D, Dent S et al (2016) Cardiotoxicity of anticancer treatments: epidemiology, detection, and management. CA Cancer J Clin 66(4):309–325
Suter TM, Ewer MS (2013) Cancer drugs and the heart: importance and management. Eur Heart J 34(15):1102–1111
Aleman BMP, Moser EC, Nuver J et al (2014) Cardiovascular disease after cancer therapy. Eur J Cancer 12(1):18–28
Von Hoff DD, Layard MW, Basa P et al (1979) Risk factors for doxorubicin-induced congestive heart failure. Ann Intern Med 91:710–717
Smith LA, Cornelius VR, Plummer CJ et al (2010) Cardiotoxicity of anthracycline agents for the treatment of cancer: systematic review and meta-analysis of randomised controlled trials. BMC Cancer 10:337
Lotrionte M, Biondi-Zoccai G, Abbate A et al (2013) Review and meta-analysis of incidence and clinical predictors of anthracycline cardiotoxicity. Am J Cardiol 112(12):1980–1984
Speyer JLGM, Zeleniuch-Jacquotte A, Wernz JC et al (1992) ICRF-187 permits longer treatment with doxorubicin in women with breast cancer. J Clin Oncol 10:117–127
Coukell AJ, Faulds D (1997) Epirubicin: an updated review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy in the management of breast cancer. Drugs. 1997(53):453–482
Piccart-Gebhart M, Proctor M, Leyland-Jones B et al (2005) Trastuzumab after adjuvant chemotherapy in HER2—positive breast cancer. NEJM 353:1659–1672
Romond E, Perez E, Bryant J et al (2005) Trastuzumab plus adjuvant chemo therapy for operable HER2—positive breast cancer. NEJM 353:1673–1684
Dennis S, Wolfgang E, Nicholas R et al (2011) Adjuvant trastuzumab in HER2—positive breast cancer. NEJM 365:1273–1283
Joensuu H, Wildiers H, Huovinen R et al (2018) Effect of adjuvant trastuzumab for a duration of 9 weeks vs 1 year with concomitant chemotherapy for early human epidermal growth factor receptor 2 positive breast cancer: the sold randomized clinical trial. JAMA Oncol 4(9):1199–1206
Citron ML, Berry DA, Cirrincione C et al (2003) Randomized trial of dose-dense versus conventionally scheduled and sequential versus concurrent combination chemotherapy as postoperative adjuvant treatment of node-positive primary breast cancer: first report of intergroup trial C9741/cancer and leukemia group b trial 9741. J Clin Oncol 21:1431–1439
Burnell M, Levine MN, Chapman JW et al (2010) Cyclophosphamide, epirubicin, and fluorouracil versus dose-dense epirubicin and cyclophosphamide followed by paclitaxel versus doxorubicin and cyclophosphamide followed by paclitaxel in node-positive or high-risk node-negative breast cancer. J Clin Oncol 28:77–78
Gray RJ (1988) A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat 16:p1141–1154
Dignam James S, Qiang Z, Kocherginsky M (2012) The use and interpretation of competing risks regression models. Clin Cancer Res 18(80):2301–2308
Steinherz LJ, Steinherz PG, Tan CT, Heller G, Murphy ML (1991) Cardiac toxicity 4 to 20 years after completing anthracycline therapy. JAMA 266(12):1672–1677
Nitz U, Gluz O, Clemens M, Malter W, Reimer T, Nuding B, Aktas B, Stefek A, Pollmanns A, Lorenz-Salehi F, Uleer C (2019) West German study planB trial: adjuvant four cycles of epirubicin and cyclophosphamide plus docetaxel versus six cycles of docetaxel and cyclophosphamide in HER2-negative early breast cancer. J Clin Oncol 37(10):799–808
Gray R, Bradley R, Braybrooke J, Liu Z, Peto R, Davies L, Dodwell D, McGale P, Pan H, Taylor C, Barlow W (2019) Increasing the dose intensity of chemotherapy by more frequent administration or sequential scheduling: A patient-level meta-analysis of 37 298 women with early breast cancer in 26 randomised trials. Lancet. https://doi.org/10.1016/S0140-6736(18)33137-4
Moja L, Tagliabue L, Balduzzi S et al (2012) Trastuzumab containing regimens for early breast cancer. Cochrane Database Syst Rev Issue, 4
Cancer Care Ontario. Ontario Breast Screening Program 2011 Report. Toronto, Canada 2013. https://www.cancercare.on.ca/common/pages/UserFile.aspx?fileId=288834 Accessed 25 July 2017
Armenain AH, Lacchette C, Barac A et al (2017) Prevention and monitoring cardiac dysfunction in survivors of adult cancers: American Society of Clinical oncology clinical practice guideline. J Clin Oncol 25(8):893–911
Curigliano G, Lenihan D, Fradley M, Ganatra S, Barac A, Blaes A, Herrmann J, Porter C, Lyon AR, Lancellotti P, Patel A (2020) Management of cardiac disease in cancer patients throughout oncological treatment: ESMO consensus recommendations. Ann Oncol 31(2):171–190
Acknowledgements
We would to thank all of the women who participated in this trial and the many clinical trials staff that helped to compile these data.
Funding
This work was funded by the Canadian Society Cancer Research Institute (CSCRI), Canadian Institute for Health Research (CIHR), and the National Cancer Institute-US.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Susan Dent—advisory board/consulting, honoraria: Pfizer, Novartis, Eli Lilly; research funding Novartis Kathleen I. Pritchard—Advisory board/consulting, honoraria: Pfizer, Roche, Amgen, Novartis, Eisai, Genomic Health Inc., Myriad Genetic Laboratories.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dent, S.F., Botros, J., Rushton, M. et al. Anthracycline-induced cardiotoxicity in patients with early-stage breast cancer: the Canadian Cancer Trials Group (CCTG) MA.21 experience. Breast Cancer Res Treat 184, 733–741 (2020). https://doi.org/10.1007/s10549-020-05887-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05887-w