Abstract
The objective of this study is to investigate health-related quality of life (HRQoL) in male breast cancer patients. Data of 20,673 patients diagnosed with primary breast cancer (male: n = 84) who completed a questionnaire after discharge from hospital were analysed. HRQoL (SF-36), age, sex, education, native language, insurance status, and partnership status were measured. Cancer staging, treatment (partial mastectomy vs. radical mastectomy), and cancer site were indicated by the clinicians. The HRQoL scores of male breast cancer patients were compared with reference populations. Differences in HRQoL scores between men and women were compared using t tests and regression analysis. Compared to female breast cancer patients, male patients scored significantly higher on seven of eight subscales (physical functioning, role functioning—physical and emotional, bodily pain, vitality, social functioning, and mental health) in the regression analysis. Compared to the reference populations (general male population, men aged 61–70, and the cancer-affected population), male breast cancer patients scored lower on SF-36 subscales on average, with major differences in emotional and physical role functioning. The results suggest that male breast cancer patients may need early interventions that specifically target role functioning, which is severely impaired compared to the male reference population. Future research needs to assess HRQoL with cancer-specific questionnaires and longitudinal designs also focussing on male patients in breast centres.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Male breast cancer is a rare disease. It accounts for less than 1% of all breast cancer cases and around 0.1% of cancer mortality in men in western countries. There is little data on the epidemiology of male breast cancer, but recent studies showed an increasing number of male breast cancer cases. In the United States, the incidence of male breast cancer significantly increased from 0.86 in 1973 to 1.08/100,000 in 1998 [1]. There were an estimated 1,970 newly diagnosed male breast cancer cases in the US in 2010, with 390 patients dying of this disease [2]. Data regarding male breast cancer have long been somewhat limited since results were mostly based on small samples. Recently, a number of studies investigated incidence and mortality trends for male patients compared to female patients [1, 3, 4] and reviewed the epidemiology and risk factors for male breast cancer [5–14]. Recent analyses in population-based surveys from the US confirmed several earlier findings: breast cancer occurs later in men than in women, with a higher stage, lower grade, and more oestrogen receptor-positive tumours [3]. Recently, mortality and survival rates for men and women have improved significantly in the US; however, this trend is clearer for women than for men [3].
If little is known about the epidemiology of male breast cancer, even less is known about the health-related quality of life (HRQoL) of male breast cancer patients. Male breast cancer treatment is referred to as a special case of female breast cancer with only limited references in the guidelines. Breast cancer is a “female disease” that is rare in men, and medical treatment and rehabilitation are adjusted to female patients. We therefore assume that men affected by breast cancer suffer specific hardships, among them impaired role functioning, impaired body image, and, in addition, lack of disease-specific psychosocial support. So far and to the best of our knowledge, there has been no study specifically investigating the HRQoL of male breast cancer patients. We aimed to clarify this issue in the present study. The purpose of our study was to describe HRQoL in a sample of German breast cancer patients and to investigate whether HRQoL differs significantly between men and women controlling for age, cancer stage, type of surgery, education, native language, partnership, and insurance status.
Methods
Participants
The investigation reported here is part of a larger programme of research designed to investigate the breast centre concept of the German federal state of North Rhine-Westphalia (NRW) (population: 17.9 million). Patients undergoing treatment for primary breast cancer in one of the 51 accredited breast centres in NRW were invited to self-administer a questionnaire at home after discharge from the hospital [15]. The overall aim of the study was to compare the quality of healthcare provided in the individual breast centres as perceived by the patients. A secondary study objective was to describe the HRQoL of the patients. The study was approved by the relevant institutional ethics committees and participants provided written consent.
Inclusion criteria were as follows: (1) patients who had undergone inpatient surgery for newly diagnosed breast cancer between 1 February and 31 July; (2) at least one malignancy; and (3) at least one post-operative histological evaluation.
Procedure
Annually, between February and June (survey period 6 months) in the years 2006–2011, cross-sectional studies were performed with samples of patients from the 51 accredited breast centre hospitals in the German state of NRW. In 2005, the survey period lasted 3 months in a sample of the accredited breast centres (pre-test). Shortly before being discharged from hospital, patients were asked by the hospital staff to give written consent to be included in the survey. Once the patients had given their consent, medical personnel from the centres provided the research team with clinical information on the patients. Patients who consented to the survey were included in the study and made up the sample of patients. The survey, which was designed according to Dillman’s total design method with three contacts [16], was sent out to the patients within a week of receiving written consent; a record was kept of the number of questionnaires that were returned. Patient perspectives were measured using the Cologne Patient Questionnaire for Breast Cancer (CPQ-BC), which is based on the Cologne Patient Questionnaire (CPQ) [17]. Like the CPQ, CPQ-BC includes, among other things, questions on HRQoL and patients’ perception of the quality of healthcare [18].
Measures
Health-related quality of life
For measuring HRQoL, the German version of the SF-36 was used in the CPQ-BC. The SF-36 items were summarized into scales according to the instructions [19]. SF-36 has seven multi-item scales exploring physical functioning, role functioning—physical, bodily pain, vitality, social functioning, role functioning—emotional, and mental health and one single item (general health perception) that fitted our purposes. The scores range from 0 to 100. Higher scores represent higher levels of HRQoL.
Patient socio-demographic data
The patients were asked to indicate date of birth, school leaving certificate, native language, insurance status, and partnership status on the questionnaire.
Clinical data
In addition to the data collected by the patient questionnaire, clinical data and type of surgery performed were contributed by the medical personnel. The cancer stage was categorized using UICC categories [20], which draws from data on tumour size, lymph node, and metastasis status as indicated by the medical personnel; type of surgery and cancer site were indicated by the medical personnel (partial mastectomy vs. radical mastectomy; right/left/both).
Statistical methods
Characteristics of the sample were summarized using tables for categorical factors and means and standard deviations for continuous variables (age, HRQoL subscales). t Tests were used to analyse differences in SF-36 scores between sexes. Multivariable linear regression models were estimated for each HRQoL subscale, including all factors (age, sex, staging, cancer site, type of surgery, school leaving certificate, native language, insurance status, and partnership status) as potential predictors. Control variables were categorized except for age.
Results
Characteristics of the study population
Of the 25,980 patients who consented to participate in the study, 22,798 completed and returned the questionnaires (87.8%). Data from 20,673 patients, 84 of whom were male, were eligible for the analyses performed here, i.e. they reported on each SF-36 subscale and indicated his or her date of birth. As shown in Table 1, male patients more frequently had a higher cancer stage at time of diagnosis than women and were 5 years older on average. The vast majority of male patients had an initial radical mastectomy, whereas over 70% of the female patients for whom data was reported received breast-conserving treatment (comparable to partial mastectomy in men). Compared to female patients, the male group contained a slightly increased proportion of non-native speakers and more highly educated members (diploma from German secondary school qualifying for university/university of applied sciences admission). Men indicated being in a relationship more often than the women.
Health-related quality of life
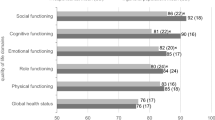
Unadjusted scores of the SF-36 subscales for male and female patients are listed in Table 2. Unadjusted scores for the men were significantly higher (i.e. better HRQoL) on six subscales: role functioning—physical, bodily pain, vitality, social functioning, role functioning—emotional, and mental health. The SF-36 scores from our sample were compared to reference populations, as shown in Fig. 1 [19]. Male breast cancer patients scored lower on all subscales than the general male population and the 61–70-year-old male population. They scored lower than the cancer patient reference population on four subscales: physical functioning, role functioning—physical, social functioning, and role functioning—emotional. Associations with age and a number of other potential predictors of HRQoL were assessed in multivariable regression analysis. Table 3 shows the results of the linear regression analyses on the eight SF-36 subscales. Controlling for age, stage, cancer site, type of surgery, school leaving certificate, native language, insurance status, and partnership status, male patients reported significantly better HRQoL scores on all subscales except for general health perception. The scores were up to 18.2 points higher (role functioning—emotional) than those of the female patients.
HRQoL of male breast cancer patients compared to the reference populations, as reported in Bullinger and Kirchberger [19]. PF physical functioning; RF-P role functioning—physical, BP bodily pain, GHP general health perceptions, VITA vitality, SF social functioning, RF-E role functioning—emotional, ME mental health
Discussion
Compared to the male reference populations, HRQoL in male breast cancer patients was lower, especially for the role functioning subscales. These results suggest that male breast cancer patients may need early psychosocial interventions that specifically target role functioning, both physical and emotional, depending on the patients’ preferences. As advocated in research on HRQoL in female breast cancer patients, more studies are needed to compare breast cancer patients with controls without breast cancer [21].
In the regression analyses, male breast cancer patients scored significantly higher on seven of the eight HRQoL subscales than female patients. This finding is not too surprising since it has been consistently reported in the literature that men show higher HRQoL scores in healthy reference populations and in most subsamples. However, one could have expected that the specific situation of men suffering from a disease usually attributed to women would bring about problems that resulted in a lower HRQoL, especially on the emotional and mental level. Provided that the HRQoL instrument applied here is adequate for our purposes, HRQoL in male breast cancer patients is not as impaired as HRQoL of female patients.
Research on HRQoL in male breast cancer patients is at a very early stage. The aim of this article was to provide the first data on this topic, bearing in mind that there are several limitations concerning the data. First, the SF-36 was used to measure HRQoL, a generic instrument which has not been tested with male breast cancer patients before. Since there is no HRQoL instrument specifically developed and validated for this patient group, the results should be considered preliminary to a certain degree. Secondly, the data were gathered in a cross-sectional design and in a local sample with inclusion and exclusion criteria. Third, although the overall number of patients surveyed was high, only 84 of the patients investigated in the analyses were male. The percentage of male patients was below 0.5%, suggesting that male patients were underrepresented. However, the response rate of the sample was consistently high. Fourth, we lacked information on co-morbidities, which might have had a significant impact on patients’ HRQoL. Additionally, we do not have any information on the relationship between stage of disease and response to the survey. On a positive note, the overall number of patients surveyed was substantially large, a high-response rate was achieved, and the data were collected with a consistent design. Compared to other studies on HRQoL in rare diseases [22–24], HRQoL was assessed shortly after surgery with low time ranges between surgery and measurement of HRQoL.
To date, this is the first comparative study of male versus female breast cancers and thus an important source of information on HRQoL. Its strengths are the large case number, the consistent survey method, and the demographic, tumour, and treatment characteristics. These preliminary results require further investigation, preferably in a larger sample and with a cancer-specific HRQoL instrument. Since case number and patient access is limited in Germany, we recommend a study in a country with nation-wide cancer registries. Bearing in mind the increasing incidence of male breast cancer, the findings of the present study suggest that the development of specific psycho-social support measures is needed.
References
Giordano SH, Cohen DS, Buzdar AU et al (2004) Breast carcinoma in men: a population-based study. Cancer 101:51–57
American Cancer Society (2010) Cancer facts & figures 2010. American Cancer Society, Atlanta
Anderson WF, Jatoi I, Tse J et al (2010) Male breast cancer: a population-based comparison with female breast cancer. J Clin Oncol 28:232–239
Stang A, Thomssen C (2008) Decline in breast cancer incidence in the United States: what about male breast cancer? Breast Cancer Res Treat 112:595–596
Contractor KB, Kaur K, Rodrigues GS et al (2008) Male breast cancer: is the scenario changing. World J Surg Oncol 6:58
Rudlowski C (2008) Male breast cancer. Breast Care 3:183–189
Speirs V, Shaaban AM (2009) The rising incidence of male breast cancer. Breast Cancer Res Treat 115:429–430
Tajima N, Tsukuma H, Oshima A (2001) Descriptive epidemiology of male breast cancer in Osaka, Japan. J Epidemiol 11:1–7
Krause W (2004) Male breast cancer-an andrological disease: risk factors and diagnosis. Andrologia 36:346–354
Weiss JR, Moysich KB, Swede H (2005) Epidemiology of male breast cancer. Cancer Epidemiol Biomarkers Prev 14:20–26
Korde LA, Zujewski JA, Kamin L et al (2010) Multidisciplinary meeting on male breast cancer: summary and research recommendations. J Clin Oncol 28:2114–2122
Hodgson NC, Button JH, Franceschi D et al (2004) Male breast cancer: is the incidence increasing? Ann Surg Oncol 11:751–755
Fentiman IS, Fourquet A, Hortobagyi GN (2006) Male breast cancer. Lancet 367:595–604
Brinton LA, Richesson DA, Gierach GL et al (2008) Prospective evaluation of risk factors for male breast cancer. J Natl Cancer Inst 100:1477–1481
Kowalski C, Würstlein R, Steffen P et al (2010) Vier Jahre Patientinnenbefragung im Rahmen der (Re-)Zertifizierung der Brustzentren in Nordrhein-Westfalen. Senologie 7:24–28
Dillman DA (1978) Mail and telephone survey. The total design method. Wiley, New York
Pfaff H, Freise D, Mager G et al (2003) Der Kölner Patientenfragebogen (KPF): Entwicklung und Validierung eines Fragebogens zur Erfassung der Einbindung des Patienten als Kotherapeuten. Sankt Augustin, Asgard-Verlag
Kowalski C, Nitzsche A, Scheibler F et al (2009) Breast cancer patients’ trust in physicians: the impact of patients’ perception of physicians’ communication behaviors and hospital organizational climate. Patient Educ Couns 77:344–348
Bullinger M, Kirchberger I (1998) SF-36. Fragebogen zum Gesundheitszustand. Handanweisung. Hogrefe-Verlag, Göttingen
Sobin LH, Wittekind C (2002) UICC TNM classification of malignant tumors, 6th edn. Wiley, New York
Avis NE, Crawford S, Manuel J (2005) Quality of life among younger women with breast cancer. J Clin Oncol 23:3322–3330
Mowry SE, LoTempio MM, Sadeghi A et al (2006) Quality of life outcomes in laryngeal and oropharyngeal cancer patients after chemoradiation. Otolaryngol Head Neck Surg 135:565–570
Major MS, Bumpous JM, Flynn MB et al (2001) Quality of life after treatment for advanced laryngeal and hypopharyngeal cancer. Laryngoscope 111:1379–1382
LoTempio MM, Wang KH, Sadeghi A et al (2005) Comparison of quality of life outcomes in laryngeal cancer patients following chemoradiation versus total laryngectomy. Otolaryngol Head Neck Surg 132:948–953
Acknowledgments
The patient survey was requested and initiated by the North Rhine Westfalian Ministry of Work, Health and Social Affairs (MAGS NRW). The costs were borne by the participating hospitals as part of the breast centre certification and benchmarking process. The hospitals asked the patients to consent to participation in the study and indicated disease characteristics as reported. Neither the ministry nor the hospitals were involved in the analysis or interpretation of data, in the writing of the report, or in the decision to submit the paper for publication.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kowalski, C., Steffen, P., Ernstmann, N. et al. Health-related quality of life in male breast cancer patients. Breast Cancer Res Treat 133, 753–757 (2012). https://doi.org/10.1007/s10549-012-1970-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-1970-3