Abstract
Expression profiling of BRCA1-deficient tumours has identified a pattern of gene expression similar to basal-like breast tumours. In this study, we examine whether a BRCA1-dependent transcriptional mechanism may underpin the link between BRCA1 and basal-like phenotype. In methods section, the mRNA and protein were harvested from a number of BRCA1 mutant and wild-type breast cancer cell lines and from matched isogenic controls. Microarray-based expression profiling was used to identify potential BRCA1-regulated transcripts. These gene targets were then validated (by in silico analysis of tumour samples) by real-time PCR and Western blot analysis. Chromatin immunoprecipitation (ChIP) assays were used to confirm recruitment of BRCA1 to specific promoters. In results, we demonstrate that functional BRCA1 represses the expression of cytokeratins 5(KRT5) and 17(KRT17) and p-Cadherin (CDH3) in HCC1937 and T47D breast cancer cell lines at both mRNA and protein level. ChIP assays demonstrate that BRCA1 is recruited to the promoters of KRT5, KRT17 and CDH3, and re-ChIP assays confirm that BRCA1 is recruited independently to form c-Myc and Sp1 complexes on the CDH3 promoter. We show that siRNA-mediated inhibition of endogenous c-Myc (and not Sp1) results in a marked increase in CDH3 expression analogous to that observed following the inhibition of endogenous BRCA1. The data provided suggest a model whereby BRCA1 and c-Myc form a repressor complex on the promoters of specific basal genes and represent a potential mechanism to explain the observed overexpression of key basal markers in BRCA1-deficient tumours.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is a heterogeneous disease that can be stratified on the basis of differential gene expression into five subtypes that differ in biology and clinical behaviour. These breast cancer subtypes include Luminal A and Luminal B, which are ERα positive, the normal-like, HER2-positive and basal-like subtypes, which typically are ERα negative. Luminal breast cancer subtypes are amenable to hormonal therapy, and HER2 tumours can now be targeted by Herceptin therapy. However, the basal-like subtype is negative for hormone receptors and HER2 and is associated with a more aggressive phenotype and poorer outcome [1–3]. Immunohistochemical studies have demonstrated that basal-like epithelial breast tumours display increased expression of KRT5 and KRT17, CDH3, cyclin E and reduced expression of KRT8, KRT18 and the CDK inhibitor p27kip1 [4–6].
BRCA1-linked breast cancers are often of high grade and triple receptor negative, indicating a high degree of similarity to tumours of the basal-like phenotype [7, 8]. In addition, several studies have reported that BRCA1-associated tumours are more likely to express markers consistent with a basal-like phenotype, and gene expression profiling reveals that BRCA1-linked tumours tend to segregate with basal-like tumours during unsupervised hierarchal cluster analysis [9]. However, the molecular mechanism underlying these striking similarities remains to be discovered. The molecular profile of BRCA1-linked breast cancers has come under increasing scrutiny, and the possible contribution that co-operating tumour suppressor and oncogenes may make in promoting the malignant phenotype of these breast tumours is of increasing interest. The c-Myc oncogene encodes a transcription factor capable of activating and repressing target gene expression. Deregulation of this gene can contribute significantly to the progression of lymphomas, lung cancer and more specifically breast cancers. Indeed, approximately 50% of BRCA1-linked inherited breast cancer cases exhibit c-Myc amplification. This is in contrast to sporadic breast cancer where c-Myc amplification occurs in only 20% of cases [10]. Whether the rate of c-Myc amplification is also high in sporadic breast tumours with epigenetic inactivation of BRCA1 requires further clarification although preliminary data would suggest that this may be the case [10]. The link between BRCA1 and c-Myc is further emphasised by the observation that both proteins interact to form a transcriptional repressor complex. This ability of BRCA1 to repress c-Myc-dependent transactivation has been documented for a number of genes including the human telomerase reverse transcriptase (hTERT) and cdc25 [11, 12]. We have previously reported that BRCA1 can co-repress the DNA damage inducible gene, psoriasin (S100A7) in a c-Myc-dependent manner [13]. This current study extends these findings and demonstrates a functional role for the BRCA1/c-Myc co-repressor complex in the regulation of several well-known basal genes delineating the well-characterised basal-like breast tumour phenotype.
Materials and methods
Microarray analysis
Total RNA (10 μg) was extracted from T47D cells as detailed below. GeneChip Two-Cycle Target Labeling and Control reagents (Affymetrix, Santa Clara, CA) were used to convert total RNA to complementary DNA (cDNA). cDNA was used as a template to generate biotinylated complementary RNA (cRNA). We used a GeneChip Instrument System (Affymetrix) to fragment the biotinylated cRNA targets and to hybridise the targets to a Breast Cancer DSA™ microarray (Almac Diagnostics, Craigavon, United Kingdom), which contains 60,856 probe sets and encodes approximately 60,000 transcripts that have been shown to be expressed in either breast cancer or normal tissue. Hybridised cRNA was stained, washed and scanned using the GeneChip Scanner 7G (Affymetrix) according to the manufacturer’s instructions. Analysis was performed as previously described [14].
Bioinformatic analysis
The raw data from an independent cohort of 17 BRCA1-mutant and 14 matched sporadic breast tumour samples were accessed through the following link (http://www.ebi.ac.uk/arrayexpress/experiments/E-TABM-366). For both cell line and tumour derived gene expression profiles, data were normalised using the Rosetta Error Model [15]. The tumour data were filtered using the following criteria: (1) Student’s unpaired t-test with statistical significance defined as a P value less than 0.05; (2) at least a 1.5-fold difference in gene expression between BRCA1-mutant and sporadic samples. The cell line data were filtered using the following criteria: (1) Student’s unpaired t-test with statistical significance defined as a P value less than 0.01; (2) at least a 1.5-fold difference in gene expression between wild type and BRCA1 siRNA knockdown samples. Hierarchical agglomerative clustering heat maps of cell line and tumour derived gene expression data were generated using Pearson correlation and average linkage.
Cell lines and siRNA transfection
HCC-EV and HCC-BR cell lines were generated and maintained as previously described [16]. T47D, MDA468, MDA231, MDA361 and MDA453 cell lines were maintained in RPMI supplemented with 10% foetal calf serum, 1 mM sodium pyruvate and 50 μg/ml penicillin/streptomycin. MCF7, ZR-75 and BT-474 cell lines were maintained in DMEM supplemented with 10% foetal calf serum, 1 mM sodium pyruvate and 50 μg/ml penicillin/streptomycin. SUM-149 cells were maintained as previously described [17].
siRNA transfection
T47D cells were transfected with BRCA1-specific siRNA;
-
BRCA1 #2 5′ CAGGAAATGGCTGAACTAGAA 3′
-
BRCA1 #3 5′ ACCATACAGCTTCATAAATAA 3′
or scrambled control oligonucleotides using oligofectamine (Invitrogen) as previously described. All other siRNA oligonucleotides were purchased from Dharmacon and target sequences are as follows:
-
BRCA1 #1 5′r(GUGUGCAGCUGAGAGGCA)(dTdT)3′,
-
c-Myc 5′r(CGACAGCAGCUCGCCCAAG)(dTdT)3′,
-
ERα 5′r(UCAUCGCAUUCCUUGCAAA)(dTdT)3′,
-
NFκB 5′r(GCCCUAUCCCUUUACGUCA)(dTdT)3′
and Sp1 5′r(GCCAAUAGCUACUCAACUA)(dTdT)3′.
All siRNA transfections were performed using 100 nM of each oligonucleotide, and cells were harvested 72 h after transfection.
RNA extraction and reverse transcription
Total RNA was extracted from cell lines using RNA Stat60 (Tel-Test, Inc) according to the manufacturer’s instructions. RNA (2 μg) was then reverse transcribed using random primers and MMLTV reverse transcriptase as described by the manufacturer (Invitrogen).
Quantitative real-time PCR
Primer sequences for KRT5, KR17, CDH3, were designed using Primer3 and NetPrimer design software. Quantitative real-time PCR was performed using SYBR Green (Finnzymes) according to the manufacturer’s instructions on the DNA Engine Opticon® 2 continuous fluorescence detection system (MJ Research). Relative mRNA expression levels were quantified for each gene using the Opticon® Monitor v2.02 software according to the standard curve method and by normalisation to the endogenous reference gene, Actin. All results obtained are the mean of triplicate experiments ± SEM.
Immunoprecipitation and Western blot assays
Immunoprecipitation and Western blot assays were performed as previously described [16] using BRCA1-AB1 (Oncogene Research Products), KRT5 (Chemicon), KRT17 (Abcam), CDH3 (BD Biosciences), c-Myc (C-19), ERα (A-20), NFκB (C-20), Sp1 (IC6) and GAPDH antibodies (all from Santa Cruz).
Chromatin immunoprecipitation assays
ChIP and Re-ChIP assays were performed as previously described [14] using BRCA1-AB1 (Oncogene Research Products), c-Myc (N262), ERα (HC-20), NFκB (C-20) and Sp1 (IC6) antibodies (all from Santa Cruz). PCR amplification was performed over 30 cycles of 95°C for 10 s, 55°C for 20 s, 72°C for 20 s using primers designed to the specific promoter regions of KRT5 (forward 5′ GTGCTGCAAGGCAAGGTTAT 3′ and reverse 5′ GAATGATTAAGTGGGCTGGG 3′), KRT17 (forward 5′ GGCTCTCGGTCTCCTCTTTC 3′ and reverse 5′ TCTTTCACCCCACACTGCTC 3′) and CDH3 (forward 5′ ACCCTTTGGCACCACTACAG 3′ and reverse 5′ GACCGCTTCTTCCATTTCAC 3′). PCR was also performed using primers designed to nonspecific promoter regions 1 kb upstream of the transcription start site of each of the genes. Two sets of primers were designed per gene, KRT5 (forward A, 5′ AGCCCTCTGGTGAGAAGTCA, forward B 5′ TGGGTCTGGTCCTGTTCTTC and reverse A 5′ GAAGAACAGGACCAGACCCA, reverse B 5′ CCTGAGTCCCTGTCACCTGT), KRT17 (forward A, 5′ CCTGAGTCCCTGTCACCTGT, forward B 5′ TGGCATTGATGAGTGAGAGG and reverse A 5′ CCTCTCACTCATCAATGCCA, reverse B 5′ AGCCGAGAGACATTCCTCAA) and CDH3 (forward A, 5′ CATGCTAGGCCTGAGAGAGC, forward B 5′ CCATGCTAGGCCTGAGAGAG and reverse A 5′ ATTCTTTGACCTTCCAGGGC, reverse B 5′ AATCTATCCGTCCTCCTCCC). All PCR amplification reactions were then electrophoresed on 2% agarose gels.
Results
Modulation of BRCA1 results in altered expression of basal genes
We utilised a breast cancer disease specific microarray, encoding approximately 60,000 transcripts to identify genes that were regulated by BRCA1 (14). Triplicate independent siRNA experiments were performed. Inhibition of endogenous BRCA1 was confirmed by Western blot analysis prior to array-based expression profiling (Fig. 1a(i)). siRNA-mediated inhibition of endogenous BRCA1 in T47D cells identified 422 transcripts that were upregulated and 453 transcripts that were downregulated (>1.5-fold) relative to T47D cells transfected with a scrambled control siRNA (P = 1.0e-5) (Fig. 1a(ii)). Further analysis of the data revealed that KRT17 and CDH3, both well-characterised basal markers, were upregulated approximately 6-fold and 1.5-fold, respectively, in the BRCA1 knockout cells (Fig. 1a(ii) and supplementary data). This was particularly interesting since it had previously been reported that both KRT17 and CDH3 protein levels are upregulated in primary BRCA1 mutant breast tumours consistent with the reported link between BRCA1 mutation and basal-like breast cancer phenotype. We extended this analysis to evaluate KRT17 and CDH3 expression in a panel of BRCA1 mutant compared to matched sporadic breast tumours [14]. Reanalysis of this data confirmed the increase in the expression of both CDH3 and KRT17 in BRCA1 mutant tumours relative to sporadic controls (Fig. 1b).
Microarray-based expression profiling. a(i) Western blot analysis confirming siRNA inhibition of BRCA1 protein expression levels in the luminal T47D breast cancer cell line (lane 2) when compared to scrambled oligonucleotide control (lane 1). A GAPDH loading control confirming equal loading is included. (ii) Microarray analysis comparing gene expression profiles between T47D breast cancer cells treated with either a scrambled control oligonucleotide (control A, B C) or a siRNA specific to BRCA1 (BRCA1-A, B, C). The 2-Dimensional agglomerative cluster tree displays 875 probe sets that were differentially expressed between wild type and BRCA1 siRNA knockdown samples according to the following selection criteria: (i) at least a 1.5-fold difference (ii) P value for difference in expression less than .01 (unpaired t-test). Red indicates higher expression and green indicates lower expression. b Microarray analysis comparing gene expression profiles between 17 BRCA1 mutant breast tumours (M_01–M_17) and 14 sporadic breast cancers (W_01–W_14) that were matched to each other by stage and grade. The 875 probe sets identified from the cell line data analysis (Fig. 1b) were mapped to the tumour data and further filtered using the following criteria: (i) at least a 1.5-fold difference between BRCA1-mutant and sporadic samples (ii) P value for difference in expression less than 0.01 (unpaired t-test) . The 2-dimensional agglomerative cluster tree displays the resulting 144 probesets. Both CDH3 and KRT17 are highlighted as being overexpressed in the BRCA1 mutant compared to sporadic control tumours. Red indicates higher expression and green indicates lower expression
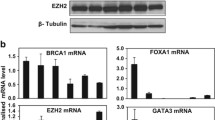
Real-time-PCR (RT-PCR) following siRNA-mediated abrogation of endogenous BRCA1 expression in T47D cells using three independent BRCA1 siRNA oligonucleotides confirmed the microarray data (Fig. 2a(i)) (KRT 17 P = 0.011, CDH3 P = 0.024). We extended the RT-PCR analysis to include KRT5, an additional basal marker previously shown to be overexpressed in BRCA1-linked breast cancers [9, 18]. Consistent with that observed for CDH3 and KRT17, inhibition of endogenous BRCA1 in T47D cells also resulted in upregulation of KRT5 mRNA levels (Fig. 2a(i) (P = 0.038) see also supplementary data). We also carried out RT-PCR in the BRCA1 reconstituted HCC1937 cell line (HCC-BR) relative to controls (HCC-EV) and observed a marked reduction in expression of all three genes; CDH3 (P = 8.0 E -4), KRT5 (P = 0.055) and KRT17 (P = 0.029) in the BRCA1 reconstituted cells relative to vector-transfected controls (Fig. 2a(ii)). In addition, we examined the protein expression levels for all three basal markers in both theT47D and HCC1937 cell line models (Fig. 2 b(i) and (ii)). Analysis of CDH3, KRT 5 and KRT17 protein expression showed an inverse relationship with BRCA1 expression consistent with that observed by RT-PCR.
Validation of target genes in a panel of Breast Cancer cell lines. a(i) Quantitative real-time PCR (qRT-PCR) analysis demonstrating increase mRNA expression levels of the basal genes (KRT17, KRT5 and CDH3) in T47D cells transfected with BRCA1 siRNA. Results were obtained from three independent experiments from three independent siRNA oligonucleotides. All values shown for qRT-PCR were normalised to actin levels and expressed as fold change in expression compared to scrambled oligonucleotide control. Statistically, significant differences were determined using Student’s t-test; * P < 0.05; ** P < 0.01; *** P < 0.001. (ii) qRT-PCR analysis demonstrating a reduction in mRNA expression levels of the basal genes (KRT17, KRT5 and CDH3) in BRCA1 reconstituted HCC1937 cells. Results were obtained from three independent experiments. All values shown for qRT-PCR were normalised to actin levels and expressed as fold change in expression compared to scrambled oligonucleotide control. Statistically, significant differences were determined using Student’s t-test; * P < 0.05; ** P < 0.01; *** P < 0.001. b(i) Western blot analysis confirming knockdown of BRCA1 protein expression after siRNA (lane 2) when compared to scrambled oligonucleotide control (lane 1) in T47D cells. Protein expression of basal genes CDH3, KRT5 and KRT17 was also analysed from the same protein lysates. GAPDH loading controls are included in each western panel and confirm equal loading. (ii) Western blot analysis confirming reconstitution of wild-type BRCA1 expression (lane 2) in BRCA1-mutated HCC1937 breast cancer cells when compared to vector-transfected control (lane 1). Protein expression of basal genes CDH3, KRT5 and KRT17 was also analysed from the same protein lysates. GAPDH loading controls are included in each western panel and confirm equal loading. c(i) qRT-PCR analysis demonstrating the endogenous levels of expressed BRCA1 mRNA in a panel of breast cancer cell lines. All values shown for qRT-PCR were normalised to actin levels. (ii) Western blot analysis confirming stable overexpression of BRCA1 in the basal-like breast cancer cell line MDA468 (lane 2) when compared to vector only control cells (lane 1). Protein expression of basal genes CDH3, KRT5 and KRT17 was also analysed from the same protein lysates. A GAPDH loading control confirming equal loading is included
We also generated a third isogenic cell line model system utilising the MDA468 breast cancer cell line. MDA468 cells have been previously described as triple negative for ERα, PR and HER2/Neu expression and were characterised as a basal-like breast cancer cell line [19]. Interestingly, this cell line has also previously been characterised as BRCA1 wild type by sequence analysis [17]. We evaluated the expression level of endogenous BRCA1 in these cells by RT-PCR relative to a panel of other breast cancer cell lines and confirmed relatively low levels of BRCA1 mRNA expression (Fig. 2c(i)). We stably expressed exogenous wild-type BRCA1 in these cells and generated corresponding isogenic control cells by transfecting with an empty vector. Consistent with a basal-like phenotype, the vector-transfected MDA468 cells (MDA-468-EV) expressed high levels of KRT5, KRT17 and CDH3. In contrast, overexpression of exogenous BRCA1 in the MDA-468-BR cells resulted in a marked reduction in expression of all three of these basal markers (Fig. 2c(ii)).
Taken together, these results demonstrate that modulation of BRCA1 expression alters the expression of three genes associated with the basal-like breast cancer phenotype at both the mRNA and protein levels. This would suggest that BRCA1 is involved in transcriptional regulation of these genes.
BRCA1 and c-Myc form a complex on the promoter of CDH3
In order to investigate the transcriptional role played by BRCA1, we carried out chromatin immunoprecipitation (ChIP) assays to evaluate whether BRCA1 is associated with the promoters of CDH3, KRT17 and KRT5. We initially used the BRCA1 mutant HCC-EV (−) and BRCA1 reconstituted HCC-BR (+) cells (Fig. 3a) and immunoprecipitated for RNA polymerase II (Pol II) using an antibody that recognises both the transcriptionally active and inactive forms (lanes 3 and 4); BRCA1, using an antibody that recognises truncated BRCA1 (lanes 5 and 6); and matched IgG as a negative control (lanes 7 and 8). The immunoprecipitated DNA was analysed following PCR amplification using primers specific to the immediate promoter regions of CDH3, KRT17 and KRT5 (panels a, d and g, respectively) and to two regions 1 kb or more upstream of the transcription start site (panels b, c, e, f, h and i, respectively). Enrichment of BRCA1 on the immediate promoter region but not at the upstream promoter regions demonstrates specific protein-DNA affinity. The assays therefore confirm recruitment of functional BRCA1 to the promoters of CDH3, KRT17 and KRT5 in the BRCA1 reconstituted HCC-BR cells but not in the BRCA1 transcriptionally inactive HCC-EV cells. Similarly, we confirmed recruitment of BRCA1 to the immediate promoter of all three basal genes in T47D breast cancer cells treated with a scrambled control oligonucleotides (+) but not in cells treated with a BRCA1 specific siRNA (−) (Fig. 3b). As a positive control, we also confirmed enrichment for BRCA1 on the ERα promoter from both cell lines (Fig. 3a, b, panel f).
BRCA1 recruitment to the promoters of CDH3, KRT17 and KRT5. a Chromatin Immunoprecipitation (ChIP) in HCC-EV (−) and HCC-BR (+) cells, and (b) in T47D cells treated with either scrambled control (+) or BRCA1 specific siRNA oligonucleotide (−). Recruitment of RNA Polymerase II (Pol II) (lanes 3 and 4) and BRCA1 (lanes 5 and 6) to basal gene promoters was assessed using primers specific to the CDH3, KRT17 and KRT5 promoters. One percent of total input DNA was used as a loading control (lanes 1 and 2), and isotype-matched IgG was used as an internal control for the immunoprecipitation (lanes 7 and 8). Panels a, d and g represent the immediate promoter regions, all other panels represent upstream promoter sequences. Panel j is an internal control to show efficient immunoprecipitation of BRCA1 on the ERα promoter
To date, there is significant evidence demonstrating that BRCA1 can function as either a transcriptional co-activator or a co-repressor by interacting with a number of sequence-specific DNA binding transcription factors [20]. We therefore examined the promoters of CDH3, KRT17 and KRT5 for consensus-binding sites of transcription factors previously known to associate with BRCA1. Promoter analysis using Alibaba and Ensembl genome browsers revealed the presence of common binding sites for c-Myc, Sp1, NFκB and ERα.
In order to explore the requirement of these transcription factors for the regulation of CDH3, we carried out siRNA-based experiments targeted against c-Myc, Sp1, NFκB and ERα in T47D cells and evaluated the effect on CDH3 expression by Western blot analysis (Fig. 4a). SiRNA-mediated inhibition of endogenous c-Myc in T47D cells resulted in a marked induction of CDH3 (Fig. 4a (i)), this is consistent with the concept that BRCA1 and c-Myc can form a transcriptional co-repressor complex. In contrast, inhibition of endogenous Sp1, NFκB or ERα resulted in no significant increase in CDH3 expression (Fig. 4a (ii, iii and iv, respectively)).
BRCA1 is forming two independent transcriptional complexes on the promoter of CDH3. a Western blot analysing effect of abrogation of transcription factors on CDH3 expression. T47D cells were transfected with either scrambled control oligonucleotides (lane 1) or siRNA oligonucleotides to (i) c-Myc, (ii) Sp1 (iii) NFκB and (iv) ER α (lane 2 in each panel, respectively). Seventy-two hours following transfection cells were harvested, and CDH3 expression was analysed by Western blot. GAPDH loading controls are included in each western panel and confirm equal loading. b ChIP–re-ChIP screening for interactions between factors recruited to the CDH3 promoter. DNA-protein complexes prepared from T47D cells were subjected to ChIP using antibodies for Pol II (lanes 2–7) and BRCA1 (lanes 8–13). The immunoprecipitated complexes (protein and DNA) were eluted from the beads and then subjected to a further immunoprecipitation with either Pol II (lanes 2 and 8), BRCA1 (lanes 3 and 9), c-Myc (lanes 4 and 10), ERα (lanes 5 and 11), NFκB (lanes 6 and 12) and Sp-1 (lanes 7 and 13) specific antibodies. One percent of total input DNA was used as a loading control (lane 1), and isotype-matched IgG was used as an internal control for the immunoprecipitation (lanes 14–19). c ChIP–re-ChIP screening for interactions between Pol II, BRCA1, c-Myc and Sp1 transcription factors on the CDH3 promoter. DNA-protein complexes were prepared as previously described and subjected to ChIP using antibodies for Pol II (lanes 1 and 2), BRCA1 (lanes 3 and 4) c-Myc (lanes 5 and 6), Sp1 (lanes 7 and 8) and HA as an internal control (lanes 9 and 10). The immunoprecipitated complexes were eluted from the beads, divided equally and subjected to a further round of immunoprecipitation for Pol II (pane 1), BRCA1 (panel 2), c-Myc (panel 3) or Sp1 (panel 4). The immunoprecipitated complexes were eluted from the beads, DNA was purified, and PCR specific for CDH3 promoter was performed. d Co-Immunoprecipitation Western blot analysis demonstrating the association of endogenous BRCA1 with c-Myc and Sp-1 in T47D cells. About 1 mg of protein lysate was immunoprecipitated with BRCA1 (lane 2), c-Myc (lane 3) or Sp-1 (lane 4) or IgG (lane 5) specific antibodies and immunoblotted for either BRCA1, c-Myc or Sp-1. About 10% of the input lysate was loaded as a positive control (lane 1). e ChIP from T47D cells treated with either scrambled control (+) or c-Myc-specific siRNA oligonucleotide (−). Recruitment of RNA Polymerase II (Pol II) (lanes 2 and 3), BRCA1 (lanes 4 and 5) and c-Myc (lanes 7 and 8) to the promoter of CDH3 was analysed. One percent of total input DNA was used as a loading control and isotype-matched IgG was used as an internal control for the immunoprecipitation (lanes 9 and 10)
To investigate the possibility of direct interactions between the transcription factors and BRCA1, we performed a re-ChIP assay (Fig. 4b). This assay involves consecutive immunoprecipitations and therefore allows the identification of protein complexes associated with specific promoters. By using the T47D cell line model, we immunoprecipitated for Pol II (as a positive control), BRCA1 and isotype-matched IgG (as a negative control). The immunoprecipitated complexes were then subjected to a second immunoprecipitation for Pol II (P), BRCA1 (B), c-Myc (C), ERα (E), NFκB (N) and Sp1 (S). The precipitated protein and DNA complexes were eluted and the DNA purified. This DNA was then analysed by PCR for CDH3 promoter amplification. The re-ChIP patterns demonstrate an interaction between Pol II and BRCA1 as expected (Fig. 4b, lanes 3 and 8), they also demonstrate an interaction between BRCA1 and c-Myc (lane 10) and between BRCA1 and Sp1 (lane 13).
In order to analyse whether BRCA1 is part of a large multi-protein transcriptional complex with both c-Myc and SP1 or whether it is forming two independent complexes involving each factor, we performed an additional re-ChIP experiment. We initially immunoprecipitated for Pol II, BRCA1, c-Myc, Sp1 and control IgG from T47D cells, which had been treated with either a scrambled control(+) or a BRCA1-specific siRNA oligonucleotides (−). We then performed a second round of immunoprecipitations with each immunoprecipitated complex. The precipitated protein and DNA complexes were eluted and the DNA purified. The DNA was then analysed by PCR for CDH3 promoter amplification (Fig. 4c). The data confirm that BRCA1 is interacting with Pol II, c-Myc and Sp1 on the promoter of CDH3 (lane 3, panels; 1, 3 and 4, respectively). It also demonstrates that BRCA1 expression does not affect Pol II, c-Myc or Sp1 recruitment as loss of BRCA1 by siRNA does not affect c-Myc or Sp1 binding to the promoter of CDH3 (lane 2 panel 1, lane 6 panel 3 and lane 8 panel 4, respectively). The data would also propose that BRCA1 is forming two distinct transcriptional complexes as c-Myc does not directly complex with Sp1 (lane 5 panel 4 and lane 7 panel 3).
To evaluate this, further we carried out co-immunoprecipitation assays from T47D cells (Fig. 4d). We found that BRCA1 co-immunoprecipitated with both c-Myc and Sp1 (lane 2); however, there was no observed direct interaction between c-Myc and Sp1 (lanes 3 and 4). This data is in agreement with our re-ChIP and would suggest that BRCA1 is present in two separate complexes on the CDH3 promoter.
The data also suggest that it is the c-Myc/ BRCA1 complex, which is repressing CDH3 expression and not the BRCA1/Sp1 complex (See Fig. 4a). We therefore wanted to ascertain whether c-Myc is required for BRCA1 recruitment to the CDH3 promoter. We performed a further ChIP experiment with T47D cells treated with either a scrambled control (+) or a c-Myc specific siRNA (−) oligonucleotides and immunoprecipitated for Pol II, BRCA1, c-Myc and control IgG (Fig. 4e). The data show that endogenous c-Myc is essential for BRCA1 recruitment to the CDH3 promoter (lane 4 compared to lane 3).
In order to confirm the requirement for BRCA1 and c-Myc in the transcriptional regulation of CDH3, we inhibited BRCA1, c-Myc and Sp1 expression by siRNA and performed RT-PCR analysis in T47D cells (Fig. 5a). Abrogation of both BRCA1 and c-Myc lead to increased mRNA expression of CDH3 (P = 0.02, P = 0.04, respectively), Sp1 knockdown had little effect. We further confirmed the requirement for BRCA1 and c-Myc by performing additional siRNA experiments either as single or as double siRNA transfections and examining CDH3 protein expression in T47D cells by Western blot (Fig. 5b). Reduced expression of either BRCA1 or c-Myc resulted in the re-expression of CDH3 (lanes 2 and 3). We extended these studies to reanalyse the effect of BRCA1 and c-Myc knockdown on the expression of KRT17 and KRT5. Consistent with that observed for CDH3 abrogation of either BRCA1 or c-Myc resulted in re-expression of both KRT 17 and KRT5. This data suggests that both proteins must be present and functional on the promoters of CDH3, KRT17 and KRT5 in order for repression to occur. We therefore propose that it is the BRCA1-c-Myc complex that is functioning to repress expression of these three basal markers.
BRCA1 and c-Myc are necessary to mediate repression of CDH3, KRT17 and KRT5. a qRT-PCR analysis demonstrating upregulation of mRNA expression levels of CDH3 in T47D cells treated with siRNA specific to BRCA1, c-Myc or Sp1. All values shown for qRT-PCR were normalised to actin levels and expressed as fold change in expression compared to scrambled oligonucleotide control. Statistically, significant differences were determined using Student’s t-test; * P < 0.05; ** P < 0.01; *** P < 0.001. b Western blot analysis confirming siRNA knockdown of BRCA1 (lane 2), siRNA knockdown of c-Myc (lane 3) and double siRNA knockdown of both BRCA1 and c-Myc (lane 4) protein expression levels in T47D breast cancer cells (lane 2) when compared to scrambled oligonucleotide control (lane 1). Abrogation of both c-Myc and BRCA1 expression in T47D cells results in re-expression of CDH3, KRT17 and KRT5. GAPDH loading controls are included in each western panel and confirm equal loading
Discussion
This study when viewed with other published data leads us to hypothesise that loss of BRCA1 expression may contribute to the development of basal-like epithelial breast tumours. We have shown in vitro that reconstitution of BRCA1 in the mutant HCC1937 breast cell line results in reduced expression of three genes associated with the basal-like phenotype; CDH3, KRT17 and KRT5. We have also shown that expression of these genes in the luminal breast cancer cell line, T47D, can be abrogated by siRNA knockdown of endogenous BRCA1. These data are consistent with several immunohistochemical studies that have shown that BRCA1-associated tumours are more likely to express genes consistent with a basal-like epithelial phenotype [4–6, 21]. For example, 88% of BRCA1-mutated tumours were shown to express KRT 5, and tumours staining positive for CDH3 were reported to be seven times more likely to arise from BRCA1 mutant patients when compared to sporadic breast cancer patients [18]. Interestingly, BRCA1 mRNA expression levels have also been reported to be twofold lower in sporadic basal-like epithelial breast cancers relative to matched controls [22]. The mechanism through which BRCA1 is epigenetically inactivated in breast cancer is still unclear although promoter methylation and repression by ID4 may play a role [22]. These observations support the concept that breast tumours may acquire a BRCA1-like phenotype either by mutation or by epigenetic inactivation of BRCA1. Consistent with these studies, the MDA468 cell line utilised in this study has previously been described as a basal-like breast cancer cell line expressing low levels of endogenous wild-type BRCA1 [17]. We demonstrated that overexpression of exogenous BRCA1 in these cells resulted in the repression of CDH3, KRT17 and KRT5 suggesting that in addition to BRCA1 mutational status, the overall expression levels of wild-type BRCA1 may also be important in the regulation of defined basal markers.
We have presented data to indicate that BRCA1 is a component of two distinct transcriptional complexes on the CDH3 promoter. The complex involving BRCA1 and Sp1 does not appear to play a crucial role in the repression of CDH3. Previous reports in the literature have shown that BRCA1 is capable of repressing the transcriptional potential of Sp1 on the insulin-like growth factor 1 receptor (IGF-IR) promoter, by preventing Sp1 recruitment to the promoter [13, 23, 24]. In the current study, it appears that the BRCA1/Sp1 complex is not involved in active repression of CDH3 under the conditions studied. It is possible, however, that the BRCA1/Sp1 complex may be involved in regulation of CDH3 following activation of as yet unknown signalling cascades.
In contrast, the data presented here does suggest that the BRCA1 is part of a transcriptional complex that includes c-Myc and Pol II, which mediates repression of defined basal markers such as CDH3. This finding is consistent with previous observations from our laboratory where we have reported that BRCA1 and c-Myc form a complex that inhibits expression of the DNA damage inducible gene, psoriasin [13]. Previous studies have reported that the ability of BRCA1 to interact with c-Myc requires the helix-loop-helix (HLH) region of c-Myc, the motif known to be responsible for Myc–Max dimerisation [24]. BRCA1 does not, however, interact with Max, so it is possible that BRCA1 may compete with Max for binding to the c-Myc HLH region [24]. Alternatively, BRCA1 has also been shown to reside in a trimeric complex with c-Myc and an adaptor molecule, N-Myc-Interacting protein (Nmi) [11]. The ability of BRCA1 to repress c-Myc-dependent hTERT promoter activity was dependent on its association with Nmi [11].
The observation that c-Myc may be preferentially amplified in BRCA1-deficient tumours is intriguing and suggests that amplification of c-Myc in a BRCA1-mutated background may confer a selective advantage to the tumour. This is in direct contrast to the correlation between BRCA1 mutations and Her2 amplification, which is a very rare event in breast cancer [25]. It may be that amplification of c-Myc in this background represents a particularly efficient means of driving proliferation, which is normally held in check by functional BRCA1. It will therefore be important in the future to more fully understand the differences in the transcriptional programme regulated by c-Myc in a BRCA1 wild type compared to a BRCA1-deficient background.
References
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, Zhu SX, Lonning PE, Borresen-Dale AL, Brown PO, Botstein D (2000) Molecular portraits of human breast tumours. Nature 406:747–752
Foulkes WD, Chappuis PO, Wong N, Brunet JS, Vesprini D, Rozen F, Yuan ZQ, Pollak MN, Kuperstein G, Narod SA, Begin LR (2000) Primary node negative breast cancer in BRCA1 mutation carriers has a poor outcome. Ann Oncol 11:307–313
Foulkes WD, Stefansson IM, Chappuis PO, Begin LR, Goffin JR, Wong N, Trudel M, Akslen LA (2003) Germline BRCA1 mutations and a basal epithelial phenotype in breast cancer. J Natl Cancer Inst 95:1482–1485
Foulkes WD, Brunet JS, Stefansson IM, Straume O, Chappuis PO, Begin LR, Hamel N, Goffin JR, Wong N, Trudel M, Kapusta L, Porter P, Akslen LA (2004) The prognostic implication of the basal-like (cyclin E high/p27 low/p53+/glomeruloid-microvascular-proliferation+) phenotype of BRCA1-related breast cancer. Cancer Res 64:830–835
Arnes JB, Brunet JS, Stefansson I, Begin LR, Wong N, Chappuis PO, Akslen LA, Foulkes WD (2005) Placental cadherin and the basal epithelial phenotype of BRCA1-related breast cancer. Clin Cancer Res 11:4003–4011
Palacios J, Honrado E, Osorio A, Cazorla A, Sarrio D, Barroso A, Rodriguez S, Cigudosa JC, Diez O, Alonso C, Lerma E, Dopazo J, Rivas C, Benitez J (2005) Phenotypic characterization of BRCA1 and BRCA2 tumors based in a tissue microarray study with 37 immunohistochemical markers. Breast Cancer Res Treat 90:5–14
Lakhani SR, Jacquemier J, Sloane JP, Gusterson BA, Anderson TJ, van de Vijver MJ, Farid LM, Venter D, Antoniou A, Storfer-Isser A, Smyth E, Steel CM, Haites N, Scott RJ, Goldgar D, Neuhausen S, Daly PA, Ormiston W, McManus R, Scherneck S, Ponder BA, Ford D, Peto J, Stoppa-Lyonnet D, Easton DF et al (1998) Multifactorial analysis of differences between sporadic breast cancers and cancers involving BRCA1 and BRCA2 mutations. J Natl Cancer Inst 90:1138–1145
Lakhani SR, O’Hare MJ, Ashworth A (2001) Profiling familial breast cancer. Nat Med 7:408–410
Sorlie T, Tibshirani R, Parker J, Hastie T, Marron JS, Nobel A, Deng S, Johnsen H, Pesich R, Geisler S, Demeter J, Perou CM, Lonning PE, Brown PO, Borresen-Dale AL, Botstein D (2003) Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 100:8418–8423
Grushko TA, Dignam JJ, Das S, Blackwood AM, Perou CM, Ridderstrale KK, Anderson KN, Wei MJ, Adams AJ, Hagos FG, Sveen L, Lynch HT, Weber BL, Olopade OI (2004) MYC is amplified in BRCA1-associated breast cancers. Clin Cancer Res 10:499–507
Li H, Lee TH, Avraham H (2002) A novel tricomplex of BRCA1, Nmi, and c-Myc inhibits c-Myc-induced human telomerase reverse transcriptase gene (hTERT) promoter activity in breast cancer. J Biol Chem 277:20965–20973
Li Y, Pan J, Li JL, Lee JH, Tunkey C, Saraf K, Garbe JC, Whitley MZ, Jelinsky SA, Stampfer MR, Haney SA (2007) Transcriptional changes associated with breast cancer occur as normal human mammary epithelial cells overcome senescence barriers and become immortalized. Mol Cancer 6:7
Kennedy RD, Gorski JJ, Quinn JE, Stewart GE, James CR, Moore S, Mulligan K, Emberley ED, Lioe TF, Morrison PJ, Mullan PB, Reid G, Johnston PG, Watson PH, Harkin DP (2005) BRCA1 and c-Myc associate to transcriptionally repress psoriasin, a DNA damage-inducible gene. Cancer Res 65:10265–10272
Hosey AM, Gorski JJ, Murray MM, Quinn JE, Chung WY, Stewart GE, James CR, Farragher SM, Mulligan JM, Scott AN, Dervan PA, Johnston PG, Couch FJ, Daly PA, Kay E, McCann A, Mullan PB, Harkin DP (2007) Molecular basis for estrogen receptor alpha deficiency in BRCA1-linked breast cancer. J Natl Cancer Inst 99:1683–1694
Weng L, Dai H, Zhan Y, He Y, Stepaniants SB, Bassett DE (2006) Rosetta error model for gene expression analysis. Bioinformatics 22:1111–1121
Quinn JE, Kennedy RD, Mullan PB, Gilmore PM, Carty M, Johnston PG, Harkin DP (2003) BRCA1 functions as a differential modulator of chemotherapy-induced apoptosis. Cancer Res 63:6221–6228
Elstrodt F, Hollestelle A, Nagel JH, Gorin M, Wasielewski M, van den Ouweland A, Merajver SD, Ethier SP, Schutte M (2006) BRCA1 mutation analysis of 41 human breast cancer cell lines reveals three new deleterious mutants. Cancer Res 66:41–45
Lakhani SR, Reis-Filho JS, Fulford L, Penault-Llorca F, van der Vijver M, Parry S, Bishop T, Benitez J, Rivas C, Bignon YJ, Chang-Claude J, Hamann U, Cornelisse CJ, Devilee P, Beckmann MW, Nestle-Kramling C, Daly PA, Haites N, Varley J, Lalloo F, Evans G, Maugard C, Meijers-Heijboer H, Klijn JG, Olah E, Gusterson BA, Pilotti S, Radice P, Scherneck S, Sobol H, Jacquemier J, Wagner T, Peto J, Stratton MR, McGuffog L, Easton DF (2005) Prediction of BRCA1 status in patients with breast cancer using estrogen receptor and basal phenotype. Clin Cancer Res 11:5175–5180
Neve RM, Chin K, Fridlyand J, Yeh J, Baehner FL, Fevr T, Clark L, Bayani N, Coppe JP, Tong F, Speed T, Spellman PT, DeVries S, Lapuk A, Wang NJ, Kuo WL, Stilwell JL, Pinkel D, Albertson DG, Waldman FM, McCormick F, Dickson RB, Johnson MD, Lippman M, Ethier S, Gazdar A, Gray JW (2006) A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10:515–527
Mullan PB, Quinn JE, Harkin DP (2006) The role of BRCA1 in transcriptional regulation and cell cycle control. Oncogene 25:5854–5863
Laakso M, Loman N, Borg A, Isola J (2005) Cytokeratin 5/14-positive breast cancer: true basal phenotype confined to BRCA1 tumors. Mod Pathol 18:1321–1328
Turner NC, Reis-Filho JS, Russell AM, Springall RJ, Ryder K, Steele D, Savage K, Gillett CE, Schmitt FC, Ashworth A, Tutt AN (2007) BRCA1 dysfunction in sporadic basal-like breast cancer. Oncogene 26:2126–2132
Abramovitch S, Glaser T, Ouchi T, Werner H (2003) BRCA1-Sp1 interactions in transcriptional regulation of the IGF-IR gene. FEBS Lett 541:149–154
Wang Q, Zhang H, Kajino K, Greene MI (1998) BRCA1 binds c-Myc and inhibits its transcriptional and transforming activity in cells. Oncogene 17:1939–1948
Palacios J, Robles-Frias MJ, Castilla MA, Lopez-Garcia MA, Benitez J (2008) The molecular pathology of hereditary breast cancer. Pathobiology 75:85–94
Author information
Authors and Affiliations
Corresponding author
Additional information
Funding bodies
Julia Gorski and Niamh Buckley: R&D Office NI, Colin James: Cancer Research UK, Jennifer Quinn: Breast Cancer Campaign, Gail Stewart: Medical Research Council, Kieran Crosbie Staunton: European Social Fund, Richard Wilson: Queens University Belfast, Richard Kennedy and Fionnuala McDyer: Almac Diagnostics, Paul Mullan: Action Cancer N. I., Paul Harkin: Breast Cancer Campaign and Cancer Research UK.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gorski, J.J., James, C.R., Quinn, J.E. et al. BRCA1 transcriptionally regulates genes associated with the basal-like phenotype in breast cancer. Breast Cancer Res Treat 122, 721–731 (2010). https://doi.org/10.1007/s10549-009-0565-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-009-0565-0