Abstract
Purpose To explore the relationship of regional cerebral blood flow and metabolism with cognitive function and past exposure to chemotherapy for breast cancer.
Patients and methods Subjects treated for breast cancer with adjuvant chemotherapy remotely (5–10 years previously) were studied with neuropsychologic testing and positron emission tomography (PET), and were compared with control subjects who had never received chemotherapy. [O-15] water PET scans was acquired during performance of control and memory-related tasks to evaluate cognition-related cerebral blood flow, and [F-18] fluorodeoxyglucose (FDG) PET scans were acquired to evaluate resting cerebral metabolism. PET scans were analyzed by statistical parametric mapping and region of interest methods of analysis.
Results During performance of a short-term recall task, modulation of cerebral blood flow in specific regions of frontal cortex and cerebellum was significantly altered in chemotherapy-treated subjects. Cerebral activation in chemotherapy-treated subjects differed most significantly from untreated subjects in inferior frontal gyrus, and resting metabolism in this area correlated with performance on a short-term memory task previously found to be particularly impaired in chemotherapy-treated subjects. In examining drug-class specific effects, metabolism of the basal ganglia was significantly decreased in tamoxifen + chemotherapy-treated patients compared with chemotherapy-only breast cancer subjects or with subjects who had not received chemotherapy, while chemotherapy alone was not associated with decreased basal ganglia activity relative to untreated subjects.
Conclusion Specific alterations in activity of frontal cortex, cerebellum, and basal ganglia in breast cancer survivors were documented by functional neuroimaging 5–10 years after completion of chemotherapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the most common cancer in women, with over 211,000 new cases per year [1]. As a result of mammographic screening, there has been a substantial shift to earlier stage at diagnosis, and the majority of women with breast cancer diagnosed today achieve long-term survival. The long-term toxicities of adjuvant treatments have thus become especially pertinent [2]. Cognitive complaints are being reported among women who have received adjuvant chemotherapy for breast cancer with concerning frequency [3–5]. Of eight recently published studies involving breast cancer survivors undergoing comprehensive neuropsychological testing of cognitive performance across multiple domains [6–13], half of them have found memory to be particularly affected [6–8, 11]. Some studies [11, 14–16], have also suggested an association specifically between tamoxifen and cognitive problems.
Understanding the basis of long-term neurocognitive effects of endocrine and chemotherapy regimens may help in seeking strategies to prevent them. Positron emission tomography (PET) has served as a powerful tool in identifying regional brain activity associated with a variety of conditions affecting cognition, including Alzheimer’s and other neurodegenerative dementias, cerebrovascular disease, hypothyroidism, and major depression [17]. Positron-emitting water, [O-15] water, has most often been used with PET to measure acute changes in cerebral blood flow that occur while carrying out specific cognitive tasks. A positron-emitting glucose analogue, [F-18]fluorodeoxyglucose (FDG), has been frequently employed with PET to assess differences in resting brain metabolism occurring between normal and cognitively impaired subjects. The present report describes the first PET studies of regional brain activity, indexed by measures of both cerebral blood flow changes and resting brain metabolism, to explore alterations in the brains of women who were treated remotely with adjuvant therapy for breast cancer.
Patients and methods
Patients
The chemotherapy-exposed group was comprised of 16 right-handed women having histories of breast cancer treated with adjuvant chemotherapy. All had undergone their last course of cytotoxic chemotherapy 5–10 years prior to enrollment in the present study. Eleven had also undergone tamoxifen therapy. A control group of eight right-handed women had never received chemotherapy (Table1). Those controls with breast cancer histories were well-matched to the chemotherapy-treated subjects in time elapsed since diagnosis (7.4 ± 1.5 and 7.4 ± 1.7 years), as well as in age at enrollment (53.2 ± 4.1 years and 50.4 ± 5.1 years), and thus in age at diagnosis (45.8 and 43.0 years, respectively). For one type of image-based analysis, comparison was also made to cerebral metabolic data that had been previously acquired from a standard reference group of an additional 10 healthy women (44 ± 13 years), none of whom were included among the control subjects described above, who had previously undergone PET studies with FDG but not with [O-15]water, and who were cognitively normal on a standard neuropsychologic testing battery. This study was approved by the UCLA Institutional Review Board and all subjects provided written informed consent for all of the procedures described in this report.
Neurocognitive assessment
In our recent report of neurocognitive performance following adjuvant chemotherapy in an investigation three times as large (n = 72) as the present imaging-focused study, the single most significant cognitive deficit (P = 0.007) among breast cancer survivors who received chemotherapy was demonstrated on the ROCF Delayed Recall task, among the 21 neuropsychologic tests which were administered [8]. In light of the large number of cognitive and imaging parameters acquired, an a priori hypothesis with respect to neurocognitive performance was valuable for precluding the necessity of a large-scale analysis that would require a harsh statistical correction for multiple comparisons. We therefore decided in advance to use the ROCF Delayed Recall test in testing the hypothesis that differences in cerebral activity demonstrated with PET which were pertinent to cognitive effects would be correlated with neuropsychologic test performance. Cognitive testing was supervised and statistically assessed by a licensed clinical neuropsychologist, as previously described [8], and conducted within 72 hours of participants undergoing brain PET imaging at the UCLA Medical Center, as outlined below.
PET imaging and analyses
PET with [O-15]water was used to assess acute changes in cerebral blood flow associated with performance of control and memory tasks. In a standard word-pair association cognitive tasking protocol, cue words were presented on a computer screen during scanning, 10 min (short-term task) or 1 day (long-term task) after having been presented paired with other words. Tasks occurred twice each, in counterbalanced fashion, i.e. a control read-repeat task, short-term read-recall task, long-term read-recall task, long-term read-recall task, short-term read-recall task and control read-repeat task. Based upon our previous observations concerning short-term memory (as reflected by another short-term memory task employed in a prior study, the Rey–Osterreith Complex Figure (ROCF) Delayed Recall task; see previous section), scans acquired during the control and short-term recall tasks employed here were contrasted in the present analysis, and scans acquired during the long-term recall task were used in a different kind of study [18].
PET was performed with 3D acquisitions, collecting 63 contiguous data planes in a 128x128 image matrix using a CTI HR + scanner (CTI, Knoxville, TN,USA). Transmission scans obtained with a positron-emitting source were used for attenuation correction. The six PET images using [O-15]water were acquired at 14-min intervals. For each scan, after administration of 555 MBq [O-15]water, PET data were obtained in six five-s frames followed by nine 10-s frames and summed for all frames after appearance of tracer into the brain (beginning approximately 25 s after injection). Scans were reconstructed with a Hann 0.5R filter and zoom factor of 3.5.
Following the conclusion of the activation study, FDG was used to assess regional cerebral metabolism during mental rest. Subjects were scanned in the supine position, 40 min following injection of 185 MBq FDG in a dimly lit room having low ambient noise, with eyes and ears unoccluded. Six five-min frames were acquired, and summed-frame images were produced, after excluding frames for patient motion, if necessary. Group-based differences in resting metabolism were evaluated by both statistical parametric and dedicated region of interest (ROI) methods described below.
Activation studies with [O-15]water PET, as well as covariate analyses of cognitive performance versus brain metabolism measured with FDG PET, were examined with statistical parametric mapping (SPM) software by methods previously described [19]. Briefly, images were coregistered and reoriented into a standardized coordinate system [20] using the nonlinear spatial transformation package in SPM2 [21], smoothed three-dimensionally at a full-width half-maximum of 16 mm, and normalized to mean global activity. Pooled data were then statistically assessed to identify the voxels which significantly differed between cognitive tasks (activation [O-15]water scans) and/or between treatment groups, or which significantly correlated with a specified neuropsychologic parameter. The study was considered to be exploratory rather than aimed at testing specific regional hypotheses. Results were reported in terms of locations of the most significant effects (regionally and/or in x,y,z Talairach-style millimeter coordinates; Z and P values) and, when statistical strength of peak voxels was high enough to survive a Bonferroni-type correction at P < 0.05, also by corrected values. While this represents a relatively conservative kind of multiple comparison adjustment, it was useful in protecting against false-positive findings in an initial study of this kind, focusing attention upon the most statistically robust findings. In ROI analyses, 42 different brain regions were defined, based on comparison to anatomical, MR and PET atlases. These included on left and right, anterior and posterior cingulate, primary sensorimotor, superior and inferior parietal, parietotemporal in the area of the angular and marginal gyri, three temporal, five frontal, primary and associative visual occipital, and cerebellar cortex, as well as left and right thalamus, putamen, caudate nucleus head, and brainstem at the level of midbrain and pons. ROI activity values were normalized to the average pixel activity value across all regions. To adjust for the number of regions assessed, a multiple-comparison statistical correction was applied, and regional activity differences were considered significant at P < 0.001.
Results
Neurocognitive Performance
Regional brain activation during recall
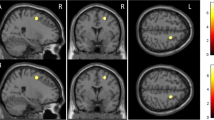
In between-group analyses, in examining the brain volume for regions in which the pattern of activation during performance of a short-term verbal memory task differed for the chemotherapy group relative to the control group, the most significant difference between the two groups (P < 0.0005) was located in the inferior frontal gyrus (−50,20,10, in the vicinity of Brodmann’s areas 44 and 45, corresponding to Broca’s area in the dominant hemisphere). Peak activation occurring in the inferior frontal gyrus corresponded to a 2.3% increase of activity during recall and was highly significant (P < 0.0005 after correction for multiple comparisons, Z = 5.95) in treated patients (Fig. 1, left), while activity in this area was only weakly increased during the cognitive task in untreated patients (P = 0.960 after correction for multiple comparisons, Z = 2.96, left inferior frontal gyrus in Brodmann’s area 45). Untreated patients, in contrast, demonstrated greatest cortical activation in the parietal cortex, with significance peaking in the vicinity of Brodmann’s area 7 (−30,−78,48, Z = 3.50), slightly lateral to the precuneus region and extending anteriorly and inferiorly towards a near-maximal focus (−28,−50,32, Z = 3.49) in the supramarginal/angular gyrus area (Brodmann’s areas 39,40, in the vicinity of Wernicke’s area in the dominant hemisphere) (Fig. 1, right), as well as the contralateral primary visual cortex (Z = 3.88); peak parietal activation in the chemotherapy-treated patients occurred slightly more anterolaterally (−36,−64,46, Brodmann’s area 40 of the inferior parietal lobule, Z = 3.85).
Activation associated with short-term recall in chemotherapy-treated (left) and untreated (right) subjects. Color scale corresponds to voxels with significant activation (P< 0.01). Peak activation occurring in the inferior frontal gyrus (bright yellow area in left image), was highly significant (P< 0.0005 after correction for multiple comparisons, Z = 5.95) in treated patients, but not in untreated patients, who showed more significant activation in the parietal cortex (bright yellow area in right image). See text for details
Chemotherapy patients also experienced significant activations in the contralateral posterior cerebellum near midline, as well as superior frontal gyrus (Z = 4.66, Z = 4.28; P = 0.010, P = 0.046 at peak voxels after multiple-comparison correction, respectively). In between-group analyses, the near-midline posterior cerebellum (peak activation located at 8,−88,−26, av. magnitude +2.6%) in fact represented the non-cortical area showing the most significant activation difference relative to untreated subjects (Z = 3.11, P = 0.001, for group-based difference).
Correlation of resting regional brain metabolism with cognitive performance
Performance on the ROCF Delayed Recall test of the chemotherapy-treated group averaged 3.2 points (13%) lower than the performance of the control group (20.6 ± 4.8 ± 1.2 vs. 23.8 ± 6.3 ± 2.2, mean ± SD ± SE, respectively). This was consistent with chemotherapy-treated patients scoring an average of 4.1 points lower on the ROCF Delayed Recall test in our larger study employing a full neuropsychological battery with three times as many subjects [8], which was the most significant difference (p < 0.007) detected between treatment arms in that study, as discussed in the Methods section, though the difference did not reach statistical significance in the present smaller group.
After statistical correction for multiple comparisons, neither ROI nor SPM methods of analysis yielded significant differences in resting metabolism between the control and adjuvant therapy groups. This is perhaps not surprising, given the range of cognitive performance levels within the chemotherapy-treated group, with Z-scores ranging from 1.7 SD above to 2.0 SD below normal mean on the ROCF Delayed Recall test and with substantial overlap between groups. A potentially more sensitive approach to that issue was to examine the brain region by region for correlations across individuals between cognitive performance and metabolism of specific regions. When this was done, the region of the brain having metabolism most significantly correlated with ROCF performance across the chemotherapy-treated subjects was located in the left inferior frontal gyrus (P < 0.0005, Z = 3.39 at voxel of peak significance, Fig. 2). Each 1 SD decline in ROCF performance corresponded to a 3% decrement in FDG activity, across a range of cognitive performance spanning almost four standard deviations of the normal mean. No such relationship was found in the untreated patients, or even for the combined group of treated and untreated patients, consistent with this correlation being related to chemotherapy-associated cognitive deficits in particular, rather than representing a generalized relationship between this cognitive task and brain metabolism.
Sagittal (left) and transaxial (right) views of statistical parametric maps identifying areas where regional brain metabolism correlated with ROCF performance across chemotherapy-treated subjects. Voxels with correlative significance of P< 0.01 are depicted in yellow, and superimposed upon an average MR T1-weighted image for anatomical reference. Red cursor lines intersect at the voxel of peak significance, located in the left inferior frontal cortex. See text for details
We also examined whether effects on brain metabolism occurred which were specific to the type of pharmacotherapy, by comparing resting metabolism among breast cancer patients treated with regimens which included tamoxifen to subjects not receiving tamoxifen-containing regimens. The most significant such effect was found in the basal ganglia. Metabolism in the lentiform nucleus was 7–8% lower in patients receiving cytotoxic chemotherapy plus tamoxifen than in patients receiving cytotoxic chemotherapy only (P < 0.01), while metabolic activity of the lentiform nucleus in subjects who received cytotoxic chemotherapy only did not differ from that of subjects who had received no therapy, which in turn did not significantly differ from metabolism in subjects having neither breast cancer nor therapy (Fig. 3).
Level of metabolism in lentiform nuclei (normalized to whole-brain activity) measured in subjects undergoing chemotherapy + tamoxifen therapy tended to be lower (by 7–8%, P< 0.01) than the level seen in all other control groups, including those subjects who received chemotherapy without tamoxifen, as well as those who received no chemotherapy for their breast cancer, and a reference group without chemotherapy or breast cancer
Discussion
In the past decade, there has been a growing body of research examining the short and long-term consequences of breast cancer treatments, and their impact upon quality of life in general, and cognitive function in particular, of the patients who receive them [22–36]. The cognitive-related complaints of these patients typically center upon their perception of mental slowness and diminished abilities to maintain attention, concentrate, and remember things. Adjuvant systemic therapies have also been associated with decreased performance on various neurocognitive tasks [3, 6, 7, 9, 11, 12, 37], and deficits may persist for years following cessation of treatment. The present study identified particular cerebral activation patterns differing in chemotherapy-treated breast cancer patients, relative to untreated subjects. Abnormal activation in inferior frontal cortex during performance of a short-term memory recall task was found in chemotherapy-treated patients. Untreated patients, in contrast, demonstrated greatest cortical activation in the parietal cortex and occipital cortex when performing the same task. Thus, overall, the altered cortical activation associated with performance of a memory task in chemotherapy-treated patients could be characterized as involving greater recruitment of frontal cortical tissue.
As the inferior frontal gyrus was also the brain region with metabolism most strongly correlated with chemotherapy subjects’ cognitive performance on a short-term memory recall task, further weight is added to the suggestion that the chemotherapy-associated changes in cerebral activation noted in this area are related to the cognitive deficits found in chemotherapy-treated patients. It also raises the possibility that the increased frontal activation during performance of the memory task may represent a compensatory response to lower resting metabolism found in this region of the brain in treated impaired patients.
In addition, women whose treatment regimens included tamoxifen had low basal ganglia activity, even relative to women who had received chemotherapy but not tamoxifen. Recently, decreases in regional cerebral metabolism were reported in a study of women who had received tamoxifen-only therapy [38]. That study focused upon effects of estrogen depletion in postmenopausal women, and differed from the present study in a number of ways. It did not include any women who had received cytotoxic chemotherapy, nor any control group with a history of breast cancer but who had not received tamoxifen. Nor did the study include [O-15]water scans or other method of examining modulation in brain function during actual performance of cognitive tasks, or report ROI analysis of FDG data comprising basal ganglia regions, or identify any correlations between neuropsychologic parameters and altered cerebral metabolism. The study, however, did provide evidence that diminished estrogen signaling is associated with decreased metabolism in certain parts of the forebrain, as seen in the present study.
While the most significant cortical memory-related activation difference between chemotherapy-treated and untreated subjects (treated > untreated) occurred in the inferior frontal gyrus in our study, the most significant non-cortical activation difference occurred in the posterior cerebellum near midline. In fact, other than the inferior frontal gyrus region, this cerebellar area was the most strongly activated among chemotherapy-treated subjects (Z = 4.66, P = 0.01, after correction for multiple comparisons). Interestingly, a previous PET study examining chronic effects of cognition-impairing substances (in that case, from smoking marijuana) on regional cerebral activations measured during performance of a verbal memory task found that the most significant activation difference (drug-exposed > controls) also was found in the posterior cerebellum near midline [39], and this particular cerebellar region has been implicated in attention, memory, and other higher cognitive processes [39,40].
A pertinent clinical question raised by the current findings is whether it may be feasible to employ the kind of neuroimaging tools used here to diminish future cognitive impact of particular treatment regimens. For example, FDG PET studies might be used to monitor cerebral response to potentially neurotoxic therapies–analogously to our current use of MUGA studies to monitor cardiac response to chemotherapy regimens containing doxorubicin or other cardiotoxic agents – taking advantage of the typical lead time (2–10 years) by which cerebral metabolic changes precede development of neurologic symptoms [41]. If useful, this approach could in fact be accomplished in a clinically practical manner, through a simple ‘add-on’ view of the brain to whole-body PET or PET/CT studies already commonly being performed for tumor staging and re-staging assessments in many breast cancer patients [42].
One limitation inherent in the design of this type of exploratory study is that it is quite possible for differences to exist between treatment groups in factors that could affect cognitive function, other than the treatment itself. Although, as seen in Table 1, the groups were found to be demographically similar in most respects, including years of education and estimated premorbid I.Q., and we are not aware of any such differences occurring between the groups that would be expected to differentially impact upon brain function, other potential differences which are not apparent here could influence cognition and associated brain activity. This limitation is probably unavoidable, since it would not be feasible or ethical to randomize patients to receive chemotherapy or not, for the purpose of studying its cognitive effects. Another limitation is the relatively small number of subjects (though not atypical of studies involving functional brain imaging measures, due to the expense and relative scarcity of scanner and cyclotron resources), which tends to make treatment subgroups particularly limited in subject number, and diminishes the ability to draw generalizable conclusions, particularly about subgroups.
Mechanistically, in addition to possible direct effects of administered therapeutics on brain tissue, agents could act by modulating levels of endogenous hormones (e.g. cortisol and estrogen) for which specific receptors are present in the nervous system [43–55]. Neurologic effects might also be exerted through responses of the immune system (e.g. release of proinflammatory cytokines) [56–67], or secondary to psychologic changes associated with the therapeutic agents (e.g. in levels of anxiety, depression and fatigue). To better understand the roles of these different possible mechanisms on cerebral functioning, it will be necessary for future studies to include longitudinal assessments which begin with baseline measures of neuropsychologic performance, hormone levels, immune function, and regional cerebral activity prior to the administration of systemic therapies.
References
Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A, Feuer EJ, Thun MJ (2005) Cancer statistics. CA Cancer J Clin 55:10–30
Ganz PA (2002) What outcomes matter to patients: a physician-researcher point of view. MedCare 40:III11-III19
Phillips KA, Bernhard J (2003) Adjuvant breast cancer treatment and cognitive function: current knowledge and research directions. J Natl Cancer Inst 95:190–197
Ahles TA, Saykin A (2001) Cognitive effects of standard-dose chemotherapy in patients with cancer. Cancer Invest 19:812–820
Meyers CA (2000) Neurocognitive dysfunction in cancer patients. Oncology 14:75–79
Ahles TA, Saykin AJ, Furstenberg CT, Cole B, Mott LA, Skalla IK, Whedon MB, Bivens S, Mitchell T, Greenberg ER, Silberfarb PM (2002) Neuropsychologic impact of standard-dose systemic chemotherapy in long- term survivors of breast cancer and lymphoma. J Clin Oncol 20:485–493
Brezden CB, Phillips KA, Abdolell M, Bunston T, Tannock IF (2000) Cognitive function in breast cancer patients receiving adjuvant chemotherapy. J Clin Oncol 18:2695–2701
Castellon SA, Ganz PA, Bower JE, Petersen L, Abraham L, Greendale GA (2004) Neurocognitive performance in breast cancer survivors exposed to adjuvant chemotherapy and tamoxifen. J Clin Exp Neuropsychol 26:955–69
Schagen SB, van Dam FS, Muller MJ, Boogerd W, Lindeboom J, Bruning PF (1999) Cognitive deficits after postoperative adjuvant chemotherapy for breast carcinoma. Cancer 85:640–650
Tchen N, Juffs HG, Downie FP, Qi-Long Y, Hu H, Chemerynsky I, Clemons M, Crump M, Goss PE, Warr D, Tweedale ME, Tannock IF (2003) Cognitive function, fatigue, and menopausal symptoms in women receiving adjuvant chemotherapy for breast cancer. Journal of Clinical Oncology, 21:4175–4183
van Dam FS, Schagen SB, Muller MJ, Boogerd W, Wall E, Droogleever Fortuyn ME, Rodenhuis S (1998) Impairment of cognitive function in women receiving adjuvant treatment for high-risk breast cancer: high-dose versus standard-dose chemotherapy. J Natl Cancer Inst 90:210–218
Wieneke MH, ER Dienst (1995) Neuropsychological Assessments of Cognitive Functioning Following Chemotherapy for Breast Cancer. Psycho-Oncology 4:61–66
Wefel JS, Lenzi R, Theriault RL, Meyers CA (2004) The cognitive sequelae of standard-dose adjuvant chemotherapy in women with breast carcinoma: Results of a prospective, randomized, longitudinal trial. Cancer 100:2292–2299
Arpels JC (1996) The female brain hypoestrogenic continuum from the premenstrual syndrome to menopause. A hypothesis and review of supporting data. J Reprod Med 41:633–639
Jenkins V, Shilling V, Fallowfield L, Howell A, Hutton S (2004) Does hormone therapy for the treatment of breast cancer have a detrimental effect on memory and cognition? A pilot study. Psychooncology 13:61–66
Paganini-Hill A, Clark LJ (2000) Preliminary assessment of cognitive function in breast cancer patients treated with tamoxifen. Breast Cancer Res Treat 64:165–176
Silverman DHS, Alavi A (2005) PET imaging in the assessment of normal and impaired cognitive function. Radiologic Clin North Am Saunders/Elsevier, Philadelphia, 43:67–77
Lai J, Dy CJ, Pio BS, Ganz PA, Castellon SA, Abraham L, Waddell K, Phelps ME, Bookheimer SY, Silverman DHS (2005) Anterior cingulate and temporal lobe regions associated with congruence of memory recall to mood. J Nucl Med 46(5, abstr. suppl):no. 29
Rasgon NL, Silverman D, Siddarth P, Miller K, Ercoli LM, Elman S, Lavretsky H, Huang S-C, Phelps PE, Small GW (2005) Estrogen use and brain metabolic change in postmenopausal women with age-associated memory decline. Neurobiology of Aging 226:229–235
Talairach J, Tournoux P (1988) Co-planar stereotaxic atlas of the human brain. 3-dimensional proportional system: An approach to cerebral imaging. New York: Thieme
Friston K, Ashburner J, Heather J, et al (2003) Statistical Parametric Mapping (SPM2). Available at www.fil.ion.ucl.ac.uk/spm (The Wellcome Department of Cognitive Neurology, University College London, London)
Partridge AH, Burstein HJ, Winer EP (2001) Side effects of chemotherapy and combined chemohormonal therapy in women with early-stage breast cancer. J Natl Cancer Inst Monogr 135–142
Ganz PA (2001a) Impact of tamoxifen adjuvant therapy on symptoms, functioning, and quality of life. J Natl Cancer Inst Monogr 130–134
Shapiro CL, Recht A (2001) Side Effects of Adjuvant Treatment of Breast Cancer. N Engl J Med 344:1997–2008
Ganz PA, Hirji K, Sim MS, Schag CA, Fred C, Polinsky ML (1993) Predicting psychosocial risk in patients with breast cancer. Med Care 31:419–431
Schag CA, Ganz PA, Polinsky ML, Fred C, Hirji K, Petersen L (1993) Characteristics of women at risk for psychosocial distress in the year after breast cancer. J Clin Oncol 11:783–793
Ganz PA, Desmond KA, Belin TR, Meyerowitz BE, Rowland JH (1999) Predictors of sexual health in women after a breast cancer diagnosis. J Clin Oncol 17:2371–2380
Ganz PA, Rowland JH, Desmond K, Meyerowitz BE, Wyatt GE (1998a) Life after breast cancer: understanding women’s health-related quality of life and sexual functioning. J Clin Oncol 16:501–514
Rowland JH, Desmond KA, Meyerowitz BE, Belin TR, Wyatt GE, Ganz PA (2000) Role of breast reconstructive surgery in physical and emotional outcomes among breast cancer survivors. J Natl Cancer Inst 92:1422–1429
Bower JE, Ganz PA, Desmond KA, Rowland JH, Meyerowitz BE, Belin TR (2000) Fatigue in breast cancer survivors: occurrence, correlates, and impact on quality of life. J Clin Oncol 18:743–753
Ganz PA, Rowland JH, Meyerowitz BE, Desmond KA (1998b) Impact of different adjuvant therapy strategies on quality of life in breast cancer survivors. Recent Results Cancer Res 152:396–411
Ganz PA, Greendale GA, Petersen L, Zibecchi L, Kahn B, Belin TR (2000) Managing menopausal symptoms in breast cancer survivors: results of a randomized controlled trial. J Natl Cancer Inst 92:1054–1064
Ganz PA, Desmond KA, Leedham B, Rowland JH, Meyerowitz BE, Belin TR (2002) Quality of life in long-term, disease-free survivors of breast cancer: a follow-up study. J Natl Cancer Inst 94:39–49
Loprinzi CL, Thome SD (2001) Understanding the Utility of Adjuvant Systemic Therapy for Primary Breast Cancer. J Clin Oncol 19:972–979
Ravdin PM, Siminoff LA, Davis GJ, Mercer MB, Hewlett J, Gerson N, Parker HL (2001) Computer Program to Assist in Making Decisions About Adjuvant Therapy for Women With Early Breast Cancer. J Clin Oncol 19:980–991
Ganz PA (1998) Cognitive dysfunction following adjuvant treatment of breast cancer: a new dose-limiting toxic effect? J Natl Cancer Inst 90:182–183
Castellon SA, Silverman DHS, Ganz P (2005) Breast cancer treatment and cognitive functioning: current status and future challenges in assessment. Breast Cancer Research and Treatment 92:199–206
Eberling JL, Wu C, Tong-Turnbeaugh R, Jagust WJ (2004) Estrogen- and tamoxifen-associated effects on brain structure and function. Neuroimage 21:364–371
Block RI, O’Leary DS, Hichwa RD, Augustinack JC, Ponot LLB, Ghoneim MM, Arndt S, Hurtig RR, Watkins GL, Hall JA, Nathan PE, Andreasen NC (2002) Effects of frequent marijuana use on memory-related regional cerebral blood flow. Pharmacol Biochem Behav 72:237–250
Schmamann JD, ed: The cerebellum and cognition. Academic Press, San Diego, 1997
Silverman DHS and Melega WP (2004) Molecular imaging of biological processes with PET: evaluating biologic bases of cerebral function. In PET: Molecular Imaging and Its Biological Applications ME Phelps, ed., Springer-Verlag, New York, pp. 509–583
Tran A, Pio BS, Khatibi B, Czernin J, Phelps ME, Silverman DHS (2005) 18F-FDG PET for staging breast cancer in patients with inner-quadrant versus outer-quadrant tumors: comparison with long-term clinical outcome. J Nucl Med 46:1455–1459
Ciocca DR, Roig LM (1995) Estrogen receptors in human nontarget tissues: biological and clinical implications. Endocr Rev 16:35–62
Hampson E (1990) Variations in sex-related cognitive abilities across the menstrual cycle. Brain Cogn 14:26–43
Komenich PKD, Lane M, Dickey RP, Stone SC (1978) Gonadal hormones and cognitive performance. Physiological Psychology 6:115–120
Jacobs DM, Tang MX, Stern Y, Sano M, Marder K, Bell KL, Schofield P, Dooneief G, Gurland B, Mayeux R (1998) Cognitive function in nondemented older women who took estrogen after menopause. Neurology 50:368–373
McLay RN, Maki PM, Lyketsos CG (2003) Nulliparity and late menopause are associated with decreased cognitive decline. Journal of Neuropsychiatry and Clinical Neurosciences 15:161–167
Kawas C, Resnick S, Morrison A, Brookmeyer R, Corrada M, Zonderman A, Bacal C, Lingle DD, Metter E (1997) A prospective study of estrogen replacement therapy and the risk of developing Alzheimer’s disease: the Baltimore Longitudinal Study of Aging. Neurology 48:1517–1521
Paganini-Hill A, Henderson VW (1996) Estrogen replacement therapy and risk of Alzheimer disease. Arch Intern Med 156:2213–2217
Maki PM, Resnick SM (2000) Longitudinal effects of estrogen replacement therapy on PET cerebral blood flow and cognition. Neurobiology of Aging 21(2):373–83
Shaywitz SE, Shaywitz BA, Pugh KR, et al (1999) Effect of estrogen on brain activation patterns in postmenopausal women during working memory tasks. JAMA 281:1197–202
LeBlanc ES, Janowsky J, Chan BK, Nelson HD (2001) Hormone replacement therapy and cognition: systematic review and meta- analysis. JAMA 285:1489–1499
Haskell SG, Richardson ED, Horwitz RI (1997) The effect of estrogen replacement therapy on cognitive function in women: a critical review of the literature. J Clin Epidemiol 50:1249–1264
Shumaker SA, Legault C, Rapp SR, Thal L, Wallace RB, Ockene JK, Hendrix SL, Jones III BN, Assaf AR, Jackson RD, Morley Kotchen J, Wassertheil-Smoller S, Wactawski-Wende J (2003) Estrogen Plus Progestin and the Incidence of Dementia and Mild Cognitive Impairment in Postmenopausal Women: The Women’s Health Initiative Memory Study: A Randomized Controlled Trial. JAMA 289:2651
Rapp SR, Espeland MA, Shumaker SA, Henderson VW, Brunner RL, Manson JE, Gass MLS, Stefanick ML, Lane DS, Hays J, Johnson KC, Coker LH, Dailey M, Bowen D (2003) Effect of Estrogen Plus Progestin on Global Cognitive Function in Postmenopausal Women: The Women’s Health Initiative Memory Study: A Randomized Controlled Trial. JAMA 289:2663
Larson SJ, Dunn AJ (2001) Behavioral effects of cytokines. Brain Behav Immun 15:371–387
Reichenberg A, Yirmiya R, Schuld A, Kraus T, Haack M, Morag A, Pollmacher T (2001) Cytokine-associated emotional and cognitive disturbances in humans. Arch Gen Psychiatry 58:445–452
Lerner DM, Stoudemire A, Rosenstein DL (1999) Neuropsychiatric toxicity associated with cytokine therapies. Psychosomatics 40:428–435
Denicoff KD, Rubinow DR, Papa MQ, Simpson C, Seipp CA, Lotze MT, Chang AE, Rosenstein D, Rosenberg SA (1987) The neuropsychiatric effects of treatment with interleukin-2 and lymphokine-activated killer cells. Ann Intern Med 107:293–300
Valentine AD, Meyers CA, Kling MA, Richelson E, Hauser P (1998) Mood and cognitive side effects of interferon-alpha therapy. Semin Oncol 25:39–47
Capuron L, Ravaud A, Gualde N, Bosmans E, Dantzer R, Maes M, Neveu PJ (2001) Association between immune activation and early depressive symptoms in cancer patients treated with interleukin-2-based therapy. Psychoneuroendocrinology 26:797–808
Singh VK, Guthikonda P (1997) Circulating cytokines in Alzheimer’s disease. J Psychiatr Res 31:657–660
Holmes C, El-Okl M, Williams AL, Cunningham C, Wilcockson D, Perry VH (2003) Systemic infection, interleukin 1beta, and cognitive decline in Alzheimer’s disease. J Neurol Neurosurg Psychiatry Jun;74(6):788–9
Papassotiropoulos A, Bagli M, Jessen F, Bayer RA, Maier W, Rao ML, Heun R (1999) A genetic variation of the inflammatory cytokine interleukin-6 delays the initial onset and reduces the risk for sporadic Alzheimer’s disease. Ann Neurol 45:666–668
Weaver JD, Huang MH, Albert M, Harris T, Rowe JW, Seeman TE (2002) Interleukin-6 and risk of cognitive decline: MacArthur studies of successful aging. Neurology 59:371–378
Maier SF (2003) Bi-directional immune-brain communication: Implications for understanding stress, pain, and cognition. Brain, Behavior & Immunity 17:69–85
Pugh RC, Fleshner M, Watkins LR, Maier SF, Rudy JW (2001) The immune system and memory consolidation: A role for the cytokine IL-1beta. Neuroscience & Biobehavioral Reviews 25:29–41
Blair JR, Spreen O: Predicting pre-morbid IQ (1989) a revision of the National Adult Reading Test. The Clinical Neuropsychologist, 3:129–136
Acknowledgements
This research was funded by the Breast Cancer Research Foundation and by an American Cancer Society Clinical Research Professorship award to Dr. Ganz. The authors have no financial interests or relationships with industry which bear on the subject matter described in the paper. We are indebted to Erin Siu for her assistance in manuscript preparation.
Author information
Authors and Affiliations
Corresponding author
Additional information
This report represents the original work of the authors. It has not been previously published and is not under submission to any other journal or publication.
Rights and permissions
About this article
Cite this article
Silverman, D.H.S., Dy, C.J., Castellon, S.A. et al. Altered frontocortical, cerebellar, and basal ganglia activity in adjuvant-treated breast cancer survivors 5–10 years after chemotherapy. Breast Cancer Res Treat 103, 303–311 (2007). https://doi.org/10.1007/s10549-006-9380-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-006-9380-z