Abstract
Birch, Ginsburg, and Jablonka lay out a very convincing case for an important transition marker: unlimited associative learning (UAL). Especially welcome are the empirical predictions. I focus here not on the question of how to infer phenomenal consciousness from this behavioral metric, but on possible novel applications of this useful and fundamental framework. Specifically, I highlight two aspects of biology that are often not considered in philosophy of mind approaches that focus on natural species and evolutionary time scales. These are (1) the ability of minds and bodies to change drastically on the time scale of an individual experiencing subject, and (2) bioengineering of novel living forms with no evolutionary history at the organism level. Both of these aspects provide interesting new contexts within which to explore UAL and its implications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Birch, Ginsburg, and Jablonka (BGJ) lay out a very convincing case for an important transition marker: unlimited associative learning (UAL). Especially welcome are the empirical predictions. I focus here not on the question of how to infer phenomenal consciousness from this behavioral metric, but on possible novel applications of this useful and fundamental framework. Specifically, I highlight two aspects of biology that are often not considered in philosophy of mind approaches that focus on natural species and evolutionary time scales. These are (1) the ability of minds and bodies to change drastically on the time scale of an individual experiencing subject, and (2) bioengineering of novel living forms with no evolutionary history at the organism level. Both of these aspects provide interesting new contexts within which to explore UAL and its implications; the biological examples are discussed in depth elsewhere (Levin 2019, 2020).
BGJ start out by drawing a clear line between “experiencing subjects” that have a subjective point of view on the world, and systems that do not. “Finding that line, and understanding how it was crossed, is a major challenge for science and philosophy”. They are referring to a phylogenetic line—a major transition during evolution. It is important however to apply this long-standing question to a new timescale: metamorphosis during the lifetime of an individual, which focuses attention on the integration of minimal subunits into a cognitive whole and on the continuity of that consciousness in the face of changes to its material substrate. BGJ’s framework focuses on the perspective of a single Subject—the owner of experience. But all cognitive agents are made of parts, and the key question is how the individual components (e.g., cells) integrate into a global whole that has a unified subjective experience and goals. Importantly, these parts can be rearranged during the lifetime of an individual, not only by evolution. One example is the transition of a caterpillar into a butterfly or moth; during this process, the animal’s brain is largely disassembled, and rebuilt to control an entirely new kind of body. Despite this drastic remodeling of the neural structures, some memories appear to be maintained (Blackiston et al. 2015). Likewise, a planarian flatworm’s head and brain can be completely removed, but upon regenerating from tissues in the tail half, memories persist (Corning 1967; Shomrat et al. 2013). What is it like to be a caterpillar changing into a butterfly, or a planarian being fragmented into several new individuals, all of which will bear the memories of the original (Neuhof et al. 2016)?
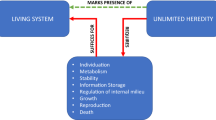
The lack of simple 1:1 identification of learning and memories with a clear, well-defined “owner” during and after remodeling events is not limited to rare corner cases of living forms. Every multicellular creature takes part in the slow, gradual assembly of a cognitive agent from parts, during the acquisition of learning capacity and unified perspective from the embryonic state. Starting as one fertilized egg cell, numerous competent subunits (embryonic cells) work together to build a somatic embodiment of unified consciousness, recapitulating the evolutionary journey to multicellularity (Levin 2019). Thus, any theory of transition markers and consciousness needs to be extended to deal with not only “intact”, behaving creatures that differ across geological timescales, but with ones whose bodies, brains, and minds are changing during the operation of their cognition. Cognitive agents not only change drastically during metamorphosis and regeneration, but are actively assembled from (non-, or minimally-conscious) components in a process that often takes weeks or months. Somatic plasticity is relevant for UAL not only because of the hardware that needs to make the associations, but also because of the questions concerning the changing substrate of the storied memories (associations formed during the UAL). In addition to the substrate, the information content is plastic as well, and can be altered by processes distinct from normal learning. For example, the transfer of memories that had belonged to different individuals (Bedecarrats et al. 2018; Pietsch et al. 1969) or indeed the wholesale inception of memories that had never belonged to any agent, using optogenetics (Ramirez et al. 2013; Vetere et al. 2019).
The question of phylogenetic transition also needs to be stretched in a different way, to account for the plasticity of life outside the normal evolutionary stream. During embryogenesis, radical changes can be induced by experimenters in the structure and function of organisms, including adding or subtracting brain tissue, rearranging the topology of the nervous system, and providing entirely new capabilities by sensory substitution experiments (Bach-y-Rita 1967; Blackiston et al. 2013). Even during adulthood, drastic changes to coherent agents can be made via transplants of brain tissue, which readily cross species (such as Drosophila neurons put into rodent or human brains (Savel'ev et al. 1994; Saveliev et al. 1997). Such work demonstrates living, functional chimeras (made at the genetic, cell, tissue, or organ-level of mixing) and constructs animals that contain material from very diverse species across the tree of life or indeed are functionally fused to inorganic sensor or effector organs. What is important about such cases is that they result in novel beings, with body and brain structures that are not anywhere on the standard phylogenetic history of life on Earth.
Multiscale chimerism is readily and routinely achieved in the laboratory and thus is firmly included in BGJ’s nomological sufficiency (not a metaphysical flight of fancy). A consideration of the plasticity and gradual assembly of minds, from diverse sources, widens the applicability of frameworks like UAL well beyond phylogenetic time scales and questions about whether a given “standard” species is on one or another side of the line demarcating conscious from non-conscious agents. A gradualist perspective is suggested by (1) the temporal continuum of developmental processes in which no single event can be identified as a magic transition from a “mere” ball of cells to a conscious being, and (2) the structural continuum of an infinite set of living, functional combinations that can be made from cells and tissues of pre-UAL and post-UAL sources. Moreover, (3) cognition is multi-scale, as even individual cells (Baluška et al. 2016; Lyon 2006, 2015) and organs (Pezzulo et al. 2015) from which an organism assembles are themselves examples of basal cognition: a given body may be home to more than one consciousness, including individual cells, two brain hemispheres with perhaps distinct sets of preferences (Gazzaniga 1970), etc. Thus, while cognitive capacities clearly feature major transitions, a fruitful conceptual approach is that of a gradual continuum—how much consciousness—not whether there is consciousness or not.
Recent advances in synthetic bioengineering require extending consciousness frameworks even further. It is now possible to create entirely novel organisms, consisting from inorganic (Chan et al. 2012) or cell-based (Kriegman et al. 2020) components. These exhibit new morphologies and functional behaviors, and will become increasingly more sophisticated (Kamm et al. 2018). One crucial aspect of these organisms is that they exhibit structure and function that, unlike the rest of life on Earth, are not explained by a long history of evolutionary selection (although, some were evolved in a virtual world inside a computer (Kriegman et al. 2020)). UAL must be sought as an emergent capacity, and will surely be a target of design for rational implementation, in synthetic organisms (Doursat et al. 2014; Macia et al. 2017; Sole et al. 2016). A number of BGJ’s proposed tests can be directly implemented in synthetic organisms, for example (Testing Claim 1) to determine whether UAL’s components form a natural cluster by emerging together in diverse synthetic forms. Likewise, stimulus–response protocols (Testing Claim 2) can readily be performed in biobots. Finally, the evolutionary emergence of UAL and/or its components in this new, parallel stream of life will serve as an important complement to the N = 1 of existing Earth lineages, and accessible long before exobiological research provides additional subjects.
The kinds of new organisms that exhibit UAL will help refine not only the BGJ framework, but will serve as an important context in which to develop philosophy of mind and cognitive science for life-as-it-could-be (Langton 1995) and mind-as-it-could-be. Especially salient are developments in the field of brain-computer interfaces (connecting devices to human or animal brains, and conversely including biological controllers inside robotic bodies) which bring up important issues of intrinsic motivation and consciousness in non-protoplasm-based AI but also hybrid biological-AI (Oudeyer et al. 2013) agents. Indeed, the intersection of questions about consciousness and the incipient creation of a pantheon of hybrid creatures containing biological and electronic/machine learning components reveal the important links of UAL to questions of ethics (Levin et al. 2020) and how we are to relate to organisms that are living but outside of the “standard” phylogenetic ladder of cognitive capacity.
References
Bach-y-Rita P (1967) Sensory plasticity. Applications to a vision substitution system. ActaNeurolScand 43:417–426
Baluška F et al (2016) On having no head: cognition throughout biological systems. Front Psychol 7:902
Bedecarrats A et al (2018) RNA from trained aplysia can induce an epigenetic engram for long-term sensitization in untrained aplysia. eNeuro. https://doi.org/10.1523/ENEURO.0038-18.2018
Blackiston DJ et al (2013) Ectopic eyes outside the head in Xenopus tadpoles provide sensory data for light-mediated learning. J ExpBiol 216:1031–1040
Blackiston D et al (2015) The stability of memories during brain remodeling: a perspective. CommunIntegrBiol 8:e1073424
Chan V et al (2012) Development of miniaturized walking biological machines. Sci Rep 2:857
Corning WC (1967) Regeneration and retention of acquired information (NASA). In: Corning WC, Ratner SC (eds) Chemistry of learning. Springer, Boston. https://doi.org/10.1007/978-1-4899-6565-3_18
Doursat R et al (2014) Growing fine-grained multicellular robots. Soft Robotics 1:110–121
Gazzaniga M (1970) The bisected brain. Appleton-Century-Crofts, New York
Kamm RD et al (2018) Perspective: the promise of multi-cellular engineered living systems. Apl Bioeng 10(1063/1):5038337
Kriegman S et al (2020) A scalable pipeline for designing reconfigurable organisms. ProcNatlAcadSci U S A 117:1853–1859
Langton CG (1995) Artificial life an overview. MIT Press, Cambridge Mass
Levin M (2019) The computational boundary of a “Self”: developmental bioelectricity drives multicellularity and scale-free cognition. Front Psychol. https://doi.org/10.3389/fpsyg.2019.02688
Levin M (2020) Life, death, and self: fundamental questions of primitive cognition viewed through the lens of body plasticity and synthetic organisms. Biochem Biophys Res Commun. https://doi.org/10.1016/j.bbrc.2020.10.077
Levin M et al (2020) Applications and ethics of computer-designed organisms. Nat Rev Mol Cell Biol 21(11):655–656
Lyon P (2006) The biogenic approach to cognition. Cogn Process 7:11–29
Lyon P (2015) The cognitive cell: bacterial behavior reconsidered. Front Microbiol 6:264
Macia J et al (2017) Synthetic associative learning in engineered multicellular consortia. J R Soc Interface. https://doi.org/10.1098/rsif.2017.0158
Neuhof M et al (2016) Vertically- and horizontally-transmitted memories - the fading boundaries between regeneration and inheritance in planaria. Biol Open 5:1177–1188
Oudeyer P-Y, Kaplan F (2007) What is intrinsic motivation? A typology of computational approaches. Front Neurorobotics 1:6
Pezzulo G et al (2015) Re-membering the body: applications of computational neuroscience to the top-down control of regeneration of limbs and other complex organs. IntegrBiol (Camb) 7:1487–1517
Pietsch P et al (1969) Brain transplantation in salamanders—an approach to memory transfer. Brain Res 14:707–715
Ramirez S et al (2013) Creating a false memory in the hippocampus. Science 341:387–391
Savel’ev SV et al (1994) Transplantation of insect embryonal nerve cells into the brain of amphibia and mammals. BiullEkspBiol Med 117:364–368
Saveliev SV et al (1997) Chimeric brain: theoretical and clinical aspects. Int J Dev Biol 41:801–808
Shomrat T et al (2013) An automated training paradigm reveals long-term memory in planarians and its persistence through head regeneration. J ExpBiol 216:3799–3810
Sole R et al (2016) Synthetic collective intelligence. Biosystems 148:47–61
Vetere G et al (2019) Memory formation in the absence of experience. Nat Neurosci 22:933–940
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Levin, M. Unlimited plasticity of embodied, cognitive subjects: a new playground for the UAL framework. Biol Philos 36, 17 (2021). https://doi.org/10.1007/s10539-021-09792-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10539-021-09792-4