Abstract
The global network of protected areas (PAs) is systematically biased towards remote and unproductive places. Consequently, the processes threatening biodiversity are not halted and conservation impact—defined as the beneficial environmental outcomes arising from protection relative to the counterfactual of no intervention—is smaller than previously thought. Yet, many conservation plans still target species’ representation, which can fail to lead to impact by not considering the threats they face, such as land conversion and climate change. Here we aimed to identify spatial conservation priorities that minimize the risk of land conversion, while retaining sites with high value for threatened plants at risk from climate change in the Brazilian Cerrado. We compared a method of sequential implementation of conservation actions to a static strategy applied at one time-step. For both schedules of conservation actions, we applied two methods for setting priorities: (i) minimizing expected habitat conversion and prioritizing valuable sites for threatened plants (therefore maximizing conservation impact), and (ii) prioritizing sites based only on their value for threatened plants, regardless of their vulnerability to land conversion (therefore maximizing representation). We found that scenarios aimed at maximizing conservation impact reduced total vegetation loss, while still covering large proportions of species’ ranges inside PAs and priority sites. Given that planning to avoid vegetation loss provided these benefits, vegetation information could represent a reliable surrogate for overall biodiversity. Besides allowing for the achievement of two distinct goals (representation and impact), the impact strategies also present great potential for implementation, especially under current conservation policies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The establishment of protected areas (PAs) around the world has a long history of decisions based on particular values and interests, such as scenic beauty and its recreational value, generally covering unproductive landscapes, with steep slopes and unfertile soils (Pressey et al. 2000, 2002; Ladle and Whittaker 2011). This practice has resulted in a systematic bias in the network of PAs towards places less likely to suffer from direct human pressures even in the absence of protection (Joppa and Pfaff 2009; Carranza et al. 2014; Devillers et al. 2014; Pfaff et al. 2014). A clear disadvantage of this bias is that processes threatening the persistence of species, such as land conversion, are not halted despite the apparent increasing conservation efforts (Pressey et al. 2000; Devillers et al. 2014). Consequently, the “conservation success” or “conservation impact” of PAs, defined as the beneficial environmental outcomes arising from protection relative to a counterfactual of no intervention (Ferraro 2009; Pressey et al. 2015), tends to be small, minimally avoiding human-induced effects (Andam et al. 2008).
Aiming to improve environmental outcomes, the scientific community has shifted prioritization approaches towards targeting biodiversity representation within PAs, with the objective of maximizing biodiversity features covered by PA networks (Possingham et al. 2000). Yet, such a strategy ignores the risk of land conversion, basing decisions solely on the conservation value of a given site, defined by measures such as species richness, irreplaceability or complementarity (see the naive myopic scenario in Costello and Polasky 2004; the myopic scenario in Drechsler 2005; and maximizing gain scenario in Visconti et al. 2010). Usually, such frameworks are also static in that they assume unrealistically that, once identified, conservation actions (e.g. ecological restoration, PA expansion, among others; Strassburg et al. 2017) can be implemented before any objectives are compromised by, for example, loss of vegetation cover upon which species depend (Meir et al. 2004). However, it is widely recognized that species distributions and human-induced threats vary not just in space but also in time (Pressey et al. 2007).
Climate change and accelerating rates of deforestation are recognized as the two major threats to species today and in the near future (Thomas et al. 2004; Pereira et al. 2010). Using species distribution models (SDMs), many studies have already predicted that climate change is likely to compromise the efficiency of current PA networks by shifting species ranges out of protected sites, with some species unable to disperse quickly enough to track their suitable climatic conditions (Thuiller et al. 2005; Brook et al. 2008; Lemes and Loyola 2013). Deforestation will probably act synergistically with climate change to increase species extinction risks (Brook et al. 2008). Therefore, unless conservation plans consider the dynamic nature of biodiversity and threats associated with it, conservation actions will continue to fail in ensuring species persistence (Pressey et al. 2007).
In a world of limited financial resources, it is also worth considering the insufficient funds designated to conservation initiatives. In order to maximize the chances of delivering effective conservation outcomes, conservationists need to plan for the practical implementation of actions (Knight et al. 2008). Scheduling conservation actions is, in most cases, a long and gradual process accompanied by changes in land use and consequent biodiversity loss (Costello and Polasky 2004; Meir et al. 2004). The complexity of addressing ecological processes and dynamic threats in conservation planning (Pressey et al. 2007; Lourival et al. 2011) has stimulated initial studies to model sequential protection in parallel with vegetation loss while targeting areas needing immediate intervention (Pressey et al. 2004; Drechsler 2005; Strange et al. 2006; O’Hanley et al. 2007; Visconti et al. 2010). However, the majority of these studies relied on non-spatially explicit simulations of land conversion and few have incorporated the effects of climate change on species (informed myopic scenario in Costello and Polasky 2004; foresighted in Drechsler 2005; and minimizing loss in Visconti et al. 2010; Mascia 2014; Santika et al. 2015; Visconti and Joppa 2015).
Because the threats to species, especially through vegetation loss and climate change, are likely to persist or even increase in the future (IPCC 2014), and given the economic and political motivations for the residual tendency of future PAs (Devillers et al. 2014; Pressey et al. 2015), we aimed to select spatial conservation priorities that minimize the risk of land conversion in the Brazilian Cerrado while also considering the effects of climate change on species distributions. This study is the first to combine land conversion and climate change to compare the impact with the representation strategy to solve a dynamic area selection problem..
Methods
Study area
Our study region was the Brazilian Cerrado, a Neotropical savanna covering a quarter of the Brazilian terrestrial area (203,644,800 ha). This region was classified as a Biodiversity Hotspot due to its high levels of plant endemism and high rates of primary vegetation loss (Mittermeier et al. 2004; Strassburg et al. 2017). Human pressure has increased in the region owing to land conversion for agricultural activities and cattle raising (Klink and Moreira 2002) which, combined with the biome’s low coverage by PAs (8.3%), has intensified the negative effects on biodiversity at many scales (Brook et al. 2008).
Overview of analyses
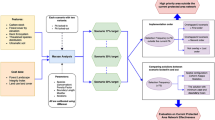
The whole method is divided in three major phases: (1) species distribution modelling, (2) systematic planning of conservation scenarios, and (3) evaluation of conservation scenarios performance in 2050 (Fig. 1). The first phase encompasses the generation of distribution models for endemic and threatened plants of Brazil, projected both into the present and future climate scenarios. In the second phase, we used the predicted species’ distributions and their respective uncertainty maps and maximum dispersal distances together with land-use models, all restricted to the Cerrado domain (n = 221 species), as inputs to the spatial prioritization analyses. This process was followed by the development of four conservation intervention scenarios varying in scheduling of action (acting now and time-step action) and setting priorities (maximizing impact or representation). Lastly, in the third phase, we evaluated the conservation impact of each intervention scenario by measuring the avoided land conversion relative to the counterfactual scenario (no action). We also calculated the average size of all species’ ranges covered by PAs and priority sites for each intervention and the no-action scenario.
Species distribution modelling
Climatic variables
To compute bioclimatic variables in intervals of five years from the present (henceforth referred to as baseline) to 2050, we obtained climatic simulations of four coupled atmosphere–ocean general circulation models (AOGCMs; Table S1) from the CMIP5 (Coupled Model Intercomparison Project Phase 5) database for the most severe representative concentration pathway (RCP 8.5). The RCPs describe four different CO2 emission scenarios, varying from the most stringent mitigation scenario (2.6) to the scenario in which emission rates continue to steeply rise throughout the twenty-first century (8.5). We analyzed only the worst-case scenario, because the lack of efforts to cut down emissions will likely lead to pathways ranging between 6.0 and 8.5 (Pachauri 2014). Besides, climatic simulations until 2050 do not significantly differ among the four pathways (RCP2.6, RCP4.5, RCP6.0, RCP8.5).
We downloaded five monthly atmospheric variables for both baseline and future scenarios: mean surface temperature (tas), maximum surface temperature (tasmax), minimum surface temperature (tasmin), precipitation flux (pr), and water evaporation flux (evspsbl). The baseline scenario represents the climate conditions contemporary to most species occurrence records, ranging from 1950 to 1999. We averaged each climatic raw data set independently for this interval to obtain a unique layer of values per climatic variable under each AOGCM. For the future scenarios, we also averaged the climatic variables under each of the AOGCMs for seven different intervals: 2018–2022, 2023–2027, 2028–2032, 2033–2037, 2038–2042, 2043–2047 and 2048–2052. Here, we refer to each time-step using the centroid of the interval, namely: 2020, 2025, 2030, 2035, 2040, 2045 and 2050. Subsequently, we downscaled the climatic variables to the resolution of 0.1° × 0.1° (latitude/longitude) for the Neotropical region using the change factor approach across an ordinary krigging process (see details in Lima-Ribeiro et al. 2015). Finally, from the downscaled averages of monthly temperature (tas, tasmin and tasmax) and precipitation (pr), we computed the 19 bioclimatic variables present in the WorldClim database (www.worldclim.org/current). The monthly values of water evaporation flux (evspsbl) were summed to generate a unique value of total evapotranspiration per interval considered.
Soil variables
We downloaded 30 surface and sub-soil variables from the Harmonized World Soil Database (http://webarchive.iiasa.ac.at/Research/LUC/External-World-soil-database/HTML/) (Table S1). To select the least correlated variables which also explained most of the data variation, we did a Factor Analysis (FA) with all the 30 variables. This analysis is similar to a PCA, but instead of combining the weighted observed variables in axes of explanation, the FA groups the variables in latent axes, also called factors, and at the same time maintains the correlation value of each one of the original variables to the factors created (Child 1990). We then selected only those variables with greatest correlation values with each factor, but also considering their biological meaning. We ended up with two soil variables: percentage of clay in the topsoil and topsoil pH in soil–water solution. These variables were upscaled to the same spatial resolution and extent as the bioclimatic variables (0.1° × 0.1° latitude/longitude and Neotropical region, respectively) using an ordinary krigging process (Lima-Ribeiro et al. 2015). Because soil aspects are restricted to the baseline period, we assumed soil conditions to be constant through time, being used in species distribution modelling as “constraint variables” to better model the environmental preferences of plant species.
Occurrence records
We gathered plant records occurring in Brazil from the Global Biodiversity Information Facility (GBIF) database (http://www.gbif.org/what-is-gbif) and from the Brazilian National Centre for Plant Conservation (CNCFlora) database (http://cncflora.jbrj.gov.br/portal/). We opted for including Brazilian endemic species (Forzza et al. 2010) under any of the three threat categories (vulnerable, endangered, and critically endangered) established by the Brazilian Red List of Threatened Plants (Martinelli and Moraes 2013) following the IUCN classification system (IUCN 2014). This selection was made considering the urgency of species protection and to represent in subsequent prioritization analyses those species moving in or out of the Cerrado in the future. Because predictive uncertainties are large in models built from few occurrence records (Pearson et al. 2007), we selected species with at least 10 occurrences, a number previously demonstrated to generate models with a very good accuracy even when they are not corrected for environmental or geographical bias (see details in Varela et al. 2014).
From a total of 2113 threatened Brazilian plant species, only 504 species remained for the subsequent prioritization analyses after the implementation of all filters. The exclusion of very rare species could be detrimental to conservation management in the sense that they can be particularly in need for protection (Engler et al. 2004). On the other hand, including only those species with minimally acceptable sample sizes could ensure less biased and more reliable predictive models (Wisz et al. 2008), besides guaranteeing a more feasible computational effort. Since our final aim was to test for different strategies of prioritization, we opted for deriving better predictive models rather than including a larger number of species.
Modelling species distribution through time
Once we had the 19 bioclimatic variables calculated, one variable of evapotranspiration, and two soil variables interpolated, we did a Factor Analysis (FA) using all of them. The variables with highest values in the FA had the highest correlations with the factors proposed by the model, which are analogous to PCA axes. We chose the main variables indicated by the FA and performed a Pearson’s correlation, to reduce the number of highly collinear predictors in the model. At the end, we ended up with eight variables to be used in the species distribution models (SDM) for each plant species: annual mean temperature, mean diurnal range, temperature seasonality, annual precipitation, precipitation seasonality, precipitation of driest quarter, evapotranspiration, and percentage of clay in the top soil. Using a correlation approach, we associated the eight environmental variables and all selected plant species occurrence records using five different modelling techniques: BIOCLIM (Busby 1991), Gower distance (Carpenter et al. 1993), Multivariate Adaptive Regression Splines (MARS; Friedman 1991), Random Forest (RF; Breiman 2001), and Support Vector Machine (SVM; Müller et al. 2001). These methods encompass a good variety of strategies of statistical adjustment (Tessarolo et al. 2014), as well as differ in complexity from simple bioclimatic envelope procedures and distance methods based on presence-only data to regression and machine-learning approaches that require presence and absence/background data (Rangel and Loyola 2012).
Because real absence data are not available for Brazilian threatened plants, we randomly generated pseudo-absences over the Neotropical extent to build the distribution models, keeping every species prevalence equal to 0.5 (i.e. number of pseudo-absences equal to species occurrences). From all occurrence and pseudo-absence data, 75% were used for training the models and 25% for testing them. To ensure robustness of our predictions (Broennimann et al. 2007), the random data partition was repeated 10 times for every combination of modelling technique and AOGCM used per time-step (4 AOGCMs × 5 modelling techniques × 10 repetitions = 200 models per species per time-step).
We assessed the predictive accuracy of each model (calibrated with the training occurrence records) by computing the True Skill Statistic (TSS) using the threshold that maximizes the sum of sensitivity and specificity to compute the confusion matrix (Zweig and Campbell 1993). For each species, from the 10 repetitions of data partitioning for each combination of AOGCM and modelling method, we chose the model with the highest TSS value to project onto all eight time-steps (1 baseline + 7 future). The projections resulted in 20 distribution maps (4 AOGCMs × 5 modelling methods) for each time-step, totaling 160 distribution maps per species (4 AOGCMs × 5 modelling methods × 8 time intervals). We computed the standard deviation (SD) among the standardized suitability values predicted by the 20 the models in each grid cell, obtaining an uncertainty map per species for each interval (Araújo and New 2007). Because SD measures the dissimilarity of model predictions per grid cell, this procedure offers a spatially explicit estimate of methodological uncertainties among the combinations of all AOGCMs and modelling methods, which should be accounted for in conservation planning (see Carvalho et al. 2011).
Next, we converted the continuous predictions generated for each species into binary maps (presence or absence) by using the same threshold aforementioned (Zweig and Campbell 1993). Finally, we combined the projections using the majority consensus; i.e. at least 50% of the models (4 AOGCMs × 5 modelling methods) should predict the species occurrence in a given grid cell for it to be converted into a presence; otherwise, the grid cell represented the absence of a species. All the analyses were done using the R package “dismo” (Hijmans et al. 2012).
Systematic planning of conservation scenarios
Species maximum dispersal distances
We used the regressive models by Tamme et al. (2014) to estimate species’ dispersal distances based on two key life-history traits: growth form (tree, shrub, and herb) and dispersal syndrome (animal, ant, wind, ballistic, and no special syndrome) (Tamme et al. 2014). The growth form information was downloaded from the Brazilian Flora 2020 website (http://floradobrasil.jbrj.gov.br/reflora/listaBrasil/PrincipalUC/PrincipalUC.do?lingua=en#CondicaoTaxonCP), using the R package “flora”, and the dispersal syndrome information was gathered from a thorough research of the literature. We estimated all individual species dispersal distances using the R package “dispeRsal” (Tamme et al. 2014).
Yearly land-use projections
Both baseline and future yearly land-use maps used for the Cerrado were extracted from Soares-Filho et al. (2016) and contained information on both croplands and presence/absence of savanna or forest vegetation. The future yearly land-use maps were built from projections of agricultural expansion for 2024 and extrapolated to 2050 based on historical trends between 1994 and 2013 (MAPA 2014). The spatial allocation of croplands throughout the Cerrado was based on information on climatic suitability for each crop and on probabilities of vegetation loss, which were estimated as a function of spatial determinants of habitat conversion, such as distance to roads and previously converted areas. Vegetation loss was constrained to areas where conversion is legal in accordance with the Forest Code (for further details see Soares-Filho et al. 2016).
To transform all land-use maps to the same resolution and extent of the SDMs, we used a resampling method called nearest neighbor, which is a method used to manipulate categorical datasets (Hijmans et al. 2016). This resampling method assigns to a new grid the value of the input grid cell whose center is closest to the output grid cell center. We also transformed all grid cells classified differently from forest/savanna into no-vegetation sites. Therefore, predictions were represented in a binary format, i.e. sites were considered to be either cleared or not. We performed the resampling and reclassification using the R package “raster” (Hijmans et al. 2016).
Prioritization analyses
We used Zonation v.4.0 (Moilanen et al. 2014) to run the spatial prioritization analyses. Through a balance across input data, such as SDMs and connectivity information, this software identifies valuable areas for retaining habitat quality for multiple species, thus generating a hierarchical ranking of conservation importance for all grid cells across the landscape.
Here, we refer to the importance attributed to each grid cell as conservation value. The calculation of this conservation value depends on the input variables considered and on the rule of choice for progressive cell removal. The first cell removed from the landscape receives the lowest conservation value, while the last cell removed scores the highest conservation value. We chose the additive benefit function as our rule of cell removal, because it usually delivers the highest average proportion of species/features retained in the landscape compared to other functions (Arponen et al. 2005), while also accounting for connectivity and species weights, among other variables (Thomson et al. 2009; Kujala et al. 2013; Moilanen et al. 2014). The input variables included in the Zonation analysis were the following: i) species-specific maximum dispersal distances, ii) potential distribution of species predicted by SDMs, (iii) uncertainty maps, and iv) a map of current PAs.
Yearly land-use predictions generated for the Cerrado were included in the prioritization analysis a posteriori (i.e. not as an input in Zonation) to identify presence/absence of vegetation remnants both in the baseline and future time-steps. This procedure enabled us to simulate the two methods of setting priorities for conservation actions: (i) one that accounts for future vegetation loss focused on conservation impact, and (ii) another that ignores vulnerability to land conversion, focused on representation.
We also incorporated in the Zonation analysis a facility called distribution smoothing. This is a two-dimensional kernel smoothing using a species-specific parameter (Moilanen et al. 2014) which, in our case, identified semi-continuous areas environmentally suitable for a given species per time-step. The level of smoothing for each species was determined by its maximum dispersal distance, assuming that species are capable of reaching distances within their maximum dispersal capacity (Moilanen et al. 2005). Therefore, areas within each species’ maximum dispersal distance received higher conservation values, contributing to an overall increase in spatial connectivity among selected priority sites.
Prioritization frameworks that attempt to account for future conditions require not only the selection of areas with high conservation values, but also penalties for high uncertainty in species occurrences, in our case derived from predictive uncertainty across SDM projections (Moilanen et al. 2006a; Faleiro et al. 2013; Lemes et al. 2013). The danger of not considering this source of uncertainty is that conservation implementation could be seriously compromised (Ascough et al. 2008), considering that the process of decision-making already has some inherent uncertainty. We applied a Zonation facility called distribution discounting (Meller et al. 2014). This analysis subtracts the distribution model information in a given grid cell (1 or 0) for each species by the SD in that grid cell for a specific period of time (see Moilanen et al. 2006b), thus targeting areas with high conservation value (i.e. high contribution to overall species representation) and low uncertainty.
From the potential distributions of 504 plant species, we kept only those occurring or predicted to occur in the Cerrado during any interval in the future. The final number of species included in subsequent analyses totaled 221 (for each one, eight SDMs each considering all time-steps) (Table S2), belonging to 55 families of angiosperms. Of these, four are classified as critically endangered, 130 as endangered, and 87 as vulnerable. In such analyses, species weights generally are used to scale the conservation value of each prioritized site. Therefore, we established the relative importance of species based on their threat categories (Martinelli and Moraes 2013). Inevitably, the weighting criteria were somewhat arbitrary, but we used the following rationale: critically endangered (CR) species were considered to have twice the priority (100% more) of non-threatened species, receiving a multiplying weight of 2. Endangered (EN) species received a weight of 1.5 (50% more than non-threatened species) and vulnerable (VU) species received a weight of 1.25 (25% more than non-threatened species).
The current network of PAs was included in the prioritization analysis as a mask file, which is a facility in Zonation that establishes an order of removal across the edge grid cells based on their pre-defined conservation importance. The sites inside PAs are assigned a higher value of conservation importance to force their removal only after all non-protected sites are ranked (Lehtomäki and Moilanen 2013). This is a way of guaranteeing that the highest priority sites (i.e. sites inside PAs) persist at all time-steps.
The land-use model had yearly predictions ranging from 2012 (referred to as baseline) until 2050 with binary information on the presence of vegetation remnants. We opted to include in the prioritization only the projections corresponding to the first year of each time interval considered in the analysis. The average rate of land conversion was 2.3% of the biome’s area every five years.
Conservation intervention scenarios
Here, we simulated four scenarios varying in methods of scheduling action and setting priorities, limited by a fixed budget (determined by a fixed 8.4% of the Cerrado’s area or 17,044,636 ha) over 35 years. We assessed these scenarios against a referential scenario of no further protection, also called the counterfactual scenario. The 8.4% conservation target was defined based on the calculation of the mean percentage area occupied by new PAs every five years (~ 1.2% of the biome) in the last 30 years in Brazil, multiplied by seven time periods. Adding the total area already covered by PAs (8.3%) to the total area targeted for prioritization (8.4%), by 2050 the four prioritization scenarios involved the allocation of 16.7% of the biome (33,970,494 ha) to some type of conservation action.
By combining methods of scheduling action and setting prioritization, we simulated four intervention scenarios: (1) acting now—maximizing representation, (2) acting now—maximizing conservation impact, (3) time-step action—maximizing representation, and (4) time-step action—maximizing conservation impact (Fig. 1, Table 1). We distinguished the terms for prioritization because representation, as typically used, has only a tenuous relationship with impact (Pressey et al. 2017). These scenarios were designed to explore how varying methods of scheduling action and setting priorities could influence the impact of each planning scenario in terms of avoided vegetation loss and biodiversity representation inside PAs and priority sites.
Since impact can be measured only against a counterfactual scenario of no intervention (Andam et al. 2008), we simulated the no-action scenario through the establishment of no conservation interventions until 2050, except for maintaining the current network of PAs. Additionally, we assumed, for all scenarios, that priority sites and PAs were effective in mitigating species’ range loss by preventing vegetation loss and facilitating range shifts caused by climate change. We also assumed that non-protected (either by priority sites or PAs) and non-converted sites were available for selection. Further explanation of scenarios is detailed in Table 1.
Evaluation of scenarios in 2050
We produced a map of priority sites for each of the four scenarios. Based on each scenario, we also produced a map for each species’ remaining distribution containing the species’ range in 2050 accumulated with their previous ranges that fell inside priority sites in any of the time-steps or that, during this period, entered the current network of PAs (Table 1, Fig. 1). Since no prioritization analysis was carried out in the counterfactual scenario, species’ remaining ranges under this scenario were a result of the species’ range in 2050 accumulated with their previous distributions that fell inside PAs in any of the time-steps. To make all five scenarios comparable, the year-base of evaluation was 2050.
Using the information on selected priority sites, we calculated the avoided vegetation loss (% impact) and the average percentage of species’ range covered by PAs and priority sites (% representation) as measures of conservation success. For each scenario, we measured % impact as (C-T)/C*100, where C is the total area converted throughout the whole time-series (baseline—2050) in the counterfactual scenario, and T is the total area converted by 2050 in one of the intervention scenarios. The second metric is the result of the average size of all species’ ranges represented within priority sites and PAs in 2050, divided by their total range size across the landscape in this same year, multiplied by 100.
Results
In scenarios aimed at targeting maximum species’ representation only (maximizing representation), priority sites were mostly distributed in the center and throughout the eastern border of Cerrado (Fig. 2a, c), while scenarios aimed at maximizing conservation impact selected a wider spread of priority sites, distributed mostly in the north-eastern Cerrado (Fig. 2b, d). Scenarios differing in their scheduling, but aimed at either maximizing conservation impact or representation, overlapped spatially between 55% and 85% of the total area selected (Table 2). Scenarios built based on the same method of scheduling priorities (immediately or sequentially through time), or that differed in both strategies of scheduling and setting priorities, overlapped spatially between 20% and 30% of the total area selected (Table 2).
Spatial distribution of priority sites and existing protected areas (PAs) in each of the intervention scenarios across the Cerrado biome: a acting now—maximizing representation, b acting now—maximizing conservation impact, c time-step action—maximizing representation and d time-step action—maximizing conservation impact
In the counterfactual scenario, an additional 15% (30,205,233 ha) of the Cerrado was converted by 2050 (Fig. S1A), to give place to various crop plantations mostly in the northern Cerrado (Soares-Filho et al. 2016). The impact or percentage avoided loss of scenarios that maximized impact was between 3.5 and 4.5 times larger than that of the representation approaches (Table 3). However, the average percentage of species’ range sizes represented within PAs and priority sites was similar among the intervention scenarios (Table 3). When analyzed by threat category, the difference among scenarios became even smaller, especially for the critically endangered species, which had almost the same level of representation inside PAs and priority sites across scenarios (Table 4).
Discussion
Here, we offer a way of accommodating spatially explicit land-use and species distribution projections under climate change within prioritization scenarios, while also providing a new perspective on predictive impact to conservation, something that has been needed for some time (Pressey et al. 2004, 2015, 2017). Our findings show that targeting conservation impact brings greater outcomes in terms of avoided vegetation loss, while still covering large proportions of species’ ranges inside PAs and priority sites (Table 3). Even though the representation approaches, aimed at maximizing species representation only, delivered marginally higher amounts of species’ coverage compared to the impact strategies, they were much less effective in stopping land conversion. Additionally, our results show that the strategies for setting priorities (maximizing impact or representation) were more important in determining the amount of avoided land conversion or the amount of species’ ranges protected than strategies varying in scheduling (time-step action or acting now) (Table 3).
The results on the spatial location of priority sites among the four intervention scenarios and the slightly smaller values of species’ representation delivered by the impact scenarios can be explained by the uneven distribution of both threatened species and vegetation loss predictions throughout the Cerrado. In this biome, there is a collection bias of plant species towards areas with easiest access routes, which are also places where species have been subject to historical threatening processes, such as road constructions, human population increases, and urban development (Oliveira et al. 2016). Because spatial biases in records inevitably affect SDMs, there is a clear species richness bias towards the southern and also most developed part of the Cerrado (Fig. S1B), where there are fewer opportunities for further land-use expansion (MMA 2007). Conversely, the northern part of the Cerrado is poorly studied, with fewer recorded occurrences of plant species, and has the largest tracts of native vegetation and the greatest opportunities for development and land conversion. Accordingly, our land-use change projections predicted that most vegetation conversion will likely take place in the north of the biome (Fig. S1A). Our projections are supported by the policy of agricultural expansion in Brazil in the so-called Matopiba region (MAPA 2014), which covers the northern Cerrado. These north–south differences explain why the impact scenarios tended to select sites with fewer species and more threatened sites in the northern region (Fig. S1E and S1G; Fig. 2b, d), while representation approaches prioritized less vulnerable and species’ rich sites in the central and southern Cerrado (Fig. S1D and S1F; Fig. 2a, c).
Yet, despite such spatial differences in the location of priority sites, the impact and representation scenarios still delivered similar average sizes of species’ ranges protected. Even after disentangling the average values of species’ range protection by their threat statuses, the results are still similar within each category, regardless of the intervention scenario (Table 4). Many works have focused on the use of species’ representation as their only measure of performance, which can be a good strategy if we are dealing with a landscape where threats have ceased, are completely unpredictable or where land conversion is not the main pressure (Arponen et al. 2005; Lourival et al. 2011; Summers et al. 2012).
Here we show that minimizing overall biodiversity loss by saving species both from climate change and land conversion could be achieved with any of the impact strategies. First, prioritizing ecosystem protection over areas targeted for their biodiversity features prevents us from relying solely on species distribution models, which although useful, have high uncertainties (Dessai and Hulme 2004; Araújo and New 2007) and are based on only a subset of regional taxa. These models are almost always generated from highly biased species records (Kadmon et al. 2004; Tessarolo et al. 2014), and tend to be especially challenging for species with small numbers of point occurrences, such as threatened and endemic species. Vegetation information, however, can be a surrogate for general biodiversity and is less subject to taxonomic and geographical bias. A second benefit of targeting threats is that we guarantee the persistence of both biological patterns and processes associated with native vegetation, which are commonly neglected relative to representation as a conservation goal (Carroll et al. 2001). A third and last benefit of the impact approach is the protection of valuable ecosystem services (e.g. water provision, erosion control) derived from essential ecological functions that otherwise would be put at risk (Hoekstra et al. 2005; Millennium Ecosystem Assessment 2005). This last benefit is particularly crucial for the Cerrado biome.
Most prioritization frameworks have focused on selecting areas that minimize future conflicts with other competing land uses, which are usually places less likely to be converted (Faleiro et al. 2013). Although such strategies might facilitate implementation of conservation actions, the selection of places with higher likelihood of being converted might, in contrast, have two positive effects: one of protecting the most biologically important and vulnerable fragments, and the other of leaving as unprotected those sites located in areas with low pressure for land conversion and least need for protection (Devillers et al. 2014; Pressey et al. 2015). Besides, by targeting vulnerable areas for protection, we could also contribute to climate change adaptation and mitigation, through ecosystem-based adaptation (Munang et al. 2013).
Until now few papers have attempted to integrate multiple threats, such as climate change and land conversion, and have rarely focused on maximizing conservation impact combining more than one species into a single prioritization framework (Santika et al. 2015; Visconti and Joppa 2015). Here, we integrated climate change and habitat loss into conservation planning by explicitly targeting the most valuable sites to multiple plant species under climate change while also minimizing land conversion. There is strong evidence that future biodiversity patterns will be a result of the interactions between both these threatening processes (Brook et al. 2008; Nepstad et al. 2008), which means they cannot be considered in isolation (Hansen et al. 2001). In fact, this paper covers a gap in the literature by accounting both for the direct effects of climate change, through shifts in species ranges, and the indirect effects of climate change through changes in crop suitability (Jones et al. 2016). Recent papers have highlighted how severe these effects can be (Segan et al. 2015) and how accounting for and trying to mitigate such stressors could help biodiversity to cope with climate change and contribute to the success of conservation plans (Selig et al. 2014).
Conservation actions able to safeguard biodiversity and mitigate threats include, but should not be restricted to, the expansion and creation of PAs. Recent evidence suggests a trend of downgrading, downsizing, and degazettement in PAs, which is generally influenced and determined by factors such as dynamic governance regimes, illegal activities that frequently go beyond PA boundaries, and petroleum and mineral extraction pressures (Mascia and Pailler 2011; Bernard et al. 2014; Mascia 2014). Although we have assumed that protected sites are efficient in completely removing the risk of biodiversity and vegetation loss, future interventions should consider improving monitoring and enforcement, increasing habitat quality outside PAs, and developing policies for endangered species (Strassburg et al. 2017). Considering other kinds of strategies could prevent us from relying on PAs as the only conservation tool possible, whose efficiency has proved to be compromised by social pressures.
The scenarios built here have potential for implementation, especially if current conservation policies are adapted to explicitly recognize impact as a goal. For example, the Convention on Biological Diversity targets could be implemented under one of the impact strategies proposed here (Joppa and Pfaff 2009). Carbon-based payment programs, such as REDD, serve as an important incentive to reduce deforestation and could be boosted by shifts in conservation policies towards maximization of conservation impact (Pfaff et al. 2014). However, as with any conservation measure, the success of the prioritization strategy depends on the gain in the targeted objective. If our objective were to restore natural vegetation, the most efficient strategy would be to select degraded sites with the highest potential for natural regeneration (Crouzeilles et al. 2015). Therefore, the prioritization recommendations presented here are not meant to be imposed on decision-makers, but rather to serve as a guideline for conservation to achieve the primary goal of minimizing biodiversity loss.
After this first attempt at incorporating climate change effects in dynamic prioritization plans, it is imperative that we work to improve and simplify the approach. It is in our interests as conservation scientists that our spatial prioritization outputs are translated into feasible planning products that can effectively separate biodiversity from threatening processes. A first step for fulfilling the “knowing-doing” gap and facilitating this bridge between scientists and practitioners (Knight et al. 2011) could be the development of interactive platforms that allow not only the execution of prioritizations scenarios (with multiple choices, ranging from representation to impact scenarios) but also the construction of species distribution models or the proper handling of other relevant input variables used in conservation planning. Due to the difficulties involved in dealing with so many variables, having a network of scientists and practitioners working on a unified project would be crucial to make it feasible. In the literature, there has been a significant progress in conservation planning but little conversation between scientists on how to integrate their advances and make them available collaboratively to society and decision-makers (Knight et al. 2008).
Our study involves some caveats that are worth mentioning for future refinements. Our model does not incorporate the possible displacement of land conversion that the establishment of PAs could cause in surrounding areas (Pfaff and Robalino 2012). However, a recent work has showed that avoided vegetation loss in the Cerrado was not offset by an increase in conversion elsewhere (Carranza et al. 2014). Therefore, although worth testing, it is not completely unrealistic to assume that, if implemented, the current approach would not necessarily generate pervasive effects. A second limitation of this work concerns the number of species included in our analysis, which decreased with increasing threat level. This pattern finds explanation in the overall inverse correlation between threat category and range size (here predicted from the number of occurrences) (Summers et al. 2012). Excluding species with less than 10 occurrences from the analysis could influence prioritization results by ignoring species with higher needs for intervention, but we were not able to quantify this effect.
Lastly, it is important to highlight that the spatial prioritizations we proposed are tools to help people make decisions grounded on available and relevant information for fulfilling the goals initially established. To deliver effective conservation actions, results such as ours must be integrated with social, political, and institutional initiatives. Nonetheless, such a theoretical approach can motivate rethinking about where to focus conservation in the near future to minimize the loss of biodiversity.
References
Andam KS, Ferraro PJ, Pfaff A et al (2008) Measuring the effectiveness of protected area networks in reducing deforestation. Proc Natl Acad Sci USA 105:16089–16094. https://doi.org/10.1073/pnas.0800437105
Araújo MB, New M (2007) Ensemble forecasting of species distributions. Trends Ecol Evol 22:42–47. https://doi.org/10.1016/j.tree.2006.09.010
Arponen A, Heikkinen RK, Thomas CD, Moilanen A (2005) The value of biodiversity in reserve selection: representation, species weighting, and benefit functions. Conserv Biol 19:2009–2014. https://doi.org/10.1111/j.1523-1739.2005.00218.x
Ascough JC, Maier HR, Ravalico JK, Strudley MW (2008) Future research challenges for incorporation of uncertainty in environmental and ecological decision-making. Ecol Modell 219:383–399. https://doi.org/10.1016/j.ecolmodel.2008.07.015
Bernard E, Penna LAO, Araújo E (2014) Downgrading, downsizing, degazettement, and reclassification of protected areas in Brazil. Conserv Biol 28:939–950. https://doi.org/10.1111/cobi.12298
Breiman L (2001) Random forests. Mach Learn 45(1):5–32. https://doi.org/10.1017/CBO9781107415324.004
Broennimann O, Treier UA, Müller-Schärer H et al (2007) Evidence of climatic niche shift during biological invasion. Ecol Lett 10:701–709
Brook BW, Sodhi NS, Bradshaw CJA (2008) Synergies among extinction drivers under global change. Trends Ecol Evol 23:453–460. https://doi.org/10.1016/j.tree.2008.03.011
Busby JR (1991) BIOCLIM: a bioclimate analysis and prediction system. In: Margules CR, Austin MP (eds) Nature conservation: cost effective biological surveys and data analysis. CSIRO, Canberra, pp 64–68
Carpenter G, Gillison AN, Winter J (1993) Domain—a flexible modeling procedure for mapping potential distributions of plants and animals. Biodivers Conserv 2:667–680. https://doi.org/10.1007/BF00051966
Carranza T, Balmford A, Kapos V, Manica A (2014) Protected area effectiveness in reducing conversion in a rapidly vanishing ecosystem: the Brazilian Cerrado. Conserv Lett 7:216–223. https://doi.org/10.1111/conl.12049
Carroll C, Noss RF, Paquet PC (2001) Carnivores as focal species for conservation planning in the rocky mountain region. Ecol Appl 11:961–980
Carvalho SB, Brito JC, Crespo EG et al (2011) Conservation planning under climate change: toward accounting for uncertainty in predicted species distributions to increase confidence in conservation investments in space and time. Biol Conserv 144:2020–2030. https://doi.org/10.1016/j.biocon.2011.04.024
Child D (1990) The essentials of factor analysis, 2nd edn. Cassel Educational Limited, London
Costello C, Polasky S (2004) Dynamic reserve site selection. Resour Energy Econ 26:157–174. https://doi.org/10.1016/j.reseneeco.2003.11.005
Crouzeilles R, Beyer HL, Mills M et al (2015) Incorporating habitat availability into systematic planning for restoration: a species-specific approach for Atlantic Forest mammals. Divers Distrib. https://doi.org/10.1111/ddi.12349
Dessai S, Hulme M (2004) Does climate adaptation policy need probabilities? Clim Policy 4:107–128. https://doi.org/10.1080/14693062.2004.9685515
Devillers R, Pressey RL, Grech A et al (2014) Reinventing residual reserves in the sea: are we favouring ease of establishment over need for protection? Aquat Conserv Mar Freshw Ecosyst 25:480–504. https://doi.org/10.1002/aqc.2445
Drechsler M (2005) Probabilistic approaches to scheduling reserve selection. Biol Conserv 122:253–262. https://doi.org/10.1016/j.biocon.2004.07.015
Engler R, Guisan A, Rechsteiner L (2004) An improved approach for predicting the distribution of rare and endangered species from occurrence and pseudo-absence data. J Appl Ecol 41:263–274. https://doi.org/10.1111/j.0021-8901.2004.00881.x
Faleiro FV, Machado RB, Loyola RD (2013) Defining spatial conservation priorities in the face of land-use and climate change. Biol Conserv 158:248–257. https://doi.org/10.1016/j.biocon.2012.09.020
Ferraro PJ (2009) Counterfactual thinking and impact evaluation in environmental policy. In: Birnbaum M, Mickwitz P (eds) Environmental program and policy evaluation: new directions for evaluation. Jossey-Bass, San Francisco, pp 75–84
Forzza RC, Leitman PM, Costa A et al (2010) Catálogo de plantas e fungos do Brasil, vol 2. Andrea Jakobsson Estúdio: Instituto de Pesquisas Jardim Botânico, Rio de Janeiro
Friedman JH (1991) Multivariate adaptive regression splines. Ann Stat 19:1–141
Hansen AJ, Neilson RP, Dale VH et al (2001) Global change in forests: responses of species, communities, and biomes. Bioscience 51:765. https://doi.org/10.1641/0006-3568(2001)051%5b0765:GCIFRO%5d2.0.CO;2
Hijmans ARJ, Phillips S, Leathwick J, Elith J (2012) Package “dismo”. Species distribution modeling
Hijmans RJ, van Etten J, Cheng J et al (2016) Package “raster”. Geographic data analysis and modeling
Hoekstra JM, Boucher TM, Ricketts TH, Roberts C (2005) Confronting a biome crisis: global disparities of habitat loss and protection. Ecol Lett 8:23–29. https://doi.org/10.1111/j.1461-0248.2004.00686.x
IPCC (2014) Climate change 2014: Synthesis report. Contribution of working groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on climate change. Core Writing Team, Geneva
IUCN (2014) Guidelines for using the IUCN Red List Categories and Criteria
Jones KR, Watson JEM, Possingham HP, Klein CJ (2016) Incorporating climate change into spatial conservation prioritisation: a review. Biol Conserv 194:121–130. https://doi.org/10.1016/j.biocon.2015.12.008
Joppa LN, Pfaff A (2009) High and far: biases in the location of protected areas. PLoS ONE 4:1–6. https://doi.org/10.1371/journal.pone.0008273
Kadmon R, Farber O, Danin A (2004) Effect of roadside bias on the accuracy of predictive maps produced by bioclimatic models. Ecol Appl 14:401–413. https://doi.org/10.1890/02-5364
Klink CA, Moreira AG (2002) Past and current human occupation, and land use. In: Oliveira PS, Marquis RJ (eds) The cerrados of Brazil: ecology and natural history of a neotropical savanna. Columbia University Press, New York, pp 69–88
Knight AT, Cowling RM, Rouget M et al (2008) Knowing but not doing: selecting priority conservation areas and the research-implementation gap. Conserv Biol 22:610–617. https://doi.org/10.1111/j.1523-1739.2008.00914.x
Knight AT, Cowling RM, Boshoff AF et al (2011) Walking in STEP: lessons for linking spatial prioritisations to implementation strategies. Biol Conserv 144:202–211. https://doi.org/10.1016/j.biocon.2010.08.017
Kujala H, Moilanen A, Araújo MB, Cabeza M (2013) Conservation planning with uncertain climate change projections. PLoS ONE. https://doi.org/10.1371/journal.pone.0053315
Ladle RJ, Whittaker RJ (2011) Social values and conservation biogeography. In: Conservation biogeography, 1st edn. Blackwell, Chichester, p 297
Lehtomäki J, Moilanen A (2013) Environmental modelling and software methods and work flow for spatial conservation prioritization using Zonation. Environ Model Softw 47:128–137. https://doi.org/10.1016/j.envsoft.2013.05.001
Lemes P, Loyola RD (2013) Accommodating species climate-forced dispersal and uncertainties in spatial conservation planning. PLoS ONE 8:e54323. https://doi.org/10.1371/journal.pone.0054323
Lemes P, Melo AS, Loyola RD (2013) Climate change threatens protected areas of the Atlantic Forest. Biodivers Conserv 23:357–368. https://doi.org/10.1007/s10531-013-0605-2
Lima-Ribeiro MS, Varela S, González-Hernández J et al (2015) Ecoclimate: a database of climate data from multiple models for past, present, and future for macroecologists and biogeographers. Biodivers Inform 10:1–21. https://doi.org/10.17161/bi.v10i0.4955
Lourival R, Drechsler M, Watts ME et al (2011) Planning for reserve adequacy in dynamic landscapes; maximizing future representation of vegetation communities under flood disturbance in the Pantanal wetland. Divers Distrib 17:297–310. https://doi.org/10.1111/j.1472-4642.2010.00722.x
Martinelli G, Moraes MA (2013) Livro Vermelho da flora do Brasil. Instituto de Pesquisas Jardim Botânico do Rio de Janeiro, Rio de Janeiro
Mascia MB (2014) Protected area downgrading, downsizing, and degazettement (PADDD) in Africa, Asia, and Latin America and the Caribbean, 1900–2010. Biol Conserv 169:355–361. https://doi.org/10.1016/j.biocon.2013.11.021
Mascia MB, Pailler S (2011) Protected area downgrading, downsizing, and degazettement (PADDD) and its conservation implications. Conserv Lett 4:9–20. https://doi.org/10.1111/j.1755-263X.2010.00147.x
Meir E, Andelman S, Possingham HP (2004) Does conservation planning matter in a dynamic and uncertain world? Ecol Lett 7:615–622. https://doi.org/10.1111/j.1461-0248.2004.00624.x
Meller L, Cabeza M, Pironon S et al (2014) Ensemble distribution models in conservation prioritization: from consensus predictions to consensus reserve networks. Divers Distrib 20:309–321. https://doi.org/10.1111/ddi.12162
Millennium Ecosystem Assessment (2005) Ecosystems and Human well-being: synthesis. Island Press, Washington, DC
Ministério da Agricultura Pecuária e Abastecimento (2014) Projeções do Agronegócio: Brasil 2013/2014 a 2023/2024—Projeções de Longo Prazo, 5nd edn. Ministério da Agricultura, Pecuária e Abastecimento. Assessoria de Gestão Estratégica, Brasília
Ministério do Meio Ambiente (2007) Cerrado e Pantanal: áreas e ações prioritárias para conservação da biodiversidade. MMA, Brasília
Mittermeier RA, Gil PR, Hoffman M et al (2004) Hotspots revisited: earth’s biologically richest and most endangered terrestrial ecoregions. Cemex, Conservation International, Agrupación Sierra Madre, Mexico City
Moilanen A, Franco AMA, Early RI et al (2005) Prioritizing multiple-use landscapes for conservation: methods for large multi-species planning problems. Proc R Soc B Biol Sci 272:1885–1891. https://doi.org/10.1098/rspb.2005.3164
Moilanen A, Runge MC, Elith J et al (2006a) Planning for robust reserve networks using uncertainty analysis. Ecol Modell 199:115–124. https://doi.org/10.1016/j.ecolmodel.2006.07.004
Moilanen A, Wintle BA, Elith J, Burgman M (2006b) Uncertainty analysis for regional-scale reserve selection. Conserv Biol 20:1688–1697. https://doi.org/10.1111/j.1523-1739.2006.00560.x
Moilanen A, Pouzols FM, Meller L et al (2014) Spatial conservation planning methods and software Zonation
Müller KR, Mika S, Rätsch G et al (2001) An introduction to kernel-based learning algorithms. IEEE Trans Neural Netw 12:181–201. https://doi.org/10.1109/72.914517
Munang R, Thiaw I, Alverson K et al (2013) Climate change and ecosystem-based adaptation: a new pragmatic approach to buffering climate change impacts. Curr Opin Environ Sustain 5:67–71. https://doi.org/10.1016/j.cosust.2012.12.001
Nepstad DC, Stickler CM, Filho BS- et al (2008) Interactions among Amazon land use, forests and climate: prospects for a near-term forest tipping point. Philos Trans R Soc Lond B Biol Sci 363:1737–1746. https://doi.org/10.1098/rstb.2007.0036
O’Hanley JR, Church RL, Keith Gilless J (2007) Locating and protecting critical reserve sites to minimize expected and worst-case losses. Biol Conserv 134:130–141. https://doi.org/10.1016/j.biocon.2006.08.009
Oliveira U, Paglia AP, Brescovit AD et al (2016) The strong influence of collection bias on biodiversity knowledge shortfalls of Brazilian terrestrial biodiversity. Divers Distrib 22:1232–1244. https://doi.org/10.1111/ddi.12489
Pachauri RK (2014) Climate Change 2014. Synthesis Report
Pearson RG, Raxworthy CJ, Nakamura M, Townsend Peterson A (2007) Predicting species distributions from small numbers of occurrence records: a test case using cryptic geckos in Madagascar. J Biogeogr 34:102–117. https://doi.org/10.1111/j.1365-2699.2006.01594.x
Pereira HM, Leadley PW, Proença V et al (2010) Scenarios for global biodiversity in the 21st century. Science 330(6010):1496–1501
Pfaff A, Robalino J (2012) Protecting forests, biodiversity, and the climate: predicting policy impact to improve policy choice. Oxf Rev Econ Policy 28:164–179. https://doi.org/10.1093/oxrep/grs012
Pfaff A, Robalino J, Lima E et al (2014) Governance, location and avoided deforestation from protected areas: greater restrictions can have lower impact, due to differences in location. World Dev 55:7–20. https://doi.org/10.1016/j.worlddev.2013.01.011
Possingham H, Ball I, Andelman S (2000) Mathematical methods for identifying representative reserve netorks. In: Ferson S, Burgman M (eds) Quantitative methods for conservation biology. Springer, New York, pp 291–305
Pressey RL, Hager TC, Ryan KM et al (2000) Using abiotic data for conservation assessments over extensive regions: quantitative methods applied across New South Wales, Australia. Biol Conserv 96:55–82
Pressey RL, Whish GL, Barrett TW, Watts MEJ (2002) Effectiveness of protected areas in north-eastern New South Wales: recent trends in six measures. Biol Conserv 106:57–69. https://doi.org/10.1016/S0006-3207(01)00229-4
Pressey RL, Watts ME, Barrett TW (2004) Is maximizing protection the same as minimizing loss? Efficiency and retention as alternative measures of the effectiveness of proposed reserves. Ecol Lett 7:1035–1046. https://doi.org/10.1111/j.1461-0248.2004.00672.x
Pressey RL, Cabeza M, Watts ME et al (2007) Conservation planning in a changing world. Trends Ecol Evol 22:583–592. https://doi.org/10.1016/j.tree.2007.10.001
Pressey RL, Visconti P, Ferraro PJ (2015) Making parks make a difference: poor alignment of policy, planning and management with protected-area impact, and ways forward. Philos Trans R Soc Lond B Biol Sci 370:20140280. https://doi.org/10.1098/rstb.2014.0280
Pressey RL, Weeks R, Gurney GG (2017) From displacement activities to evidence-informed decisions in conservation. Biol Conserv 212:337–348. https://doi.org/10.1016/j.biocon.2017.06.009
Rangel TF, Loyola RD (2012) Labeling ecological niche models. Nat Conserv 10:119–126
Santika T, Mcalpine CA, Lunney D et al (2015) Assessing spatio-temporal priorities for species’ recovery in broad-scale dynamic landscapes. J Appl Ecol 52:832–840. https://doi.org/10.1111/1365-2664.12441
Segan DB, Hole DG, Donatti CI et al (2015) Considering the impact of climate change on human communities significantly alters the outcome of species and site-based vulnerability assessments. Divers Distrib 21:1101–1111. https://doi.org/10.1111/ddi.12355
Selig ER, Turner WR, Troëng S et al (2014) Global priorities for marine biodiversity conservation. PLoS ONE 9:1–11. https://doi.org/10.1371/journal.pone.0082898
Soares-Filho B, Rajâo R, Merry F et al (2016) Brazil’s market for trading forest certificates. PLoS ONE 11:1–17. https://doi.org/10.1371/journal.pone.0152311
Strange N, Thorsen BJ, Bladt J (2006) Optimal reserve selection in a dynamic world. Biol Conserv 131:33–41. https://doi.org/10.1016/j.biocon.2006.02.002
Strassburg BBN, Brooks T, Feltran-Barbieri R et al (2017) Moment of truth for the Cerrado hotspot. Nat Ecol Evol 1:13–15. https://doi.org/10.1038/s41559-017-0099
Summers DM, Bryan BA, Crossman ND, Meyer WS (2012) Species vulnerability to climate change: impacts on spatial conservation priorities and species representation. Glob Chang Biol 18:2335–2348. https://doi.org/10.1111/j.1365-2486.2012.02700.x
Tamme R, Götzenberger L, Zobel M et al (2014) Predicting species’ maximum disperal distances from simple plant traits. Ecology 95:505–513. https://doi.org/10.1890/13-1000.1
Tessarolo G, Rangel TF, Araújo MB, Hortal J (2014) Uncertainty associated with survey design in species distribution models. Divers Distrib 20:1258–1269. https://doi.org/10.1111/ddi.12236
Thomas CD, Cameron A, Green RE et al (2004) Extinction risk from climate change. Nature 427:145–148. https://doi.org/10.1038/nature02121
Thomson JR, Moilanen AJ, Vesk PA et al (2009) Where and when to revegetate: a quantitative method for scheduling landscape reconstruction. Ecol Appl 19:817–828. https://doi.org/10.1890/08-0915.1
Thuiller W, Lavorel S, Araújo MB et al (2005) Climate change threats to plant diversity in Europe. PNAS 102:8245–8250
Varela S, Anderson RP, García-Valdés R, Fernández-González F (2014) Environmental filters reduce the effects of sampling bias and improve predictions of ecological niche models. Ecography (Cop) 37:1084–1091. https://doi.org/10.1111/j.1600-0587.2013.00441.x
Visconti P, Joppa L (2015) Building robust conservation plans. Conserv Biol 29:503–512. https://doi.org/10.1111/cobi.12416
Visconti P, Pressey RL, Segan DB, Wintle BA (2010) Conservation planning with dynamic threats: the role of spatial design and priority setting for species’ persistence. Biol Conserv 143:756–767. https://doi.org/10.1016/j.biocon.2009.12.018
Wisz MS, Hijmans RJ, Li J et al (2008) Effects of sample size on the performance of species distribution models. Divers Distrib 14:763–773. https://doi.org/10.1111/j.1472-4642.2008.00482.x
Zweig MH, Campbell G (1993) Receiver-operating characteristics (ROC) plots—a fundamental evaluation tool in clinical medicine. Clin Chem 39:561–577
Acknowledgements
Lara M. Monteiro received a master scholarship from CAPES. Robert L. Pressey’s research is funded by CNPq (Grant 308532/2014-7), O Boticário Group Foundation for Nature Protection (Grant PROG_0008_2013), and CNCFlora (Grant 065/2016). Fernanda Thiesen Brum received a postdoctoral scholarship from CNPq (Grant 152172/2016-5) and currently holds an industrial and technological development scholarship (DTI-A) by CNPq (Grant 381106/2017-9). Robert L. Pressey acknowledges the support of the Australian Research Council. Leonor Patricia C. Morellato is funded by FAPESP, the São Paulo Research Foundation (Grants #2010/52113-5 and #2013/50155-0 FAPESP-Microsoft Research Virtual Institute) and receives a Research Productivity Fellowship from CNPq. This paper is a contribution of the Brazilian Network on Global Climate Change Research funded by CNPq (Grant 437167/2016-0) and FINEP (Grant 01.13.0353.00) and of the INCT in Ecology, Evolution and Biodiversity Conservation founded by MCTIC/CNPq/FAPEG (Grant 465610/2014-5).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Guarino Rinaldi Colli.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Monteiro, L.M., Brum, F.T., Pressey, R.L. et al. Evaluating the impact of future actions in minimizing vegetation loss from land conversion in the Brazilian Cerrado under climate change. Biodivers Conserv 29, 1701–1722 (2020). https://doi.org/10.1007/s10531-018-1627-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-018-1627-6