Abstract
Global oil palm expansion has caused substantial ecological damage to tropical biodiversity. We quantified wild mammal richness in large oil palm plantation estates and semi-traditional oil palm smallholdings in Peninsular Malaysia. We sampled 41 plantation estates and 14 smallholdings, and used line-transect surveys coupled with semi-structured interviews to develop a database of the native mammals found in oil palm landscapes. Semi-structured interviews revealed a total of 32 mammal species, including 13 IUCN Red Listed taxa of high conservation value. Our results showed that human activity and the size of patches of remnant rainforest were important factors influencing the richness of mammal species in oil palm landscapes. More carnivorous and herbivorous species were reported in smallholdings than plantation estates, most probably as a response to greater habitat heterogeneity in smallholdings. All species, irrespective of conservation status, were more likely to be recorded in oil palm plantation estates and smallholdings that supported large areas of native forest. Our findings suggest that biodiversity conservation in oil palm landscapes will require a variety of conservation approaches. Minimizing poaching, reducing disturbance from human activity, and protecting existing forest patches appear particularly important. Strategies to promote the persistence of both high and low conservation value species should be adopted as part of a strengthened certification scheme for oil palm production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Southeast Asia’s tropical rainforests are rapidly disappearing because large areas are being destroyed by the expansion of commercial agriculture (Donald 2004; Koh and Wilcove 2007; Asner et al. 2009; Rudel et al. 2009). Millions of hectares of rainforest have been converted to industrial oil palm Elaeis guineensis in Southeast Asia (Lambert and Collar 2002; Linkie et al. 2003; Fitzherbert et al. 2008; Danielsen et al. 2009) leading to mammal extinctions (Brooks et al. 1999; Ceballos and Ehrlich 2002; Kinnaird et al. 2003; Steinmetz et al. 2006; Nantha and Tisdell 2009; Sodhi et al. 2009). To date, few studies have examined mammal species diversity in Southeast Asian oil palm plantations (Maddox et al. 2007; Bernard et al. 2009), with most researchers instead having focused on birds (Aratrakorn et al. 2006; Peh et al. 2006; Koh 2008; Edwards et al. 2010; Najera and Simonetti 2010; Azhar et al. 2011). Although mammal diversity will undoubtedly be highest in natural forest systems, carefully designed oil palm landscapes may provide some opportunities for the conservation of forest mammals outside protected areas. To the best of our knowledge, no previous studies have examined the effect of different management systems of existing oil palm cultivation on mammal diversity, abundance, and feeding guilds.
Plantation estates and smallholdings are the two most common management systems employed in oil palm cultivation in Southeast Asia. Plantation estates differ greatly from smallholdings in terms of infrastructure and other characteristics (Azhar et al. 2013a). Unlike smallholdings, plantation estates are equipped with palm oil mills, paved roads, housing settlements, and perimeter fences. Production of palm oil is typically more intensive in plantation estates than smallholdings. In Malaysia alone, in 2011, oil palm established in plantation estates and smallholdings covered 4,271,653 ha and 689,200 ha, respectively (Malaysia Palm Oil Board 2011). These cultivation systems are relevant to other geographical regions where oil palm has expanded rapidly (Butler and Laurance 2009; Lee et al. 2013).
To address the knowledge gaps on mammal biodiversity in established oil palm landscapes and quantify the impacts of different management regimes on mammals, we studied forest mammals (excluding bats and rodents) in oil palm landscapes in Peninsular Malaysia. Our overarching goal was to identify new ways to improve palm oil certification standards (e.g. Roundtable on Sustainable Palm Oil’s principles and criteria). Specifically, we asked: (1) Does the management system of oil palm cultivation influence the species richness of wild mammals? We predicted that smallholdings would provide habitat for native species that would not be able to survive in large-scale plantation estates. This is because of greater stand structural complexity and plant diversity in smallholdings (Azhar et al. 2011). (2) Which landscape-level factors influence the richness of wild mammals in oil palm landscapes? We predicted that the number of high conservation value mammals would increase with increasing area of natural forest patches within oil palm landscapes. This was because we expected that endangered and forest species would benefit from the retention of natural forest patches within oil palm cultivation areas (Koh 2008). (3) How do forest mammals respond to human activity? We predicted that all mammal species would be sensitive to human activity because such activity can disturb animal habitats and may introduce poachers to oil palm areas (Azhar et al. 2013b; Luskin et al. 2013). We defined human activity as any activity related to oil palm cultivation that consistently takes place inside plantation estates and smallholdings. These activities include fruit harvesting, replanting, weeding and insecticide spraying, fertilizer application, infrastructure maintenance, and security patrolling. (4) How do mammal species belonging to different feeding guilds respond to management systems, human activity, and landscape-level attributes? We predicted that omnivorous, carnivorous, and herbivorous mammals would exhibit different levels of sensitivity to predictor variables because members of different groups of mammals have different resource requirements.
Methods
Study site
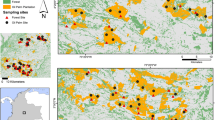
We investigated oil palm landscapes in the states of Selangor, Perak and Negeri Sembilan in Peninsular Malaysia (between 4°29′09″N, 100°42′47″E and 2°29′00″N, 101°56′35″E). Over a period of nine months (January–September in 2009), we used line transects to sample animals in 41 plantation estates and 14 smallholdings. All plantations had been established at least 5 years before surveys commenced. The oldest site was established in 1917, at Tennamaran Estate in Selangor. Because of logistical problems (e.g. accommodation, safety and permission to access a given site), we also conducted interviews in 27 plantation estates and nine smallholdings to obtain further data on the presence of mammals (Fig. 1).
Plantation estates and their management
We defined plantation estates as oil palm landscapes that covered more than 50 ha of oil palm monoculture and were operated by plantation companies (Azhar et al. 2011). The plantation estates were managed by six companies: Sime Darby Plantation, United Plantation (UP), the Federal Land and Development Agency (FELDA), Malaysian Airport’s Agriculture and Horticulture, the Federal Land Consolidation and Rehabilitation Authority (FELCRA), and Perbadanan Kemajuan Pertanian Selangor (PKPS). The total area of these estates was approximately 91,000 ha of planted area, with planted areas ranging in size from 155 to 16,000 ha. Plantation management (e.g. harvesting) was supported by both manual work and modern machinery. The perimeters of plantation estates were fenced and trenched, and guarded by security staff to deter theft of oil palm fruit and intrusion by wildlife poachers.
Smallholdings and their management
We defined smallholdings as semi-traditional cultivation areas covering less than 50 ha that were owned by individuals and not plantation companies (Roundtable of Sustainable Palm Oil 2014). In comparison with oil plantation estates, smallholdings were less dependent on modern infrastructure, and fruit was manually harvested rather than by machinery. The total area of smallholdings in the study region was approximately 15,000 ha. Smallholdings typically supported multiple-age stands of oil palm in which oil palms were intercropped with other commercial plants (e.g. bananas, cassavas, coffee, pineapples, or indigenous fruit trees) (Amoah et al. 1995). Old oil palm plants in smallholdings were not always clear-cut at the end of productive cycle (25–30 years), unlike in plantation estates. Similar to oil palm plantation estates, harvesting of oil palm fruit took place every 2 weeks in smallholdings.
Line transect sampling
We surveyed mammals by walking variable-length transects (Anderson et al. 1979; Gibbons and Gregory 2006) between 0700 and 1200 h and again between 1530 and 1900 h on days of clear and fine weather. Two observers walked a total of 470 transects once, with 418 transects and 52 transects located in plantation estates and smallholdings, respectively. We spaced transects >500 m apart to ensure spatial independence of animal observations. We avoided surveying along plantation roads due to potential interference by vehicles. We included only those mammals that were detected visually or identified by footprints. We recorded the abundance of each species where possible.
Field interviews
To supplement our field surveys, we gathered local ecological knowledge (LEK) (Anadon et al. 2009) via interviews with local experts—an approach that has been used successfully in other parts of the world (East et al. 2005; Anderson et al. 2007; Moore et al. 2010; Rasalato et al. 2010; Li et al. 2013). We interviewed 238 and 108 respondents in plantation estates and smallholdings, respectively. For various reasons, another 36 workers declined to participate when we first met them, and 16 more withdrew during or after the interviews.
A key prerequisite when using local expert knowledge is to ensure that local people are in fact qualified to provide the information they are asked to provide. We reasoned that meaningful local experts were those individuals who spent large amounts of time within oil palm plantations, and therefore could be meaningfully assumed to regularly encounter conspicuous mammal species (see also Anderson et al. 2007). Based on this rationale, we interviewed plantation workers, including managers, security officers, field supervisors, and labourers. Identified experts typically worked 6 days in a week in oil palm plantation estates or smallholdings. We interviewed independent smallholders and those who were hired as labourers in smallholdings.
We sought permission from each respondent for an interview which took on average 15 min to complete. All respondents were kept anonymous for confidentiality and in an attempt to ensure they would give honest answers. We also recorded respondents’ background information such as age, working experience, position, and personal interest in wildlife hunting (Table 1).
We did not assume that local experts held detailed technical knowledge. To obtain information from them about local mammal species, we provided local names of animals (in Malay and Tamil languages) and illustrations of each mammal species (Francis 2008) to the respondents, asking them whether they had seen these animals in oil palm landscapes. Small mammals and bats were excluded because they were not easily identified. We also asked respondents about the species occurrence, specific location, and perceived trends in abundance for each species (increasing or decreasing over the last 5 years). We allowed respondents to talk about other related issues such as poaching and animal behaviour. We interviewed only respondents who had worked for more than 6 months in a particular location.
Open and closed questions (regarding mammal identification) were asked by researchers during interviews with oil palm workers. Interviewees were allowed to reject any questions that they did not feel comfortable with or for which they felt uncertain of providing accurate answers. We calculated human activity values within each of the sites as the average number of hours/year that respondents worked within each site (Anderson et al. 2007).
We attempted to conduct interviews at all of the 55 sites where we surveyed mammals on transect lines. However, we ultimately obtained interview data from only 36 sites because: (1) we did not meet any respondents at some sites, (2) some workers were not eligible to be interviewed (e.g. they were recently appointed workers), or (3) some refused to answer survey questions. Notably, for those sites where we did obtain data, we are confident that there is no systematic observer difference between smallholdings and plantation estates. In both cases, the local experts we consulted spent large amounts of time within the relevant plantation, and thus could be assumed to be equally knowledgeable about the species living within it.
Each management regime is characterized by different features including the amount of manpower. Plantations hire many more workers than smallholdings for a range of other purposes, including as labourers, guards, truck drivers, and agronomy research assistants. Therefore, we do not think there is a clear bias with people working in smallholdings necessarily spending more time in such plantations.
Landscape metric measurements
Data on the area of oil palm cultivation were provided by plantation managers. We also measured two landscape-level attributes: (1) the cumulative area of surrounding natural forest patches (within 5 km of sites where interviews were conducted), and (2) the distance to the nearest continuous forest that covered >50,000 ha (Table 1). We calculated landscape metrics from a land use dataset (Malaysian Agriculture Department 2006) using ArcGIS 9.3 software (ESRI, Redlands, CA, USA).
Statistical analyses
We used data from local ecological knowledge in all statistical analyses. This was because line-transect surveys provided insufficient data. To examine the potential problem arising from multi-collinearity between predictor variables, we performed Spearman’s rank correlation tests between variables. To address the first three questions related to the influence of management system of oil palm cultivation, landscape-level factors, and human activity on species richness, we employed generalized linear mixed models (GLMMs) (Schall 1991; Paterson and Lello 2003). We classified species as being of high conservation value (HCV), or low conservation value (LCV). We defined HCV species as those classified as critically endangered, endangered, or vulnerable according to IUCN Red List (Francis 2008). We used a quasi-Poisson distribution with a log-link function in the modelling process. We incorporated work experience (log-transformed) as an offset in regression models. To take account of the effect of multiple respondents nested within a given site, we specified site as a random effect (Bolker et al. 2009). Ideally, we would have included time since plantation establishment in our analysis, but unfortunately, a lack of accurate historical records coupled with changes in ownership prevented us from obtaining adequate data on this.
GLMMs were used to quantify relationships between management systems and landscape-level attributes and overall species richness, number of HCV species, and number of LCV species. We included five fixed effects: management system (plantation estates or smallholdings), area of oil palm planting (ha), area of forest patch (ha), isolation from continuous forest (kilometer), and disturbance from human activity (hours per year). We log-transformed predictor variables prior to modeling. We selected the most parsimonious models based on Akaike information criterion (AIC) values, adjusted R2, and the Akaike weight of models (Burnham and Anderson 2002; Grafen and Hails 2002). We used AIC values in model selection as the value of (n/K), where n = sample size and K = number of parameters, is large (>40).
Lastly, for our fourth question on the sensitivity of mammals from different feeding guilds (Francis 2008) to different management regimes and landscape-level attributes, we used GLMMs and repeated similar model selection procedures as for the preceding questions to investigate guild responses (based on animal abundance) to management systems and landscape-level attributes. We used GenStat15 (VSN International, Hemel Hempstead, UK) for all data analyses.
Results
We found that the age of respondents and the amount of work experience were correlated (Spearman’s rank correlation coefficient = 0.70, p < 0.001). Respondents working in smallholdings had more work experience than respondents working in plantation estates (Table 1). The majority of respondents were labourers (>45 %). In term of nationality, approximately 64 % of respondents were Malaysian, 29 % Indonesian, 6 % Bangladeshi, and 1 % comprised other nationalities. Our results from correlation tests revealed weak correlations between variables (the highest correlation coefficient was −0.34, and that was between the amount of human activity and planting area).
Species richness of forest mammals
Using variable length transects, we detected only seven species of forest mammals. These were wild boar Sus scrofa, long-tailed macaque Macaca fascicularis, pig-tailed macaque Macaca nemestrina, dusky langur Trachypithecus obscures, silver leaf monkey Trachypithecus cristatus, Malay civet Viverra tangalunga, and leopard cat Prionailurus bengalensis.
By contrast, the use of local ecological knowledge revealed the occurrence of 32 mammal species in oil palm landscapes, including 13 species of high conservation value (Table 2). High conservation value species were sighted by respondents only infrequently and none were detected in line-transect surveys. Notably, large mammals such as Malayan tiger Panthera tigris, sun bear Helarctos malayanus, and Asiatic tapir Tapirus indicus were reported only in oil palm landscapes that were located within 5 km of natural forest.
Responses classified by conservation status
We found that all species, irrespective of conservation status, were more likely to be reported by respondents in sites with decreasing level of human activity (Wald statistic = 122.05; p < 0.001) and increasing natural forest patch area within five kilometers of the site where interviews were conducted (Wald statistic = 18.22; p < 0.001) (Table 3; Fig. 2). There was no significant difference in overall species richness between smallholdings (back-transformed mean = 2.24 species) and plantation estates (back-transformed mean = 2.39 species) (Wald statistic = 1.02; p = 0.316). Our results indicated that the most parsimonious model for explaining variation in overall mammal species richness was one in which human activity and cumulative area of natural forest patches were the explanatory variables. This model accounted for 57.9 % of the Akaike weights in the model set and explained 31.4 % of variation in mammal species richness across the study sites (Table 4).
Analyses of interview data revealed that the richness of HCV species increased at sites with decreasing level of human activity (Wald statistic = 14.22; p < 0.001) and increasing natural forest patch area (Wald statistic = 5.84; p = 0.022) (Table 3; Fig. 1). Our results also indicated that richness of HCV species in smallholdings (back-transformed mean = 0.50 species) did not differ significantly from that in plantation estates (back-transformed mean = 0.35 species) (Wald statistic = 0.06; p = 0.808). The most parsimonious model for HCV species included both human activity and cumulative area of natural forest patches within 5 km from sighting location. This model accounted for 44.6 % of the Akaike weights in the model set and explained 5.0 % of the variation in the occurrence of HCV species (Table 4).
Respondents also reported LCV species richness was higher at sites with decreasing level of human activity (Wald statistic = 148.16; p < 0.001) and increasing area size of forest patches (Wald statistic = 19.56; p < 0.001) (Table 3; Fig. 1). With respect to richness of LCV species, we found no significant differences between smallholdings (back-transformed mean = 1.86 species) and plantation estates (back-transformed mean = 1.90 species) (Wald statistic = 0.01; p = 0.944). The most parsimonious model for LCV species was one that included both human activity and cumulative area of natural forest patches. This model accounted for 58.5 % of the Akaike weights in the model set and explained 35.3 % of the variation in the occurrence of LCV species (Table 4). Neither planting area of oil palm nor isolation from major expanses of native forest explained any significant further variance.
Feeding guilds
Respondents reported the occurrence of 13, 11, and eight species of omnivorous, carnivorous, and herbivorous mammals, respectively. GLMM analyses revealed that the number of omnivores decreased at sites with decreasing level of human activity (Wald statistic = 91.54; p < 0.001) and increasing natural forest patch area (Wald statistic = 25.32; p < 0.001) (Table 3). No significant differences were found between smallholdings (back-transformed mean = 14.57 animals) and plantation estates (back-transformed mean = 12.37 animals) with respect to the number of omnivorous mammals reported by respondents. The most parsimonious model for the abundance of omnivores was one that included both increasing level of disturbance from human activity and cumulative area of natural forest patches. This model accounted for 44.8 % of the Akaike weights in the model set and explained 35.8 % of the variation in the occurrence of omnivorous animals (Table 4; Fig. 2).
The abundance of carnivores was influenced significantly by management system (Wald statistic = 6.01; p = 0.015) (Table 3). The number of carnivorous mammals reported by respondents from smallholdings and plantation estates was 2.00 animals and 1.02 animals (back-transformed means on the original scale), respectively. Further analysis showed that the most parsimonious model for carnivore abundance was one that included management system, increasing cumulative area of natural forest patches, and decreasing isolation from continuous forests. This model accounted for 37.2 % of the Akaike weights in the model set and explained 2.78 % of the variation in the occurrence of carnivorous species (Table 4).
Oil palm management system significantly influenced the abundance of herbivores in oil palm landscapes (Wald statistic = 7.63; p = 0.006) (Table 3). The abundance of herbivorous mammals reported by respondents from smallholdings and plantation estates was 3.18 animals and 0.47 animals (back-transformed means on the original scale), respectively. The most parsimonious model for the abundance of herbivores included management system, increasing area size of oil palm cultivation, decreasing cumulative area of natural forest patches, and increasing isolation from continuous forests. This model accounted for 42.7 % of the Akaike weights in the model set and explained 9.25 % of the variation in the occurrence of herbivorous species (Table 4).
Discussion
Our study produced three key findings that can inform palm oil management: (1) The number of forest mammals, HCV species, and LCV species in oil palm landscapes was significantly lower in areas with more human activity; (2) the number of all species, regardless of conservation status, was influenced by the amount of natural forest cover within the boundaries of oil palm plantation estates and smallholdings and; (3) smallholdings supported a greater abundance of carnivores and herbivores than plantation estates.
Relationships between management system and mammal species richness
Our findings demonstrate that, in comparison with plantation estates, smallholdings sustained higher numbers of carnivorous and herbivorous mammals. These findings imply that smallholdings may offer better refuge habitats to these groups than plantation estates (Azhar et al. 2013a). This may be because oil palm smallholdings were planted with numerous tree species or other crops besides oil palm. Structural complexity of commercial crop agriculture may increase mammal diversity (Ramírez and Simonetti 2011). Several mammal species may temporarily venture into oil palm habitats in search of food. For example, the Malayan tiger P. tigris may follow prey species like the wild boar from natural forest areas into oil palm landscapes (Ickes 2001; Kawanishi and Sunquist 2004). Another possible explanation for the comparatively higher levels of richness of carnivores and herbivores in smallholdings is that such areas are usually characterised by stands of oil palm of different ages, creating both spatial and temporal habitat heterogeneity (Luskin and Potts 2011).
Landscape context effects
Overall mammal species richness, high conservation value species richness, and low conservation value species richness, increased significantly with the cumulative area of natural forest patches within 5 km of each site. This may be because many mammals (especially forest-associated species) mainly inhabit forest patches, but can forage in neighboring oil palm habitats. Other studies from around the world also have shown that nearby forested areas can influence species richness and abundance in adjacent agricultural and/or plantation areas (Lindenmayer et al. 2002; Anand et al. 2008; Lees and Peres 2008; Reino et al. 2009; Tubelis et al. 2007; Yamaura et al. 2009).
Some high conservation value species occasionally used oil palm landscapes located close to large forest patches (e.g. Malayan tiger, sun bear H. malayanus, melanistic leopard Panthera pardus, tapir T. indicus, and pig-tailed macaque Macaca nemestrina). Hunting may drive some mammal species out of their natural forest habitats and force them to temporarily take refuge in oil palm areas, particularly in plantation estates, which are normally off limits to poachers. Some of our interviewees reported that some forest mammals (e.g. wild boar, Malayan porcupine Hystrix brachyura, and Sunda pangolin Manis javanica) were poached by local hunters in oil palm landscapes.
The responses of omnivorous, carnivorous and herbivorous mammals
We found more omnivorous mammals in oil palm landscapes (irrespective of management system) than carnivorous and herbivorous mammals. The highest number of species and highest levels of abundance in oil palm landscapes were reported for omnivorous mammals. This may have been because omnivorous mammals exploit a wide range of food resources in oil palm landscapes. For example, the long-tailed macaque M. fascicularis was the only primate species recorded in oil palm landscapes irrespective of management system. A possible reason for this is that the species can feed on unripe oil palm fruits and small animals. The presence of large forest patches also may provide both food resources and sheltering habitat for omnivorous mammals. Our findings suggest that carnivorous mammals are unlikely to occupy large plantation estates, possibly because their prey is less abundant in plantation estates than smallholdings. Another important finding was that the abundance of herbivores increased with the loss of natural forest patches. This may be attributed to greater food production at edge habitats in small patches than in large ones (Bender et al. 1998).
Conservation implications
Based on our analyses, we make three recommendations to improve the conservation of forest mammals in existing oil palm landscapes. We argue that these measures should be included as part of strengthened guidelines in certification schemes for sustainable palm oil production. These strengthened measures are: (1) Poaching should be prohibited in smallholdings because without deterrent measures (e.g. guards and fences), poachers can easily access smallholdings. Compared to plantation estates, smallholdings supported more species, and therefore hold some promise for supporting some species of wild mammals. Because many smallholders are not very wealthy, it is necessary to carefully consider whether government assistance may be required to control poaching. (2) Disturbance from human activity inside oil palm landscapes should be minimized. Human activity can lead to poaching, being committed regularly or opportunistically by some oil palm workers (Azhar et al. 2013b). (3) Existing natural forest patches within the boundaries of oil palm landscapes should be retained and protected. Studies in coffee, cacao, and other kinds of plantations have shown that wildlife habitats, corridors, stepping stones, and can act as a “soft” matrix between natural forest patches (Munoz et al. 2006; Williams-Guillen et al. 2006). However, forest patches may not be a long term solution for perpetuating wildlife because they were highly accessible to hunters and exposed to edge effects and fires (Canale et al. 2012).
Mammal species richness in oil palm landscapes is much lower than in undisturbed forest (Maddox et al. 2007). Natural forest should not be cleared to establish new areas of oil palm because of the substantial negative impacts on biodiversity (Brooks et al. 1999; Sodhi et al. 2004; Wilcove and Koh 2010). However, we have shown that a number of forest species are able to use oil palm environments (though not necessarily as their primary habitat). Similarly to Bali et al. (2007), we suggest that carefully designed oil palm agro-ecosystems could provide a useful buffer around protected areas and may help to facilitate connectivity between such areas. However, the outstanding issues of forest conversion and poaching must be addressed if oil palm landscapes are to form high-quality matrix around reserves.
References
Amoah FM, Nuertey BN, Baidoo-Addo K, Oppong FK, Osei-Bonsu K, Asamoah TEO (1995) Underplanting oil palm with cocoa in Ghana. Agrofor Syst 30:289–299
Anadon JD, Gimenez A, Ballestar R, Perez I (2009) Evaluation of local ecological knowledge as a method for collecting extensive data on animal abundance. Conserv Biol 23:617–625
Anand MO, Krishnaswamy J, Das A (2008) Proximity to forests drives bird conservation value of coffee plantations: implications for certification. Ecol Appl 18:1754–1763
Anderson DR, Laake JL, Crain BR, Burnham KP (1979) Guidelines for line-transect sampling of biological populations. J Wildl Manag 43:70–78
Anderson J, Rowcliffe JM, Cowlishaw G (2007) Does the matrix matter? A forest primate in a complex agricultural landscape. Biol Conserv 135:212–222
Aratrakorn S, Thunhikorn S, Donald PF (2006) Changes in bird communities following conversion of lowland forest to oil palm and rubber plantations in southern Thailand. Bird Conserv Int 16:71–82
Asner GP, Rudel TK, Aide TM, Defries R, Emerson R (2009) A contemporary assessment of change in humid tropical forests. Conserv Biol 23:1386–1395
Azhar B, Lindenmayer DB, Wood J, Fischer J, Manning A, McElhinny C, Zakaria M (2011) The conservation value of oil palm plantation estates, smallholdings and logged peat swamp forest for birds. For Ecol Manag 62:2306–2315
Azhar B, Lindenmayer DB, Wood J, Fischer J, Manning A, McElhinny C, Zakaria M (2013a) The influence of agricultural system, stand structural complexity and landscape context on foraging birds in oil palm landscapes. Ibis 155:297–312
Azhar B, Lindenmayer DB, Wood J, Fischer J, Manning A, McElhinny C, Zakaria M (2013b) Contribution of illegal hunting, culling of pest species, road accidents and feral dogs to biodiversity loss in established oil-palm landscapes. Wildl Res 40:1–9
Bali A, Kumar A, Krishnaswamy J (2007) The mammalian communities in coffee plantations around a protected area in the Western Ghats, India. Biol Conserv 139:93–102
Bender DJ, Contreras TA, Fahrig L (1998) Habitat loss and population decline: a meta-analysis of the patch size effect. Ecology 79:517–533
Bernard H, Fjeldsa J, Mohamed M (2009) A case study on the effects of disturbance and conversion of tropical lowland rain forest on the non-volant small mammals in north Borneo: management implications. Mamm Study 34:85–96
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Stevens MHH, White JSS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24:127–135
Brooks TM, Pimm SL, Kapos V, Ravilious C (1999) Threat from deforestation to montane and lowland birds and mammals in insular South-east Asia. J Anim Ecol 68:1061–1078
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, USA
Butler RA, Laurance WF (2009) Is oil palm the next emerging threat to the Amazon? Trop Conserv Sci 2:1–10
Canale GR, Peres CA, Guidorizzi CE, Gatto CAF, Kierulff MCM (2012) Pervasive defaunation of forest remnants in a tropical biodiversity hotspot. PLoS One 7(8):e41671. doi:10.1371/journal.pone.0041671
Ceballos G, Ehrlich PR (2002) Mammal population losses and the extinction crisis. Science 296:904–907
Danielsen F, Beukema H, Burgess ND, Parish F, Bruhl CA, Donald PF, Murdiyarso D, Phalan B, Reijnders L, Struebig M, Fitzherbert EB (2009) Biofuel plantations on forested lands: double jeopardy for biodiversity and climate. Conserv Biol 23:348–358
Donald PF (2004) Biodiversity impacts of some agricultural commodity production systems. Conserv Biol 18:17–37
East T, Kumpel NF, Milner-Gulland EJ, Rowcliffe JM (2005) Determinants of urban bushmeat consumption in Rio Muni, Equatorial Guinea. Biol Conserv 126:206–215
Edwards DP, Hodgson JA, Hamer KC, Mitchell SL, Ahmad AH, Cornell SJ, Wilcove DS (2010) Wildlife-friendly oil palm plantations fail to protect biodiversity effectively. Conserv Lett 3:236–242
Fitzherbert EB, Struebig MJ, Morel A, Danielsen F, Brulh CA, Donald PF, Phalan B (2008) How will oil palm expansion affect biodiversity? Trends Ecol Evol 23:538–545
Francis CM (2008) A field guide to the mammals of south-east asia. New Holland, London
Gibbons DW, Gregory RD (2006) Birds. In: Sutherland WJ (ed) Ecological census techniques, 2nd edn. Cambridge University Press, Cambridge, pp 308–344
Grafen A, Hails R (2002) Modern statistics for the life sciences. Oxford University Press, Oxford
Ickes K (2001) Hyper-abundance of native wild pigs (Sus scrofa) in a lowland dipterocarp rain forest of Peninsular Malaysia. Biotropica 33:682–690
Kawanishi K, Sunquist ME (2004) Conservation status of tigers in a primary rainforest of Peninsular Malaysia. Biol Conserv 120:329–344
Kinnaird MF, Sanderson EW, O’ Brien TG, Wibisono HT, Woolmer G (2003) Deforestation trends in a tropical landscape and implications for endangered large mammals. Conserv Biol 17:245–257
Koh LP (2008) Can oil palm plantations be made more hospitable for forest butterflies and birds? J Appl Ecol 45:1002–1009
Koh LP, Wilcove DS (2007) Cashing in palm oil for conservation. Nature 448:993–994
Lambert FR, Collar NJ (2002) The future for Sundaic lowland forest birds: long-term effects of commercial logging and fragmentation. Forktail 18:127–146
Lee JSH, Abood S, Ghazoul J, Barus B, Obidzinski K, Koh LP (2013) Environmental impacts of large-scale oil palm enterprises exceed that of smallholdings in Indonesia. Conserv Lett. doi:10.1111/conl.12039
Lees AC, Peres CA (2008) Conservation value of remnant riparian forest corridors of varying quality for Amazonian birds and mammals. Conserv Biol 22:439–449
Li J, Yin H, Wang D, Jiagong Z, Lu Z (2013) Human-snow leopard conflicts in the Sanjiangyuan region of the Tibetan plateau. Biol Conserv 166:118–123
Lindenmayer DB, Cunningham RB, Donnelly CF, Nix H, Lindenmayer BD (2002) Effects of forest fragmentation on bird assemblages in a novel landscape context. Ecol Monogr 72:1–18
Linkie M, Martyr DJ, Holden J, Yanuar A, Hartana AT, Sugardjito J, Leader-Williams N (2003) Habitat destruction and poaching threaten the Sumatran tiger in Kerinci Seblat National Park, Sumatra. Oryx 37:41–48
Luskin MS, Potts MD (2011) Microclimate and habitat heterogeneity through the oil palm lifecycle. Basic Appl Ecol 12:540–551
Luskin, Christina ED, Kelley LC, Potts MD (2013) Modern hunting practices and wild meat trade in the oil palm plantation-dominated landscapes of Sumatra, Indonesia. Hum Ecol. doi:10.1007/s10745-013-9606-8
Maddox T, Priatna D, Gemita E, Salampessy A (2007) The conservation of tigers and other wildlife in oil palm plantations, Jambi Province, Sumatra, Indonesia. ZSL conservation report No. 7. The Zoological Society of London, London
Malaysia Palm Oil Board (2011) Economic and statistics. http://www.mpob.gov.my. Accessed 5 Dec 2012
Moore JE, Cox TM, Lewison RL, Read AJ, Bjorkland R, McDonald SL, Crowder LB, Aruna E, Ayissi I, Espeut P, Joynson-Hicks C, Pilcher N, Poonian CNS, Solarin B, Kiszka J (2010) An interview-based approach to assess marine mammal and sea turtle captures in artisanal fisheries. Biol Conserv 143:3795–3805
Munoz D, Estrada A, Naranjo E, Ochoa S (2006) Foraging ecology of howler monkeys in a cacao (Theobroma cacao) plantation in Comalcalco, Mexico. Am J Primatol 68:127–142
Najera A, Simonetti JA (2010) Enhancing avifauna in commercial plantations. Conserv Biol 24:319–324
Nantha HS, Tisdell C (2009) The orangutan-oil palm conflict, economic constraints and opportunities for conservation. Biodivers Conserv 18:487–502
Paterson S, Lello J (2003) Mixed models: getting the best use of parasitological data. Trends Parasitol 19:370–375
Peh KSH, Sodhi NS, de Jong J, Sekercioglu CH, Yap CAM, Lim SLH (2006) Conservation value of degraded habitats for forest birds in southern Peninsular Malaysia. Divers Distrib 12:572–581
Ramírez PA, Simonetti JA (2011) Conservation opportunities in commercial plantations: the case of mammals. J Nat Conserv 19:351–355
Rasalato E, Maginnity V, Brunnschweiler JM (2010) Using local ecological knowledge to identify shark river habitats in Fiji (South Pacific). Environ Conserv 37:90–97
Reino L, Beja P, Osborne PE, Morgado R, Fabiao A, Rotenberry JT (2009) Distance to edges, edge contrast and landscape fragmentation: interactions affecting farmland birds around forest plantations. Biol Conserv 142:824–838
Roundtable of Sustainable Palm Oil (2014) Definition of smallholders. http://www.rspo.org/en/definition. Accessed 10 Jan 2014
Rudel TK, Defries R, Asner GP, Laurance WF (2009) Changing drivers of deforestation and new opportunities for conservation. Conserv Biol 23:1396–1405
Schall R (1991) Estimation in generalized linear-models with random effects. Biometrika 78:719–727
Sodhi NS, Koh LP, Brook BW, Ng PKL (2004) Southeast Asian biodiversity: an impending disaster. Trends Ecol Evol 19:654–660
Sodhi NS, Lee TM, Koh LP, Brook BW (2009) A meta-analysis of the impact of anthropogenic forest disturbance on Southeast Asia’s biotas. Biotropica 41:103–109
Steinmetz R, Chutipong W, Seuaturien N (2006) Collaborating to conserve large mammals in Southeast Asia. Conserv Biol 20:1391–1401
Tubelis DP, Lindenmayer DB, Cowling A (2007) Bird populations in native forest patches in south-eastern Australia: the roles of patch width, matrix type (age) and matrix use. Landsc Ecol 22:1045–1058
Wilcove DS, Koh LP (2010) Addressing the threats to biodiversity from oil-palm agriculture. Biodivers Conserv 19:999–1007
Williams-Guillen K, McCann C, Sanchez JCM, Koontz F (2006) Resource availability and habitat use by mantled howling monkeys in a Nicaraguan coffee plantation: can agroforests serve as core habitat for a forest mammal? Anim Conserv 9:331–338
Yamaura Y, Ikeno S, Sano M, Okabe K, Ozaki K (2009) Bird responses to broad-leaved forest patch area in a plantation landscape across seasons. Biol Conserv 142:2155–2165
Acknowledgments
This study was supported by the Ministry of Higher Education Malaysia through the Fundamental Research Grant Scheme, the Wildlife Conservation Society’s Research Fellowship Grant 2009, The Australian National University’s Field Trip Grant 2009, and funds from Prof. D. Lindenmayer. We are very grateful to a number of people for assistance with various aspects of this study, namely, Adrian Manning, S. Holzknecht, J. Stein, K. Ikin, E. Yusof, A. Jambari, and N. L. Ibrahim. We also thank all plantation managers and staff from Sime Darby Plantation, United Plantation, FELDA, FELCRA, and Perbadanan Kemajuan Pertanian Selangor.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David L. Hawksworth.
Rights and permissions
About this article
Cite this article
Azhar, B., Lindenmayer, D.B., Wood, J. et al. Ecological impacts of oil palm agriculture on forest mammals in plantation estates and smallholdings. Biodivers Conserv 23, 1175–1191 (2014). https://doi.org/10.1007/s10531-014-0656-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-014-0656-z