Abstract
The impacts of invasive plants on arthropod communities are often reported to be negative and have predominantly been explored aboveground, but there is a paucity of information regarding what happens belowground. To address this gap, we compared soil properties and soil fauna communities associated with two native plant species (Leptospermum scoparium—mānuka and Chionochloa rubra—red tussock) and two invasive species (non-N-fixing Calluna vulgaris—European heather and N-fixing Cytisus scoparius—Scotch broom) in the Central Plateau of New Zealand. We expected that (1) at individual plant level soil properties would be different under invasive and native plant species, with higher soil nutrient concentrations under invasive species, especially N-fixing broom; (2) total abundance of soil fauna would be higher under invasive plant species, as generally positive impact of invasive plants on soil invertebrates is indicated in the literature; (3) invasive plants, and especially N-fixing broom, will be associated with greater abundances of soil decomposer groups. We found that soil properties and soil fauna assemblages did not cluster by plant invasive status as initially predicted. At individual plant level, there was similarity in soil conditions between mānuka and broom, and between red tussock and heather. The invasive N-fixer (broom) had positive effects on soil N availability, with higher N pool and lower C/N ratio in soil under this species. There were no consistent differences in total soil fauna abundance between invasive and native plants. Broom and mānuka were associated with higher abundances of Collembola, Oligochaeta and Diplopoda; heather and red tussock had higher abundances of Hymenoptera and Hemiptera. Significantly more Oligochaeta and Collembola under broom matched the prediction of invasive plants (and especially N-fixing invasives) being associated with greater abundances of decomposers. However, another important decomposer group—oribatid mites—did not show the same tendency. These results evidence that simplified generalizations regarding the impacts of invasive plants are unlikely to be justified, since the ecological effects of plant invasions are complex and do not always follow the same pattern. Therefore, we need to take into consideration the ecological context and the traits of individual plant species and target organisms in an unbiased manner to fully understand the impacts of plant invasions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Invasive plants affect their invaded ecosystems in multiple ways, such as by altering plant community composition, aboveground and below-ground arthropod composition, above and below-ground chemical environments, and soil properties (Levine et al. 2003; Litt et al. 2014, 2024; Clavijo McCormick et al. 2023). Invasive plants can displace native plants through competition and by means of various abiotic and biotic mechanisms: these include chemical alterations and physical disturbances to the soil, changes in soil microorganism composition and relationships, allelopathy, disruptions to pollinator interactions, and altered plant–herbivore dynamics (Levine et al. 2003; Vilà et al. 2011).
Over time, invasive species may become dominant in the invaded ecosystem. Therefore, it is not surprising that invaded systems are often associated with changes in aboveground arthropod abundance and richness when compared to similar uninvaded systems (Blayney 2012; Litt et al. 2014). A decline in aboveground arthropods is often attributed to the decrease in plant biodiversity (Ebeling et al. 2018) and to the replacement of native plant resources by novel plant resources. However, invasive plants can also affect fauna communities by altering plant-arthropod linkages associated with pollination (Muñoz and Cavieres 2008) or by providing new food sources or habitat features that can be utilized by native groups (Galappaththi et al. 2023).
The effect of invasive plants on above-ground arthropods can vary in size and direction depending on the specific invader and the recipient habitat, and which arthropod taxa are considered (Keesing 1995; Litt et al. 2014; Effah et al. 2020a). A recent review (Litt et al. 2024) has concluded that although published papers often report invasive plants having a negative influence on arthropod populations, arthropod responses to alien plants are far from uniform. While some functional groups with close association with native plants, such as specialist herbivores, pollinators, and their specialist predators are often negatively affected by alien plant invasion; generalist arthropods may benefit from enhanced resource abundance (Galappaththi et al. 2023; Litt et al. 2024). Our growing understanding of the complexity of ecological interactions mediated by invasive plants, suggests that generalising the effects of invasive plants as solely negative may be an oversimplification, and urges the need to develop other theoretical frameworks that allow for better predictive tools.
As well as above-ground, plants can alter below-ground fauna assemblages since plants provide the main input into soil food webs, either through input and subsequent decomposition of leaf litter or through root material and the exudation of organic compounds by the roots that alter soil properties (Wolfe and Klironomos 2005; Weidenhamer and Callaway 2010; Potapov et al. 2019). Recent evidence suggests that root material and exudates, rather than leaf litter, supply the greatest proportion of this input (Pollierer et al. 2007; Bluhm et al. 2017; Fu et al. 2017; Sprunger et al. 2019).
Invasive plants are often associated with greater soil pools of organic matter and increased rates of litter decomposition and nutrient cycling than native plant species (Liao et al. 2008). This trend is often ascribed to the tendency in invasive plant taxa to present higher values for performance-based plant traits, such as leaf nutrient concentrations and net primary productivity, which are linked to nutrient cycling rates (Stefanowicz et al. 2018). For example, nitrogen-fixing plant species are disproportionately represented among invasive plants and are associated with higher rates of nitrogen (N) cycling than native or invasive plants lacking this trait (Ehrenfeld 2003; Vilà et al. 2011; Souza-Alonso et al. 2015).
The effect of invasive plants on soil properties is not consistent and the magnitude as well as the direction of the effect can depend on numerous invader-related and ecosystem-related variables. For example, Dassonville et al. (2008) found that invasive plants had positive effects on soil nutrient pools when the initial soil was nutrient-poor and negative effects when the initial soil was nutrient-rich. A meta-analysis considering impacts of 113 invasive plant species across the world on N cycle (Castro-Díez et al. 2014) found that, overall, plant invasions increase soil N pools, even when excluding N-fixing plants. However, the impacts were heterogeneous and reflected differences in climate and other properties of the invaded sites, and phylogenetic and functional characteristics of the invading and native plants. The impact on soil N pools was greater in warm climates and when the invasive plants and the natives differed in N-fixation ability (Castro-Díez et al. 2014).
The literature exploring the impacts of invasive plants on below-ground arthropod communities is still scarce but suggests variable impacts depending on the invasive plant identity and the recipient system (Belnap and Phillips 2001; Kappes et al. 2007; Tanner et al. 2013). Higher primary productivity and leaf nutrient concentrations, often associated with invasive plants, are expected to stimulate the below-ground decomposer communities which are able to utilize a new resource. A meta-analysis (Meisner et al. 2014) suggested that plant invasion generally enhanced C cycling and increased abundance of soil meso-invertebrates (unspecified taxa) and nematodes, while having variable effects on other soil biota and processes. Detritivores generally increase in response to higher litter input from alien plants but in some cases can also show decline due to unfavourable changes in litter quality and phenology (Litt et al. 2024). Wolkovich et al. (2009) found that in a semi-arid system, extensively invaded by alien grasses, soil N and litter input have increased in areas of high grass invasion but most arthropod taxa in detritus-based food web have declined. The lack of bottom-up effects was linked to changes in habitat structure and litter quality, altered microclimate, and disruption of trophic relationships (Wolkovich et al. 2009). Similarly, a review of effects of alien plant invasions in riparian zones (Castro-Díez and Alonso 2017) found either no effect or negative effect on abundance and diversity of soil micro- and macro-arthropods.
To address the paucity of information on the impacts of invasive plants on below-ground arthropod communities, we used a model system in the North Island Central Plateau (NICP) of New Zealand—home to Tongariro National Park, a UNESCO dual cultural and natural World Heritage site—consisting of two invasive plants: heather Calluna vulgaris (L.) Hull and Scotch broom Cytisus scoparius (L.) Link (henceforth broom); and two native plants: mānuka Leptospermum scoparium (J.R. Forst et G. Forst.) and red tussock Chionochloa rubra (Zotov). The aim of the study was to investigate soil physicochemical properties and soil fauna assemblages associated with individual plants growing under natural conditions in the NICP. Based on existing literature data, we predicted that: (1) soil properties would cluster for invasive and native plants, with separation between these two groups explained by higher soil nutrient concentrations under invasive plants, especially for broom, as it is a N-fixing species; (2) total abundance of soil fauna would be higher under invasive plant species, as generally positive impact of invasive plants on soil invertebrates is indicated in the literature; (3) invasive plants, and especially N-fixing broom, will be associated with greater abundances of soil decomposer groups due to more labile leaf litter (lower C/N ratio).
Previous work exploring the impact of plant invasions on arthropod communities in the Central Plateau has mainly focused on above-ground effects (Keesing 1995; Effah et al. 2020a), but below-ground impacts remain poorly understood. This work adds to previous efforts to understand and mitigate the impacts of plant invasion on the NICP (e.g., Effah et al. 2020a, b, c, 2022a) and will help to unveil the association between soil fauna and invasive plants in this valuable ecosystem.
Methods
Site description
The North Island Central Plateau (NICP) is a high-altitude volcanic region of the North Island, New Zealand. Exotic plant invasion is one of the main risks threatening the natural and cultural assets of this region (Department of Conservation 2006). The native vegetation in the area is dominated by red tussock C. rubra grassland and by tussock shrubland with Dracophyllum spp. and mānuka L. scoparium, there are also local areas of disturbed ground and scoria slopes (Chapman and Bannister 1990; Effah et al. 2020a, b, c). Two important invasive species of the area are European heather C. vulgaris, family Ericaceae, henceforth heather, and broom C. scoparius, family Fabaceae, both species originally from Europe. From 1912 to 1923, heather was deliberately planted and its spread otherwise facilitated to provide suitable habitat for exotic game fowl (Bagnall 1982). After planting ceased, heather continued to spread and is now the most widespread weed species of the area, covering more than 50,000 ha of the TNP and greater NICP (Effah et al. 2020b) Broom was introduced to the area in the 1960s, and while it is not as widespread as heather, its range has expanded in recent years. This species is able to fix nitrogen, and thus has the potential to alter soil nutrients and other soil properties (Landcare Research 2022).
Red tussock is a native grass species common in the central North Island. It was selected for this study partly as a species of distinctive native character to the area, and partly because it is sensitive to displacement by invasive plants. Red tussock grassland communities are considered highly at risk due to heather invasion, which resulted in a large decline in red tussock between 1960 and 1984 (Chapman and Bannister 1990). Mānuka is a widespread native New Zealand shrub; it is typically a mid-successional plant species but it persists in areas that do not support succession to climax forest, such as the NICP (Stephens et al. 2005; Hayes et al. 2013; Effah et al. 2020c). This species is of economic interest due to the medicinal properties of its honey and it is a taonga (treasured) species of cultural value to Māori (Effah et al. 2022b; Morgan et al. 2019).
The field sites were located inside the Waiouru military training area: 39° 18′ 44.064″ S, 175° 44′ 21.551″ E (site 1); 39° 18′ 34.091″ S, 175° 44′ 20.184″ E (site 2); 39° 18′ 8.711″ S, 175° 44′ 41.927″ E (site 3). The region has a Kӧppen climate classification of “cfb” (temperate oceanic climate) (Kottek et al. 2006). The mean daily temperature ranges from 5 to 15 °C (Chappell 2015), with an annual rainfall in excess of 1500 mm (DOC n.d). The region has low fertility tephric recent and orthic allophanic soils mostly formed from volcanic ash (Hewitt 2010)—Andosols, in the World Soil Classification system. The sites were chosen based on the presence of healthy populations of heather, broom, mānuka and red tussock, as well as being reasonably distant from one another while located on the same soil type. At each of the three sites, five healthy individual plants of broom, heather, mānuka and red tussock were selected, making up 60 plants over the entire study. Individual plants were selected to have similar phenology within each species. Plants were only selected if they were not in proximity (less than one meter) from any individuals of the other study species. Sampling was conducted in late February/early March 2021 (Southern Hemisphere summer).
Soil sampling
We sampled the soil adjacent to the bases of individual plants; the plants themselves were not lifted. At each individual sampling point, soil temperature and soil volumetric water content were recorded. A digital soil thermometer was used to collect three measurements around the target plant which were then averaged. A time domain reflectometry soil moisture meter (TDR Field Scout 100, Spectrum Technologies, Inc.) was used to collect three measurements of soil volumetric water content (VWC) around the target plant that were then averaged. Soil samples (5 cm length × 5 cm width × 8 cm depth) were taken close to the base of every plant in this study and kept on ice in the field. These soil cores were used to extract mesofauna as detailed below. Following mesofauna extraction air-dry soil cores were sent to Hill Laboratories in Hamilton, New Zealand for analysis, which included pH, Olsen phosphorous (Olsen P), anaerobically mineralisable nitrogen (AMN, measure of N mineralised in anaerobic incubation at 40 °C for 7 days), total nitrogen (TN, measured using the Dumas combustion method), organic matter (OM, derived from total C measured by the Dumas combustion method), carbon/nitrogen ratio (C/N), potassium (K+), calcium (Ca2+), magnesium (Mg2+), sodium (Na+), cation exchange capacity (CEC), total base saturation (TBS), and volume weight (VW).
Soil fauna sampling
For this study, we sampled soil macrofauna (defined as any organism larger than 2 mm on its longest axis), plus the important mesofauna decomposers—oribatid mites (Acari: Oribatida) and springtails (Collembola) (Potapov et al. 2022). One macrofauna soil sample (30 cm length, 20 cm width, 15 cm depth) was collected with a shovel directly close to the base of each target plant; the plant itself was not removed. A total of 60 macrofauna samples were collected for this study. Samples were bagged and put on ice in the field and stored at 4 °C in the lab until sorting. Macrofauna were hand-sorted from the samples and stored in 70% ethanol until being identified to the order level.
Mesofauna were extracted from the soil analysis cores using a modified Berlese-Tullgren apparatus for 10 days using a mesh size of ~ 2 mm. Mesofauna were extracted into 70% ethanol. Following mesofauna extraction, soil cores were resealed in their original zip-lock bags and sent to Hill Laboratories for soil analysis. Samples were sorted under a dissection microscope and the dominant decomposers—oribatid mites and springtails—were counted.
Data analysis
One soil sample was a suspected urine patch, indicated by abnormally high nitrogen content; however, excluding this sample from analysis did not change the results, so the sample was left included. Soil property data were standardised to unit variance (subtracted the mean and divided by standard deviation) prior to principal component analysis (PCA) using FactoMineR and factoextra packages in R (version 4.1.3). The PCA biplot and corresponding table of variable contributions were then used to visualise differences in soil properties under different plant species (heather, broom, mānuka and red tussock). Permutational multivariate analysis (PERMANOVA) in PRIMER 7 (Clarke et al. 2014) in a nested model with site as a random factor was used to test for differences in the composition of soil properties under different plant species. When the main effect was significant, multiple comparisons were performed between individual plant species. If clusters were identified from the PCA ordination, PERMANOVA was used to compare the soil properties between the clusters using a contrast statement.
Generalized linear models (GLMs) in R were used to test differences in specific soil properties between vegetation types. For each soil variable, a GLM with normal, gamma or inverse-gaussian distribution family was generated, and the model with the best fit to the data (lowest AIC value) was used. The LRTest function from the lmtest package was used to assess the significance of the predictor variable for each of the chosen models. When the model was significant, Tukey’s honestly significant difference test (Tukey HSD) was performed for post-hoc pairwise analysis.
Total abundance and taxon richness for each sample were calculated. Simpson’s diversity and Shannon’s diversity indices (Morris et al. 2014) were calculated using the vegan package in R. GLMs were used to compare soil fauna abundance and diversity under different plant species (heather, broom, mānuka and red tussock). For total abundance and taxon richness, GLMs with Poisson and negative-binomial distributions were generated, and the best fitting models were selected using AIC. For Simpson’s index and Shannon’s index, GLMs with normal, gamma or inverse-Gaussian distribution family were generated and the model with the best fit was selected using AIC. The LRTest was used to assess the significance of the predictor variable for each of the chosen models. When the model was significant, a Tukey’s HSD test was performed for post-hoc means analysis.
Linear discriminant analysis (LDA) was performed to visualise differences in soil fauna communities under the four plant species. For this analysis, counts of Diplura, Blattodea and Achatinoidea were combined into “Other” to reduce the number of zeros in the taxon matrix, taxa that were not present in more than two samples were omitted, and the dataset was square root-transformed. PERMANOVA with Bray–Curtis distance using nested model with site as a random factor was performed on this dataset to test if the composition of the soil fauna community varied significantly among plant species. When significant results were detected, multiple comparisons were performed using PERMANOVA with Bray–Curtis distance. Individual groups were then compared between plant species using GLMs (selected by AIC from Poisson and negative binomial distributions) and a likelihood ratio test. When models were significant, TukeyHSD were performed for post-hoc means comparison.
Results
Soil properties
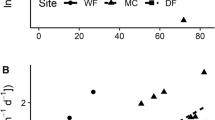
The PCA ordination showed overlap between soil properties under mānuka and broom as well as between red tussock and heather, forming two distinctive clusters, mānuka/broom and red tussock/heather (Fig. 1). The first and second principal components (PC1 and PC2) collectively explained 58.3% of the variation in soil properties between vegetation types. PCA ordination (Fig. 1) suggests that red tussock and heather were associated with acidic and nutrient-poor but wetter soils, whereas broom and mānuka were associated with soils with higher pH and higher soil nutrient availability. The soil properties contributing most to the dissimilarity between plant species were nitrogen (AMN, TN), Ca2+, Mg2+, TBS, pH, and carbon/nitrogen ratio (C/N). Despite being identified as contributors to dissimilarity, models for soil cation exchange capacity and organic matter between individual plant species were not significant.
PCA biplot showing the soil properties under Scotch broom (Cytisus scoparius), European heather (Calluna vulgaris), mānuka (Leptospermum scoparium) and red tussock (Chionochloa rubra), NZ Central Plateau, 2021. Large circles indicate the centroids of the respective groups. Abbreviations: volume weight (VW), volumetric water content (VWC), Olsen phosphorous (Olsen P), anaerobically mineralizable nitrogen (AMN), total nitrogen (TN), organic matter (OM), carbon/nitrogen ratio (C.N), cation exchange capacity (CEC), total base saturation (TBS)
The overall composition of soil properties was significantly different among the plant species (PERMANOVA; Pseudo-F3,48 = 3.32, P = 0.003). There was no significant difference in soil properties between manuka and broom, or between heather and red tussock, which clustered together in the PCA (Fig. 1). Soil properties composition was significantly different between heather/red tussock and broom/mānuka clusters (Pseudo-F1,48 = 5.63, P = 0.010), as well as between broom and heather (P = 0.015), broom and red tussock (P = 0.029), mānuka and heather (P = 0.024), mānuka and red tussock (P = 0.019).
Table 1 summarizes the soil properties associated with the four tested plant species. The mean soil pH values were slightly acidic for all treatments but were significantly more acidic in the soil associated with heather and red tussock than with broom and mānuka. Total N and anaerobically mineralizable N were significantly higher under broom than under other plant species; C/N ratio was significantly lower under broom (Table 1). Among cations, K+ was highest under broom, but Ca2+, Mg2+ and Na+ were highest under mānuka.
Soil fauna
Hymenoptera (ants), Collembola and Oribatida were the most abundant taxa across all sites. LDA ordination showed some separation between soil fauna assemblages under the four vegetation types (Fig. 2). Broom and mānuka were associated with higher abundances of Collembola, Oligochaeta, Diplopoda and Amphipoda, and with lower abundances of Hymenoptera and Hemiptera than heather and red tussock.
Soil fauna community composition significantly differed under plant species (PERMANOVA; Pseudo-F3,48 = 1.81, P = 0.036). However, significant differences in overall community composition were detected only between heather and mānuka (P = 0.026), and between red tussock and mānuka (P = 0.001).
Total fauna abundance was significantly different among the four plant species (LRTest; χ2 = 13.02, P = 0.005) and was highest under broom (Fig. 3, Table 2). Among individual taxa, we found significantly higher abundance of Collembola and Oligochaeta under broom, and higher abundance of Hemiptera under heather and red tussock. Hymenoptera (ants) were most abundant under red tussock (Fig. 3 and Table 2). Taxon richness, Simpson’s and Shannon’s indices for soil fauna showed no significant difference among the vegetation types.
Abundance of selected soil fauna taxa under Scotch broom (Cytisus scoparius), European heather (Calluna vulgaris), mānuka (Leptospermum scoparium) and red tussock (Chionochloa rubra), NZ Central Plateau, 2021 (see Table 2 for the full list of taxa). Values are total counts. The median for each plant species is indicated by the line across the box. The mean is indicated by a diamond. Different letters indicate significant differences of the means (Tukey’s HSD, α = 0.05)
Discussion
Invasive plants are becoming increasingly significant modifiers of ecosystems alongside other anthropogenic stressors (e.g., IPBES 2023). Many reports (mainly above-ground) link invasive plants with negative impacts on arthropod communities, but a growing body of evidence suggests that arthropod responses to invasive plants are complex and can vary in magnitude and direction depending on the studied system (Litt et al. 2024). In this study, we explored the below-ground impacts of two invasive plants (one N-fixer and one non-N fixer) on the soil properties and arthropod fauna of an iconic New Zealand ecosystem, when compared to those properties under two native plants. We found (contrary to our initial prediction) that soil properties and soil fauna assemblages did not cluster by plant invasive status, highlighting the importance of understanding the context and traits of each invasive plant species and the risk of hasty generalisation when predicting the ecological impacts of invasive plants.
Soil properties
Regarding soil properties, we found that native red tussock (C. rubra) clustered together with invasive non N-fixer heather (C. vulgaris), while native mānuka (L. scoparium) and invasive N-fixer Scotch broom (C. scoparius), and that soil property composition significantly differed between these two clusters. Mānuka and broom were associated with less acidic, more nutrient-rich soils, whereas soils under heather and red tussock were more acidic and nutrient-poor. The soil properties contributing most to the dissimilarity between plant species were nitrogen, pH, potassium, calcium, magnesium and C/N ratio. Liao et al. (2008) showed that woody N-fixing invasives have greater impacts on soil carbon and nitrogen cycling. N-fixing invasives are associated with greater impacts on soil nitrogen cycles than non-N-fixing invasives (Ehrenfeld 2003; Vilà et al. 2011). Indeed, we found that broom was associated with higher soil nutrient concentrations and had significantly higher soil total N and AMN, potassium, and lower soil C/N ratio than any other study plant species. Our findings for soil properties are not fully consistent with reports for other invasive plants, which suggest that invasives tend to have higher rates of nutrient cycling, presumably due to higher values for performance-based plant traits relative to native plants (Rothstein et al. 2004; Liao et al. 2008; Stefanowicz et al. 2018); in our study site (NICP) this was true for broom but not for heather.
Different plant species can have varying effects on soil properties, and pre-existing soil conditions can influence the germination and success of these species in distinct ways (van der Putten et al. 2013). Therefore, the relationship observed between the plant species studied and soil properties may reflect the individual species ability to thrive under specific soil conditions, rather than their ability to modify them (van der Putten et al. 2013). Plant species are adapted to different soil habitats, and certain species may have competitive advantages under specific soil conditions. Our findings indicate that heather and red tussock are associated with lower soil nutrient levels compared to mānuka and broom. Heather, an early successional species, typically establishes in nutrient-poor soils in its native range (Keesing 1995). Its adaptations for low-nutrient habitats could enable it to thrive in nutrient-poor areas, such as those in the NICP. In contrast, mānuka is associated with relatively nutrient-rich soils. As a mid-successional species, mānuka is adapted to disturbed sites and those with soil conditions that impede forest succession (Stephens et al. 2005). Mid-successional species typically colonize areas with moderate soil nutrient levels, suggesting that mānuka may preferentially establish in nutrient-rich patches. Given the observational nature of this study, it is challenging to determine whether plant species influence soil properties or vice versa, or if both processes are occurring concurrently. Consequently, caution is warranted when interpreting higher soil nitrogen concentrations under broom as a response to broom invasion rather than a driver.
Soil fauna
Regarding soil fauna abundance, soil macrofauna assemblages did not cluster by invasive status as was predicted. In fact, our results showed no consistent differences in total soil fauna abundance between invasive and native plants. Instead, we found that the mean total abundance of fauna under invasive N-fixing plant (broom) was similar to a native plant on low-fertility soil (red tussock) but was significantly higher when compared to another native species (mānuka). We found significantly more Oligochaeta and Collembola under broom, which matched our predictions of invasive plants (and especially N-fixing invasives) being associated with greater abundances of decomposers. However, another important decomposer group—oribatid mites—did not show the same tendency. Collembola are opportunistic, generalist consumers which feed on variety of decomposing organic materials and microorganisms in the soil; their abundance reflects the food supply available in the soil food web (Potapov et al. 2016). Oribatids, on the other hand, are known to have highly specific habitat preferences and may be responding to other environmental drivers (Schatz and Behan-Pelletier 2007).
Many soil fauna groups are sensitive to soil physico-chemical properties (Hopkin and Read 1992; Birkhofer et al. 2012; Hoeffner et al. 2021; Singh et al. 2020). For example, Oligochaeta are linked to numerous soil properties, including soil organic matter, pH and moisture (Baker et al. 1998; Baker and Whitby 2003; De Wandeler et al. 2016; Hoeffner et al. 2021; Jiménez et al. 2011; Perreault and Whalen 2006; Singh et al. 2020). De Wandeler et al. (2016) showed that Oligochaeta abundance increased with pH as it moved from acidic to neutral. Diplopoda have been linked to soil concentrations of calcium and magnesium, as they require these elements for development of their exoskeletons (Hopkin and Read 1992). We found that mānuka and broom were associated with higher abundances of Oligochaeta and Diplopoda than red tussock and heather, and soil analysis showed that mānuka and broom were associated with higher pH (closer to neutral) and greater Mg2+ and Ca2+ concentrations. As pH, Mg2+ and Ca2+ have been linked to the distribution of Oligochaeta and Diplopoda, the soil properties may be driving the soil fauna community compositions.
Our findings for soil fauna communities are only partially consistent with the literature, which suggests that decomposer groups could be stimulated by invasive plants relative to native plants due to their higher performance-based traits (Meisner et al. 2014; Williamson and Fitter 1996). In our study, we found this to be true for broom but not for heather. However, our findings are consistent with conclusions from a previous study by Effah et al. (2020a), who suggested that individual plant identity, rather than invasive status, was important in predicting an invasive plants effect on the above-ground arthropod community in the Central Plateau. Below-ground, this appears to be true of both soil properties and soil fauna.
Conclusion
Our findings highlight the need to consider plant species, rather than simply the invasive status when predicting the impact of plant invasion on the soil biotic and abiotic factors. As Scotch broom is associated with high soil nutrients and is likely engineering these conditions through its N-fixing ability (Liao et al. 2008), it may have a greater impact on the native habitat of the NICP area relative to European heather, which is associated with low soil fertility, as is native red tussock. By increasing the nutrient richness of the soil, broom may facilitate secondary invasion by disproportionately benefitting other non-native plants suited to nutrient-rich soils and reducing the advantage of the native plants of the area which are adapted to very low nutrient soils. As well as secondary invasion, this could accelerate the rate of succession in the Central Plateau tussock grassland ecosystems.
The relatively low nutrient soils associated with heather in our study could imply a relatively tame impact upon the soil ecosystem. Since heather favors a similar composition of soil properties as red tussock, we can predict that the removal of this invasive plant could be followed by revegetation by red tussock and other native plants that are adapted to the naturally low soil fertility of the area. The similarity in the soil properties under broom and mānuka could, however, bode well for the revegetation by mānuka following broom removal, especially if management encourages such establishment. On the other hand, habitat heterogeneity is an important aspect of the Central Plateau plant communities, so the preservation of low nutrient soil habitats may be threatened by the spread of broom.
The impacts of invasive plants on soil fauna are complex and can depend on multiple drivers such as the feeding guild, diet breadth (generalist vs. specialist), and sensitivity to environmental factors (such as soil pH and nutrients) of the target organism. For instance, we observed that generalist detritivores such as collembola to benefit from the environment N-fixing invasive plant (presumably due to higher food supply), and found a link between soil pH, Mg2+ and Ca2+ and the distribution of Oligochaeta and Diplopoda. Thus, a higher taxonomic resolution for fauna (and corresponding information on their biology and ecology) would allow better interpretation of the drivers of changes.
Altogether, the findings of this study highlight the need for future research into a variety of plant species and target arthropod feeding guilds, taxa, or species. Although we did not observe a separation of soil properties and fauna based on invasive and native plant species, this does not disprove the trends of invasive and native plants in literature, but rather highlights the limitations in using these trends alone in predicting the traits of an invasive and native subset. Instead, there may be a good cause to investigate the species-specific traits of important invasive plants and of native plants susceptible to displacement. To better quantify the magnitude of the effect invasive plants can have on the soil system, longitudinal studies should be conducted to include the pre-invasion soil properties, as well as measurements of the soil over the plants life cycle and the changes throughout various control measures. An ecosystem approach involving multiple trophic levels is highly encouraged for future studies.
Data availability statement
The data set underpinning the results can be accessed by contacting BP or MM.
References
Bagnall A (1982) Heather at Tongariro. A study of a weed introduction. Tussock Grassl Mt Lands Inst Rev 41:17–21
Baker GH, Whitby WA (2003) Soil pH preferences and the influences of soil type and temperature on the survival and growth of Aporrectodea longa (Lumbricidae): the 7th international symposium on earthworm ecology · Cardiff · Wales · 2002. Pedobiologia 47:745–753
Baker GH, Carter PJ, Curry JP, Cultreri O, Beck A (1998) Clay content of soil and its influence on the abundance of Aporrectodea trapezoides Dugès (Lumbricidae). App Soil Ecol 9:333–337
Belnap J, Phillips SL (2001) Soil biota in an ungrazed grassland: response to annual grass (Bromus tectorum) invasion. Ecol Appl 11:1261–1275
Birkhofer K, Schöning I, Alt F, Herold N, Klarner B, Maraun M et al (2012) General relationships between abiotic soil properties and soil biota across spatial scales and different land-use types. PLoS ONE 7(8):e43292
Blayney A (2012) The ecosystem effects of the biocontrol of heather (Calluna vulgaris) with the heather beetle (Lochmaea suturalis). MSc thesis, Massey University, Palmerston North
Bluhm SL, Ammerschubert S, Polle A, Scheu S (2017) Root-derived carbon and nitrogen from beech and ash trees differentially fuel soil animal food webs of deciduous forests. PLoS ONE 12:e0189502
Castro-Díez P, Alonso Á (2017) Effects of non-native riparian plants in riparian and fluvial ecosystems: a review for the Iberian Peninsula. Limnetica 36:525–541. https://doi.org/10.23818/limn.36.19
Castro-Díez P, Godoy O, Alonso A, Gallardo A, Saldaña A (2014) What explains variation in the impacts of exotic plant invasions on the nitrogen cycle? A meta-analysis. Ecol Lett 17:1–12
Chapman H, Bannister P (1990) The spread of heather, Calluna vulgaris (L.) Hull, into indigenous plant communities of Tongariro National Park. NZ J Ecol 14:7–16
Chappell PR (2015) The climate and weather of Manawatu-Wanganui. NIWA Sci Technol Ser 66:1–40
Clarke KR, Gorley RN, Somerfield PJ, Warwick RM (2014) Change in marine communities: an approach to statistical analysis and interpretation, 3rd edn. PRIMER-E, Plymouth
Clavijo McCormick A, Effah E, Najar-Rodriguez A (2023) Ecological aspects of volatile organic compounds emitted by exotic invasive plants. Front Ecol Evol 11:1059125
Dassonville N, Vanderhoeven S, Vanparys V, Hayez M, Gruber W, Meerts P (2008) Impacts of alien invasive plants on soil nutrients are correlated with initial site conditions in NW Europe. Oecologia 157:131–140
De Wandeler H, Sousa-Silva R, Ampoorter E, Bruelheide H, Carnol M, Dawud SM et al (2016) Drivers of earthworm incidence and abundance across European forests. Soil Biol Biochem 99:167–178
Department of Conservation (2006) Tongariro National Park management plan. NZ Department of Conservation
DOC (n.d). Tongariro weather. NZ Department of Conservation. https://www.doc.govt.nz/parks-and-recreation/places-to-go/central-north-island/places/tongariro-national-park/about-tongariro-national-park/tongariro-weather/. Accessed 30 Oct 2023
Ebeling A, Hines J, Hertzog LR, Lange M, Meyer ST, Simons NK, Weisser WW (2018) Plant diversity effects on arthropods and arthropod-dependent ecosystem functions in a biodiversity experiment. Basic Appl Ecol 26:50–63
Effah E, Barrett DP, Peterson PG, Potter MA, Holopainen JK, Clavijo McCormick A (2020a) Effects of two invasive weeds on arthropod community structure on the Central Plateau of New Zealand. Plants 9(7):919
Effah E, Barrett DP, Peterson PG, Godfrey AJR, Potter MA, Holopainen JK, Clavijo McCormick A (2020b) Natural variation in volatile emissions of the invasive weed Calluna vulgaris in New Zealand. Plants 9(2):283
Effah E, Barrett DP, Peterson PG, Potter MA, Holopainen JK, Clavijo McCormick A (2020c) Seasonal and environmental variation in volatile emissions of the New Zealand native plant Leptospermum scoparium in weed-invaded and non-invaded sites. Sci Rep 10(1):11736
Effah E, Svendsen L, Barrett DP, Clavijo McCormick A (2022a) Exploring plant volatile-mediated interactions between native and introduced plants and insects. Sci Rep 12(1):15450
Effah E, Min Tun K, Rangiwananga N, Clavijo McCormick A (2022b) Mānuka clones differ in their volatile profiles: potential implications for plant defence, pollinator attraction and bee products. Agronomy 12(1):169
Ehrenfeld JG (2003) Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems 6:503–523
Fu X, Guo D, Wang H, Dai X, Li M, Chen F (2017) Differentiating between root- and leaf-litter controls on the structure and stability of soil micro-food webs. Soil Biol Biochem 113:192–200
Galappaththi HSD, de Silva WPP, Clavijo McCormick A (2023) A mini-review on the impact of common gorse in its introduced ranges. Trop Ecol 64:1–25
Hayes L, Fowler SV, Paynter Q, Groenteman R, Peterson P, Dodd S et al (2013) Biocontrol of weeds: achievements to date and future outlook. In: Dymond JR (ed) Ecosystem services in New Zealand—conditions and trends. Manaaki Whenua Press, Lincoln, pp 375–385
Hewitt AE (2010) New Zealand soil classification. Manaaki Whenua Press, Lincoln
Hoeffner K, Santonja M, Monard C, Barbe L, Le Moing M, Cluzeau D (2021) Soil properties, grassland management, and landscape diversity drive the assembly of earthworm communities in temperate grasslands. Pedosphere 31:375–383
Hopkin SP, Read HJ (1992) The biology of millipedes. Oxford University Press, Oxford
Jiménez J-J, Decaëns T, Amézquita E, Rao I, Thomas RJ, Lavelle P (2011) Short-range spatial variability of soil physico-chemical variables related to earthworm clustering in a neotropical gallery forest. Soil Biol Biochem 43:1071–1080
Kappes H, Lay R, Topp W (2007) Changes in different trophic levels of litter-dwelling macrofauna associated with giant knotweed invasion. Ecosystems 10:734–744
Keesing VF (1995) Impacts of invasion on community structure: habitat and invertebrate assemblage responses to Calluna vulgaris (L.) Hull invasion in Tongariro National Park, New Zealand. Ph.D. thesis, Massey University, Palmerston North
Kottek M, Grieser J, Beck C, Rudolf B, Rubel F (2006) World map of the Köppen-Geiger climate classification updated. Meteorol Z 15:259–263
Landcare Research (2022) How damaging is the broom gall mite? https://www.landcareresearch.co.nz/publications/weed-biocontrol/weed-biocontrol-articles/how-damaging-is-the-broom-gall-mite/. Accessed 30 Oct 2023
Levine JM, Vila M, Antonio CMD, Dukes JS, Grigulis K, Lavorel S (2003) Mechanisms underlying the impacts of exotic plant invasions. Proc R Soc B 270:775–781
Liao C, Peng R, Luo Y, Zhou X, Wu X, Fang C et al (2008) Altered ecosystem carbon and nitrogen cycles by plant invasion: a meta-analysis. New Phytol 177:706–714
Litt AR, Cord EE, Fulbright TE, Schuster GL (2014) Effects of invasive plants on arthropods. Conserv Biol 28:1532–1549
Litt AR, Mitchell AB, Tallamy DW (2024) Alien plants and insect diversity. In: Rodríguez J, Pyšek P, Novoa A (eds) Biological invasions and global insect decline. Academic Press, pp 119–142. https://doi.org/10.1016/B978-0-323-99918-2.00005-7
Meisner A, Hol G, de Boer W, Krumins J, Wardle D, Putten W (2014) Plant–soil feedbacks of exotic plant species across life forms: a meta-analysis. Biol Invasions. https://doi.org/10.1007/s10530-014-0685-2
Morgan E, Perry NB, Chagné D (2019) Science at the intersection of cultures—Māori, Pākehā and mānuka. NZ J Crop Hortic Sci 47:225–232
Morris EK, Caruso T, Buscot F, Fischer M, Hancock C, Maier TS et al (2014) Choosing and using diversity indices: insights for ecological applications from the German Biodiversity Exploratories. Ecol Evol 4:3514–3524
Muñoz AA, Cavieres LA (2008) The presence of a showy invasive plant disrupts pollinator service and reproductive output in native alpine species only at high densities. J Ecol 96:459–467
Perreault JM, Whalen JK (2006) Earthworm burrowing in laboratory microcosms as influenced by soil temperature and moisture. Pedobiologia 50:397–403
Pollierer MM, Langel R, Körner C, Maraun M, Scheu S (2007) The underestimated importance of belowground carbon input for forest soil animal food webs. Ecol Lett 10:729–736
Potapov AM, Semenina EE, Korotkevich AY, Kuznetsova NA, Tiunov AV (2016) Connecting taxonomy and ecology: trophic niches of collembolans as related to taxonomic identity and life forms. Soil Biol Biochem 101:20–31
Potapov AM, Tiunov AV, Scheu S (2019) Uncovering trophic positions and food resources of soil animals using bulk natural stable isotope composition. Biol Rev 94:37–59
Potapov AM, Beaulieu F, Birkhofer K, Bluhm SL et al (2022) Feeding habits and multifunctional classification of soil-associated consumers from protists to vertebrates. Biol Rev 97:1057–1117
Rothstein DE, Vitousek PM, Simmons BL (2004) An exotic tree alters decomposition and nutrient cycling in a Hawaiian montane forest. Ecosystems 7:805–814
IPBES (2023) Summary for policymakers of the thematic assessment report on invasive alien species and their control of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. In: Roy HE, Pauchard A, Stoett P, Renard Truong T, Bacher S, Galil BS, Hulme PE, Ikeda T, Sankaran KV, McGeoch MA, Meyerson LA, Nuñez MA, Ordonez A, Rahlao SJ, Schwindt E, Seebens H, Sheppard AW, Vandvik V (edss). IPBES secretariat, Bonn, Germany. https://doi.org/10.5281/zenodo.7430692
Schatz H, Behan-Pelletier V (2007) Global diversity of oribatids (Oribatida: Acari: Arachnida). In: Balian EV, Lévêque C, Segers H, Martens K (eds) Freshwater animal diversity assessment. Developments in hydrobiology, vol 198. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-8259-7_35
Singh S, Sharma A, Khajuria K, Singh J, Vig AP (2020) Soil properties changes earthworm diversity indices in different agro-ecosystem. BMC Ecol 20:1–14
Souza-Alonso P, Guisande-Collazo A, González L (2015) Gradualism in Acacia dealbata Link invasion: impact on soil chemistry and microbial community over a chronological sequence. Soil Biol Biochem 80:315–323
Sprunger CD, Culman SW, Peralta AL, DuPont ST, Lennon JT, Snapp SS (2019) Perennial grain crop roots and nitrogen management shape soil food webs and soil carbon dynamics. Soil Biol Biochem. https://doi.org/10.1016/j.soilbio.2019.107573
Stefanowicz AM, Majewska ML, Stanek M, Nobis M, Zubek S (2018) Differential influence of four invasive plant species on soil physicochemical properties in a pot experiment. JSS 18:1409–1423
Stephens J, Molan PC, Clarkson BD (2005) A review of Leptospermum scoparium (Myrtaceae) in New Zealand. NZ J Bot 43:431–449
Tanner RA, Varia S, Eschen R, Wood S, Murphy ST, Gange AC (2013) Impacts of an invasive non-native annual weed, Impatiens glandulifera, on above-and below-ground invertebrate communities in the United Kingdom. PLoS ONE 8(6):e67271
van der Putten WH, Bardgett RD, Bever JD, Bezemer TM, Casper BB, Fukami T et al (2013) Plant–soil feedbacks: the past, the present and future challenges. J Ecol 101:265–276
Vilà M, Espinar JL, Hejda M, Hulme PE, Jarošík V, Maron JL et al (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14:702–708
Weidenhamer JD, Callaway RM (2010) Direct and indirect effects of invasive plants on soil chemistry and ecosystem function. J Chem Ecol 36:59–69
Williamson M, Fitter A (1996) The varying success of invaders. Ecology 77:1661–1666
Wolfe BE, Klironomos JN (2005) Breaking new ground: soil communities and exotic plant invasion. Bioscience 55:477–487
Wolkovich EM, Bolger DT, Holway DA (2009) Complex responses to invasive grass litter by ground arthropods in a Mediterranean scrub ecosystem. Oecologia 161:697–708. https://doi.org/10.1007/s00442-009-1425-7
Acknowledgements
We are grateful to Evans Effah, Paul Barrett, Paul Peterson, Mari Nakano, Ruby Mountford and Fern Kumeroa their assistance with field work and data processing. Thanks to Massey technical staff – Cleland Wallace, Shaun Nielsen and Tracy Harris – for all the help provided to BP with equipment and documentation. We also thank the anonymous reviewers for their comments which helped to improve the manuscript.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This project was possible thanks to a Marsden Fast Start Grant (Royal Society of New Zealand – Te Apārangi) to ACM.
Author information
Authors and Affiliations
Contributions
AR, MM and ACM designed this study. BP collected the data, analysed the data and made the figures. AR, MM and ACM supervised BP. BP and MM wrote the original draft. ACM provided funding for fieldwork and chemical analysis. All authors contributed to interpretation of results and commented on the manuscript drafts.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pearson, B.M., Minor, M.A., Robertson, A.W. et al. Plant invasion down under: exploring the below-ground impact of invasive plant species on soil properties and invertebrate communities in the Central Plateau of New Zealand. Biol Invasions (2024). https://doi.org/10.1007/s10530-024-03441-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10530-024-03441-z