Abstract
Few conifers are considered invasive in Europe, yet recent studies indicate that several species used for forestry display abundant regeneration and spread into surrounding natural habitats. Three species were identified as being particularly at risk in Belgium, but data is lacking regarding their dispersal. We characterized the recruitment curves of Tsuga heterophylla, Abies grandis and Thuja plicata. Isolated plantations were monitored and realized dispersal (i.e. seedlings and recruited regeneration) was recorded and measured over 750 m in different directions. We calculated the wave expansion rate and frontier expansion rate for each planting site and fitted dispersal kernels for each site and species. Regeneration was classified in three size categories (seedlings, saplings and trees above 1.5 m), and the recruitment distances were analyzed for each size class. The effect of the forest type (deciduous, coniferous, open or mixed) on the density of regeneration was also investigated with regression models. The recruitment curves varied greatly across sites, showing heterogeneous habitat suitability and uneven post-germination processes. Considering the frontier expansion rate, the three conifers appear to spread beyond documented threshold rate of invasiveness. Regeneration density was higher in coniferous forest type, as well as open areas for Tsuga heterophylla. An escape effect was noticed as mean and maximal dispersal distances of saplings and taller trees were greater than those of seedlings. Our study indicates that Tsuga heterophylla displays the highest risk of rapid spread into adjacent natural habitats, followed by Abies grandis. Thuja plicata faces more recruitment limitations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Introduced tree species can be significant invaders, leading to various large-scale impacts on ecosystems (Richardson and Rejmánek 2011; Pyšek 2016). Conifers, in particular, are troublesome invaders in the Southern Hemisphere, with notable examples including Pinus species in New Zealand and South Africa, and Cryptomeria japonica (L. f.) D.Don on Réunion Island (Richardson and Rejmánek 2004; Richardson et al. 2004; Edwards et al. 2021). However, in Europe, reports of conifer invasions are relatively scarce, despite the extensive use of introduced conifers for timber production.
The relatively low number of documented conifer invasions in Europe could be attributed to the recent history of their introduction, as well as the phylogenetic proximity of introduced species to native conifers (Kowarik 1995; Carrillo-Gavilán and Vilà 2010). Alternatively, this scarcity may reflect a lack of awareness or insufficient research on ongoing spread. Silviculture is recognized as a major pathway for the introduction of invasive tree species (Pyšek 2016) and Essl et al. (2010) demonstrated that conifers introduced for timber production have a higher likelihood of escaping cultivation compared to those introduced for other purposes.
Nevertheless, certain conifer species have exhibited invasive behavior in temperate forests of Europe. For instance, Picea sitchensis (Bong.) Carrière is considered invasive in Norway, dense regeneration of Tsuga heterophylla (Raf.) Sarg. has been reported in Scotland, and studies in Germany have recommended avoiding the planting of Pseudotsuga menziesii (Mirb.) Franco near sensitive natural habitats (Forestry Commission Scotland 2015; Nygaard and Øyen 2017; Bindewald et al. 2021). These examples underscore the potential for conifer species to become invasive in European temperate forests, highlighting the need for ongoing monitoring and research to better understand and mitigate their spread.
Dispersal is a critical component of the invasion process, playing a fundamental role in determining spatial patterns and rates of species spread (Clobert et al. 2001; Cousens et al. 2008). A detailed examination of dispersal and recruitment patterns in potential invaders can thus enhance our understanding of colonization dynamics, help identify species that are rapidly spreading, and support efforts to prevent further expansion (Kot et al. 1996; Hastings et al. 2005; Davies and Sheley 2007; Monty and Mahy 2010).
Seed dispersal is influenced by species traits like the propagule size and mass and its terminal velocity and plant height (Caplat et al. 2012; Moravcová et al. 2015; Johnson et al. 2019). Tamme et al. (2014) demonstrated that decent predictions of maximum dispersal distances can be achieved using a combination of simple traits, such as dispersal syndrome, growth form, seed mass, seed release height and terminal velocity. Typically, the majority of wind-dispersed seeds fall near the parental trees, but the smaller fraction of seeds dispersed over longer distances is important for species spread.
While studying seed dispersal is crucial for understanding a species' ability to reach long distances, the germination and recruitment of seedlings also play a critical role in determining the abundance and spatial structure of natural regeneration, as well as population dynamics (Amm et al. 2012; Beckman et al. 2020). Realized dispersal, which combines seed dispersal with seedling establishment, is influenced by abiotic and biotic factors that affect the latter process (Bullock et al. 2006). The distribution of surviving seedlings can be impacted by the spatial arrangement of suitable micro-habitats, post-dispersal predation or diseases, intra- or inter-specific competition as well as the seedling mortality rate (Amm et al. 2012). Vulnerability to predators and pathogens is often believed to be greater near parent trees, a distance-dependent process known as the Janzen-Connell effect (Janzen 1970; Connell 1971). As a result, seedling recruitment is expected to be proportionally higher farther from parent trees—a concept more simply referred to as the "escape hypothesis" by Howe and Smallwood (1982). Martin et al. (2010) demonstrated a density-dependent effect for recruitment curves for both native and exotic species in closed temperate forests of Connecticut, where the mean dispersal distance of seedlings was found to be greater than that of seeds. The escape hypothesis was also supported in a study on Pinus halepensis in Israel, where Nathan et al. (2000) found that the probability of seedlings’ survival increased with the distance from the plantation.
In a previous study we performed in old forest arboreta of Southern Belgium (Fanal et al. 2021), we identified several conifer species displaying an important natural regeneration density and surpassing the rather subjective but commonly used threshold of dispersal over 100 m from plantings in 50 years proposed by Richardson et al. (2000). However, the measurement of dispersal in that study was limited to a 100-m buffer around the arboreta, resulting in truncated recruitment curves and an incomplete understanding of dispersal patterns beyond this distance.
In the present study, we selected the three species presenting the highest invasive potential identified by Fanal et al. (2021), based on the combination of their regeneration density, dispersal distance and size structure: Tsuga heterophylla, Abies grandis (Douglas ex D.Don) Lindl. and Thuja plicata Donn ex D.Don. These three species were planted in several forest trials across Southern Belgium over the last century, but to a very limited extent. Taking advantage of the existence of these isolated stands, we aimed to describe the recruitment curves of these species to better document their invasive potential and inform decision-making for forest managers. In addition, we tested for a potential escape effect and assessed whether the surrounding forest type influenced dispersal distances and regeneration densities.
Material and method
Study species
Tsuga heterophylla (Western hemlock) is an evergreen coniferous tree from the Pinaceae family that can reach a height of 80 m in its area of origin. This area extends from southern Alaska to northern California (Christy and Mack 1984). It is a highly shade-tolerant specie, and seedlings grow well under their own cover (Schrader 1998). Tsuga heterophylla seeds are winged and their total length is about 7 mm. Mean seed weight is 1.75 g (Turner 1985). They are primarily dispersed by wind. Age of first fecundity is 25 years (Petit et al. 2017). The mature tree produces seeds every year with a peak production every three to four years (Christy and Mack 1984). Western hemlock was introduced in Belgium for its high growth rate in regions with sufficient water supply (Galoux 1951). Its appearance in Belgian arboreta dates back to the beginning of the twentieth century. Despite a fairly sustained growth, this species was not popularized in Southern Belgium. Very few stands have been planted, mainly in the region of the Ardennes for site adequacy.
Abies grandis (Vancouver fir) is an evergreen conifer tree from the Pinaceae family reaching 100 m in height in its native rage, in western North America (Riou-Nivert 2001). It is a shade-tolerant specie. Age of first fecundity is 25 years (Petit et al. 2017). Seeds are winged and their total length is about 17 mm (Riou-Nivert 2001). Mean seed weight is 25.0 g (Turner 1985). They are primarily dispersed by wind. Abies grandis was introduced in Western Europe during the nineteenth century. In Southern Belgium, the species has been planted in several arboreta, but forest plantations are rare (OEWB 2021).
Thuja plicata (Western Red-cedar) is an evergreen tree from the Cupressaceae family, originating from western North America (Riou-Nivert 2001). It is a shade-tolerant specie. Age of first fecundity is 20 years (Petit et al. 2017). Seeds are about 6 mm long, with lateral wings about as wide as the body (Riou-Nivert 2001). Mean seed weight is 1.10 g (Turner 1985). They are primarily dispersed by wind. Thuja plicata was introduced in Western Europe during the nineteenth century. In Southern Belgium, the species has been planted in several arboreta and different cultivars are used for ornamental purposes in gardens (Riou-Nivert 2001). Forest plantations are rare.
Based on species traits, Richardson and Rejmánek (2011) predicted high invasiveness for Tsuga heterophylla and Thuja plicata, but not for Abies grandis. However, all three species have been documented as invasive in Great Britain at least once (Clement and Foster 1994; Welch et al. 2001). Tsuga heterophylla has shown particularly vigorous regeneration around plantings in Scotland and Norway, where few native species can survive beneath its canopy (Oyen 2001; Harmer et al. 2011; Forestry Commission Scotland 2015). In Germany, it has also been reported to outcompete native species (Frischbier et al. 2017).
Study sites
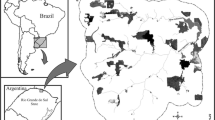
A total of 14 sites were selected across Southern Belgium, comprising 6 sites for Tsuga heterophylla, 4 sites for Abies grandis, and 4 sites for Thuja plicata (Fig. 1). These sites consisted of isolated, monospecific, and even-aged small stands, each less than 1 hectare in size and at least 50 years old, with planting dates ranging from 1919 to 1970. All sites were located at least 2 km from any other plantings of the same or closely related species to avoid confusion at the seedling stage. The elevation of the sites ranged from 197 to 634 m above sea level, with exact locations provided in Table 1 and illustrated in Fig. 1.
Location of the study sites for each species in Wallonia, Belgium. Background on the map is the tree cover in 2000 (Hansen et al. 2013)
The surrounding environments varied depending on location but were primarily forested areas, ranging from native beech forests to intensively planted spruce stands, with occasional open clear-cuts or grasslands. Western sites (Froidchapelle, Viroinval, Rochefort) were predominantly surrounded by native oak and beech forests, while southern sites (Melreux, Laidprangeleux, Saint-Michel, Mirwart) were characterized by beech and spruce forests on brown-acidic soils. Northern sites (Baelen, Jalhay) were situated on peaty soils and were bordered by beech, spruce, or open grasslands.
Data collection
At each site, three directions were randomly chosen. A circle sector of 10° and 750 m long was delimited for each direction, starting 20 m inside of the planting to describe the regeneration under the parent trees as well (Fig. 2). This methods ensures a constant sampling effort along increasing distance (Bullock and Clarke 2000). For each 10-m interval, the number of individuals originating from the natural regeneration was counted and assigned to one of the three size classes: 0 to 0.3 m (A), 0.3 to 1.5 m (B) and more 1.5 m (C). The first class represent seedlings, the second one saplings and the third one established trees (Laroque et al. 2001).The type of forest cover (deciduous, coniferous, mixed or open) was also noted for each 10-m interval.
Fieldwork took place from 2018 to 2022, during summer and autumn. Each sector was visited once. Sites with Tsuga heterophylla were visited either in 2018 or 2019. For the site of Mirwart, one of the three sampled sectors could not be completed and was therefore replaced by one additional sector in the site of Vecmont. Sites with Abie grandis and Thuja plicata were visited in 2020 and/or 2022 (see details in Table 1).
Data analyses
While dispersal curves are built to model seed dispersal over the distance from a seed source, recruitment curves describe the distribution of actual descendants at increasing distance from the parent trees. The former corresponds either to the predicted density of seeds as a function of distance, or to probability density function, also called dispersal kernels, representing the distribution of post-dispersal locations at different distances from the parent trees (Nathan et al. 2012). Several standardized dispersal kernels are described in the literature, which perform relatively well according to the studied growth form or dispersal mode (Bullock et al. 2017). Wald, 2DT, Lognormal and mixed models (models encompassing two different kernels to explain both short- and long-distance dispersal) are often preferred for wind-dispersed species (Bullock and Clarke 2000; Greene et al. 2004; Martin and Canham 2010; Norghauer et al. 2011; Loebach and Anderson 2018; Wyse and Hulme 2021). However, the preliminary choice of a function is quite subjective, and the best approach is to fit several functions and assess their relevance with a best-fit approach and examination of the credibility of the tail prediction (Bullock et al. 2006).
Richardson et al. (2000) suggested the threshold of 100 m in 50 years for seed-dispersed alien plants to be considered invasive. As an application of this definition, Nygaard and Øyen (2017) described the dispersal of established regeneration Picea sitchensis around plantings in Norway with two indicators: the Wave Expansion Rate (WER), which is the median spread distance divided by the parent stand age minus their maturity age (Eq. 1), and the Frontier Expansion Rate (FER) is the maximum spread distance divided by the parent stand age minus their maturity age (Eq. 2). These indicators allow to discuss invasiveness with empirical and objective quantitative data.
Regeneration density was calculated based on the number of individuals and the area of each 10-m interval of the circle sectors. The regeneration density values in the 20 m inside the plantings were used in histograms but excluded for the rest of the analyses.
The “dispfit” package was used to model the probability density functions for each species. The “dispfit” package fits and compares several parameterized functions usually used in dispersal studies to describe dispersal curves and predict dispersal distances (Proença-Ferreira et al. 2023). For each site as well as the combination of all sites per species, the best function was selected based on AIC (Bozdogan 1987). The parameters value, mean, skewness and kurtosis were extracted with the “dispfit” package. The predicted values were plotted with the “ggplot2” package (Wickham 2016).
We fitted a linear mixed model to test if the maximum dispersal distance (provided for each sector) differs significantly with the species. The model included the site, the time since planting and the year of sampling as random effects.
To test the influence of the forest cover type (deciduous, coniferous, mixed or open) on the regeneration, zero-inflated Poisson regressions were fitted on count of trees for each species with the “glmmTMB” package (Brooks et al. 2017) Forest cover type and distance were set as fixed effects, site, time since planting and year of sampling as random effects. The distance was also used as covariate for the zero-inflated part of the model. Overdispersion and zero-inflation were checked with the “performance” package. AIC of the models with and without the time since planting and the year of sampling as random effect were calculated, and the best model selected.
We used an alpha level of 0.05 for all statistical tests. All analyses were performed in R Studio 4.3.1 (R Core Team 2022).
Results
On average, the number of individuals recorded per sector was 614.7 ± 323.6 (mean ± SE) for Tsuga heterophylla, 2202.9 ± 829.5 for Abies grandis and 89.2 ± 46.37 for Thuja plicata. If we remove the first 20 m inside the plantings, the numbers drop to 271.6 ± 192.0, 1528.9 ± 643.6 and 45.8 ± 27 respectively, which means that 44%, 69% and 51% of the counted regeneration was situated under the planting. However, 92 to 93% of this under-parent’s regeneration was under 0.3 m high. Maximum regeneration densities reached 5.27 trees.m−2 for Tsuga heterophylla, 55.2 trees.m−2 for Abies grandis and 3.02 trees.m−2 for Thuja plicata.
The raw dispersal data, expressed as the regeneration density against the distance from plantings, is shown for the three species in Fig. 3A, 4A and 5A.
A Raw data of dispersal of Tsuga heterophylla per site. Y axis is the mean of the regeneration densities between sectors. X axis is the distance from the edge of the plantation. Colors indicate the proportion of trees in the size classes: A for seedlings < 0.3 m, B for saplings between 0.3 and 1.5 m, C for young trees > 1.5 m. B Comparison of predicted values of dispersal (probability density functions) for each site and for the combination of all sites. Best model, based on AIC, is used each time. C) Proportion of recruited trees found under each forest cover type
A Raw data of dispersal of Abies grandis per site. Y axis is the mean of the regeneration densities between sectors. X axis is the distance from the edge of the plantation. Colors indicate the proportion of trees in the three size classes: A for seedlings less than 0.3 m, B for saplings between 0.3 and 1.5 m, C for young trees above 1.5 m. B Comparison of predicted values of dispersal (probability density functions) for each site and for the combination of all sites. Best model, based on AIC, is used each time. C Proportion of recruited trees found under each forest cover type
A Raw data of dispersal of Thuja plicata per site. Y axis is the mean of the regeneration densities between sectors. X axis is the distance from the edge of the plantation. Colors indicate the proportion of trees in the three size classes: A for seedlings less than 0.3 m, B for saplings between 0.3 and 1.5 m, C for young trees above 1.5 m. B Comparison of predicted values of dispersal (probability density functions) for each site and for the combination of all sites. Best model, based on AIC, is used each time. C Proportion of recruited trees found under each forest cover type
The mean dispersal distance across all sectors was 51.9 ± 15.6 m (mean ± SE) for Tsuga heterophylla, 22.7 ± 3.8 m for Abies grandis and 29.5 ± 11.9 m for Thuja plicata. Maximum dispersal distances were 565 m for T. heterophylla, 355 m for A. grandis and 265 m for T. plicata. When comparing across sectors, maximum dispersal distance of T. plicata were significantly lower than the maximum dispersal values of T. heterophylla and A. grandis (p = 0.04, estimate = -141.6). The model's total explanatory power was substantial (conditional R2 = 0.44) and the part related to the fixed effects alone (marginal R2) was of 0.16. Time since planting was not retained in the final model.
Among sites, WER ranged from 0.14 to 6.21 m year−1 for Tsuga heterophylla, with an average of 1.41 m year−1. It ranged from 0.11 to 1.22 m year−1 for Thuja plicata, with an average of 0.63 m year−1 and from 0.17 to 1 m year−1 for Abies grandis, with an average of 0.44 m year−1. FER varied from 0.38 to 17.12 m year−1 for Tsuga heterophylla, with an average of 8.20 m year−1. It ranged from 1.49 to 6.02 m year−1 for Thuja plicata, with an average of 3.56 m year−1 and from 2.93 to 10.14 m year−1 for Abies grandis, with an average of 7.04 m year−1.
The probability density functions show right-skewed, leptokurtic recruitment curves, with parameters varying greatly between sites (Table S1, supplementary material). Particularly, the curve of Mirwart presents a remarkably flat shape and fat tail and differs greatly from all the other recruitment curves of Tsuga heterophylla (Fig. 3). In most cases, most of the seeds fall in the first 100 m of the seed source. Among all the tested functions, the Wald function was the most often selected, followed by the 2Dt function.
Dispersal parameters also varied according to the considered size class. Looking at Table S2 (supplementary material), mean and median distances are often higher for trees above 0.3 m (B) and 1.5 m (C) than for seedlings under 0.3 m (A). If we consider the 95th percentile of distance, saplings and well-established trees are found further from plantings than seedlings: 201.9 (B) and 195.3 m (C) against 93 m for seedlings of Tsuga heterophylla, 85.9 (B) and 82.7 m (C) against 55 m for seedlings of Abies grandis and 89.3 (B) and 71.5 m (C) against 48.3 m for seedlings of Thuja plicata.
The influence of forest cover type on regeneration count was evaluated, and although zero-inflation did not reach statistical significance, residual graphical analysis from the tested models indicated that a zero-inflated model provided a superior fit. Overdispersion was not detected. Deciduous covers were primarily beech- or oak-dominated stands, while coniferous covers mainly consisted of Norway spruce plantations. Open areas comprised clear-cuts or small forest clearings. A significant effect of forest cover type on regeneration count was observed, with the exception of Thuja plicata, for which model convergence was not achieved. Although this species tended to favor open areas, the low sapling count led to high error margins, precluding the identification of a definitive pattern. For Tsuga heterophylla, regeneration densities were significantly higher in open areas (p < 0.001) and under coniferous cover (p < 0.001) compared to deciduous cover (Fig. 3C). Abies grandis demonstrated a clear preference for coniferous cover (p < 0.001) (Fig. 4C). Detailed model parameters are presented in Table S3 (supplementary material).
Discussion
Before an exotic tree species can be considered as invasive, it is necessary to document its ability to disperse at significant distance from the planted parent trees and to regenerate abundantly (Richardson et al. 2000; Nygaard and Øyen 2017). Also, its offspring must be capable of reaching maturity in order to reproduce and, in turn, create new populations.
The three conifer species under study showed variable spread patterns and regeneration capacities, with different proportion of established trees in the regeneration.
Considering spread capacities, the observed values of WER for the three species were relatively modest, at least if compared to the 2 m year−1 benchmark corresponding to the proposed dispersal threshold of 100 m from parent trees in less than 50 years by Richardson et al. (2000). WER was above this threshold only for Tsuga heterophylla in one site. However, the FER can be considered a more reliable estimate of species actual spread (Skarpaas and Shea 2007). Considering FER, the three species crossed the invasive threshold in the majority of sites. On average, Tsuga heterophylla FER was 8.20 m year−1 but it even reached 17 m year−1 in the most invaded site of Mirwart. Abies grandis FER reached 10 m year−1, against 6 m year−1 for Thuja plicata.
Wald and 2Dt were the most selected functions for the dispersal kernels, two distributions that tend to produce flat tails (Nathan et al. 2012). Tsuga heterophylla displayed the most important dispersal distances with one site characterized by a really flat dispersal curve, with half of the regeneration exceeding 200 m. For the rest of the sites and species, curves were very right-skewed with half of the regeneration in the first 50 m around the plantings. This is consistent with other observations of recruited dispersal made in similar studies; in Amm et al. (2012), the mean of the realized dispersal distance of A. alba is 5 to 25 m from parent trees. In Nygaard and Øyen (2017), median spread distance of Picea sitchensis was often under 50 m, except for a few sites where it reached up to 200 m. As our three species are wind-dispersed, wind and topography likely affected dispersal, and therefore our observations may vary according to the chosen sampling direction. We did expect dispersal at further distance in the direction of strongest winds during the period of seeds release, and we tested this assumption (data not shown). However, the test was inconclusive because of the great variations in the direction of strongest winds between years and sites. Though, considering this effect, we did sample at least three sectors per site in varying directions and managed to have a balanced representation of directions for each species.
Long-distance dispersal events influence the rate of invasions by allowing the settlement of new populations at far distances, and these events are not as rare as one might think (Horn et al. 2001). Tsuga heterophylla seeds have large wings and relatively small mass, enabling them to be dispersed over long distances (Owens and Molder 1984; Turner 1985). In open and moderately windy areas of its native range, dispersal up to 1150 m from the parent tree has been reported. In denser stands, however, most seeds are known to fall much closer to the parents (Owens and Molder 1984). Despite large wing-like features, the seeds of Abies grandis are much heavier than the two other species under study (Turner 1985). Thuja plicata seeds are small but have small wings, and observed dispersal in the native range does not exceed 122 m from the parent trees (Arno and Hammerly 1977). Long-dispersal is difficult to detect on experimental setups and requires extensive samplings. In our case, we did find some regeneration at far distance from the plantings, exceeding 200 m for Thuja plicata, 300 m for Abies grandis and even 500 m for Tsuga heterophylla. These maximal dispersal distances are congruent with the information available from the native range.
High densities of regeneration were observed for the studied conifer species, especially for Abies grandis for which the maximum density measured was 55 trees/m2. These high values are usually observed in the first 50 m after the planting edge. If a high density of Tsuga heterophylla and Abies grandis were found in our sampling, much lower densities were observed for Thuja plicata. According to Minore (1990), average annual seed crops of Thuja plicata in its native range vary greatly but reach several thousands of seeds per ha. Seed production may be lower in its introduced range, or seed or seedling survival be low. In its native range, Thuja plicata seeds remain viable for several years (Minore 1990) and Schopmeyer (1974) reported germination rates between 34 and 90%, but seedling survival is low. Drought damage seedlings grown in full sunlight and Fungi, birds, insects, and smothering by fallen leaves of deciduous species are causes of seedling mortality (Arno and Hammerly 1977). In contrast, Tsuga heterophylla seeds are viable only into the first growing season after release and appears to germinate and grow well on many seedbeds whether rotten wood, litter, or bare soil (Williamson 1976). For Abies grandis, germination is variable but relatively low. Over 40% of seedlings die during the first two tears, due to biotic agents and drought (Foiles et al. 1990).
For Tsuga heterophylla, 61% of the stems were saplings between 0.3 and 1.5 m high, and 13% were above 1.5 m. Amongst these were cone-bearing mature trees. This clearly shows that not only do the seeds germinate, but the development of the descendants is common. For Abies grandis, trees over 1.5 m high represent less than 1% of the regeneration (13 individuals) and saplings 5% (931 individuals nonetheless). Finally, only 2 trees over 1.5 m were found for Thuja plicata (< 1%) and 34 saplings (9%), against 342 seedlings. The mortality of seedlings in Southern Belgium seems to be far more important for Abies grandis and Thuja plicata than for Tsuga heterophylla.
Not only are recruited trees present for Tsuga heterophylla and Abies grandis, but taller trees are found further from plantings. The probability of seedlings recruitment is therefore higher with increased distance from parent trees, which validates the escape hypothesis for these studied species. This suggests that the shape of the recruitment curves is dissimilar to the seed shadow, both because of varying favorable micro-sites availability and distance-dependent survival rates. However, we do not have the necessary data to clarify the process under this escape effect, i.e. if it is due to competition or herbivory and pest pressures. Though, regeneration was also dense inside the plantations, under the parent trees, but most of this regeneration was under 0.3 m high: dense planting densities and lack of light reaching the understory likely induce competition that hinders seedling development.
Both seed dispersal and environmental characteristics play an important role in the recruitment of seedlings (Amm et al. 2012). The great variation of spread observed in the different sites is probably due to abiotic and biotic filters. On the seed dispersal stage, time since fecundity, topography and local wind conditions can influence the seed rain spatial distribution (Norghauer et al. 2011; Caplat et al. 2012). Availability of favorable microsites (vegetation type, disturbances…) affects the germination of seeds, while the recruitment stages is highly dependent on the resource availability and the structure and composition of the receiving community (competition, herbivory, pests). The invasibility of the habitats surrounding the plantings of exotic species therefore plays a role in the spread of an exotic tree species (Nygaard and Øyen 2017). In our previous study (Fanal et al. 2021) on the regeneration of exotic conifers in old arboreta, it appeared that the density of regeneration was in general higher under coniferous stands and in forest clearings than under deciduous tree species. In Amm et al. (2012), the regeneration of Abies alba was affected by tall beech trees but facilitated by pine plantations. Calviño-Cancela and Rubido-Bará (2013) demonstrated that native forests are more resistant to invasions from exotic eucalyptus species than pine plantations. In our study, we found that regeneration of Abies grandis is facilitated by conifer plantations, while Tsuga heterophylla favors open areas (such as forest clearings) and coniferous stands as well. Climax native beech forest appear to be particularly resistant to invasions from exotic conifers, that struggle to find favorable germination conditions and may suffer from low light availability (Ligot et al. 2013). Thuja plicata also displayed low regeneration in the region of the “Hautes Fagnes” (sites of Jalhay and Baelen), likely because of dense layers of graminoids like purple moor-grass (Molinia caeruleae). This detrimental competition with graminids was also observed for Sitka spruce in Coastal Norway (Nygaard and Øyen 2017).
Performing a thorough data collection of the realized dispersal around isolated, mature plantings provides valuable information on the dispersal process and potential of a species, which is an indubitable asset in assessing its invasive potential. Still, this method is particularly time-consuming. A manpower of almost 100 man day was necessary to conduct this study, and the important variation between locations emphasizes the need of multiplying monitoring sites. Yet, management of an exotic species must be undertaken before its exponential spread to be effective. Given the urgency to detect species at risk at an early stage of invasion, simplified protocols could be developed to assess the potential of regeneration and spread. Wyse and Hulme (2021) demonstrated that modelled dispersal potential is the strongest predictors of spread rates of exotic pines in New-Zealand, far more efficient that currently used risk assessment tools. Dispersal models could become part of risk assessment combining on-site monitoring of mature plantings with simpler predictors such as species traits or introduction pathways. Growth and dispersal traits have already been identified as predictors of invasiveness (Grotkopp et al. 2002; Richardson and Rejmánek 2004; van Kleunen et al. 2010; Fanal et al. 2022, 2023). In 2014, Tamme et al. (2014) demonstrated that maximum dispersal distances can be reasonably predicted using a simple model based on growth form, dispersal syndrome, and mean seed mass. For our three species, it estimates maximum dispersal distances of 408 m (CI = 234–712 m) for Tsuga heterophylla, 325 m (CI = 189–558 m) for Abies grandis, and 274 m (CI = 150–502 m) for Thuja plicata. These estimates are in close agreement with our observed maximum dispersal distances of 585 m, 375 m, and 285 m, respectively, with most observations within sites falling within the predicted confidence intervals. This alignment suggests that even simple trait-based models can be effective in predicting the potential spread of exotic species, underscoring their value in ecological risk assessments and management strategies. Examined in parallel with the vulnerability of the receiving ecosystem, and the foreseen exotic species range evolution under climate change (Thurm et al. 2018; Puchałka et al. 2023), such risk assessment tools would provide helpful information for a smart selection of species used in future forest plantings.
Conclusion
Given the numerous post-germination events affecting the recruitment of seedlings and the spatial heterogeneity of micro-sites suitable for germination, multiplying sites and transects is a necessity to assess the invasive potential of exotic conifers based on their spread. Tsuga heterophylla, Abies grandis and Thuja plicata all spread outside plantings, but considering the dispersal distances, measured size classes and regeneration densities, Tsuga heterophylla is the species the most at risk of becoming invasive in suitable receiving communities. Models predicting maximum dispersal distances based on functional traits may prove useful when on-site time-consuming monitoring is not feasible.
References
Amm A, Pichot C, Dreyfus P, Davi H, Fady B (2012) Improving the estimation of landscape scale seed dispersal by integrating seedling recruitment. Ann for Sci 69:845–856. https://doi.org/10.1007/S13595-012-0208-1/FIGURES/3
Arno SF, Hammerly RP (1977) Northwest trees. The Montaineers, Seattle
Beckman NG, Aslan CE, Rogers HS (2020) Introduction to the special issue: the role of seed dispersal in plant populations: perspectives and advances in a changing world. AoB PLANTS. https://doi.org/10.1093/AOBPLA/PLAA010
Bindewald A, Miocic S, Wedler A, Bauhus J (2021) Forest inventory-based assessments of the invasion risk of Pseudotsuga menziesii (Mirb.) Franco and Quercus rubra L. in Germany. Eur J Forest Res 140:883–899. https://doi.org/10.1007/S10342-021-01373-0/FIGURES/5
Bozdogan H (1987) Model selection and Akaike’s Information Criterion (AIC): The general theory and its analytical extensions. Psychometrika 52:345–370. https://doi.org/10.1007/BF02294361/METRICS
Brooks M, Kristensen K, van Benthem K, Magnusson A, Berg C, Nielsen A, Skaug H, Maechler M, Bolker B (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.32614/RJ-2017-066
Bullock JM, Clarke RT (2000) Long distance seed dispersal by wind: measuring and modelling the tail of the curve. Oecologia 124:506–521. https://doi.org/10.1007/PL00008876/METRICS
Bullock JM, Shea K, Skarpaas O (2006) Measuring plant dispersal: An introduction to field methods and experimental design. Plant Ecol 186:217–234. https://doi.org/10.1007/s11258-006-9124-5
Bullock JM, Mallada González L, Tamme R, Götzenberger L, White SM, Pärtel M, Hooftman DAP (2017) A synthesis of empirical plant dispersal kernels. J Ecol 105:6–19. https://doi.org/10.1111/1365-2745.12666
Calviño-Cancela M, Rubido-Bará M (2013) Invasive potential of Eucalyptus globulus: seed dispersal, seedling recruitment and survival in habitats surrounding plantations. For Ecol Manage 305:129–137. https://doi.org/10.1016/J.FORECO.2013.05.037
Caplat P, Nathan R, Buckley YM (2012) Seed terminal velocity, wind turbulence, and demography drive the spread of an invasive tree in an analytical model. Ecology 93:368–377. https://doi.org/10.1890/11-0820.1
Carrillo-Gavilán MA, Vilà M (2010) Little evidence of invasion by alien conifers in Europe. Divers Distrib 16:203–213. https://doi.org/10.1111/j.1472-4642.2010.00648.x
Christy EJ, Mack RN (1984) Variation in Demography of Juvenile Tsuga Heterophylla Across the Substratum Mosaic. The Journal of Ecology 72:75. https://doi.org/10.2307/2260007
Clement EJ, Foster MC (1994) Alien plants of the British Isles. Botanical Society of the British Isles, London
Clobert J, Danchin E, Dhondt AA (2001) Dispersal. Oxford University Press, New York
Connell JH (1971) On the Role of Natural Enemies in Preventing Competitive Exclusion in Some Marine Animals and in Rain Forest Trees. In: Den Boer PJ, Gradwell GR (eds) Dynamics of Populations. Centre for Agricultural Publishing and Documentation, Wageningen
Cousens R, Dytham C, Law R (2008) Dispersal in plants : a population perspective. Oxford University Press, Oxford, p 221
Davies KW, Sheley RL (2007) A conceptual framework for preventing the spatial dispersal of invasive plants. Weed Sci 55:178–184. https://doi.org/10.1614/WS-06-161
Edwards P, Sprague R, Stahlmann-Brown P (2021) Removing invasive conifers - considerations, complexity and costs. Environ Res Commun 3:071004. https://doi.org/10.1088/2515-7620/AC13C7
Essl F, Moser D, Dullinger S, Mang T, Hulme PE (2010) Selection for commercial forestry determines global patterns of alien conifer invasions. Divers Distrib 16:911–921. https://doi.org/10.1111/J.1472-4642.2010.00705.X
Fanal A, Mahy G, Fayolle A, Monty A (2021) Arboreta reveal the invasive potential of several conifer species in the temperate forests of western Europe. NeoBiota 64:23–42. https://doi.org/10.3897/NEOBIOTA.64.56027
Fanal A, Mahy G, Monty A (2022) Can we foresee future maple invasions? A comparative study of performance-related traits and invasiveness of eight Acer species. Plant Ecol 223:1181–1192. https://doi.org/10.1007/S11258-022-01266-1/FIGURES/4
Fanal A, Porté A, Mahy G, Monty A (2023) Fast height growth is key to non-native conifers invasiveness in temperate forests. Biol Invasions 26:857–874. https://doi.org/10.1007/S10530-023-03214-0/TABLES/6
Foiles MW, Graham RT, Olson DFJ (1990) Abies grandis (Dougl. ex D. Don) Lindl. grand fir. In: Burns, Russell M.; Honkala, Barbara H., technical coordinators. Silvics of North America. Volume 1. Conifers. Agric. Handb. 654. U.S. Department of Agriculture, Forest Service, Washington, DC, 52–59.
Forestry Commission Scotland (2015) Managing invasive and non-native forestry species. Available from: http://scotland.forestry.gov.uk/images/corporate/pdf/managing-invasive-and-non-native-forestry-species.pdf (March 14, 2018).
Frischbier N, Damm C, Wohlwend M, Aas G, Wagner S (2017) Zur Naturverjüngung der Westlichen Hemlocktanne (Tsuga heterophylla (Rai) Sarg.) in Kleinbeständen in Thüringen. Forstarchiv 88:131–135. https://doi.org/10.4432/0300-4112-88-131
Galoux A (1951) Les Principales Essences Forestières de l’Amérique Septentrionale Tempérée. Leur introduction en Belgique. Available from: https://www.milieuinfo.be/dms/d/d/workspace/SpacesStore/3859d45b-758d-4ef7-a6ec-25aa16de12e0/170426.pdf.
Greene DF, Canham CD, Coates KD, Lepage PT (2004) An evaluation of alternative dispersal functions for trees. J Ecol 92:758–766
Grotkopp E, Rejmánek M, Rost TL (2002) Toward a causal explanation of plant invasiveness: seedling growth and life-history strategies of 29 pine (Pinus) species. Am Nat 159:396–419. https://doi.org/10.1086/338995
Hansen M, Potapov P, Moore R, Hancher M, Turubanova S, Tyukavina A, Thau D, Stehman S, Goetz S, Loveland T, Kommareddy A, Egorov A, Chini L, Justice C, Townshend J (2013) High-resolution global maps of 21st-century forest cover change. Science 342:850–853. https://doi.org/10.1126/science.1244693
Harmer R, Beauchamp K, Morgan G (2011) Natural regeneration in western hemlock plantations on ancient woodland sites. Available from: https://www.forestry.gov.uk/pdf/FCRN011.pdf/$FILE/FCRN011.pdf (March 14, 2018).
Hastings A, Cuddington K, Davies KF, Dugaw CJ, Elmendorf S, Freestone A, Harrison S, Holland M, Lambrinos J, Malvadkar U, Melbourne BA, Moore K, Taylor C, Thomson D (2005) The spatial spread of invasions: new developments in theory and evidence. Ecol Lett 8:91–101. https://doi.org/10.1111/J.1461-0248.2004.00687.X
Horn HS, Nathan R, Kaplan SR (2001) Long-distance dispersal of tree seeds by wind. Ecol Res 16:877–885. https://doi.org/10.1046/J.1440-1703.2001.00456.X
Howe F, Smallwood J (1982) Ecology of seed dispersal. Annu Rev Ecol Syst 13:201–228. https://doi.org/10.1146/ANNUREV.ES.13.110182.001221
Janzen DH (1970) Herbivores and the number of tree species in tropical forests. Am Nat 104:501–528. https://doi.org/10.1086/282687
Johnson JS, Cantrell RS, Cosner C, Hartig F, Hastings A, Rogers HS, Schupp EW, Shea K, Teller BJ, Yu X, Zurell D, Pufal G (2019) Rapid changes in seed dispersal traits may modify plant responses to global change. AoB PLANTS 11:1–20. https://doi.org/10.1093/AOBPLA/PLZ020
Kot M, Lewis MA, Van Den Driessche P (1996) Dispersal data and the spread of invading organisms. Ecology 77:2027–2042. https://doi.org/10.2307/2265698
Kowarik I (1995) Time lags in biological invasions with regard to the success and failure of alien species. In: Pyšek P, Prach K, Rejmánek M, Wade M (eds) Plant Invasions - General Aspects and Special Problems. SPB Academic Publishing, pp 15–38
Laroque CP, Lewis DH, Smith DJ (2001) Treeline dynamics on southern Vancouver island, British Columbia. Western Geography 10:43–63
Ligot G, Balandier P, Fayolle A, Lejeune P, Claessens H (2013) Height competition between Quercus petraea and Fagus sylvatica natural regeneration in mixed and uneven-aged stands. For Ecol Manage 304:391–398
Loebach CA, Anderson RC (2018) Measuring short distance dispersal of Alliaria petiolata and determining potential long distance dispersal mechanisms. PeerJ. https://doi.org/10.7717/PEERJ.4477/FIG-3
Martin PH, Canham CD (2010) Dispersal and recruitment limitation in native versus exotic tree species: Life-history strategies and Janzen-Connell effects. Oikos 119:807–824. https://doi.org/10.1111/J.1600-0706.2009.17941.X
Martin PH, Canham CD, Kobe RK (2010) Divergence from the growth-survival trade-off and extreme high growth rates drive patterns of exotic tree invasions in closed-canopy forests. J Ecol 98:778–789. https://doi.org/10.1111/j.1365-2745.2010.01666.x
Minore D (1990) Thuja plicata Donn ex D. Don western redcedar. In: Burns, Russell M.; Honkala, Barbara H., technical coordinators. Silvics of North America. Volume 1. Conifers. Agric. Handb. 654. U.S. Department of Agriculture, Forest Service, Washington, DC, 590–600.
Monty A, Mahy G (2010) Evolution of dispersal traits along an invasion route in the wind-dispersed Senecio inaequidens (Asteraceae). Oikos 119:1563–1570. https://doi.org/10.1111/J.1600-0706.2010.17769.X
Moravcová L, Pyšek P, Jarošík V, Pergl J (2015) Getting the right traits: reproductive and dispersal characteristics predict the invasiveness of herbaceous plant species. PLoS ONE. https://doi.org/10.1371/journal.pone.0123634
Nathan R, Safriel UN, Noy-Meir I, Schiller G (2000) Spatiotemporal variation in seed dispersal and recruitment near and far from Pinus halepensis trees. Ecology 81:2156–2169. https://doi.org/10.1890/0012-9658
Nathan R, Klein E, Robledo-Arnuncio JJ, Revilla E (2012) Dispersal kernels: review. In: Clobert J, Baguette M, Benton TG, Bullock JM (eds) Dispersal Ecology and Evolution. Oxford University Press, Oxford, pp 187–210
Norghauer JM, Nock CA, Grogan J (2011) The importance of tree size and fecundity for wind dispersal of big-leaf mahogany. PLoS ONE 6:e17488. https://doi.org/10.1371/JOURNAL.PONE.0017488
Nygaard PH, Øyen B-H (2017) Spread of the Introduced Sitka Spruce (Picea sitchensis) in Coastal Norway. Forests. https://doi.org/10.3390/f8010024
OEWB (2021) PanoraBois Wallonie 2021. Available from: www.oewb.be (May 12, 2023).
Owens JN, Molder M (1984) The reproductive cycles of western and mountain hemlock. Ministry of Forests, Information Services Branch, Victoria, BC.
Oyen B-H (2001) Vestamerikansk hemlokk: Gjokungen blant innforte bartraer i Vest-Norge? [Western Hemlock: an invasive introduced conifer in Western Norway.]. Blyttia 59:208–216
Petit S, Cordier S, Claessens H, Ponette Q, Vincke C, Marchal D, Weissen F (2017) Fichier écologique des essences. Forêt.Nature, UCLouvain-ELIe, ULiège-GxABT, SPWARNE-DNF. Available from: https://www.fichierecologique.be/#!/ (May 6, 2020)
Proença-Ferreira A, Borda-de-Água L, Porto M, Mira A, Moreira F, Pita R (2023) dispfit: An R package to estimate species dispersal kernels. Ecol Inform 75:102018. https://doi.org/10.1016/j.ecoinf.2023.102018
Puchałka R, Paź-Dyderska S, Jagodziński AM, Sádlo J, Vítková M, Klisz M, Koniakin S, Prokopuk Y, Netsvetov M, Nicolescu V-N, Zlatanov T, Mionskowski M, Dyderski MK (2023) Predicted range shifts of alien tree species in Europe. Agric for Meteorol 341:109650. https://doi.org/10.1016/J.AGRFORMET.2023.109650
Pyšek P (2016) What determines the invasiveness of tree species in central Europe? In: Krumm F, Vítková L (eds) Introduced tree species in European forests: opportunities and challenges. European Forest Institute, pp 68–77
R Core Team (2022) R: A Language and Environment for Statistical Computing. Available from: https://www.r-project.org.
Richardson DM, Rejmánek M (2004) Conifers as invasive aliens: a global survey and predictive framework. Divers Distrib 10:321–331. https://doi.org/10.1111/j.1366-9516.2004.00096.x
Richardson DM, Rejmánek M (2011) Trees and shrubs as invasive alien species - a global review. Divers Distrib 17:788–809. https://doi.org/10.1111/j.1472-4642.2011.00782.x
Richardson DM, Pysek P, Rejmanek M, Barbour MG, Panetta FD, West CJ (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107. https://doi.org/10.1046/j.1472-4642.2000.00083.x
Richardson DM, Rouget M, Rejmánek M (2004) Using Natural Experiments in the Study of Alien Tree Invasions: Opportunities and Limitations. In: Bartol SM, Gordon MS (eds) Experimental Approaches to Conservation Biology. Univerity of California Press Ltd., Los Angeles, California, pp 180–201. https://doi.org/10.1525/california/9780520240247.003.0012
Riou-Nivert P (2001) Les résineux: Tome I: Connaissance et reconnaissance. Institut pour le développement forestier, 256 pp.
Schopmeyer CS (1974) Thuja L. Arborvitae. In: Schopmeyer CS (Ed), Seeds of woody plants in the United States. Agriculture Handbook No. 450. U. S. Department of Agriculture, Forest Service, Washington D.C., 805–809.
Schrader B (1998) Structural development of late successional forests in the central Oregon Coast Range : abundance, dispersal, and growth of western hemlock (Tsuga heterophylla) regeneration. Oregon State University
Skarpaas O, Shea K (2007) Dispersal Patterns, Dispersal Mechanisms, and Invasion Wave Speeds for Invasive Thistles. 170: 421–430. https://doi.org/10.1086/519854
Tamme R, Götzenberger L, Zobel M, Bullock JM, Hooftman DAP, Kaasik A, Pärtel M (2014) Predicting species’ maximum dispersal distances from simple plant traits. Ecology 95:505–513. https://doi.org/10.1890/13-1000.1
Thurm EA, Hernandez L, Baltensweiler A, Ayan S, Rasztovits E, Bielak K, Zlatanov TM, Hladnik D, Balic B, Freudenschuss A, Büchsenmeister R, Falk W (2018) Alternative tree species under climate warming in managed European forests. For Ecol Manage 430:485–497. https://doi.org/10.1016/J.FORECO.2018.08.028
Turner D (1985) Successional relationships and a comparison of biological characteristics among six northwestern conifers. Bull Torrey Bot Club 112:421–428
van Kleunen M, Weber E, Fischer M (2010) A meta-analysis of trait differences between invasive and non-invasive plant species. Ecol Lett 13:235–245. https://doi.org/10.1111/J.1461-0248.2009.01418.X
Welch D, Carss DN, Gornall J, Manchester SJ, Marquiss M, Preston CD, Telfer MG, Arnold H, Holbrook J (2001) An audit of alien species in Scotland. Scottish Natural Heritage Review 139:1–225
Wickham H (2016) Springer-Verlag New York ggplot2: Elegant Graphics for Data Analysis. Available from: https://ggplot2.tidyverse.org.
Williamson RL (1976) Natural regeneration of western hemlock. Western hemlock management conference: Proceedings. University of Washington College of Forest Resources, Seattle, WA, pp 166–169
Wyse SV, Hulme PE (2021) Dispersal potential rather than risk assessment scores predict the spread rate of non-native pines across New Zealand. J Appl Ecol 58:1981–1992. https://doi.org/10.1111/1365-2664.13947
Acknowledgements
We are grateful to Louis Abandonné, Guillaume Baralle, Pauline Crasson, Adrien Delforge, Maïké Dellicour, Emilie Guilmin, Vladimir Joassin, Marie Patinet, Thomas Pollet, Lise Pouilloux and Eric Rathle for their help in the field. We also thank the “Département de la Nature et des Forêts” for providing information on stand locations and granting access to study sites.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This work was supported by the University of Liege. The authors have no relevant financial or non-financial interests to disclose. All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Aurore Fanal and Arnaud Monty. The first draft of the manuscript was written by Aurore Fanal and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The datasets generated during this study are available on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fanal, A., Mahy, G. & Monty, A. Recruitment curves of three non-native conifers in European temperate forests: implications for invasions. Biol Invasions (2024). https://doi.org/10.1007/s10530-024-03438-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10530-024-03438-8