Abstract
Hydropower dams are one of the largest sources of anthropogenic impacts on freshwater systems in the world. In addition to the modifications in environmental dynamics (i.e., fish migration barrier, water flow reduction, depth increase, flood regulation), the novel environments are favorable to the dissemination of non-native species in the basin. The lack of native species adapted to the changed environment creates occupations opportunity (i.e., available resources) for non-native species, which end up finding in these environments ideal areas to complete their life cycle. Thus, the aim of the work was identify which reservoir characteristics provide as well benefits for non-native fish species. Specifically, we tested the hypothesis that the spatial structure, reservoir productivity and morphology, and chronological characteristics are factors related to the composition and abundance of non-native fish species in reservoir. Using novel statistical techniques (Principal coordinates of neighbour matrices and Distance-based linear models), it was possible to identify the spatial patterns and to understand how the characteristics of the reservoirs influence the composition of the abundance of these species. Our results show that some reservoir characteristics provide benefits to non-native fish species, thus being localities within the hydrologic basins that can be considered as sources of non-native fish species propagules. In general, our results showed that larger and older reservoirs have a greater abundance of non-native fish species. Also, it was possible to identify spatial patterns, where in smaller scales neighboring reservoir tend to be more similar as to the composition and abundances of non-native fish species and this similarity can reach basin level.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Currently, there has been a great deal of discussion on the consequences of the introduction of non-native species in ecosystems (Vitule et al. 2009; Vilà et al. 2011; Gallardo et al. 2016). Impacts of introduced non-native species are context-dependent, ranging from direct competition with native species to homogenization and extinction of the biota (Wilcove et al. 1998; Ceradini and Chalfoun 2017). There are studies describing shifts in food webs of the fish fauna and changes in the structure of the native community, leading to ecosystem shifts (Olowo and Chapman 1999; Attayde et al. 2007). The introduction and subsequent success in the establishment of non-native species depend on a host of factors such as: distance from water bodies to urban centers (i.e., greater the proximity greater the propagule pressure; Spear et al. 2013; Dostálek et al. 2014); resident native species richness (i.e., the biotic resistance/acceptance; Elton 1958; Fridley et al. 2007); and system productivity (i.e., dynamic equilibrium model; Huston 1979, 2004). Therefore, understanding the mechanisms that lead to success in the establishment of non-native species in environments is a challenge for modern ecologists and many hypotheses have been developed (Catford et al. 2009).
Some studies show that anthropogenic-modified environments are more susceptible to invasion because they have dramatically altered habitats and food web structure, causing extensive environmental changes (Havel et al. 2005; Ortega et al. 2018). Some modifications are so fast that they do not provide enough time for evolutionary responses of native species (Byers 2002). In this context, in freshwater ecosystems, reservoirs are recent anthropogenic-modified environments in the landscape and are in constant biological modification (i.e., biological succession; Agostinho et al. 1999, 2008, 2015). Among the environmental modifications caused by the construction of dams are the alteration in flow regime, sediment retention along the reservoir and remodeling of aquatic communities (Agostinho et al. 1999; Miranda and Krogman 2015). Most native fish species cannot remain in these regions due to lack of pre-adaptation to lentic environments or fail to maintain viable populations over time, due to new system dynamics, for example, long distance migratory species such as Salminus brasiliensis and Pseudoplatystoma corruscans (Agostinho et al. 2015). Thus, over time, richness and abundance of native species decrease (Bailly et al. 2016; Ortega et al. 2018), which may favor the appearance of “resource gaps” for non-native species (Elton 1958; Davis et al. 2000). In addition to the aging of the reservoirs, other variables such as reservoir area and productivity can influence the invasion process, because they are related to the amount of resource available in the environment. Larger and more productive reservoirs tend to have greater non-native fish species richness because they have more environmental heterogeneity (e.g., different amount of habitat) and trophic resources availability, respectively (Huston 1979, 2004; Melbourne et al. 2007).
Once established, populations of non-native species represent a constant propagules source for upstream and downstream regions of reservoirs. Because dams are built in regions with high hydropower potential (i.e., to supply the energy demand, water supply and other purposes, these buildings are designed to capture as much water as possible), associated reservoirs have high connectivity with the river networks where they are located, facilitating dispersion of these species (i.e., the configuration of the space has great influence on the dispersion of non-native species; Havel et al. 2005). When reservoirs are in a cascade, this situation can be aggravated because non-native species find favorable regions for their establishment throughout the basin. Thus, the position of the reservoir in space (i.e., distance and/or insertion in the same basin), in relation to the reservoirs that already have established non-native species, influences the probability of the reservoir to suffer with propagule pressure of these species. Neighboring reservoirs that are inserted in the same basin are more likely to have the same composition (i.e., abundance and richness of species) of non-native species than those further away or that are inserted in different basins.
In this context, this paper aims to specify which reservoir characteristics provide benefits for invasive species. Specifically, we tested the hypothesis that the spatial structure, reservoir productivity morphology and chronological characteristics are factors related to the composition of non-native species in reservoir. We expect that the position of the reservoir in the basin (spatial structure) influence non-native species composition and abundance, due to fish dispersion and introduction by humans. Such influence may be restricted to smaller scales (i.e., neighboring reservoirs; close reservoirs with similar non-native species composition and abundance); or to reach larger scales (i.e., reservoirs inserted into the same basin, with similar non-native species composition and abundance). It is also expected that, the chronological age of reservoir has a positive influence in the abundance of non-native species, once this variable provides insights of the time of propagule-pressure. The same is expected for the reservoir’s morphology and productivity, as they are associated with the amount of resource available.
Materials and methods
Study area
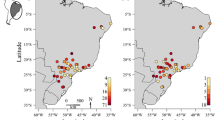
We studied 29 reservoirs located in rivers of the Paraná State and neighbouring states (Fig. 1), south Brazil. Seven reservoirs are on the Paranapanema River; two on the Tibagi River, a tributary of the Paranapanema; two on the Ivaí River; two on the Piquiri River; thirteen on the Iguaçu River; and four in the Litorânea basin (Online Resource B). Therefore, out of the 29 studied reservoirs, 25 belonged to the Paraná River basin (Paranapanema, Tibagi, Ivaí, Piquiri and Iguaçu rivers), and four to the Atlantic basin. However, the Iguaçu basin is separated from the Paraná by an insurmountable barrier for fish, the Iguaçu Falls.
Biological data
Fish assemblages were sampled in the lacustrine region of the reservoirs, in different depth (littoral, surface—pelagic and bottom—bathypelagic) using gill-nets of different mesh sizes (2.4–14.0 cm between opposing knots), exposed for 24 h, the fish were collected in the morning, afternoon and night. The collection were carried out in 2001 in the drought and rain period, which comprise the months of July and November, respectively. The data on numerical abundance of each species captured by sample were indexed by the catch per unit effort (CPUE; number of individuals in 1000 m2 of gillnet during 24 h). Taxonomic identification was based on Reis et al. (2003), except for families Clariidae and Ictaluridae, for which we used Burges (1989), Centrarchidae (Singler and Singler 1987) and Cyprinidae (Cavender and Coburn 1992).
Environmental data
The environmental variables measured along the fish samplings were used as proxy for productivity, such as: turbidity (NTU), total suspended material (mg/L; Teixeira et al. 1965), total phosphorus (µg/L; APHA 2005), orthophosphate (µg/L; APHA 2005), total nitrogen (µg/L; Mackereth et al. 1978), chlorophyll-a (µg/L; Nusch 1980), nitrate (µg/L; Mackereth et al. 1978), dissolved organic carbon (µg/L; Shimazdu TOC-5000A Total Organic Carbon (TOC) Analyzer), phosphate (µg/L; APHA 2005), total dissolved phosphorus (µg/L; APHA 2005), and biovolume of phytoplankton (mm3/L). The morphological characteristics (area and depth) of the reservoirs were taken from Gubiani et al. (2011). The reservoirs’ age was obtained by the difference between the year of sampling and the year of the formation of the reservoir. Although recognized as important, some variables such as retention time, discharge, variation in water level, were not used because they were not available for several reservoirs.
Spatial data
For the construction of the spatial data matrix we used the hydrological distance between the reservoirs. The hydrologic distance is calculated on a shapefile that represents the hydrographic network, using as starting and finishing points the geographical coordinates of each reservoir (i.e., latitude and longitude). The calculation of the distances is performed with the Dijkstra algorithm, which measures the smallest distances between two points (Dijkstra 1959; Loro et al 2015). We performed the calculation with the QNEAT3 complement (Qgis Network Analysis Toolbox; Raffler 2018), implemented in Qgis 3.0 (QGIS Development Team 2018).
We used PCNM (Principal Coordinates of Neighbour Matrices) to summarize the spatial structure. This method allows ecologists to model spatial structure at multiple spatial scales and to incorporate this representation in statistical analysis (Borcard et al. 2011). The spatial structure, derived from the the hydrological distances among reservoirs were summarized in a resemblance matrix (Euclidean distance) and this matrix was truncate to retain only the distances among close neighbours. Posteriorly, a PCoA (Principal Coordinates Analysis) of the truncated distance matrix was conducted to summarize the spatial structure in PCNMs (axes generated in the PCoA). The eigenvectors were then used as spatial explanatory variables in a model (Online Resource A). The scores of the first PCNM represent the greatest scale in the sample sites, while the last represents the smaller scale (i.e., the PCNM produces a spectral decomposition of space and can model spatial structure at all the spatial scale that can be perceived by the data set; Borcard et al. 2004). This procedure was performed in program R, using the pcnm function of the vegan package (Online Resource A).
Data analysis
In order to summarize the productivity proxy variables in a single variable, we performed a principal component analysis (PCA) of all the productivity proxies (variables) to use the scores of the first and second axes (the ones that explain most of the variation) as latent variables. We decided to use latent variables because it minimizes the problems caused by collinearity in models (Dormann et al. 2013). Thus, we summarized all productivity variables in two axes (the scores of the first two PCA axes).
Prior to analysis, we transformed the non-native fish assemblage data to express species abundance as square-root transformed proportionate abundances in each sampling site. This transformation reduces the weight of the most abundant species in the analysis. We also transformed the variable area by taking its natural logarithm. Due to the great number of environmental variables and possible correlations among them, we used the variation inflation factor (VIF) between each possible pair of variables to exclude one of those with VIF > 10 (Neter et al. 1996). When VIFs exceeding 10 there is a signal of serious multicollinearity, requiring correction.
In order to choose the most appropriate PCNM scores to include in the general model, twenty four PCNM (and their scores) were generated and summarized the spatial structure of our sampling sites (Resource Online D). After the generation of the scores of the axes, a matrix was created containing the twenty-four axes of PCNMs and the others environmental variables as area, age, PCA1, PCA2 and sampling month.
To evaluate the influence of environmental and spatial variables on the variability of composition of all non-native fishes and to select the best explanatory model, we applied a DISTLM (Distance-Based Linear Model) using Akaike’s information criterion (AIC) (Anderson et al. 2008). The forward selection was performed to select only the PCNMs and environmental variables that had significant influences on the structure of the composition of the non-native fish species (Blanchet et al. 2008). Distance-based redundancy analysis (dbRDA) was used to examine the influence of predictors on the spatial distribution of samples (Anderson et al. 2008). In all analysis, 9999 permutations were made. The variable sampling month (July and November) was included in the model as a dummy variable to quantify the temporal effects in the samplings. Finally, to evaluate which species were most related to the patterns observed in the model, a Spearman correlation was performed between species CPUEs (original composition of non-native fish data matrix) and the first two axes of dbRDA.
Results
We recorded 17 non-native species in the reservoirs of which Oreochromis niloticus and Geophagus brasiliensis were found in all the basins (Online Resource F). Of the all the basins only Atlantic and Piquiri basins did not present exclusive species. The exclusive species of Iguaçu basins were: Characidium sp., Clarias gariepinus, Ctenopharyngodon idella, Leporinus macrocephalus, Odontesthes bonariensis; in the Paranapanema basin were found three exclusive species: Astronotus ocellatus, Odontostilbe sp., Plagioscion squamosissimus; in the Tibagi and Ivai only one specie was found: Colossoma macropomum and Hoplias lacerdae, respectively.
The first PCA axis (eigenvalue = 1.52) was positively correlated with total phosphorus (r = 0.75) and total nitrogen (r = 0.75), and represented approximately 50.7% of the total variability, while the second PCA axis (eigenvalue = 0.91) was positively correlated with chlorophyll (r = 0.93), and represented approximately 30.4% of total variability (Online Resource C). Thus, when the PCA 1 and 2 scores increased, the values of total phosphorus, total nitrogen and chlorophyll also increase (Fig. 2). Therefore, we consider that higher values of the scores in PCA1 and PCA2 indicate higher productivity.
Result of the principal component analysis with the proxy productivity variables (chlorophyll-a, total phosphorus and total nitrogen concentrations). Each point on the graph represents a reservoir at a given collection. Black triangles: Tibagi; light grey triangles: Paranapanema; black squares: Litorânea; dark grey quadrilaterals: Iguaçu; light gray crosses: Ivaí; dark grey circles: Piquiri. The arrow represents the direction of the effect (positive correlation) of the summarized environmental variables on an axis
The linear model selected by DISTLM included PCNM1, PCNM2, PCNM4, PCNM7, PCNM8, PCNM9, PCNM11, PCNM12, PCNM14, PCNM15, PCNM22, PCNM24, area, collection and age as determinants of non-native species composition and abundance in reservoir. Although all variables in the model presented significant relationships, the percentage of variation explained by each of them was different (Table 1). The variable that presented the highest correlation was PCNM1, followed by age, PCNM6, sampling month, PCNM2, PCNM12, PCNM8, PCNM22, Area, PCNM15, PCNM4, PCNM24, PCNM11, PCNM9, PCNM7 and PCNM14. The graphic representation of DISTLM results was represented by a dbRDA. The percentage of total variation in the first axes was 33.15% and the second axes was 11.63% of total variation.
The significant PCNM1, age, PCNM6, sampling month, PCNM2, PCNM12, PCNM8, PCNM22, Area, PCNM15, PCNM4, PCNM24, PCNM11, PCNM9, PCNM7 and PCNM14 effects are visible in the ordination biplot (Fig. 3). The first axis was positively correlated with area (reservoirs of the Paranapanema River have larger areas), PCNM1 and PCNM7 and negatively correlated with age and PCNM2. The second axis was positively correlated with PCNM1, PCNM6 and PCNM24 and negatively correlated with sampling month, PCNM12 and PCNM4 (Table 2). Some PCNM axes such as PCNM8, PCNM22, PCNM15, PCNM11, PCNM9 and PCNM14 despite having significant relationships in the DISTL model, showed very low relationship values with the first two axes (> 0.2). The species that showed significant positive correlation with the first axis of the dbRDA was P. squamosissimus, while C. carpio, G. brasiliensis and C. rendalli presented significant negative correlations (Table 3). While G. brasiliensis and C. rendalli were positively correlated with second axis and O. bonariensis was negatively correlated with second axis (Table 3).
Result of the distance-based redundancy analysis (dbRDA) with the predictor variables (age, area, sample month and PCNMs) showing the greatest importance for the linear model. Black triangles: Tibagi; light gray triangle: Paranapanema; black squares: Litorânea; dark grey quadrilaterals: Iguaçu; light gray crosses: Ivaí; dark gray circle: Piquiri
Discussion
Our results indicate that reservoirs are suitable environments for the establishment of non-native species, and some environmental variables are closely related for this process. Among the species found in the reservoirs are Cyprinus carpio and Micropterus salmoides, which fall within the eight most widely-introduced freshwater fishes in Europe (García-Berthou et al. 2005). Among the variables that influence the establishment of species in the reservoirs are the spatial arrangement in fine and broad scales, the area of the reservoir and age.
The space was the variable that together (i.e., broad and fine scale) had the greatest influence on the composition of non-native species in the reservoirs. This influence was detected in most of the study reservoirs (e.g., PCNM1, PCNM2 and PCNM7 were responsible for the separation of the reservoirs in the first axis and the PCNM1, PCNM6, PCNM4, PCNM12 and PCNM24 were responsible for the separation of the reservoirs in the second axis). In the first axis we can observe that the reservoirs of Paranapanema basin were separated from the others and their composition and abundance of the non-native species had a great influence on the broad scale. This result can be explained by the fact that the species P. squamosissimus, among the studied basins, is restricted to the Paranapanema basin and it was present and abundant in all reservoirs of this basin (Online Resource E). It was possible to observe an influence of fine and broad scales on our data, since the model selected the first and last axes of the PCNM. These results strengthen the idea that reservoirs act as step-stones for biological invasions (Havel et al. 2005). At fine scales (i.e., represented by PCNM6 PCNM12 and PCNM24), the reservoirs appear to act as a source of propagules for neighboring reservoirs, mainly for C. rendalli, G. brasiliensis, C. carpio and O. bonariensis. This propagule pressure can occur “naturally” in the direction downstream a reservoir through the active and passive dispersion of individuals, eggs and larvae, or in an “artificial” way through human introduction. The “artificial” pressure can occur in the reservoir-upstream direction as well. Since the population of the non-native species establishes in the reservoir, the probability of invasion in neighboring reservoirs increases. In contrast, at broad scale, the PCNM1 and 2 (i.e., the variables responsible for the greatest proportion of the variation in the composition of non-native species), separated the Paranapanema sub-basin from the others, while Paranapanema was in the positive side of dbRDA1, Iguaçu, Litorânea, Tibagi, Piquiri and Ivai were in the negative side. The selection of the variable PCNM1 and 2 by the model shows that the dispersion of the non-native species can reach the basin level, especially for P. squamosissimus from Paranapanema basin and G. brasiliensis. C. rendalli, C. carpio and O. bonariensis for the other basins, especially for Iguaçu and Litorânea basin where their abundance shows the highest values (Online Resource E). Still, this result captures the differences in history of non-native species introduction in the basins. The history of introduction is spatially dependent, and it related to the incentive of public policies (very common in Brazil; see Agostinho et al. 2007, 2010) for the introduction of non-native species along the country.
As expected, the area of the reservoirs presented positive relationships with the composition and abundance of non-native species, particularly the species P. squamosissimus had the greatest influence on the results. This relationship is also found for native fish species in these reservoir (Bailly et al. 2016; Ortega et al. 2018) and is related to the size and diversity of available habitats (Drakare et al. 2006). Therefore, larger reservoirs tend to support greater abundance of non-native species (Gido and Brown 1999). Since this relationship is found for both native and non-native species, it is an indication that either there is biotic acceptance in these environments or that interspecific relationships are so weak that the introduction of species in these environments does not interfere with the composition of pre-existing species. Nevertheless, more work needs to be done to test these hypotheses in these environments.
Another variable that had an influence on the non-native species composition and abundance was reservoir age. There are some studies that reveal that aging is a good predictor of fish assemblages (Bailly et al. 2016; Santos et al. 2017; Ortega et al. 2018). However, age seems to affect composition of native and non-native species in different ways. While, the age had a negative influence on native species (i.e., over time there is a reduction in the richness of native species; Agostinho et al. 2015; Ortega et al. 2018), our results suggest that the same is not true for the main non-native fish species (e.g., C. rendalli, C. carpio and G. brasiliensis). Older reservoirs showed greater abundance of these species. These results may have two possible explanations: older reservoirs suffered more propagule pressure and/or the non-native species had a longer period to increase their populations.
Although our results identify spatial patterns in the composition of non-native fish species in reservoirs, the study presents some limitations. It is important to emphasize that the history of species introduction (i.e., the period between the introduction and collection of data can not be controlled) and the distance of reservoir to urban centers affects our results, because its influence is spatially dependent (e.g., in the 1970s there was great incentive to stocking P. squamosissimus in reservoirs of the Paranapanema river) and affects the composition of non-native species in the reservoirs. However, since we used abundance data in the analyses, it was possible to identify some trends regarding the success of establishment of these species. Secondly, the status of some of these species in the environment was not considered, for instance if indeed they can be considered invasive species (i.e., non-native species that spread beyond the introduction site and become abundant; Rejmánek et al. 2002). This requires long-term community monitoring (i.e., temporal analysis). However, some of these species are recognized as one of the most widely introduced aquatic species in the world, as for example: C. carpio, C. idella and O. niloticus (García-Berthou et al. 2007). Another limitation is that the trophic state of reservoirs is very dependent on the water retention time and can vary greatly over the year. Although our results agree with the literature, we believe that more specific studies are needed to test the hypothesis that invasive species became dominant in disturbed and productive environments (Dynamic equilibrium model; Huston 1979, 2004).
Thus, our results show that some reservoir characteristics provide benefits to non-native species, thus being localities within the hydrologic basins that can be considered as sources of non-native species propagules. In general, our findings showed that larger and older reservoirs have a greater abundance of non-native species. Also, it was possible to identify spatial patterns, where in smaller scales neighboring reservoir tend to be more similar as to the composition and abundance of non-native species and this similarity can reach basin level. Since fish invasive species are characterized by great dispersal ability and tolerance to a wide range of environmental conditions (Moyle 1986; Sakai et al. 2001), reservoirs can act as passports to increase the amplitude of invaded area for these species. Therefore, it is worth emphasizing possible negative synergistic effects of the presence of reservoirs in the basin.
References
Agostinho AA, Miranda LE, Bini LM, Gomes LC, Thomaz SM, Suzuki HI (1999) Patterns of colonization in neotropical reservoirs, and prognoses on aging. In: Tundisi JG, Straskraba M (eds) Theoretical reservoir ecology and its applications. Backhuys Publishers, Rio de Janeiro, pp 227–265
Agostinho AA, Gomes LC, Pelicice FM (2007) Ecologia e Manejo de Recursos Pesqueiros em Reservatórios do Brasil. Eduem, Maringá
Agostinho AA, Pelicice F, Gomes LC (2008) Dams and the fish fauna of the Neotropical region: Impacts and management related to diversity and fisheries. Braz J Biol 68:1119–1132
Agostinho AA, Pelicice FM, Gomes LC, Júlio HF Jr (2010) Reservoir fish stocking: when one plus one may be less than two. Nat Conserv 8:103–111
Agostinho AA, Gomes LC, dos Santos NCL, Ortega JCG, Pelicice F (2015) Fish assemblages in Neotropical resevoirs: colonization patterns, impacts and management. Fish Res 173:26–36
Anderson MJ, Gorley RN, Clarke KR (2008) Permanova + for primer: guide to software and statistical methods. Primer-E, Plymouth
APHA, AWWA, WEF (2005) Standard methods for the examination of water and wasterwater. American Public Health Association, Washington
Attayde JL, Okun N, Brasil J, Menezes R, Mesquita P (2007) Impactos da introdução da tilápia do Nilo, Oreochromis niloticus, sobre a estrutura trófica dos ecossistemas aquáticos do Bioma Caatinga. Oecol Bras 11:450–461
Bailly D, Cassemiro FA, Winemiller KO, Diniz-Filho JAF, Agostinho AA (2016) Diversity gradients of Neotropical freshwater fish: evidence of multiple underlying factors in human-modified systems. J Biogeogr 43:1679–1689
Blanchet FG, Legendre P, Borcard D (2008) Forward selection of explanatory variables. Ecology 89:2623–2632
Borcard D, Legendre P, Avois-Jacquet C, Tuomisto H (2004) Dissecting the spatial structure of ecological data at multiple scale. Ecology 85:1826–1832
Borcard D, Gillet F, Legendre P (2011) Spatial analysis of ecological data. In: Borcard D, Gillet F, Legendre P (eds) Numerical ecology with R. Springer, New York, pp 227–292
Burges WE (1989) An atlas of freshwater and marine catfishes: a preliminar survey of the Siluriformes. T.F.H. Publications, Neptune City
Byers JE (2002) Impact of non-indigenous species on natives enhanced by anthropogenic alteration of selection regimes. Oikos 97:449–458
Catford JA, Jansson R, Nilsson C (2009) Reducing redundancy in invasion ecology by integrating hypotheses into a single theoretical framework. Divers Distrib 15:22–40
Cavender TM, Coburn MM (1992) Phylogenetic relationships of North Americn cyprinidae. In: Mayder RL (ed) Systematics, historical ecology and North American freshwater fishes. Stanford University Press, Stanford
Ceradini JP, Chalfoun AD (2017) Species’ traits help predict small mammal responses to habitat homogenization by an invasive grass. Ecol Appl 27:1451–1465
Davis MA, Grime JP, Thompson K (2000) Fluctuating resources in plant communities: a general theory of invisibility. J Ecol 88:528–534
Dijkstra EW (1959) A note on two problems in connexion with graphs. Numer Math 1:269–271
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, McClean C, Osborne PE, Reineking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46
Dostálek J, Frantík T, Silarová V (2014) Changes in the distribution of alien plants along roadsides in relation to adjacent land use over the course of 40 years. Plant Biosyst 150:442–448
Drakare S, Lennon JJ, Hillebrand H (2006) The imprint of the geographical, evolutionary and ecological context onespecies–area relationships. Ecol Lett 9:215–227
Elton CS (1958) The ecology of invasions by animals and plants. Methuen, London
Fridley JD, Stachowicz JJ, Naeem S et al (2007) The invasion paradox: reconciling pattern and process in species invasions. Ecology 88:3–17
Gallardo B, Clavero M, Sánchez MI, Vilà M (2016) Global ecological impacts of invasive species in aquatic ecosystems. Glob Chang Biol 22:151–163
García-Berthou E, Alcaraz C, Pou-Rovira Q, Zamora L, Coenders G, Feo C (2005) Introduction pathways and establishment rates of invasive aquatic species in Europe. Can J Fish Aquat Sci 62:453–463
García-Berthou E, Boix D, Clavero M (2007) Non-indigenous animal species naturalized in Iberian inland waters. In: Gherardi F (ed) Biological invaders in inland waters: profiles, distribution and threats. Springer, Dordrecht, pp 123–140
Gido KB, Brown JH (1999) Invasion of the North American drainages by alien fish species. Freshw Biol 42:387–399
Gubiani EA, Angelini R, Vieira LCG, Gomes LC, Agostinho AA (2011) Trophic models in Neotropical reservoirs: testing hypotheses on the relationship between aging and maturity. Ecol Model 222:3838–3848
Havel JE, Lee CE, Zanden MJV (2005) Do reservoir facilitate invasion into landscapes? Bioscience 55:518–525
Huston MA (1979) A general hypothesis of species diversity. Am Nat 113:81–101
Huston MA (2004) Management strategies for plant invasions: manipulating productivity, disturbance, and competition. Divers Distrib 10:167–178
Loro M, Ortega E, Arce RM, Geneletti D (2015) Ecological connectivity analysis to reduce the barrier effect of roads. An innovative graph-theory approach to define wildlife corridors with multiple paths and without bottlenecks. Landsc Urban Plan 139:149–162
Mackereth FJH, Heron J, Talling JF (1978) Water analysis: some revised methods for limnologists. Scient Public, London
Melbourne BA, Cornell HV, Davies KF, Dugaw CJ, Elmendorf S, Freestone AL, Hall RJ, Harrison S, Hastings A, Holland M, Holyoak M, Lambrinos J, Moore K, Yokomizo H (2007) Invasion in a heterogeneous world: resistance, coexistence or hostile takeover? Ecol Lett 10:77–94
Miranda LE, Krogman RM (2015) Functional age as an indicator of reservoir senescence. Fisheries 40:170–176
Moyle PB (1986) Fish introductions into North America: patterns and ecological impact. In: Mooney HA, Drake JA (eds) Ecology of biological invasions of North America and Hawaii. Springer, New York, pp 27–43
Neter J, Kutner MH, Nachtsheim CJ, Wasserman W (1996) Applied linear statistical models. Irwin, Chicago
Nusch EA (1980) Comparision of different methods for chlorophyll and phaepigment determination. Arch Hydrobiol Beih Ergebn Limnol 14:14–36
Olowo JP, Chapman LJ (1999) Trophic shifts in predatory catfishes following the introduction of Nile perch into Lake Victoria. Afr J Ecol 37:457–470
Ortega JC, Agostinho AA, Santos NC, Agostinho KD, Oda FH, Severi W, Bini LM (2018) Similarities in correlates of native and introduced fish species richness distribution in Brazilian reservoirs. Hydrobiologia 817:167–177
Reis RE, Kullander SO, Ferraris CJ (2003) Check list of the freshwater fishes of South and Central America. EDIPUCRS, Porto Alegre
Rejmánek M, Richardson DM, Barbour MG, Crawley MJ, Hrusa GF, Moyle PB, Randall JM, Simberloff D, Williamson M (2002) Biological invasions: politics and the discontinuity of ecological terminology. Bull Ecol Soc Am 83:131–133
Sakai AK, Allendorf FW, Holt JS, Lodge DM et al (2001) The population biology of invasive species. Annu Rev Ecol Syst 32:305–332
Santos NCL, Santana HS, Ortega JCG, Dias RM, Stegmann LF, Araújo IMS, Severi W, Bini LM, Gomes LC, Agostinho AA (2017) Environmental filters predict the trait composition of fish communities in reservoir cascades. Hydrobiologia 802:245–253
Singler WE, Singler JW (1987) Fishes of the great basin: a natural history. University of Nevada Press, Reno
Spear D, Foxcroft LC, Bezuidenhout H, McGeoch MA (2013) Human population density explains alien species richness in protected areas. Biol Conserv 159:137–147
Teixeira C, Tundisi JG, Kutner MB (1965) Plankton studies in a mangrove environment II: the standing stock and some ecological factors. Boletim do Instituto Oceanográfico 24:23–41
Vilà M, Espinar JL, Hejda M, Hulme PE, Jarošík V, Maron JL et al (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14:702–708
Vitule JRS, Freire CA, Simberloff D (2009) Introduction of non-native freshwater fish can certainly be bad. Fish Fish 10:98–108
Wilcove DS, Rothstein D, Dubow J, Phillips A, Losos E (1998) Quantifying threats to imperiled species in the United States. Bioscience 48:607–615
Acknowledgements
The authors thank Fagner M. de Oliveira Junior for valuable comments on the analysis and ecological interpretation. We express our appreciation to the PRONEX-MCT/CNPq for financial support and to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and to Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the fellowships to graduate students. AAA and LCG received scientific productivity fellowships from CNPq.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Muniz, C.M., Ganassin, M.J.M., Agostinho, A.A. et al. Spatial and environmental factors predict the composition of non-native fish assemblages in Neotropical reservoirs. Biol Invasions 22, 499–508 (2020). https://doi.org/10.1007/s10530-019-02105-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-019-02105-7