Abstract
Despite early reports of its presence, no recent data exist on the distribution of the American bullfrog in Europe, the causes of introduction, or the trends of populations. We monitored the European situation at two spatial scales. In SW France, we performed call surveys over 2,500 wetlands. We found bullfrogs over about 2,000 km2, apparently the European area in which the strongest expansion of bullfrogs is taking place. In addition, we used questionnaires to investigate the situation at the continental scale. At least 25 independent introductions occurred in Europe; eradication attempts were successful three times, and bullfrog populations are present in five countries. Education programs and monitoring are necessary to reduce the rate of introduction and to start management action as soon as possible.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The bullfrog Rana catesbeiana is native to eastern North America, but has been introduced throughout the world during the past two centuries (Lever 2003). The bullfrog is considered to be one of the most harmful invasive species, since it negatively affects native amphibians through competition and predation (Lowe et al. 2000; Kats and Ferrer 2003; Beebee and Griffiths 2005). Moreover, both native and introduced bullfrog populations are frequently infected by the fungus Batrachochytrium dendrobatidis (Hanselmann et al. 2004; Garner et al. 2006). This fungus is the agent of chytridiomycosis, an emergent amphibian disease that causes extinction of amphibian populations at a global scale (Berger et al. 1998; Bosch et al. 2001; Lips et al. 2006; Pounds et al. 2006). Since bullfrogs can be infected by B. dendrobatidis without developing chytridiomycosis, the introduction of bullfrogs can be an important vector of this disease (Hanselmann et al. 2004; Garner et al. 2006). Knowledge of the pattern of bullfrog invasion is, therefore, extremely important in planning conservation strategies aiming to control the invasion.
Bullfrogs were introduced into Europe several times during the 20th century. Up to the 1990s, acclimatised bullfrog populations were recorded in Italy, Holland, and France (Albertini and Lanza 1987; Lanza and Ferri 1997). However, since the end of the 20th century, several new introductions have occurred and the European situation has dramatically changed. Despite attempts to clarify the distribution of introduced bullfrogs (Lever 2003), no comprehensive reports on the situation in Europe exist. We, therefore, investigated the distribution of the bullfrog at two spatial scales. First, we performed intensive field monitoring to evaluate the distribution of R. catesbeiana in western France, an area in which bullfrog expansion in Europe is progressing rapidly. In addition, we gathered information, including data from both successful and failed introductions, from herpetologists throughout Europe to obtain a picture of the present situation at the continental scale.
Methods
Distribution in SW France
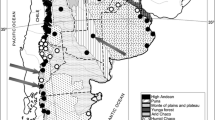
We performed field sampling in an area of southwest France (35,500 km2) including six departments: Gironde, Dordogne, Landes, Lot et Garonne, Charente, and Charente Maritime (Fig. 1). The study site was selected on the basis of anecdotal records and preliminary surveys (Detaint and Coïc 2001), and includes coastline, lowland, and hills, with a high density of wetlands. The study area was divided into a 10 × 10 km grid (total: 355 squares) and we identified water bodies for each square on the basis of 1:25000 IGN maps. Since, during late spring–summer male bullfrogs produce easily identifiable calls, we used calling surveys to evaluate the distribution of the species. We performed surveys in seven randomly selected wetlands per square, from May to August of 2003, 2004, and 2005, and from May to June of 2006. In a few cases, less than seven wetlands were present and we monitored all wetlands. We performed surveys at least 15 min after sunset, under suitable weather conditions (no heavy rain or wind), and each survey was repeated 15 days later. We performed further additional surveys in squares where we detected the presence of R. catesbeiana (total: 2,505 wetlands surveyed). The reproduction was determined on the basis of presence of tadpoles or egg masses.
Distribution at the continental scale
Monitoring of bullfrogs at the continental scale is not feasible; therefore, we used the community of European herpetologists as a source of primary information about the distribution of bullfrogs. First, we contacted the atlas committees and members of the all the European herpetological societies, and a large number of field herpetologists whom we knew to be interested in bullfrog-related problems. Moreover, during the winter of 2005–2006 we posted e-mail messages asking feedback from anyone knowing the presence of bullfrogs in Europe. These messages were posted on two major herpetological mailing lists: Herpnet (479 subscribers, Europe-wide coverage) and erpetologia (345 subscribers, mainly Italian herpetologists). We particularly focused on the Italian situation since Italy was the first country where successful introductions of R. catesbeiana occurred, and it is the country suffering the largest number of introductions. Furthermore, in several localities of northern Italy where the species is historically known to be present (Albertini and Lanza 1987), we performed additional point counts and dip-netting to confirm the bullfrog presence.
We subsequently sent questionnaires to all people reporting current or past presence of bullfrogs. The questions included the date and the causes of introduction, the origin of individuals, the current trend and status of the population, and any available information about eradication protocols. For areas of early introduction and extinction, we also obtained data from old literature and unpublished reports (Albertini 1970; Veenvliet and Veenvliet 2002). We considered as introduction events all cases in which at least one reproduction was recorded, or where a very large number of adults were observed. We did not consider as introduction events those in which only isolated adults were observed.
Invasion success was classified as follows: 0, extinct population; 1, established, but apparently not invasive population (population did not expand from the locality of introduction); 2, invasive population (expansion from the locality of introduction) (Kolar and Lodge 2001). We did not quantify invasion success for populations successfully eradicated after introduction.
Since it has been frequently assumed that species-rich communities better resist invasion than species-poor communities (Shea and Chesson 2002), we calculated the Spearman’s correlation between invasion success and the number of amphibian species present in the area, measured as the number of species recorded in the same grid square in the European herpetological atlas (Gasc et al. 1997).
Results and discussion
Distribution in southwest France
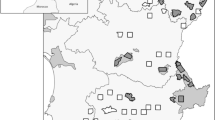
The surveys detected the presence of R. catesbeiana in 123 out the 2,505 wetlands (Fig. 1b); we observed successful reproduction in 30 wetlands. Breeding populations of R. catesbeiana are present in two geographic areas: Gironde (west of the study area; at least 10 breeding wetlands, presence of the species in an area of about 1,800 km2) and Dordogne (at least 20 breeding wetlands, presence of the species in an area of about 200 km2). It should be noted that we did not perform surveys in all the wetlands of the study area; therefore, the real number of water bodies where bullfrogs are present or breeding is probably larger than these figures.
The bullfrog distribution in the study area was not continuous. For example, the populations in the northwest corner of the study area (Dordogne) are about 90 km from the other populations (Fig. 1b), a distance far exceeding the possibility of dispersal of this species estimated on the basis of mark-recapture and microsatellite data (Austin et al. 2004; Smith and Green 2005). This suggests that secondary translocations into private wetlands as a pet or source of food (Albertini 1970; Yiming et al. 2006) can substantially increase the rate of expansion of this invasive species. Interviews of local people confirmed that, within the study area, translocations were performed at least in one case.
Our results show that R. catesbeiana is present in a large area of southwest France, and that this is the second largest area in Europe where R. catesbeiana is present. Moreover, in this area, the bullfrog seems to be particularly invasive compared to other European areas. For instance, in northern Italy, R. catesbeiana is present over some 5,000 km2, but the present situation is very similar to that recorded during the 1980s (Albertini and Lanza 1987; Ferri 2006). Moreover, Italian populations were introduced during the 1930s, and were the first acclimatised in Europe, while French populations were introduced 30 years later (Albertini and Lanza 1987; Detaint and Coïc 2001). This first monitoring of the situation in southwest France suggests an alarming expansion of the species compared to other European areas. The increasing abundance of bullfrogs in this area also increases the risk that individuals will be captured and translocated elsewhere. Although breeding populations exist in only three French areas (see below, Fig. 1a), isolated individuals have been observed in several further localities, suggesting that translocations have been frequent.
To hinder this expansion, a large-scale eradication plan is currently ongoing in southwest France, including direct capture and trapping of both adults and tadpoles, and education of local people to the problems caused by biological invasions (Detaint and Coïc 2006).
Distribution at the continental scale
We found evidence of at least 25 different introductions of R. catesbeiana in Europe (Appendix S1; Fig. 1a). Introductions have been observed in Belgium, France, Germany, Greece, Holland, Italy, Spain, and the United Kingdom. Free-ranging populations are present in Belgium, France, Germany, Greece, and Italy. Therefore, the situation is radically different from that recorded in the 1990s, when populations were present only in France, Holland, and Italy (Lanza and Ferri 1997). Successful eradication was performed in at least three cases, while in 11 further cases, bullfrogs disappeared after the introduction (Appendix S1). Two of the successful eradications (UK, Germany-Bonn) coped killing of individuals (both adults and tadpoles) and complete drainage of ponds where the population was breeding. In the third successful eradication (Germany-Stuttgart), a complete fencing of the breeding pond was performed in addition to the killing of individuals (Thiesmeier et al. 1994).
The first introductions occurred during the 1930s (Italy) and 1960s (France), but 60% of introductions occurred during the 1980s and 1990s. At least two introductions apparently occurred after 1997, the year when the introduction of R. catesbeiana was forbidden in Europe (law of the European Council 2551/1997) (Appendix S1). Information about the causes of introduction is frequently lacking; however, in most cases, bullfrogs were introduced as pets or because of personal initiatives with unknown causes. Only five introductions were performed as attempts at commercial farming. This is in contrast to observations in other continents, where the bullfrog is usually introduced for the production of food (Lever 2003; Yiming et al. 2006).
The history of the Italian invasions was more complex than those depicted by our paper (Fig. 1; Appendix S1), including multiple secondary translocations. The early dynamics of the Italian invasion has been described in detail by Albertini (1970, 1983) and Albertini and Lanza (1987).
Population invasiveness was positively related to the number of amphibians species recorded in the community (r s = 0.479, N = 21, P = 0.028). This is contrary to what would be observed if the richest communities were more resistant to invasion. Such a pattern can be accounted for if other extrinsic factors are positively related to both richness of native communities and environmental suitability for R. catesbeiana (Shea and Chesson 2002). For example, climatic features such as temperature and precipitation are extremely important in determining the distribution of European amphibians (Araujo et al. 2006), and may play a role also in the suitability of European localities for R. catesbeiana. At least five bullfrog populations from France, Italy, and the United Kingdom are or were infected by the fungus B. dendrobatidis (Garner et al. 2006). The positive association of bullfrog invasions with areas of high richness of amphibians is, therefore, a cause of particular concern for the fate of native species.
Conclusion
Our study shows an alarming increase of the presence of R. catesbeiana in Europe, probably caused by the combined effect of multiple introductions from North America, secondary translocations within European countries, and natural expansion. The number of countries where bullfrogs are present has almost doubled during the last 10 years (Lanza and Ferri 1997), and the species is present over a large area of southwest France. Legislation already forbids new introductions and environmental agencies promote eradication plans. However, translocation performed as personal initiatives seems to be the main cause of introductions. It is, therefore, very difficult to avoid concluding that new introductions will be performed in the future. Moreover, eradication can be difficult; the three successful actions have been performed at early stages of invasion and by means of strenuous destruction or fencing of all breeding wetlands. Therefore, we suggest promotion of educational programs to reduce the risk of new introductions and translocations from established populations. Careful monitoring is necessary for the early detection and management of newly established populations.
References
Albertini G (1970) Sulla diffusione della rana toro (Rana catesbeiana Shaw) importata nel mantovano. Atti Mem Accad Agricolt Sci Lett Verona 20(1968–1969):67–106
Albertini G (1983) La Rana catesbeiana Shaw nella bassa pianura reggiano-modenese. Atti Mem Accad Agricolt Sci Lett Verona 33(1981–1982):125–154
Albertini G, Lanza B (1987) Rana catesbeiana Shaw, 1802 in Italy. Alytes 6:117–129
Araujo MB, Thuiller W, Pearson RG (2006) Climate warming and the decline of amphibians and reptiles in Europe. J Biogeogr 33:1712–1728
Austin JD, Lougheed SC, Boag PT (2004) Controlling for the effects of history and nonequilibrium conditions in gene flow estimates in northern bullfrog (Rana catesbeiana) populations. Genetics 168:1491–1506
Beebee TJC, Griffiths RA (2005) The amphibian decline crisis: a watershed for conservation biology? Biol Conserv 125:271–285
Berger L, Rick Speare R, Daszak P, Earl Green D, Cunningham AA, Louise Gogging C, Slocombe R, Ragan MA, Hyatt AD, McDonald KA, Hines HB, Lips KR, Marantelli G, Parkes H (1998) Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc Natl Acad Sci USA 95:9031–9036
Bosch J, Martinéz-Solano I, Garcia-Paris M (2001) Evidence of a chytrid fungus infection involved in the decline of the common midwife toad (Alytes obstetricans) in protected areas of central Spain. Biol Conserv 97:331–337
Detaint M, Coïc C (2001) Invasion de la Grenouille taureau (Rana catesbeiana) en France: Synthèse bibliographique, suivi 2000–2001, perspectives. Cistude Nature, Le Haillan
Detaint M, Coïc C (2006) La Grenouille taureau Rana catesbeiana dans le sud-ouest de la France. Premiers résultats du programme de lutte. Bull Soc Herpetol France 117:41–56
Ferri V (2006) Rana catesbeiana Shaw, 1802. In: Sindaco R, Doria G, Razzetti E, Bernini F (eds) Atlas of Italian amphibians and reptiles. Polistampa, Firenze, pp 330–333
Garner TWJ, Perkins MW, Govindarajulu P, Seglie D, Walker S, Cunningham AA, Fisher MC (2006) The emerging amphibian pathogen Batrachochytrium dendrobatidis globally infects introduced populations of the North American bullfrog, Rana catesbeiana. Biol Lett 2:455–459
Gasc JP, Cabela A, Crnobrnja-Isailovic J, Dolmen D, Grossenbacher K, Haffner P, Lescure J, Martens H, Martínez Rica JP, Maurin H, Oliveira ME, Sofianidou TS, Veith M, Zuiderwijk A (1997) Atlas of the amphibians and reptiles in Europe. Societas Herpetologica Europaea & Museum National d’Histoire Naturelle, Paris
Hanselmann R, Rodriguez A, Lampo M, Fajardo-Ramos L, Aguirre A, Kilpatrick M, Rodriguez JP, Daszak P (2004) Presence of an emerging pathogen of amphibians in introduced bullfrogs Rana catesbeiana in Venezuela. Biol Conserv 120:115–119
Kats LB, Ferrer RP (2003) Alien predators and amphibian declines: review of two decades of science and the transition to conservation. Divers Distrib 9:99–110
Kolar CS, Lodge DM (2001) Progress in invasion biology: predicting invaders. Trends Ecol Evol 16:199–204
Lanza B, Ferri V (1997) Rana catesbeiana. In: Gasc JP, Cabela A, Crnobrnja-Isailovic J et al (eds) Atlas of the amphibians and reptiles in Europe. Societas Herpetologica Europaea & Museum National d’Histoire Naturelle, Paris, pp 132–133
Lever C (2003) Naturalized amphibians and reptiles of the world. Oxford University Press, New York
Lips KR, Brem F, Brenes R, Reeve JD, Alford RA, Voyles J, Carey C, Livo L, Pessier AP, Collins JP (2006) Emerging infectious disease and the loss of biodiversity in a neotropical amphibian community. Proc Natl Acad Sci USA 103:3165–3170
Lowe S, Browne M, Boudjelas S, De Poorter M (2000) 100 of the world’s worst invasive alien species. A selection from the Global Invasive Species Database. ISSG, SSC and IUCN. Available at www.issg.org/booklet.pdf
Pounds JA, Bustamante MR, Coloma LA, Consuegra JA, Fogden MPL, Foster PN, La Marca E, Masters KL, Merino-Viteri A, Pushendorf R, Ron SR, Sanchez-Azofeifa GA, Still CJ, Young BE (2006) Widespread amphibian extinctions from epidemic disease driven by global warming. Nature 439:161–167
Shea K, Chesson P (2002) Community ecology theory as a framework for biological invasions. Trends Ecol Evol 17:170–176
Smith MA, Green DM (2005) Dispersal and the metapopulation paradigm in amphibian ecology and conservation: are all amphibian populations metapopulations? Ecography 28:110–128
Thiesmeier B, Jäger O, Fritz U (1994) Erfolgreiche Reproduktion des Ochsenfrosches (Rana catesbeiana) im nördlichen Landkreis Böblingen (Baden-Württemberg). Zeitschrift für Feldherpetologie 1:169–176
Veenvliet P, Veenvliet JK (2002) Review of the status of Rana catesbeiana in the European Union. In: Adrados LC, Briggs L (eds) Study of application of EU wildlife trade regulations in relation to species which form an ecological threat to EU fauna and flora, with case studies of American bullfrog (Rana catesbeiana) and red-eared slider (Trachemys scripta elegans). Unpublished report to the European Commission, Amphi Consult, Denmark
Yiming L, Zhengjun W, Duncan RP (2006) Why islands are easier to invade: human influences on bullfrog invasion in the Zhoushan archipelago and neighboring mainland China. Oecologia 148:129–136
Acknowledgements
We thank all the people who helped during field sampling, and all the herpetologists who provided information about the situation in Europe. P. Veenvliet provided valuable unpublished information; the comments of B.R. Anholt and A. Bonin improved an earlier version of the manuscript. Monitoring of the French populations is funded by La Fédération des AAPPMA de la Gironde, de la Dordogne, du Lot et Garonne, Le Conseil Général de la Gironde, du Lot et Garonne et de la Dordogne, l’Agence de l’Eau Adour-Garonne, la Région Aquitaine and the European Council.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below are the electronic supplementary materials.
Rights and permissions
About this article
Cite this article
Ficetola, G.F., Coïc, C., Detaint, M. et al. Pattern of distribution of the American bullfrog Rana catesbeiana in Europe. Biol Invasions 9, 767–772 (2007). https://doi.org/10.1007/s10530-006-9080-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-006-9080-y