Abstract
This study aimed to characterize the epidemiological aspects of PrEP use and barriers to accessing this prophylaxis. This cross-sectional study was conducted between January 2021 and April 2022, encompassing 140 PrEP users treated at the Testing and Counseling Center (CTA) in Campo Grande, Mato Grosso do Sul. Data on sociodemographic characteristics and factors associated with PrEP discontinuation were obtained using a standardized questionnaire. Most PrEP users were cisgender men (92.00%), predominantly white (51.00%), over 30 years of age (56.50%), homosexual-oriented (76.50%), and had a minimum of 12 years of education (77.50%). Approximately 60.00% admitted to inconsistent condom use in recent sexual encounters, primarily involving anal intercourse. Approximately 88.00% perceived themselves as at risk of contracting STIs in the upcoming year. Regarding new presentation forms, 54.00% indicated a willingness to use “on-demand PrEP,” and 92.00% expressed interest in using “injectable PrEP.” After 6 months of follow-up, 43.60% (95.00% CI: 35.50–52.00) discontinued PrEP use, primarily due to changes in sexual behavior (38.30%) and difficulties accessing healthcare services (21.28%). This study underscores the need to involve diverse key populations and highlights the significance of PrEP as an ongoing monitoring strategy for HIV/STI prevention in addition to the importance of incorporating new formulations such as daily oral PrEP into the Brazilian National Health System (SUS).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the first reported case of Acquired Immunodeficiency Syndrome (AIDS) worldwide in 1981, the infection caused by the Human Immunodeficiency Virus (HIV) has reached pandemic proportions. According to data from 2022, it is estimated that approximately 39 million individuals are living with HIV globally [1]. In Brazil, since the first HIV infections, 1,124,063 cases of AIDS have been detected. By 2022, the detection rate was 17.1 cases per 100,000 inhabitants. In the state of Mato Grosso do Sul, this rate was 21.5 cases per 100,000 inhabitants. However, in Campo Grande, the rate was higher than that of the state, reaching 28.5 cases [2].
In 2015, the World Health Organization (WHO) recommended daily oral pre-exposure prophylaxis (PrEP) for individuals at substantial risk of HIV infection [3]. PrEP entails the daily use of a combination of two antiretroviral drugs (ARV) before engaging in sexual practices and has demonstrated an efficacy of 96% in protecting against HIV infection [4,5,6,7,8,9,10]. In December 2017, the Brazilian National Health System approved and included in the local guidelines free access to daily oral PrEP with emtricitabine and tenofovir disoproxil fumarate (FTC/TDF) for populations at substantial risk for HIV infection including men who have sex with men (MSM) and transgender women, sex workers, and serodiscordant couples.
In the state of Mato Grosso do Sul (MS), Campo Grande was the first city to dispense PrEP in 2019. Currently, PrEP is available in 11 of the 79 municipalities in the state [19].
Despite the consensus regarding the individual/population benefits and clinical parameters of the safe use of PrEP, there remain inquiries about its compensatory effects on sexual practices (e.g., decreased condom use), the relationship between social vulnerability and adherence, and the operational challenges in ensuring broad, safe, and effective access to this prophylaxis [11,12,13].
Medication adherence can significantly affect the preventive effect of PrEP. For cisgender women, nearly perfect adherence to pills (six or seven per week) is necessary to ensure maximum protection against HIV. Conversely, for cisgender men, 4–7 pills per week are sufficient, allowing PrEP use on demand. Therefore, it is crucial to identify medication adherence determinants, along with their barriers, and intervene with individuals exhibiting risky behaviors to reduce the likelihood of HIV infection [7, 9, 14,15,16,17].
Therefore, this study aimed to describe the sociodemographic and behavioral aspects of PrEP users, identify barriers to access to the prevention strategy as perceived by users, identify factors associated with non-adherence to PrEP in this population, and understand the interest in new PrEP modalities (injectable and on-demand). These findings are essential for implementing strategies by healthcare services, to ensure the success of this preventive method, reduce access barriers, and consequently, decreasing HIV transmission rates.
Materials and Methods
This is a descriptive cohort study, prospective, based on primary data conducted between January 2021 and April 2022 at the Guidance and Counseling Centers (CTA—Centro de Testagem e Aconselhamento) in the municipality of Campo Grande, Mato Grosso do Sul State, in the context of the Brazilian Public Health System (SUS—Sistema Único de Saúde). CTA is a testing and counseling center that provides diagnoses for HIV, syphilis, and other STIs, offering orientation and healthcare services.
The study included individuals who initiated the use of Pre-Exposure Prophylaxis at the CTA within the specified research period, aged 18 years or older, and reported one or more high-risk criteria for HIV infection acquisition, such as inconsistent condom use, evidence of sexual contact with an HIV-infected partner, self-reported diagnosis of sexually transmitted infections (STI), and repeated use of Post-Exposure Prophylaxis (PEP) for HIV. Individuals with renal or hepatic abnormalities in laboratory tests or severe medical comorbidities, as per the Clinical Protocol for Therapeutic Guidelines (PCDT) for PrEP in 2018 [18], were excluded. It is important to highlight that the only modality of PrEP offered by Brazil’s public policy during the research period was a daily oral regimen. This study employed a non-probabilistic convenience sampling method based on unit demand.
Of the 208 patients who started PrEP during the research period, 140 agreed to participate in the study after signing an informed consent form. Of these individuals, after 6 months, 61 discontinued PrEP use after 6 months. Through the phone numbers in the patients’ records, we were able to contact 47 participants. It was not possible to contact 14 participants because of outdated contact information in the system.
Participants were interviewed using a standardized questionnaire, created by the research team. The questionnaire comprised structured questions covering sociodemographic information, history of drug and alcohol use, medical history, presence of signs and symptoms related to sexually transmitted diseases, sexual behavior, living conditions, income, education, and other variables deemed relevant to the study. We aimed to identify the participants’ profiles and assess the behavioral risk and/or protective factors to which the study population is exposed. The evaluated variables were entered into the database using RedCap software. Starting from the sixth month of follow-up, an active search was conducted in the Logistics Control System for Medicines (SICLOM) to identify patients who discontinued prophylaxis (for more than 30 days without withdrawing the medication). We contacted patients via telephone to ascertain the reasons for discontinuation.
The variables were analyzed using the Stata software (version 13.0; Stata Corporation, College Station, TX, USA). The interview data were presented as categorical variables and as absolute and percentage frequencies. Continuous variables were expressed as medians and ranges. The prevalence was calculated using a 95% confidence interval. Chi-square and Fisher’s exact tests were used to evaluate differences between proportions. Logistic regression was used to determine the association between exposure to infection and each independent variable by estimating the odds ratios (OR). Risk factors were estimated using odds ratios in the univariate analysis. Variables with p < 0.10 in the univariate analysis were included in the multiple logistic regression according to the number of events per variable (EPV). Statistical significance was set at p < 0.05.
This study was approved by the Ethical Committee on Human Research of the Universidade Federal de Mato Grosso do Sul (CEP/UFMS), using the protocol CAAE number 3798562.2.0000.0021.
Results
In total, 140 PrEP users were included in this study (Fig. 1). Approximately 65,000 individuals use PrEP regularly in Brazil, with 701 individuals in Campo Grande, MS [18]. The most prevalent age group among the participants was over 30 years old, representing 43.57% of the sample. Most users self-identified as cisgender men (92.00%) and reported homosexual orientation (76.43%). Regarding education, 77.44% of the participants had more than 12 years of schooling. Regarding income, 57.14% earned between one and three times the minimum wage, whereas 40.71% earned more than three times the minimum wage. Among participants, 7.14% identified themselves as sex workers. Self-reported racial identification indicated that 50.71% of participants were non-white. Furthermore, most patients were single (80.29%) (Table 1).
The research revealed that irregular condom use was prevalent, affecting 59.69% of participants. Unprotected anal intercourse was reported by 58.46% of respondents, while 93.08% did not employ condoms during oral sex. The reasons for not using condoms were trust in partners (25.38%), lack of availability (11.54%), perceived unnecessary use (10.77%), and dislike for usage (9.23%). Furthermore, 19.29% of the participants reported experiencing symptoms of STI in the last six months. Concerning substance consumption, a noteworthy finding is that 86.43% of interviewees reported frequent alcohol use, ranging from “occasionally” to “daily.” Moreover, 18.57% of participants acknowledged the use of illicit drugs.
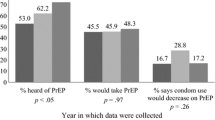
Most respondents (87.86%) acknowledged the risk of acquiring STIs in the coming years. The research revealed that information and experiences regarding PrEP were primarily shared through friends (35.71%), the Internet (33.57%), and healthcare services (27.86%). Among the barriers to PrEP adherence, embarrassment (36.43%), lack of awareness (30.71%), and fear of side effects (16.43%) were most common. Surprisingly, 12.86% of interviewees stated that they would discontinue using condoms after commencing PrEP and 10.00% would contemplate increasing the number of sexual partners.
Regarding the new modalities of PrEP, only 10.00% of the participants were aware of PrEP on demand, and 46.00% expressed a willingness to adopt it if available through the SUS. Injectable PrEP was unknown to 88.50% of the respondents, yet 92.00% showed interest in using it if provided by the SUS, meaning that it would be free of charge and approved by the country's health regulatory agency.
After six months of follow-up, 43.57% of the participants ceased using PrEP. The primary reasons for discontinuation included changes in sexual behavior (38.30%), challenges in attending appointments (21.28%), relocation without access to PrEP (14.89%), and side effects (nausea, headache, and genital itching) (6.38%) (Table 2).
Discussion
To the best of our knowledge, this is the first study to identify the reasons for and predictors of PrEP discontinuation in Central Brazil. Characteristics such as irregular condom use, history of STIs, and alcohol and substance use provide evidence that PrEP is reaching gay/cisgender men who face a higher risk of HIV seroconversion. However, significant challenges remain in reaching individuals under 30 years of age, transgender, and non-white populations.
Individuals belonging to different key populations, who are already considered vulnerable and are in other complex or unfavorable situations, can see their vulnerabilities compounded, leading to what Richard Parker categorized as a “synergy of vulnerabilities” [18]. This can significantly increase the risk of exposure to various infections, such as HIV and other sexually transmitted infections [19]. By recognizing the “synergy of vulnerabilities,” more comprehensive and effective approaches can be adopted to address public health challenges. This involves not only focusing on individual vulnerabilities but also understanding the complex interactions among social, economic, and health factors that contribute to the greater exposure of these groups to different diseases [20, 21]. Thus, interventions can be targeted more precisely and holistically to reduce risks and promote health in complex and multifaceted contexts.
It’s important to highlight that restricting access to HIV prevention and treatment during the COVID-19 pandemic has resulted in changes in the living and health conditions of individuals [22, 23]. COVID-19 intensified and exacerbated existing vulnerabilities and invisibilities in the LGBTQIA + population [24]. The pandemic has led to loss or reduction in income, suspension of medication use, increased engagement in high-risk sexual activities, missed medical appointments, barriers to accessing healthcare services, and other challenges faced by this population [25].
The majority of PrEP users in Brazil are cisgender Gay/MSM individuals (82.00%) of Caucasian ethnicity (56.00%), with an average age of 30–39 years (42.00%), and educational attainment exceeding 12 years (98.00%) [26], corroborating the characteristics found in our study. This pattern has also been observed in other studies, indicating that most PrEP users are highly educated, Caucasian, and gay/MSM [27, 28]. This warrants attention, as PrEP, as a public policy, should be accessible to all priority groups, especially marginalized populations, to ensure greater service equity. For example, in large cities with only one distribution point for prophylaxis in the central area, access to people from peripheral regions is hindered. Glidden et al. reported that individuals with higher educational attainment exhibit greater engagement with prophylaxis, underscoring the importance of catering to populations with lower educational backgrounds [29]. Individuals with less social power and legal protection are at a higher risk of HIV infection [30].
Only 7.14% of the participants identified themselves as sex workers. Sex workers represent an exceedingly vulnerable group to STIs and HIV [31]. Recent Brazilian epidemiological data indicate that cisgender female sex workers have an HIV prevalence rate of 5.30% [32]. They face an HIV acquisition risk that is 30 times higher than that of adult women (aged 15–49 years) in the general population [30]. In a study conducted in India, sex workers exhibited a favorable inclination toward the use of oral PrEP, primarily attributed to engaging in unprotected intercourse with regular partners/spouses, affluent clients, or often due to coercion and duress [33]. Initiating campaigns to enhance awareness and engage this group in PrEP is crucial.
Another disconcerting finding was that only 1.50% of participants identified themselves as transgender. A systematic review encompassing data from 15 countries revealed an HIV prevalence rate of 19.10% among transgender women [34]. More recently, a meta-analysis estimated a prevalence of 25.90% for HIV among transgender women [35]. The UNAIDS [30] underscores that this heightened risk is 14 times greater for HIV acquisition than for adult women. In Brazil, these numbers are even more alarming. The Transcender Project reported an HIV prevalence of 31.20% among cross-dresser and transgender women in Rio de Janeiro and Baixada Fluminense [36]. PrEP has proven to be effective among transgender women, not only increasing life expectancy but also demonstrating high cost-effectiveness [37, 38]. A review by Pacifico de Carvalho et al. unveiled substantial interest in using oral PrEP among transgender women, yet significant barriers to adherence persist, particularly among transgender women [39].
Veloso et al. found an early loss to follow-up of 8.40% in MSM and transgender women in Latin America [40]. The risk was higher in transgender women, participants aged 18–24 years, and those with a lower educational attainment. They further emphasized that by implementing PrEP on the same day, eligibility was confirmed to strengthen adherence to prophylaxis [40, 41].
In this study, we observed that approximately 60.00% did not consistently use condoms, with the primary reason being “trust in the partner” (25.00%). The literature suggests that these unprotected sexual encounters often occur within contexts of greater intimacy and trust, frequently involving individuals in long-term relationships [42, 43]. Nevertheless, it is imperative to underscore the importance of condom use, irrespective of emotional or sexual bonds, because individuals' narratives are influenced by diverse factors and contexts. A Brazilian study indicated that the majority of the Brazilian population reported not using condoms during sexual encounters, with the main reasons associated with trust in the partner, the use of alternative methods, or a preference for condom use during sex [44]. Emphasizing the significance of combined prevention is essential for providing alternative protection options for those facing challenges in condom use.
Regarding the history of STIs, 19.29% reported experiencing symptoms in the last 6 months, and approximately 88.00% perceived themselves as being at risk of acquiring an STI in the coming year. The diagnosis of STIs significantly increases the risk of HIV seroconversion [45]. One of the key issues related to the use of PrEP is risk compensation, which refers to the potential decrease in condom use owing to a reduced perception of HIV risk. This could lead to an increase in STI acquisition of sexually transmitted infections [11, 13, 46].
Available information on sexual behavior and STI incidence among MSM using PrEP is conflicting. Most studies reported a reduction in condom use; however, some indicated an increase in STI incidence, particularly anorectal STI [12, 47, 48], whereas others did not identify significant changes in this regard [8, 49, 50]. It is important to consider that the regular testing required by PrEP protocols may lead to an apparent increase in STI. Hence, services offering PrEP should have essential requirements, such as ongoing education to maintain condom use, testing, and treatment of highly prevalent STI, including syphilis, chlamydia, and gonorrhea. The need for further research to evaluate biomedical strategies for preventing bacterial STIs, such as PrEP and antibiotic PEP (doxycycline) [51, 52], as well as the use of mouthwash with antimicrobial activity [53], should also be emphasized.
The abuse of alcohol and other drugs, even when sporadic, can represent a risk factor for health issues, especially concerning sexual transmission, as it often leads to the relaxation of preventive methods [54,55,56,57]. Alcohol use was reported by 86.43% of the participants in this study, with 18.57% using illicit drugs. In a Brazilian study, excessive alcohol, tobacco, and illegal drug use were more prevalent among participants who had engaged in unprotected sexual intercourse than among those who had used condoms [58]. HIV prevalence rates among people who use drugs are high and distinct from those in the general population, with rates of 5.90% in 2009 and 5.00% among crack cocaine users [59]. For people who use injectable drugs, the risk of acquiring HIV is 35 times higher than that for non-users [30]. In our research, one person (0.71%) reported the use of injectable drugs.
When multivariate analysis was performed, only the age group between 18–25 years (AOR: 2.80; CI 1.03–7.61) remained statistically significant. Every day, four thousand people—including 1100 young people aged 15–24, are infected with HIV [30]. It is worth noting that between 2011 and 2021, a total of 52,513 young people with HIV, aged 15–24 years, of both sexes, progressed to AIDS, highlighting the importance of disease development in this age group and the need to make efforts to link to services and adhere to HIV prevention [60].
The discontinuation rate of PrEP found in this study was 43.57%, which is higher than the current rate in Brazil, reported as 27% by the Ministry of Health [26]. Other studies reported varying discontinuation rates. In the Demo Project, 22.00% discontinued after 12 months of use [55]. Studies in community clinics in the United States have reported discontinuation rates of 21.00% [28] and 43.00% [61]. In the Combina Study, the rate was slightly higher than 12.00% after six months of use [10]. In a cohort study in Canada, approximately 40.00% of participants were lost to PrEP follow-up after six months, with factors such as greater distance from residence, younger age, and lower income associated with lower PrEP care retention [27].
The Implementation PrEP (ImPrEP) study conducted in Brazil, Mexico, and Peru also aimed to investigate the barriers and difficulties associated with its use. Concerning barriers, lack of information, fear of side effects, and doubts about the effectiveness of prophylaxis emerged as the main concerns [50, 62]. In this context, participants reported that before starting PrEP, their main fears were “shame” (36.43%), “lack of publicity” (30.71%), and “fear of side effects” (16.43%). There are gaps in HIV prevention campaigns and dissemination directed at MSM in Latin America, resulting in low utilization of biomedical prevention methods [63]. These data highlight, once again, that knowledge of drug safety and efficacy improves adherence and increases interest in PrEP.
The PrEP strategy can have a pattern of intermittent use that varies according to individual perceptions of HIV infection risk [27, 64]. However, it is important to emphasize that these intervals were associated with the use of other methods. Nevertheless, a significant number of HIV infections occur during these intervals, suggesting that risky practices persist after discontinuing PrEP [14, 65]. Many recent studies have shown that the main reasons for discontinuation are side effects, stigma, entering a monogamous relationship, difficulty accessing services, and a reduced perception of HIV risk [27, 66,67,68]. In our analysis, a change in sexual behavior (reduction in sexual partnerships or starting a monogamous relationship) stood out, accounting for 38.3%. Difficulty attending the clinic (schedule incompatibility or lack of time) was the barrier found in 21.28%, and moving to a city/country that did not offer PrEP accounted for 14.89%. The variable of medication cost was not considered because in Brazil, since 1996, all antiretroviral drugs have been provided free of charge through the Unified Health System (SUS).
Studies involving potential PrEP users indicate that many individuals do not consider daily oral medication an acceptable option and would prefer alternative methods of PrEP administration, such as implants, microbicides, or injectables [69, 70]. Saray et al. also highlighted the preference for “injectable” PrEP among sex workers [33]. In our study, we found that 92% of the participants would use “injectable” PrEP if it were available through the public health system. Long-acting injectable cabotegravir may further contribute to HIV infection prevention compared to oral PrEP. However, for its economic viability in low and middle-income countries, it must be affordable [71]. To a lesser extent, 46.00% would opt for “on-demand” PrEP, corroborating the findings of Broussard et al. [72].
From the analysis of the reasons for discontinuation found in this study, those linked to user behavior and structure stand out. We can observe some weaknesses in service provision (limited service hours, inadequate publicity of prophylaxis, and extensive territory of the city, which hampers user mobility). This set of aspects is crucial for comprehensive care [72]. We must create potential adjustments and plan methods that can reach the target audience more effectively, always proposing greater comprehensiveness, equity, and strengthening of PrEP, thereby confronting and overcoming the HIV epidemic.
The impact on healthcare services limits access to HIV testing and prevention technologies (PrEP and PEP), particularly in vulnerable populations [73]. Thus, COVID-19 further exposed the LGBTQIA + population to rights denial, violence, discrimination, and other forms of injustice, contributing to the creation of health inequalities and inequities, and consequently influencing the sample size of the present study. In addition, approaches aimed at improving risk perception, such as the use of online risk assessment tools [74] and sexual activity logs [75], have the potential to positively influence decisions related to the initiation and discontinuation of PrEP. Printed and digital media such as social networks and dating apps are essential tools for reaching younger audiences. Facilitating access to healthcare services with flexible and extended hours, teleconsultations, and medication delivery could reduce the barriers identified in this study. The idea of offering “itinerant” PrEP could reach populations in vulnerable situations, such as sex workers or transgender individuals. Furthermore, continuous education of healthcare professionals is fundamental, as they are potential prescribers and recruiters of PrEP users.
It is important to acknowledge certain limitations. Unfortunately, the COVID-19 pandemic prevented us from enrolling more participants in this study, thereby restricting our sample size. Additionally, there is a potential for underreporting of risk behaviors during the interviews, which serves as another limitation of this cross-sectional study.
In conclusion, to succeed with PrEP in public health, we need to combine interventions to promote acceptance, support adherence, and prevent increases in risky behaviors. PrEP is not solely concerned with distributing antiretrovirals; it is a comprehensive monitoring strategy. Healthcare services should view each encounter as an opportunity to instill an accurate perception of risk to the user. Therefore, if patients decide to discontinue prophylaxis, alternative methods may be used to maintain protection against HIV and other STI.
Data Availability
All relevant and original data presented in this study have been included in this article.
References
UNAIDS. Global HIV & AIDS statistics—Fact sheet 2023. Geneva: UNAIDS, 2023. Disponível em: https://www.unaids.org/en/resources/fact-sheet. Acessado em: 20 agosto 2023.
BRASIL. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância, Prevenção e Controle das Infecções Sexualmente Transmissíveis, do HIV/aids e das Hepatites Virais. Boletim Epidemiológico HIV/AIDS 2022. Brasília: Ministério da Saúde, 2022a. Disponível em: https://www.gov.br/aids/pt-br/centrais-de-conteudo/boletins-epidemiologicos/2022/hiv-aids/boletim_epidemiologico_hiv_aids_-2022.pdf/view.
World Health Organization. Guideline on when to start antiretroviral therapy and on pre-exposure prophylaxis for HIV. Geneva: World Health Organization; 2015.
Anderson PL, Glidden DV, Liu A, Buchbinder S, Lama JR, Guanira JV, Mcmahan V, Bushman LR, Casapia M, Montoya-Herrera O, Veloso VG, Mayer KH, Chariyalertsak S, Schechter M, Bekker LG, Kallás EG, Grant RM, Iprex Study Team. Emtricitabine-tenofovir exposure and pre-exposure efficacy in men who have sex with men. Science Transl Med. 2012;4:1–17.
Baeten JM, Donnell D, Ndase P, Mugo NR, Campbell JD, Wangisi J, Tappero JW, Bukusi EA, Cohen CR, Katabira E, Ronald A, Tumwesigye E, Were E, Fife KH, Kiarie J, Farquhar C, John-Stewart G, Kakia A, Odoyo J, Mucunguzi A, Nakku-Joloba E, Twesigye R, Ngure K, Apaka C, Tamooh H, Gabona F, Mujugira A, Panteleeff D, Thomas KK, Kidoguchi L, Krows M, Revall J, Morrison S, Haugen H, Emmanuel-Ogier M, Ondrejcek L, Coombs RW, Frenkel L, Hendrix C, Bumpus NN, Bangsberg D, Haberer JE, Stevens WS, Lingappa JR, Celum C, Partners PrEP Study Team. Antiretroviral prophylaxis for HIV prevention in heterosexual men and women. N Engl J Med. 2012;367(5):399–410. https://doi.org/10.1056/NEJMoa1108524.
Choopanya K, Martin M, Suntharasamai P, Sangkum U, Mock PA, Leethochawalit M, Chiamwongpaet S, Kitisin P, Natrujirote P, Kittimunkong S, Chuachoowong R, Gvetadze RJ, McNicholl JM, Paxton LA, Curlin ME, Hendrix CW, Vanichseni S, Bangkok Tenofovir Study Group. Antiretroviral prophylaxis for HIV infection in injecting drug users in Bangkok, Thailand (the Bangkok Tenofovir Study): a randomized, double-blind, placebo-controlled phase 3 trial. Lancet. 2013;381(9883):2083–90. https://doi.org/10.1016/S0140-6736(13)61127-7.
Grant RM, Lama JR, Anderson PL, McMahan V, Liu AY, Vargas L, Goicochea P, Casapía M, Guanira-Carranza JV, Ramirez-Cardich ME, Montoya-Herrera O, Fernández T, Veloso VG, Buchbinder SP, Chariyalertsak S, Schechter M, Bekker LG, Mayer KH, Kallás EG, Amico KR, Mulligan K, Bushman LR, Hance RJ, Ganoza C, Defechereux P, Postle B, Wang F, McConnell JJ, Zheng JH, Lee J, Rooney JF, Jaffe HS, Martinez AI, Burns DN, Glidden DV, iPrEx Study Team. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363(27):2587–99. https://doi.org/10.1056/NEJMoa1011205.
McCormack S, Dunn DT, Desai M, Dolling DI, Gafos M, Gilson R, Sullivan AK, Clarke A, Reeves I, Schembri G, Mackie N, Bowman C, Lacey CJ, Apea V, Brady M, Fox J, Taylor S, Antonucci S, Khoo SH, Rooney J, Nardone A, Fisher M, McOwan A, Phillips AN, Johnson AM, Gazzard B, Gill ON. Pre-exposure prophylaxis to prevent the acquisition of HIV-1 infection (PROUD): effectiveness results from the pilot phase of a pragmatic open-label randomised trial. Lancet. 2016;387(10013):53–60. https://doi.org/10.1016/S0140-6736(15)00056-2.
Thigpen MC, Kebaabetswe PM, Paxton LA, Smith DK, Rose CE, Segolodi TM, Henderson FL, Pathak SR, Soud FA, Chillag KL, Mutanhaurwa R, Chirwa LI, Kasonde M, Abebe D, Buliva E, Gvetadze RJ, Johnson S, Sukalac T, Thomas VT, Hart C, Johnson JA, Malotte CK, Hendrix CW, Brooks JT, TDF2 Study Group. Antiretroviral preexposure prophylaxis for heterosexual HIV transmission in Botswana. N Engl J Med. 2012;367(5):423–34. https://doi.org/10.1056/NEJMoa1110711.
Zucchi EM, Grangeiro A, Ferraz D, Pinheiro TF, Alencar T, Ferguson L, Estevam DL, Munhoz R, Equipe do Estudo Combina! Da evidência à ação: desafios do Sistema Único de Saúde para ofertar a profilaxia pré-exposição sexual (PrEP) ao HIV às pessoas em maior vulnerabilidade [From evidence to action: challenges for the Brazilian Unified National Health System in offering pre-exposure prophylaxis (PrEP) for HIV to persons with the greatest vulnerability]. Cad Saude Publica. 2018;34(7): e00206617. https://doi.org/10.1590/0102-311X00206617. (Portuguese).
Fonner VA, Dalglish SL, Kennedy CE, Baggaley R, O’Reilly KR, Koechlin FM, Rodolph M, Hodges-Mameletzis I, Grant RM. Effectiveness and safety of oral HIV preexposure prophylaxis for all populations. AIDS. 2016;30(12):1973–83. https://doi.org/10.1097/QAD.0000000000001145.
Blumenthal J, Haubrich RH. Will risk compensation accompany pre-exposure prophylaxis for HIV? Virtual Mentor. 2014;16(11):909–15. https://doi.org/10.1001/virtualmentor.2014.16.11.stas1-1411.
Hoornenborg E, Coyer L, Achterbergh RCA, Matser A, Schim van der Loeff MF, Boyd A, van Duijnhoven YTHP, Bruisten S, Oostvogel P, Davidovich U, Hogewoning A, Prins M, de Vries HJC, Amsterdam PrEP Project team in the HIV Transmission Elimination AMsterdam (H-TEAM) Initiative. Sexual behaviour and incidence of HIV and sexually transmitted infections among men who have sex with men using daily and event-driven pre-exposure prophylaxis in AMPrEP: 2 year results from a demonstration study. Lancet HIV. 2019;6(7):e447–55. https://doi.org/10.1016/S2352-3018(19)30136-5.
Grant RM, Anderson PL, McMahan V, Liu A, Amico KR, Mehrotra M, Hosek S, Mosquera C, Casapia M, Montoya O, Buchbinder S, Veloso VG, Mayer K, Chariyalertsak S, Bekker LG, Kallas EG, Schechter M, Guanira J, Bushman L, Burns DN, Rooney JF, Glidden DV, iPrEx study team. Uptake of pre-exposure prophylaxis, sexual practices, and HIV incidence in men and transgender women who have sex with men: a cohort study. Lancet Infect Dis. 2014;14(9):820–9. https://doi.org/10.1016/S1473-3099(14)70847-3.
Grinsztejn B, Torres TS, Hoagland B, Jalil EM, Moreira RI, O’Malley G, Shade SB, Benedetti MR, Moreira J, Simpson K, Pimenta MC, Veloso VG, ImPrEP CAB-Brasil Study Team. Long-acting injectable cabotegravir for HIV preexposure prophylaxis among sexual and gender minorities: protocol for an implementation study. JMIR Public Health Surveill. 2023;9: e44961. https://doi.org/10.2196/44961.
Molina JM, Capitant C, Spire B, Pialoux G, Cotte L, Charreau I, Tremblay C, Le Gall JM, Cua E, Pasquet A, Raffi F, Pintado C, Chidiac C, Chas J, Charbonneau P, Delaugerre C, Suzan-Monti M, Loze B, Fonsart J, Peytavin G, Cheret A, Timsit J, Girard G, Lorente N, Préau M, Rooney JF, Wainberg MA, Thompson D, Rozenbaum W, Doré V, Marchand L, Simon MC, Etien N, Aboulker JP, Meyer L, Delfraissy JF, ANRS IPERGAY Study Group. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N Engl J Med. 2015;373(23):2237–46. https://doi.org/10.1056/NEJMoa1506273.
Mugwanya KK, Wyatt C, Celum C, Donnell D, Mugo NR, Tappero J, Kiarie J, Ronald A, Baeten JM, Partners PrEP Study Team. Changes in glomerular kidney function among HIV-1-uninfected men and women receiving emtricitabine-tenofovir disoproxil fumarate preexposure prophylaxis: a randomized clinical trial. JAMA Intern Med. 2015;175(2):246–54. https://doi.org/10.1001/jamainternmed.2014.6786.
Parker RG. Na contramão da AIDS: sexualidade, intervenção, política. 1ª ed. São Paulo, SP, Brasil: Rio de Janeiro, RJ, Brasil: Editora 34; ABIA - Associação Brasileira Interdisciplinar de AIDS, 2000.
BRASIL. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância, Prevenção e Controle das Infecções Sexualmente Transmissíveis, do HIV/aids e das Hepatites Virais. Agenda Estratégica para Ampliação do Acesso e Cuidado Integral das Populações-Chave em HIV, Hepatites Virais e outras Infecções Sexualmente Transmissíveis. Brasília: Ministério da Saúde; 2018d.
Castel R. A dinâmica dos processos de marginalização: da vulnerabilidade a “desfiliação.” Caderno CRH. 2006;10(26):19–40.
Davis A. Mulheres, raça e classe. São Paulo: Boitempo; 2016.
Sousa AR, Cerqueira CFC, Porcino C, Simões, KJF. Pessoas LGBTI+ e a COVID-19: Para pensarmos questões sobre saúde. Revista Baiana de Enfermagem. 2020; 35.
Stover J, Kelly SL, Mudimu E, Green D, Smith T, Taramusi I, Bansi-Matharu L, Martin-Hughes R, Phillips AN, Bershteyn A. The risks and benefits of providing HIV services during the COVID-19 pandemic. PLoS ONE. 2021;16(12): e0260820. https://doi.org/10.1371/journal.pone.0260820.
Miranda MHG, Grangeão FN, Montenegro FFPA. A Pandemia do Covid-19 e o Descortinamento das Vulnerabilidades da População LGBTQI+ Brasileira. Revista Interdisciplinar em Cultura e Sociedade. 2020;6(2):102–19.
Santos MA, Oliveira WA, Oliveira-Cardoso EA. Inconfidências de Abril: Impacto do Isolamento Social na Comunidade Trans em Tempos de Pandemia de COVID-19. Psicologia & Sociedade. 2020. https://doi.org/10.1590/1807-0310/2020v32240339.
BRASIL. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância, Prevenção e Controle das Infecções Sexualmente Transmissíveis, do HIV/aids e das Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para Profilaxia Pré-Exposição (PrEP) de risco à infecção pelo HIV. Brasília: Ministério da Saúde; 2018.
BRASIL. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Doenças de Condições e Infecções Sexualmente Transmissíveis. Painel PrEP. Brasília: Ministério da Saúde, 2022c. https://www.gov.br/aids/pt-br/assuntos/prevencao-combinada/prep-profilaxia-pre-exposicao/painel-prep. Acessado em: 20 out. 2022.
Greenwald ZR, Maheu-Giroux M, Szabo J, Robin JAB, Boissonnault M, Nguyen VK, Thomas R. Cohort profile: l’Actuel Pre-Exposure Prophylaxis (PrEP) Cohort study in Montreal, Canada. BMJ Open. 2019;9(6): e028768. https://doi.org/10.1136/bmjopen-2018-028768.
Hojilla JC, Vlahov D, Crouch PC, Dawson-Rose C, Freeborn K, Carrico A. HIV pre-exposure prophylaxis (PrEP) uptake and retention among men who have sex with men in a community-based sexual health clinic. AIDS Behav. 2018;22(4):1096–9. https://doi.org/10.1007/s10461-017-2009-x.
Hojilla JC, Satre DD, Glidden DV, McMahan VM, Gandhi M, Defechereux P, Guanira JV, Mehrotra M, Grant RM, Carrico AW. Brief report: cocaine use and pre-exposure prophylaxis: adherence, care engagement, and kidney function. J Acquir Immune Defic Syndr. 2019;81(1):78–82. https://doi.org/10.1097/QAI.0000000000001972.
UNAIDS. In Danger: UNAIDS Global AIDS Update 2022. [s.l.] United Nations, 2022. Disponível em. https://www.unaids.org/sites/default/files/media_asset/2022-global-aids-update_en.pdf. Acessado em: 20 julho 2023.
Plummer FA, Nagelkerke NJ, Moses S, Ndinya-Achola JO, Bwayo J, Ngugi E. The importance of core groups in the epidemiology and control of HIV-1 infection. AIDS. 1991;5(Suppl 1):S169-176.
Szwarcwald CL, Damacena GN, de Souza-Júnior PRB, Guimarães MDC, de Almeida WDS, de Souza Ferreira AP, Ferreira-Júnior ODC, Dourado I, Brazilian FSW Group. Factors associated with HIV infection among female sex workers in Brazil. Medicine Baltimore. 2018;97(1S Suppl 1):S54–61. https://doi.org/10.1097/MD.0000000000009013.
Sahay S, Verma A, Shewale S, Bangar S, Bijeshkumar A, Angolkar M, Subramanian T, Chandhiok N. Understanding issues around use of oral pre exposure prophylaxis among female sex workers in India. BMC Infect Dis. 2021;21(1):930. https://doi.org/10.1186/s12879-021-06612-8.
Baral SD, Poteat T, Strömdahl S, Wirtz AL, Guadamuz TE, Beyrer C. Worldwide burden of HIV in transgender women: a systematic review and meta-analysis. Lancet Infect Dis. 2013;13(3):214–22. https://doi.org/10.1016/S1473-3099(12)70315-8.
Stutterheim SE, van Dijk M, Wang H, Jonas KJ. The worldwide burden of HIV in transgender individuals: an updated systematic review and meta-analysis. PLoS ONE. 2021;16(12): e0260063. https://doi.org/10.1371/journal.pone.0260063.
Grinsztejn B, Jalil EM, Monteiro L, Velasque L, Moreira RI, Garcia AC, Castro CV, Krüger A, Luz PM, Liu AY, McFarland W, Buchbinder S, Veloso VG, Wilson EC, Transcender Study Team. Unveiling of HIV dynamics among transgender women: a respondent-driven sampling study in Rio de Janeiro, Brail. Lancet HIV. 2017;4(4):e169–76. https://doi.org/10.1016/S2352-3018(17)30015-2.
Deutsch MB, Glidden DV, Sevelius J, Keatley J, McMahan V, Guanira J, Kallas EG, Chariyalertsak S, Grant RM, iPrEx investigators. HIV pre-exposure prophylaxis in transgender women: a subgroup analysis of the iPrEx trial. Lancet HIV. 2015;2(12):e512–9. https://doi.org/10.1016/S2352-3018(15)00206-4.
Luz PM, Osher B, Grinsztejn B, Maclean RL, Losina E, Stern ME, Struchiner CJ, Parker RA, Freedberg KA, Mesquita F, Walensky RP, Veloso VG, Paltiel AD. The cost-effectiveness of HIV pre-exposure prophylaxis in men who have sex with men and transgender women at high risk of HIV infection in Brazil. J Int AIDS Soc. 2018;21(3): e25096. https://doi.org/10.1002/jia2.25096.
Avelino-Silva VI, Vasconcelos R, Cerqueira NB, Marcus U, Schmidt AJ, Veras MA. Predictors of knowledge of and access to biomedical prevention among MSM and transgender men in Latin America: results from the Latin American internet survey. HIV Med. 2022;23(7):764–73. https://doi.org/10.1111/hiv.13238.
Veloso VG, Cáceres CF, Hoagland B, Moreira RI, Vega-Ramírez H, Konda KA, Leite IC, Bautista-Arredondo S, Vinícius de Lacerda M, Valdez Madruga J, Farias A, Lima JN, Zonta R, Lauria L, Tamayo CVO, Flores HJS, Santa Cruz YMC, Aguayo RMM, Cunha M, Moreira J, Makkeda AR, Díaz S, Guanira JV, Vermandere H, Benedetti M, Ingold HL, Pimenta MC, Torres TS, Grinsztejn B, ImPrEP Study Group. Same-day initiation of oral pre-exposure prophylaxis among gay, bisexual, and other cisgender men who have sex with men and transgender women in Brazil, Mexico, and Peru (ImPrEP): a prospective, single-arm, open-label, multicentre implementation study. Lancet HIV. 2023;10(2):e84–96. https://doi.org/10.1016/S2352-3018(22)00331-9.
Pacífico de Carvalho N, Mendicino CCP, Cândido RCF, Alecrim DJD, Menezes de Pádua CA. HIV pre-exposure prophylaxis (PrEP) awareness and acceptability among trans women: a review. AIDS Care. 2019;31(10):1234–40. https://doi.org/10.1080/09540121.2019.1612014.
Li D, Li C, Wang Z, Lau JT. Prevalence and associated factors of unprotected anal intercourse with regular male sex partners among HIV negative men who have sex with men in China: a cross-sectional survey. PLoS ONE. 2015;10(3): e0119977. https://doi.org/10.1371/journal.pone.0119977.
Lachowsky NJ, Saxton PJ, Hughes AJ, Dickson NP, Milhausen RR, Dewey CE, Summerlee AJ. Frequent condom use with casual partners varies by sexual position among younger gay and bisexual men in New Zealand: national behavioural surveillance 2006–2011. Sex Health. 2016;13(1):81–6. https://doi.org/10.1071/SH14220.
Felisbino-Mendes MS, Araújo FG, Oliveira LVA, Vasconcelos NM, Vieira MLFP, Malta DC. Sexual behaviors and condom use in the Brazilian population: analysis of the National Health Survey, 2019. Rev Bras Epidemiol. 2021;24(suppl 2):e210018. https://doi.org/10.1590/1980-549720210018.supl.2. (English, Portuguese).
Solomon MM, Mayer KH, Glidden DV, Liu AY, McMahan VM, Guanira JV, Chariyalertsak S, Fernandez T, Grant RM, iPrEx Study Team. Syphilis predicts HIV incidence among men and transgender women who have sex with men in a preexposure prophylaxis trial. Clin Infect Dis. 2014;59(7):1020–6. https://doi.org/10.1093/cid/ciu450.
Kojima N, Davey DJ, Klausner JD. Pre-exposure prophylaxis for HIV infection and new sexually transmitted infections among men who have sex with men. AIDS. 2016;30(14):2251–2. https://doi.org/10.1097/QAD.0000000000001185.
Ong JJ, Baggaley RC, Wi TE, Tucker JD, Fu H, Smith MK, Rafael S, Anglade V, Falconer J, Ofori-Asenso R, Terris-Prestholt F, Hodges-Mameletzis I, Mayaud P. Global epidemiologic characteristics of sexually transmitted infections among individuals using preexposure prophylaxis for the prevention of HIV infection: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(12): e1917134. https://doi.org/10.1001/jamanetworkopen.2019.17134.
Traeger MW, Schroeder SE, Wright EJ, Hellard ME, Cornelisse VJ, Doyle JS, Stoové MA. Effects of pre-exposure prophylaxis for the prevention of human immunodeficiency virus infection on sexual risk behavior in men who have sex with men: a systematic review and meta-analysis. Clin Infect Dis. 2018;67(5):676–86. https://doi.org/10.1093/cid/ciy182.
Koechlin FM, Fonner VA, Dalglish SL, O’Reilly KR, Baggaley R, Grant RM, Rodolph M, Hodges-Mameletzis I, Kennedy CE. Values and preferences on the use of oral pre-exposure prophylaxis (PrEP) for HIV prevention among multiple populations: a systematic review of the literature. AIDS Behav. 2017;21(5):1325–35. https://doi.org/10.1007/s10461-016-1627-z.
Freeborn K, Portillo CJ. Does pre-exposure prophylaxis for HIV prevention in men who have sex with men change risk behaviour? A systematic review. J Clin Nurs. 2018;27(17–18):3254–65. https://doi.org/10.1111/jocn.13990.
Celum C, Luetkemeyer AF. Doxycycline for sexually transmitted infection prevention: evolving evidence and implementation perspectives. Sex Transm Dis. 2021;48(9):620–1. https://doi.org/10.1097/OLQ.0000000000001501.
Molina JM, Charreau I, Chidiac C, Pialoux G, Cua E, Delaugerre C, Capitant C, Rojas-Castro D, Fonsart J, Bercot B, Bébéar C, Cotte L, Robineau O, Raffi F, Charbonneau P, Aslan A, Chas J, Niedbalski L, Spire B, Sagaon-Teyssier L, Carette D, Mestre SL, Doré V, Meyer L, ANRS IPERGAY Study Group. Post-exposure prophylaxis with doxycycline to prevent sexually transmitted infections in men who have sex with men: an open-label randomised substudy of the ANRS IPERGAY trial. Lancet Infect Dis. 2018;18(3):308–17. https://doi.org/10.1016/S1473-3099(17)30725-9.
Chow EPF, Williamson DA, Hocking JS, Law MG, Maddaford K, Bradshaw CS, McNulty A, Templeton DJ, Moore R, Murray GL, Danielewski JA, Wigan R, Chen MY, Guy RJ, Zhang L, Donovan B, Grulich AE, Kaldor JM, Whiley DM, Cornelisse VJ, Howden BP, Lewis DA, Read TRH, Fairley CK. Antiseptic mouthwash for gonorrhoea prevention (OMEGA): a randomised, double-blind, parallel-group, multicentre trial. Lancet Infect Dis. 2021;21(5):647–56. https://doi.org/10.1016/S1473-3099(20)30704-0.
Schwitters A, Sabatier J, Seth P, Glenshaw M, Remmert D, Pathak S, Bock N. HIV and alcohol knowledge, self-perceived risk for HIV, and risky sexual behavior among young HIV-negative men identified as harmful or hazardous drinkers in Katutura, Namibia. BMC Public Health. 2015;15:1182. https://doi.org/10.1186/s12889-015-2516-5.
Irarrázabal LP, Ferrer L, Villegas N, Sanhueza S, Molina Y, Cianelli R. Mujeres que Consumen Sustancias y su Vulnerabilidad frente al VIH en Santiago de Chile (Women Who Consume Substances and Their Vulnerability to HIV in Santiago of Chile). Hisp Health Care Int. 2016;14(2):89–93. https://doi.org/10.1177/1540415316647978.
Liu Y, Ruan Y, Strauss SM, Yin L, Liu H, Amico KR, Zhang C, Shao Y, Qian HZ, Vermund SH. Alcohol misuse, risky sexual behaviors, and HIV or syphilis infections among Chinese men who have sex with men. Drug Alcohol Depend. 2016;168:239–46. https://doi.org/10.1016/j.drugalcdep.2016.09.020.
Wirtz AL, Zelaya CE, Latkin C, Stall R, Peryshkina A, Galai N, Mogilniy V, Dzhigun P, Kostetskaya I, Beyrer C. Alcohol use and associated sexual and substance use behaviors among men who have sex with men in Moscow. Russia AIDS Behav. 2016;20(3):523–36. https://doi.org/10.1007/s10461-015-1066-2.
Sanchez ZM, Nappo SA, Cruz JI, Carlini EA, Carlini CM, Martins SS. Sexual behavior among high school students in Brazil: alcohol consumption and legal and illegal drug use associated with unprotected sex. Clinics (Sao Paulo). 2013;68(4):489–94. https://doi.org/10.6061/clinics/2013(04)09.
Rowan SE, Patel RR, Schneider JA, Smith DK. Same-day prescribing of daily oral pre-exposure prophylaxis for HIV prevention. Lancet HIV. 2021;8(2):e114–20. https://doi.org/10.1016/S2352-3018(20)30256-3.
Broussard, J.; Bena J.; Crouch, P. C.; Taylor B.; Chavez, M.; Grant, R. M. PrEP 2–1–1 education increases PrEP uptake and preserves effective PrEP coverage in a large nurse-led community-based sexual health clinic in San Francisco. In: INTERNATIONAL AIDS CONFERENCE, 23. 2020, San Francisco. Anais eletrônicos [...] on-line: International AIDS Society, 2020.
Chan PA, Mena L, Patel R, Oldenburg CE, Beauchamps L, Perez-Brumer AG, Parker S, Mayer KH, Mimiaga MJ, Nunn A. Retention in care outcomes for HIV pre-exposure prophylaxis implementation programmes among men who have sex with men in three US cities. J Int AIDS Soc. 2016;19(1):20903. https://doi.org/10.7448/IAS.19.1.20903.
Pimenta MC, Bermúdez XP, Go AMM, Maksud I, Benedetti M, Kauss B, Torres TS, Hoagland B, Pereira GFM, Grinsztejn B, Veloso VG. Barreiras e facilitadores do acesso de populações vulneráveis à PrEP no Brasil: Estudo ImPrEP Stakeholders [Barriers and facilitators for access to PrEP by vulnerable populations in Brazil: the ImPrEP Stakeholders Study]. Cad Saude Publica. 2022;38(1): e00290620. https://doi.org/10.1590/0102-311X00290620. (Portuguese).
Elsesser SA, Oldenburg CE, Biello KB, Mimiaga MJ, Safren SA, Egan JE, Novak DS, Krakower DS, Stall R, Mayer KH. Seasons of risk: anticipated behavior on vacation and interest in episodic antiretroviral pre-exposure prophylaxis (PrEP) among a large national sample of U.S. men who have sex with men (MSM). AIDS Behav. 2016;20(7):1400–7. https://doi.org/10.1007/s10461-015-1238-0.
Greenwald Z, Beauchemin M, Benomar K, Landry G, Hamel A, Boissonnault M, Charest L, Longpré D, Lavoie S, Thomas R. High seroconversion rates following PrEP discontinuance in a Montreal clinic. J Virus Erad. 2018;4:12. https://doi.org/10.1016/S2055-6640(20)30378-2.
Kyongo JK, Kiragu M, Karugu FR, Ochieng C, Ngunjiri A, Wachihi C. How long will they take it? Oral preexposure prophylaxis (PrEP) retention for female sex workers, men who have sex with men and young women in a demonstration project in Kenya. In: International AIDS Conference. 22, 2018 Amsterdam. Abstract [...] Amsterdam; 2018.
Mcgrory E, Murungu J, Aguolu R, Alagi M, Alary M, Baeten J. Delivering PrEP: lessons from early demonstration projects. In: HIV research for prevention, 2018, Madrid. Poster [...] Madrid; 2018.
Pillay D, Stankevitz K, Lanham M, Ridgeway K, Murire M, Briedenhann E, Jenkins S, Subedar H, Hoke T, Mullick S. Factors influencing uptake, continuation, and discontinuation of oral PrEP among clients at sex worker and MSM facilities in South Africa. PLoS ONE. 2020;15(4): e0228620. https://doi.org/10.1371/journal.pone.0228620.
Bastos FIPM, Bertoni N. (Org.). Pesquisa Nacional sobre o uso de crack: quem são os usuários de crack e/ou similares do Brasil? Quantos são nas capitais brasileiras? Rio de Janeiro: ICICT; 2014.
Greene GJ, Swann G, Fought AJ, Carballo-Diéguez A, Hope TJ, Kiser PF, Mustanski B, D’Aquila RT. Preferences for long-acting pre-exposure prophylaxis (PrEP), daily oral PrEP, or condoms for HIV prevention among U.S. men who have sex with men. AIDS Behav. 2017;21(5):1336–49. https://doi.org/10.1007/s10461-016-1565-9.
Meyers K, Rodriguez K, Brill AL, Wu Y, La Mar M, Dunbar D, Koblin B, Margolis D, Sobieszczyk ME, Van Tieu H, Frank I, Markowitz M, Golub SA. Lessons for patient education around long-acting injectable PrEP: findings from a mixed-method study of phase II trial participants. AIDS Behav. 2018;22(4):1209–16. https://doi.org/10.1007/s10461-017-1871-x.
Jamieson L, Johnson LF, Nichols BE, Delany-Moretlwe S, Hosseinipour MC, Russell C, Meyer-Rath G. Relative cost-effectiveness of long-acting injectable cabotegravir versus oral pre-exposure prophylaxis in South Africa based on the HPTN 083 and HPTN 084 trials: a modelled economic evaluation and threshold analysis. Lancet HIV. 2022;9(12):e857–67. https://doi.org/10.1016/S2352-3018(22)00251-X.
Hargreaves J, Davey C, Group for lessons from pandemic HIV prevention for the COVID-19 response. Three lessons for the COVID-19 response from pandemic HIV. Lancet HIV. 2020;7(5):e309–11. https://doi.org/10.1016/S2352-3018(20)30110-7.
Scott LJ. Dolutegravir/lamivudine single-tablet regimen: a review in HIV-1 infection. Drugs. 2020;80(1):61–72. https://doi.org/10.1007/s40265-019-01247-1.
Wray TB, Luo X, Ke J, Pérez AE, Carr DJ, Monti PM. Using smartphone survey data and machine learning to identify situational and contextual risk factors for HIV risk behavior among men who have sex with men who are not on PrEP. Prev Sci. 2019;20(6):904–13. https://doi.org/10.1007/s11121-019-01019-z.
Acknowledgements
We would like to express our gratitude to the Municipal Health Secretary, Secretaria Municipal de Saúde (SESAU), for their assistance in this study, as well as to the “A Hora é Agora” Project for their support. This study was conducted with support from the Federal University of Mato Grosso do Sul (UFMS/MEC), Brazil.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Conceptualization and designed the experiments: RPBJ, GAC, ARCMC; performed the experiments: RPBJ, CA, GAC; analyzed the data: ARCMC, CA; contributed materials/analysis tools: ARCMC, LMB; writing—original draft preparation, review, and editing: RPBJ, GAC, CA, ASPS, ARCMC. Supervision: ARCMC. All authors have read and agreed to this version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interests
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Braz Junior, R.P., Cesar, G.A., Amianti, C. et al. Behind Prep Decisions: Understanding User Patterns and Discontinuation Factors in Real-World. AIDS Behav 28, 2979–2989 (2024). https://doi.org/10.1007/s10461-024-04383-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-024-04383-2