Abstract
Alley cropping systems present tree rows alternating with crop alleys. Tree rows are uncultivated and often present an understory vegetation strip (UVS), whose impacts have been less studied than those of the trees. Spatial variations within UVSs and between UVSs and crop alleys were investigated for vegetation biomass and necromass, soil organic carbon and nutrient contents, microbial biomass and respiration, and macro-invertebrate density, taxonomic diversity and functional community structure. The first 20 cm of soil were sampled in two young alley cropping sites in South-West France with sown UVSs. Tree distance did not influence studied variables within the UVS. Soil organic carbon and available phosphorus contents were significantly higher in the UVS than in the crop alley, but not soil microbial biomass and macro-invertebrate density and diversity, except for woodlice density. Earthworm distribution in ecomorphological categories differed between the UVS and the crop alley. Microbial biomass and macro-invertebrate diversity were significantly reduced 1 m away from the UVS in both sites. It was concluded that these sown UVSs were not strongly impacted by tree proximity, enriched the first 20 cm of soil with organic carbon, and provided a habitat for specific macro-invertebrates, probably due to reduced disturbance and permanent vegetation. However, UVSs could have a negative impact on soil organisms in their vicinity. Further studies on the role of UVS type, field management and seasonal variation are needed to quantify the role of UVSs on soil properties.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In temperate regions, alley cropping is a common silvoarable system, in which tree rows alternate with crop alleys (Eichhorn et al. 2006). Tree rows are most of the time covered with a spontaneous or sown understory vegetation strip (UVS, Boinot et al. 2019), mostly herbaceous. The specific composition of the UVS is almost neither detailed, but when it is sown it is often with grasses (Udawatta et al. 2008; Cardinael et al. 2017), maybe because grasses have been shown to establish easily in field margins (Critchley et al. 2006). The UVS has been shown to increase pollinator abundance and diversity in the field by providing resources and nest sites (Varah et al. 2013), but much less is known about soil biota and their functions in decomposition and food web regulation in spatially heterogeneous alley cropping systems (Marsden et al. 2019).
Detrital food webs are strongly driven by primary resources availability (Dyer and Letourneau 2003) and their quality and quantity (Porazinska et al. 2003). The UVS brings additional vegetation and its associated litter in the tree row, and is thus likely to have a strong impact on soil organisms. In addition, with its permanent ground cover, it could be an attractive habitat for soil biota that are sensitive to disturbance, which is frequent in the crop alleys. This can be expected to lead to the differentiation of UVS and crop alley soil biota communities, with distinct densities and composition. However, the UVS effect may extend further than expected based on aboveground vegetation development. On a young alley cropping site, Battie-Laclau et al. (2019) found that herbaceous roots from the UVS extended up to 2.5 m into the adjacent crop alley, suggesting that UVSs can influence nearby crop alley soil by adding living and dead organic matter. In addition, grass margins or other semi-natural habitats have often been described as over-wintering habitats for insects then able to colonize the field (Médiène et al. 2011), and the UVS has recently been shown to play the same role (Boinot et al. 2019). The specific impact of UVSs could be especially visible in young sites where tree impact is still limited.
A small number of studies have focused on soil organisms in alley cropping systems, with varying results. Cardinael et al. (2019) showed that earthworm density was enhanced in tree rows compared to crop alleys in 13 French silvoarable sites, but did not observe the gradient of earthworm density from the tree row towards the center of the crop alley that was described by Price and Gordon (1999) on a Canadian site. Abundance of soil invertebrates (0–6 mm in size) in the tree row and 1 m from the tree row was reported to be roughly double that observed more than 3 m from the tree row in an English site (Park et al. 1994), but mite abundance and species density were not related to distance from tree rows on another Canadian site (Doblas-Miranda et al. 2014). Microbial biomass has been reported to progressively decline from the tree row to the middle of the crop alley (Seiter et al. 1999), while it was not significantly different between tree rows and crop alleys in another study (Udawatta et al. 2008). Soil organic matter content, however, appears to be consistently increased in tree rows, and a role of the UVS itself in this increase has been suggested (Udawatta et al. 2008; Cardinael et al. 2017). To our knowledge, nothing is known about the variability of soil and vegetation characteristics in the UVS itself, particularly according to distance from the tree.
To investigate the changes in vegetation, soil organic carbon (SOC) and nutrient contents and the associated microorganisms and macro-invertebrates within the UVS and its vicinity in the crop alley, two young alley cropping systems were studied in the South of France. These systems presented UVSs sown with grasses of Festuca species. The sampling scheme aimed to take into account spatial heterogeneity within the UVS and between the UVS and the crop alley. Vegetation biomass and necromass from both UVSs and crops were measured, as well as microorganism biomass carbon, nitrogen and phosphorus. Studied macro-invertebrate parameters were density, diversity and functional community structure. We hypothesized that (1) soil fertility is increased in the UVS compared to the crop alley, (2) in the crop alley, soil fertility is particularly increased in the vicinity of the UVS, and (3) vegetation biomass and necromass in the UVS is negatively impacted by tree vicinity, creating spatial heterogeneity within the UVS.
Material and methods
Site description
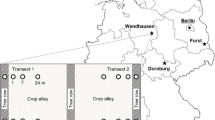
The study was conducted on two alley cropping sites in South-West France during spring of 2017. The aim was not to compare these sites, but to see if similar trends could be found in two different sites. Site A is a 5 ha plot located 6 km north-east of the town of Auch (43.682°, 0.620°), and was sampled at the beginning of March 2017. Site B is a 4.5 ha plot located 27 km east of site A (43.521°, 0.933°), and was sampled at the beginning of April 2017. This area presents an altered oceanic climate, with an annual temperature of 13 °C and an annual rainfall of 698 mm (mean values over the last 10 years for the Auch station, French Institute of Meteorology Météo France). Soil properties are presented in Table 1. In both sites, trees of 12 different species were planted in north–south oriented rows (Prunus avium (L.) L., Juglans regia L., Sorbus domestica L., Acer campestre L., Fraxinus excelsior L., Pyrus communis L., Quercus petraea (Matt.) Llebl., Ulmus ‘Nanguen’, Sorbus torminalis (L.) Crantz, Morus alba L., Tilia cordata Mill., Quercus pubescens Willd). A winter bread wheat crop (Triticum aestivum L.) was present in the crop alleys during sampling (sown in October 2016 for site A and December 2016 for site B). Both sites have been managed with reduced tillage for at least 15 years.
In site A, trees were planted in 2011 in 4 rows and spaced 5 m apart. A 2.3 m-wide UVS was sown after planting with Festuca rubra L.. Crop alleys are 36 m wide and were fertilized in February 2017 with a granular mineral fertilizer (7.5% of nitric nitrogen, 18.5% of ammoniacal nitrogen and 32.5% of sulfur trioxide) at the rate of 78 kg N ha−1.
In site B, trees were planted in 2007 in 7 rows and spaced 6–6.5 m apart. A 1.7 m-wide UVS was sown with Festuca arundinacea Schreb. after planting. Crop alleys are 20 m wide. This field has been managed according to organic farming practices since 2010, with no mineral fertilizers or pesticides.
Sampling design
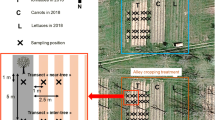
Four positions were defined as shown in Fig. 1. Two positions were sampled in the UVS, the first position at 1 m from the reference tree (UVS1) and the second at an equal distance between the reference tree and the next tree in the north direction (UVS2). Two other positions were sampled in the crop alley on a transect perpendicular to the UVS and starting from UVS2. The first position was 1 m from the UVS (CA1) and the second in the middle of the crop alley (CA2). There were 5 replications of this sampling design for each site. All analyses described below were carried out for each position and replicate for the 0–20 cm soil depth, as it was the maximum tillage depth reached more than 15 years ago, both sites being since under reduced tillage. All reference trees were of Prunus avium species.
Vegetation characteristics
Ground cover by vegetation and associated litter was determined in 1 × 1 m frames. Aboveground vegetation and associated litter were collected within 50 × 50 cm frames (Tadmor et al. 1975). Alive and dead roots, as well as litter incorporated into the soil, were collected in soil cores of 25 cm wide and 20 cm depth. Roots and litter found belowground were washed with water in order to remove residual soil. All vegetation and associated litter were then oven-dried one week at 65 °C to determine biomass and necromass. Roots that could be attributed to the tree were weighed separately. In the UVS of site A, a very dense mat of roots and belowground litter was present, and living fine roots could not be separated from dead roots and belowground litter.
SOC and nutrient contents
Soil was sampled from the previously described soil cores and air-dried for two days to reach around 10% of gravimetric water content. Soil was then sieved to 2 mm and stored at 4 °C.
SOC and total N contents were determined with an elemental analyzer in 15 mg samples of soil ground under 200 µm and oven-dried at 40 °C during 24 h (Thermo Fisher Scientific Flash 2000, sensitivity 0.5 µg). Available P was extracted from 10 g equivalent dry soil using the Olsen method, and measured by the Malachite green method (Ohno and Zibilske 1991) using an absorbance microplate reader at 630 nm (Dialab ELx808). Soil mineral N was extracted from 10 g equivalent dry soil with 40 mL of 1 M KCl solution and measured by continuous flow colorimetry (SKALAR SA 3000 flow analyser). Soil bulk density and soil fine-element bulk density (excluding particles bigger than 2 mm in diameter) were determined following the cylinder method (Blake and Hartge 1986) at two depths (0–10 cm and 10–20 cm soil layers). The SOC stock (Mg ha−1) in the 0–20 cm soil layer is the product of SOC content with soil fine-element mass, itself the product of fine-element bulk density and volume of the 0–20 cm layer. For each site, SOC stocks were adjusted to an equivalent soil mass basis (Ellert and Bettany 1995).
Microbial biomass
Microbial biomass C, N and P were determined using the fumigation-extraction method as described by Vance et al. (1987) for C, Brookes et al. (1985) for N and Brookes et al. (1982) for P, from soil sieved at 2 mm and stored at 4 °C. Briefly, around 10 g of equivalent dry soil were exposed to chloroform vapors during 24 h. Soluble C and N were extracted with 40 ml of K2SO4 solution at 0.025 M in fumigated and non-fumigated soils while soluble P was extracted with 200 ml of NaHCO3 at 0.5 M. The extracts were then shaken for one hour at ambient temperature and centrifuged at 4000 RPM. C and N contents were determined using a dissolved carbon analyzer (SHIMADZU TOC-V CSH/CSN, sensitivity at 5%) and P was determined using the Malachite green method (Ohno and Zibilske 1991) using an absorbance microplate reader at 630 nm (Dialab ELx808). Microbial biomass C, N and P per g of soil were obtained from the difference in soluble C, N and P between the fumigated and non-fumigated samples using an extraction efficiency coefficient for microbial biomass of 0.45 for C and N and 0.40 for P (Jenkinson et al. 2004).
Macro-invertebrate communities
Macro-invertebrates were hand-sorted (Smith et al. 2008b), identified and counted in previously described soil cores. We kept soil macrofauna in 70% ethanol.
All macro-invertebrates were at minimum identified to family level, except for Arachnida, Heteroptera, and Geophilomorpha that were identified to order level (see Table S1, online resources). Gastropoda, Isopoda and Dermaptera, as well as Polydesmus sp. and Lithobiomorpha, were identified to genus level according to Germain (1931), Vilvens et al. (2008), Vandel (1960) and Séchet and Noël (2015), respectively. Adult Lumbricidae and Carabidae were identified to species level (Jeannel 1942; Bouché 1972; Horellou 2006). Unidentified larvae were classified into morphological groups following Paulian (1956). We calculated density (number of individuals per m2) for each class level.
Taxonomic diversity was studied at the genus and family level, through taxonomic richness, Shannon–Weaver index and Pielou evenness index. In order to remove bias due to differences in macrofauna density, a rarefied taxonomic richness was calculated (Hurlbert 1971).
Functional community structure was studied through feeding group distribution. We classified macro-invertebrates into litter-feeder, animal-feeder and plant-feeder classes (see Table S1, online resources). Following Clough et al. (2007), recorded Staphylinidae subfamilies were all considered as animal-feeders. Carabidae were separated into animal-feeders and plant-feeders (seed-eating) following Honek et al. (2003). As earthworms presented large differences in size with other macro-invertebrates found, their functional community structure was studied separately. They were classified into endogeic, anecic and epigeic ecomorphological categories (see Table S1, online resources).
Statistical analyses
Two-way analyses of variance were used to test the effect of the interaction between factors position and site for studied variables (see Fig. S1, online resources for an overview of the data analysis strategy). When the interaction between site and position had a significant effect, we assumed that patterns of response to position differed between sites. Position effect was then tested for each site separately in a one-way analysis of variance. On the contrary, when the interaction between site and position had no significant effect, we assumed that patterns of response to the position were similar between sites. Position effect was then tested for both sites together on a linear mixed model, with site used as a random factor. For macro-invertebrate abundance data, generalized linear models fitted with the quasi Poisson law and generalized linear mixed models fitted with the binomial negative law were used instead of linear models as advised for discrete count data. Chi2 tests were performed on these models to determine significant effects of the factors studied. Post-hoc Tukey tests were performed for multiple comparisons. Significance levels were fixed at 5%. All analyses were performed using car, lme4 and vegan packages of R statistical software (version 3.3.2).
Results
Vegetation characteristics
Roots and litter were mostly from herbaceous vegetation, and tree roots were almost only present in UVS1 (Table 2). The interaction between the factors site and position was not significant for vegetation biomass and necromass (see Table S2, online resources). Aboveground and belowground vegetation biomass and necromass were significantly higher in the UVS than in the crop alley in both sites (Table 3). Ground cover was also higher in the UVS (between 90 and 100%) than in the crop alley (around 50%). In the UVS of site A, vegetation was almost only composed of Festuca rubra as other species represented less than 2% of the ground cover on average. In The UVS of site B, vegetation was almost only composed of Festuca arundinacea as other species represented less than 3% of the ground cover on average. No significant difference was found between the two positions of the UVS, or between the two positions of the crop alley for aboveground or belowground vegetation biomass and necromass (Table 3).
SOC and nutrients contents
Site A presented higher values for SOC and nutrient contents than site B (Table 2). However, the interaction between the factors site and position was only significant for mineral N content (see Table S2, online resources). SOC and Olsen P contents were significantly higher in the UVS than in the crop alley (Table 3). They were respectively 12% and 23% higher in the UVS than in the crop alley in site A, and respectively 11% and 106% higher in the UVS than in the crop alley in site B (Table 2). No significant difference was found between the two positions of the crop alley or between the two positions of the UVS for SOC and Olsen P contents (Table 3). A similar response was observed for SOC stock in the 0–20 cm soil layer (Table 3). Soil mineral N varied between positions for site A only, with increasing values from the UVS to the middle of the crop alley (Table 2).
Microbial biomass
Soil microbial biomass C was higher in site A (548 mg C kg−1 in the UVS and 476 mg C kg−1 in the crop alley) than in site B (405 mg C kg−1 in the UVS and 377 mg C kg−1 in the crop alley) as shown in Fig. 2. Microbial N was however similar in both sites (60 mg kg−1 in average in the UVS and 48 mg kg−1 in the crop alley) (Fig. 2). There was no significant interaction between the factors site and position on soil microbial biomass C, N and P (see Table S2, online resources). Microbial C and N were significantly higher only in UVS2 than in CA1 for both sites (Table 3). Similar patterns were observed for different substrate-induced microbial respirations measured on the same samples (presented in online resources, see Table S2, Table S4 and Fig. S2). Microbial P was not significantly different between positions (Table 3). It was around 13 mg kg−1 in site A and 9 mg kg−1 in site B (Fig. 2).
Mean values of soil microbial biomass C, N and P determined by the fumigation-extraction method for each position at site A and site B (0–20 cm depth). Error bars represent standard error. Positions UVS1 and UVS2 are in the understory vegetation strip (at 1 m from the tree and at equal distance between two trees respectively). Positions CA1 and CA2 are in the crop alley (at 1 m from the understory vegetation strip and in the middle of the crop alley respectively)
Macro-invertebrate communities
Macro-invertebrate density was similar in both sites, with 514 individuals per m2 on average in site A and 603 individuals per m2 on average in site B (Table 4). The interaction between the factors site and position was significant (see Table S2, online resources). Indeed, macro-invertebrate density was significantly lower in CA1 than in the UVS in site A (p value < 0.001), but not in site B (Table 4).
No difference was observed between positions at the family level for rarefied taxonomic richness and Shannon–Weaver index. In both sites, rarefied taxonomic richness index and Shannon–Weaver index at the genus level were significantly higher in the UVS than in CA1 (p value < 0.05) (see Table S3, online resources). Pielou evenness index did not vary with position, either at genus or at the family level (see Table S3, online resources).
The interaction between the factors site and position was significant for earthworm densities (see Table S2, online resources). In site A, mean earthworm density was significantly lower in CA1 than in CA2 (p value < 0.05), while in site B no significant difference was found (Table 4). Epigeic earthworms were scarce and found only in the UVS of site B (Fig. 3). Endogeic earthworms dominated in the crop alley in site A, and in all positions in site B. The percentage of endogeics in the earthworm density was significantly lower in the UVS (47% in site A and 70% in site B) compared to the crop alley (85% in site A and 87% in site B). Oppositely, the percentage of anecics in earthworm density was significantly higher in the UVS (53% in site A and 25% in site B), compared to the crop alley (15% in site A and 13% in site B). In site A, the shift was abrupt between the UVS and the crop alley. In site B, this shift was more progressive with the suggestion of intermediate values in CA1 (Fig. 3).
Percentage of earthworms belonging to endogeic, anecic and epigeic categories for each position and each site. A significant effect of position after Chi2 tests on linear mixed models (site used as a random factor) is indicated by asterisks. Significance level is 5%. A significant effect is due to significant differences between the understory vegetation strip and the crop alley (after a Tukey multiple comparison test). Positions UVS1 and UVS2 are in the understory vegetation strip (at 1 m from the tree and at equal distance between two trees respectively). Positions CA1 and CA2 are in the crop alley (at 1 m from the understory vegetation strip and in the middle of the crop alley respectively). **p value < 0.01, ***p value < 0.001
Woodlice were the only other taxon at class level to present significant differences in density between positions. Woodlice presented a significantly higher density in the UVS compared to the crop alley in both sites (p value < 0.001, Table 4). Feeding group distribution (earthworms excepted) was different between the UVS and the crop alley for site A (Fig. 4). This was due to a significant increase in the percentage of plant-feeders in CA2 compared to the UVS which was dominated by litter-feeders. Position CA1 presented intermediate values. For site B, the feeding group distribution of the macro-invertebrate community was not clearly distinct between positions, except for position CA1 that showed a lower variability and was almost entirely composed of animal-feeders (Fig. 4).
Feeding group distribution of the macro-invertebrate community (earthworms excluded) in each site. Percentages of individuals belonging to three feeding groups can be read on the triangle sides. Positions are represented by the polygons, with individual samples being the corners of the polygons. Points represent median values for each position. Positions UVS1 and UVS2 are in the understory vegetation strip (at 1 m from the tree and at equal distance between two trees respectively). Positions CA1 and CA2 are in the crop alley (at 1 m from the understory vegetation strip and in the middle of the crop alley respectively). *Significant effect of position after a Chi2 test on a linear mixed model (site used as a random factor) (p value < 0.05)
Discussion
Vegetation characteristics of sown UVSs
While the herbaceous vegetation was expected to be visibly affected by tree root competition close to the tree, the UVS biomass and necromass did not vary with distance from the nearest tree. This is in agreement with results of Battie-Laclau et al. (2019) who measured herbaceous root length density in two young French alley cropping sites, including site B of this study. This could be explained because both sites were sown just after tree plantation with Festuca sp. It colonized more than 90% of the ground cover, underlying possible competitiveness. Festuca rubra, planted in site A, is particularly known to form a dense network of vegetation at the soil surface and to tolerate shade and stress (Bergkvist et al. 2010). In addition, root biomass and root length density of grass buffers planted with three herbaceous species, including the species planted in site B Festuca arundinacea, were not affected by the presence or absence of 6 year-old trees in Missouri (Kumar et al. 2010). Competition with grass species has been shown to induce a deeper tree rooting system in a French silvopasture planted with young Prunus avium trees (Dawson et al. 2001). Grass species planted in UVSs could have outcompeted trees in the 20 first soil centimeters in the present study. However, sampling took place in early Spring while the vegetation usually reaches a peak in biomass around late Spring. This experiment does not allow to conclude on the effect of tree distance on the maximum growth of vegetation of the UVS.
Do soil fertility and organisms under UVSs present specific properties?
SOC contents in the 0–20 cm soil layer of 14.2 g kg−1 and 13.2 g kg−1 (in site A and B respectively) for the crop alley, and of 16 g kg−1 and 14.5 g kg−1 (in site A and B respectively) for the UVS were consistent with values found by Cardinael et al. (2017) for six year-old French alley cropping sites. Similarly to this study, they found significantly less SOC in the crop alley compared to the UVS in the first 10 soil cm, as did Udawatta et al. (2008) in a Missouri site. SOC content and stocks are long-term indicators because they change slowly (Powlson et al. 1987) and reflect changes over years to decades. These results could indicate an early legacy effect of the accumulation of non-exported organic matter from the herbaceous vegetation. In agreement with that suggestion, Van Vooren et al. (2017) found in a meta-analysis an increase in soil carbon stock by 25% in grassy field margins compared to adjacent fields, as did Cardinali et al. for SOC content (2014). In addition, grassy field margins also strongly reduce erosion (Van Vooren et al. 2017), which could reduce carbon losses. The UVS alone is thus likely able to drive an increase in SOC content.
Interestingly, soil microbial biomass C, N and P did not differ significantly between the UVS and the middle of the crop alley, as microbial respiration. This is surprising as UVSs and crop alleys differ in terms of quantity and quality of organic matter additions to the soil, which are known to affect microorganism biomass and activity (Porazinska et al. 2003; Liang et al. 2017). Contrary to us, Beuschel et al. (2019) found an increase of 59% in microbial biomass C and respiration in the UVS compared to the crop alley for the first 5 soil centimeters. The increase in microbial biomass could be restricted to the very first centimeters, and the effect may have been diluted by the higher depth sampled here. Guillot et al. (2019) reported higher values in the UVS compared to the crop alley for microbial biomass C, N and P for the 15 first cm depth in a 20 year-old alley cropping site in the south of France, meaning an effect on a higher depth could be visible for older sites. They found similar values to this study in the UVS (427 mg C kg−1), but values were divided by two in the crop area in their study compared to this study (219 mg C kg−1). The effect of the UVS could be more visible in soils initially poor in microbial biomass, where even a slight increase in organic matter addition to the soil would increase microbial growth, while in soils already rich in microbial biomass an increase in organic matter addition could have only little effect.
Similarly, macro-invertebrate density, taxonomic richness and diversity were not significantly higher in the UVS positions than in the middle of the crop alley. The high variability in macro-invertebrate density could explain this lack of significance. However, observed patterns suggest a decrease in macro-invertebrate density in the crop alley, with densities decreasing from 682 individuals per m2 in the UVS to 464 individuals per m2 in CA2 in site A, and from 629 individuals per m2 to 470 individuals per m2 in site B. In agreement with this suggestion, an overall increase in macro-invertebrate density (Smith et al. 2008c) and diversity (Smith et al. 2008a) was shown in grassy field margins compared to crop fields in the UK with a similar method. Woodlice, which are litter-feeders, were the only taxonomic group with significantly higher abundance in the UVS than in the crop alley in sites studied. Hassall and Tuck (2007) explained that woodlice shelter in buffered microclimates because they are sensitive to rapid drops in temperature and seek to avoid desiccation. UVSs, with a dense ground cover, large inputs of organic matter to the soil and less disturbance than the crop alley, could be interesting habitats for woodlice. The effect of the UVS in changing macro-invertebrate functional community structure is emphasized in site A, which presented a higher percentage of plant-feeders in crop alleys and of litter-feeders in UVSs. This was less obvious in site B, where macro-invertebrate feeding group distribution was highly variable in the center of the crop alley, maybe due to the highly heterogeneous distribution in this site of Staphylinidae animal-feeders capable of flight dispersal.
Mean values of 190 and 382 earthworms per m2 were found in the crop alley for sites A and B respectively. The values found by Cardinael et al. (2019) in different agroforestry plots in France were quite similar with 289 ± 85 earthworms per m2. No significant differences were found in earthworm densities between the UVS and the middle of the crop alley, even if the middle of the crop alley presented a trend of lower values in site B. These results are in contradiction with several studies (Crittenden et al. 2015; Cardinael et al. 2019) that found higher earthworm densities in field margins or UVSs than in crop fields. However, Lagerlöf et al. (2002) found higher earthworm density in a Swedish field than in field margins planted with grasses and forbs. Smith et al. (2008a, c) also found variable results for earthworm densities between grassy field margins and crop fields.
Earthworm distribution in ecomorphological categories was affected by position, with a shift from anecic and epigeic worms in the UVS to endogeic worms in the crop alley. As anecic and epigeic earthworms feed on surface plant litter, they are probably favored by the high ground cover and vegetation biomass in the UVS. Soil tillage in the crop alleys may be negatively impacting anecic earthworms but increasing the incorporation of residues and the consequent population of endogeic earthworms, such as was observed in crop alleys in the present study, in agreement with Chan (2001).
Do UVSs influence soil fertility and organisms in their vicinity?
The two crop alley positions had similar SOC contents, irrespective of their distance from the UVS. Pardon et al. (2017) found higher SOC values close to old boundary UVSs in Belgium and Bambrick et al. (2010) found an increase in SOC content with UVS proximity in Quebec and Ontario in fields of less than 10 years, but only up to 0.75 m from the UVS. In addition Pardon et al. (2017) found no effect of UVS distance on SOC values in young alley cropping sites. The sites studied could still be too young to detect any effect of UVS proximity. In addition, D’Acunto et al. (2014) found that the SOC stock increased close to woody field margins but not close to grassy field margins, suggesting a limited effect of herbaceous vegetation alone on soil in its vicinity.
Microbial biomass C and N were higher in the UVS compared to the closest position in the crop alley, as microbial respiration. This means that 1 m away from the UVS, there was a specific decrease in microorganism biomass compared to the UVS. Nii-Annang et al. (2009) did not find any variation of microbial biomass and respiration between UVS and crop alley positions in a German 9 year-old agroforestry plot, and similar results were found in German sites from 5 to 8 years old (Beuschel et al. 2019). The decrease in microorganism biomass 1 m away from the UVS could be restricted to the close vicinity of the UVS, or to site-specific conditions. In site A, mineral nitrogen fertilizer was applied. Lower values were found for mineral N close to the UVS compared to the middle of the crop alley. This could be due to competition for nutrients between the crop, the UVS, and to a lesser extent the tree. Gikas et al. (2016) suggested that mineral N is taken up by the tree for its growth in its vicinity in Greek agroforestry plots. However, there was no additional roots close to the UVS in site A. The decrease in mineral N 1 m away from the UVS could also be due to an edge effect, with less mineral N applied in the edges of the crop alley. This edge effect is also likely to happen for other amendments, like manure, and for soil tillage. This could explain the decrease observed for microbial parameters 1 m away from the UVS for site A. This edge effect is supported by several studies performed at harvest time (Chirko et al. 1996; Peng et al. 2009; Battie-Laclau et al. 2019; Swieter et al. 2019) which found a reduction in aboveground biomass crop yield close to the UVS in young alley cropping systems.
Macro-invertebrate density was lower 1 m away from the UVS compared to the UVS in site A. This was particularly due to a decrease in earthworm density in this position. Movements of earthworms by density-dependent or low-quality habitat dispersal were described by Mathieu et al. (2010) in mesocosm experiments. Macro-invertebrates could be attracted by the lack of perturbations, higher ground cover and higher resource availability in the UVS, thus potentially leading to lower densities in its vicinity as observed in site A. In site B, earthworm distribution and density 1 m away from the UVS was intermediate between that of the UVS and of the middle of the crop alley. This is in agreement with Price and Gordon (1999), who noted a decrease in earthworm abundance in a transect leading from the UVS to the center of the crop alley in an 11 year-old plot in Ontario. In this site, high densities in the UVS could have induced movements of earthworms in their vicinity. The dynamics of macro-invertebrate communities and the potential role of the UVS as a sink or a source for soil fauna is thus a key element to study in order to understand spatial variation in soil functioning in alley cropping systems. In addition, soil organisms and particularly macro-invertebrates present seasonal variations, as does the herbaceous vegetation. The results of this study suggest a possible response of these parameters for early Spring, but the effects of the UVS could depend on the season, due to changes in temperature, soil moisture, and respective development of the crop and UVS vegetation.
Conclusions
Sown UVSs induced a global homogeneity in the 20 first soil centimeters of the tree rows of two young temperate alley cropping systems, independently of tree distance. This herbaceous vegetation provided an undisturbed environment with a high ground cover, which presented an increased SOC content compared to the crop alleys. These UVSs hosted a large and specific community of soil macro-invertebrates, especially surface litter-feeders sensitive to disturbance (woodlice, epigeic and anecic earthworms). However, we did not detect a strong increase in soil microbial biomass and activity in the UVS, maybe because microbial parameters were impacted by UVS presence in the very first soil centimeters only. UVS vicinity negatively impacted microbial parameters while macro-invertebrates presented variable answers to UVS proximity within the crop alley. It remains unclear if this edge effect is due to agricultural practices or to the tree row vegetation. The role of tree rows as sources or sinks for organisms requires clarification, pleading for studies of seasonal dynamics according to distance from the UVS.
References
Bambrick AD, Whalen JK, Bradley RL et al (2010) Spatial heterogeneity of soil organic carbon in tree-based intercropping systems in Quebec and Ontario, Canada. Agrofor Syst 79:343–353. https://doi.org/10.1007/s10457-010-9305-z
Battie-Laclau P, Taschen E, Plassard C et al (2019) Role of trees and herbaceous vegetation beneath trees in maintaining arbuscular mycorrhizal communities in temperate alley cropping systems. Plant Soil. https://doi.org/10.1007/s11104-019-04181-z
Bergkvist G, Adler A, Hansson M, Weih M (2010) Red fescue undersown in winter wheat suppresses Elytrigia repens. Weed Res 50:447–455. https://doi.org/10.1111/j.1365-3180.2010.00789.x
Beuschel R, Piepho H-P, Joergensen RG, Wachendorf C (2019) Similar spatial patterns of soil quality indicators in three poplar-based silvo-arable alley cropping systems in Germany. Biol Fertil Soils 55:1–14. https://doi.org/10.1007/s00374-018-1324-3
Blake GR, Hartge KH (1986) Bulk density. In: Society of Agronomy—Soil Science Society of America A (ed) Methods of soil analysis, part 1. Physical and mineralogical methods—Agronomy monograph no. 9, 2nd edn. Madison, Wisconsin, pp 363–375
Boinot S, Poulmarc’h J, Mézière D et al (2019) Distribution of overwintering invertebrates in temperate agroforestry systems: implications for biodiversity conservation and biological control of crop pests. Agric Ecosyst Environ 285:1–11. https://doi.org/10.1016/j.agee.2019.106630
Bouché MB (1972) Lombriciens de France: écologie et systématique. Inra, Paris
Brookes PC, Powlson DS, Jenkinson DS (1982) Measurement of microbial biomass phosphorus in soil. Soil Biol Biochem 14:319–329. https://doi.org/10.1016/0038-0717(82)90001-3
Brookes PC, Landman A, Prudent G et al (1985) Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842. https://doi.org/10.1016/0038-0717(85)90144-0
Cardinael R, Chevallier T, Cambou A et al (2017) Increased soil organic carbon stocks under agroforestry: a survey of six different sites in France. Agric Ecosyst Environ 236:243–255. https://doi.org/10.1016/j.agee.2016.12.011
Cardinael R, Hoeffner K, Chenu C et al (2019) Spatial variation of earthworm communities and soil organic carbon in temperate agroforestry. Biol Fertil Soils 55:171–183. https://doi.org/10.1007/s00374-018-1332-3
Cardinali A, Carletti P, Nardi S, Zanin G (2014) Design of riparian buffer strips affects soil quality parameters. Appl Soil Ecol 80:67–76. https://doi.org/10.1016/j.apsoil.2014.04.003
Chan KY (2001) An overview of some tillage impacts on earthworm population abundance and diversity—implications for functioning in soils. Soil Tillage Res 57:179–191. https://doi.org/10.1016/S0167-1987(00)00173-2
Chirko CP, Gold MA, Nguyen PV, Jiang JP (1996) Influence of orientation on wheat yield and photosynthetic photon flux density (Q(p)) at the tree and crop interface in a Paulownia-wheat intercropping system. For Ecol Manag 89:149–156. https://doi.org/10.1016/S0378-1127(96)03853-4
Clough Y, Kruess A, Tscharntke T (2007) Organic versus conventional arable farming systems: functional grouping helps understand staphylinid response. Agric Ecosyst Environ 118:285–290. https://doi.org/10.1016/j.agee.2006.05.028
Critchley CNR, Fowbert JA, Sherwood AJ, Pywell RF (2006) Vegetation development of sown grass margins in arable fields under a countrywide agri-environment scheme. Biol Conserv 132:1–11. https://doi.org/10.1016/j.biocon.2006.03.007
Crittenden SJ, Huerta E, de Goede RGM, Pulleman MM (2015) Earthworm assemblages as affected by field margin strips and tillage intensity: an on-farm approach. Eur J Soil Biol 66:49–56. https://doi.org/10.1016/j.ejsobi.2014.11.007
D’Acunto L, Semmartin M, Ghersa CM (2014) Uncropped field margins to mitigate soil carbon losses in agricultural landscapes. Agric Ecosyst Environ 183:60–68. https://doi.org/10.1016/j.agee.2013.10.022
Dawson LA, Duff EI, Campbell CD, Hirst DJ (2001) Depth distribution of cherry (Prunus avium L.) tree roots as influenced by grass root competition. Plant Soil 231:11–19. https://doi.org/10.1023/A:1010383506446
Doblas-Miranda E, Paquette A, Work TT (2014) Intercropping trees’ effect on soil oribatid diversity in agro-ecosystems. Agrofor Syst 88:671–678. https://doi.org/10.1007/s10457-014-9680-y
Dyer LA, Letourneau D (2003) Top-down and bottom-up diversity cascades in detrital vs. living food webs. Ecol Lett 6:60–68. https://doi.org/10.1046/j.1461-0248.2003.00398.x
Eichhorn MP, Paris P, Herzog F et al (2006) Silvoarable systems in Europe—past, present and future prospects. Agrofor Syst 67:29–50. https://doi.org/10.1007/s10457-005-1111-7
Ellert BH, Bettany JR (1995) Calculation of organic matter and nutrients stored in soils under contrasting management regimes. Can J Soil Sci 75:529–538. https://doi.org/10.4141/cjss95-075
Germain L (1931) Faune de France 21–22. Mollusques terrestres et fluviatiles, Lechevallier, Paris
Gikas GD, Tsihrintzis VA, Sykas D (2016) Effect of trees on the reduction of nutrient concentrations in the soils of cultivated areas. Environ Monit Assess 188:327. https://doi.org/10.1007/s10661-016-5325-0
Guillot E, Hinsinger P, Dufour L et al (2019) With or without trees: resistance and resilience of soil microbial communities to drought and heat stress in a Mediterranean agroforestry system. Soil Biol Biochem 129:122–135. https://doi.org/10.1016/j.soilbio.2018.11.011
Hassall M, Tuck JM (2007) Sheltering behavior of terrestrial isopods in grasslands. Invertebr Biol 126:46–56. https://doi.org/10.1111/j.1744-7410.2007.00075.x
Honek A, Martinkova Z, Jarosik V (2003) Ground beetles (Carabidae) as seed predators. Eur J Entomol 100:531–544. https://doi.org/10.14411/eje.2003.081
Horellou A (2006) Les Amara de France continentale et de Corse
Hurlbert SH (1971) The nonconcept of species diversity: a critique and alternative parameters. Ecology 52:557–586. https://doi.org/10.1016/S0140-6736(00)02805-1
Jeannel R (1942) Faune de France 39–40. Coléoptères Carabiques - Tomes I et II, Lechevallier, Paris
Jenkinson DS, Brookes PC, Powlson DS (2004) Measuring soil microbial biomass. Soil Biol Biochem 36:5–7. https://doi.org/10.1016/j.soilbio.2003.10.002
Kumar S, Udawatta RP, Anderson SH (2010) Root length density and carbon content of agroforestry and grass buffers under grazed pasture systems in a Hapludalf. Agrofor Syst 80:85–96. https://doi.org/10.1007/s10457-010-9312-0
Lagerlöf J, Goffre B, Vincent C (2002) The importance of field boundaries for earthworms (Lumbricidae) in the Swedish agricultural landscape. Agric Ecosyst Environ 89:91–103. https://doi.org/10.1016/S0167-8809(01)00321-8
Liang X, Yuan J, Yang E, Meng J (2017) Responses of soil organic carbon decomposition and microbial community to the addition of plant residues with different C:N ratio. Eur J Soil Biol 82:50–55. https://doi.org/10.1016/j.ejsobi.2017.08.005
Marsden C, Martin-Chave A, Cortet J et al (2019) How agroforestry systems influence soil fauna and their functions—a review. Plant Soil. https://doi.org/10.1007/s11104-019-04322-4
Mathieu J, Barot S, Blouin M et al (2010) Habitat quality, conspecific density, and habitat pre-use affect the dispersal behaviour of two earthworm species, Aporrectodea icterica and Dendrobaena veneta, in a mesocosm experiment. Soil Biol Biochem 42:203–209. https://doi.org/10.1016/j.soilbio.2009.10.018
Médiène S, Valantin-Morison M, Sarthou JP et al (2011) Agroecosystem management and biotic interactions: a review. Agron Sustain Dev 31:491–514. https://doi.org/10.1007/s13593-011-0009-1
Nii-Annang S, Grünewald H, Freese D et al (2009) Microbial activity, organic C accumulation and 13C abundance in soils under alley cropping systems after 9 years of recultivation of quaternary deposits. Biol Fertil Soils 45:531–538. https://doi.org/10.1007/s00374-009-0360-4
Ohno T, Zibilske LM (1991) Determination of low concentrations of phosphorus in soil extracts using Malachite green. Soil Sci Soc Am 55:892–895. https://doi.org/10.2136/sssaj1991.03615995005500030046x
Pardon P, Reubens B, Reheul D et al (2017) Trees increase soil organic carbon and nutrient availability in temperate agroforestry systems. Agric Ecosyst Environ 247:98–111. https://doi.org/10.1016/j.agee.2017.06.018
Park J, Newman SM, Cousins SH (1994) The effects of poplar (P. trichocarpa X deltoides) on soil biological properties in a sylvoarable system. Agrofor Syst 25:111–118. https://doi.org/10.1007/BF00705671
Paulian R (1956) Atlas des larves d’insectes de France. Boubée N. & Cie, Paris
Peng X, Zhang Y, Cai J et al (2009) Photosynthesis, growth and yield of soybean and maize in a tree-based agroforestry intercropping system on the Loess Plateau. Agrofor Syst 76:569–577. https://doi.org/10.1007/s10457-009-9227-9
Porazinska DL, Bardgett RD, Blaauw MB et al (2003) Relationships at the aboveground-belowground interface: plants, soil biota, and soil processes. Ecol Monogr 73:377–395. https://doi.org/10.1890/0012-9615(2003)073[0377:RATAIP]2.0.CO;2
Powlson DS, Prookes PC, Christensen BT et al (1987) Measurement of soil microbial biomass provides an early indication of changes in total soil organic matter due to straw incorporation. Soil Biol Biochem 19:159–164. https://doi.org/10.1016/0038-0717(87)90076-9
Price GW, Gordon AM (1999) Spatial and temporal distribution of earthworms in a temperate intercropping system in southern Ontario, Canada. Agrofor Syst 44:141–149. https://doi.org/10.1023/A:1006213603150
Séchet E, Noël F (2015) Catalogue commenté des crustacés Isopodes terrestres de la France métropoloitaine (Crustacea, Isopoda, Oniscidea) Mémoires de la Société Linnéenne de Bordeaux, tome 16. Société linnéenne de Bordeaux, Bordeaux
Seiter S, Ingham ER, William RD (1999) Dynamics of soil fungal and bacterial biomass in a temperate climate alley cropping system. Appl Soil Ecol 12:139–147. https://doi.org/10.1016/S0929-1393(98)00163-2
Smith J, Potts S, Eggleton P (2008a) The value of sown grass margins for enhancing soil macrofaunal biodiversity in arable systems. Agric Ecosyst Environ 127:119–125. https://doi.org/10.1016/j.agee.2008.03.008
Smith J, Potts S, Eggleton P (2008b) Evaluating the efficiency of sampling methods in assessing soil macrofauna communities in arable systems. Eur J Soil Biol 44:271–276. https://doi.org/10.1016/j.ejsobi.2008.02.002
Smith J, Potts SG, Woodcock BA, Eggleton P (2008c) Can arable field margins be managed to enhance their biodiversity, conservation and functional value for soil macrofauna ? J Appl Ecol 45:269–278. https://doi.org/10.1111/j.1365-2664.2007.01433.x
Swieter A, Langhof M, Lamerre J, Greef JM (2019) Long-term yields of oilseed rape and winter wheat in a short rotation alley cropping agroforestry system. Agrofor Syst 93:1853–1864. https://doi.org/10.1007/s10457-018-0288-5
Tadmor NH, Brieghet A, Noy-meir I et al (1975) An evaluation of the calibrated weight-estimate method for measuring production in annual vegetation. J Range Manag 28:65–69. https://doi.org/10.2307/3897582
Udawatta RP, Kremer RJ, Adamson BW, Anderson SH (2008) Variations in soil aggregate stability and enzyme activities in a temperate agroforestry practice. Appl Soil Ecol 39:153–160. https://doi.org/10.1016/j.apsoil.2007.12.002
Van Vooren L, Reubens B, Broekx S et al (2017) Ecosystem service delivery of agri-environment measures: A synthesis for hedgerows and grass strips on arable land. Agric Ecosyst Environ 244:32–51. https://doi.org/10.1016/j.agee.2017.04.015
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707. https://doi.org/10.1016/0038-0717(87)90052-6
Vandel A (1960) Faune de France 64–66. Isopodes terrestres, Lechevallier, Paris
Varah A, Jones H, Smith J, Potts SG (2013) Enhanced biodiversity and pollination in UK agroforestry systems. J Sci Food Agric 93:2073–2075. https://doi.org/10.1002/jsfa.6148
Vilvens C, Maree B, Meulemane B et al (2008) Mollusques terrestres et dulcicoles de Belgique. Société Belge de Malacologie, Tomes I, II, III
Acknowledgements
We thank Carlos Trives, Josiane Abadie, Nancy Rakotondrazafy, Jean-Marc Souquet, Jean Larvy-Delarivière and Joëlle Toucet for their technical assistance in fieldwork and laboratory analyses. We are grateful to Jack De Lozzo and Vincent Blagny for allowing us to sample their fields. Camille D’Hervilly’s Ph.D. thesis was funded by La Fondation de France, and analyses were funded by the Eco&Sols research unit.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
D’Hervilly, C., Marsden, C., Hedde, M. et al. Sown understory vegetation strips impact soil chemical fertility, associated microorganisms and macro-invertebrates in two temperate alley cropping systems. Agroforest Syst 94, 1851–1864 (2020). https://doi.org/10.1007/s10457-020-00501-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-020-00501-w