Abstract
Different sources of nitrogen pose diverse effects to algal community, but the mechanism of inhibitory effects of nitrogen sources on freshwater diatoms is not fully understood. The purpose of this study was to compare biomass, photosynthetic activity, and morphological structure of three common freshwater diatoms (Cyclotella meneghiniana, Nitzschia sp., and Gomphonema parvulum) under different nitrogen sources (NO3 − or NH4 +). The sorption characteristic of each diatom was investigated, and chlorophyll a (Chl-a) content and oxygen evolution rate were analyzed to investigate stress of different nitrogen sources on each diatom in the batch experiments. Ammonium lowered the growth rate of C. meneghiniana and Nitzschia sp. when it was supplied in addition to growth-saturating nitrate concentrations, suggesting a combined effect of inhibition of nitrate uptake and direct ammonium stress. Oxygen evolution rate of Nitzschia sp. showed that the direct ammonium stress on the photosynthetic activity can be alleviated by coexistence of nitrate in the nitrogen enriched treatment, but not for C. meneghiniana and G. parvulum, which may be caused by a different nitrate transporter system within algal cells. Transmission electron microscopy was used to assess the toxicity of ammonium on ultrastructural chloroplast of each diatom. Ultrastructural changes in chloroplasts showed undefined electron-dense granules and lipid droplets, but the membrane integrity of cell was maintained, suggesting an adaptation to adjustment to ammonia stress. Results showed that Cyclotella meneghiniana and Nitzschia sp. were more sensitive to ammonium stress than Gomphonema parvulum on growth, but the mechanism remains unclear.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The outbreak of cyanobacteria blooms in Taihu Lake has attracted worldwide attention on the eutrophication problems in China (Guo 2007). Ammonia concentration in Taihu Lake reached 1.77 mg/L before cyanobacteria blooms (Zhang et al. 2011), and ammonia contamination observed in channel segment of Grand Canal at the downstream of Taihu Lake was significantly higher than any other part, with a maximum concentration of 7.18 mg/L during the wet season (Wang et al. 2010). Dai et al. (2012) experimentally compared more than a dozen common phytoplankton species’ responses to ammonia stress and suggested that phytoplankton species succession in shallow lakes may be associated with their tolerance to ammonia stress. The high concentration of ammonia, typically observed in spring in shallow lakes in China (the average and maximum ammonia concentration were 1.12 mg/L, and 10.08 mg/L), may inhibit the growth of some bloom-forming cyanobacteria taxa (e.g., Microcystis aeruginosa), while the same nutrient in low concentration in summer is often associated with cyanobacteria blooms (Dai et al. 2012; Ni et al. 2012). Nitrate can also enhance as well as inhibit the growth of M. aeruginosa and reduce its photosynthesis under certain high concentrations (from 49.98 to 300.02 mg/L) (Chen et al. 2009). In contrast, stress of different nitrogen sources on diatoms is not as well known as cyanobacteria (but see Dai et al. 2012). The use of diatoms as water quality indicators has been reported by a number of earlier studies (Quinlan et al. 2011). The nutrient conditions of different water bodies could have a distinct effect on the abundance of specific diatom species, because tolerance differences exist among various species (Bennion et al. 1996; Leland et al. 2001). For instance, Gomphonema parvulum was found to be dominant in hypereutrophic lakes, Melosira varians and Synedra spp., dominated the community in eutrophic water bodies (Pei et al. 2010), and M. varians was sensitive to organic water pollution (Szczepocka and Szulc 2009).
Few studies paid attention to the effects of physical and chemical parameters on photosynthetic activities of diatoms. Recently, ammonia (NH3) has been confirmed as an inhibitory of O2 evolution at alkaline pH, by interacting with carboxylate groups coupled to the Mn4CaO5 cluster as direct ligands or proton transfer mediators, causing inhibition of O2 evolving reaction in the center of Photosystem II(Tsuno et al. 2011; Hou et al. 2011). However, an understanding of the effect of ammonia on the photosynthetic rate of various freshwater diatoms remains unknown. Clonal cultures of algae are widely used in laboratory experiments and have contributed greatly to our knowledge of microbial ecosystems (Thessen et al. 2009). Among the multitude of diatom species, Gomphonema parvulum and Cyclotella meneghiniana represent the most widely distributed species (Swift and Wheeler 1991; Rimet et al. 2009), both G. parvulum and C. meneghiniana have been frequently observed as dominant species in eutrophic water bodies (Finlay et al. 2002; Rimet et al. 2009; Pei et al. 2010), C. meneghiniana has even been studied as a structure template in material science (Lenoci and Camp 2008). Because many physiological characteristics vary drastically among different species, Nitzschia sp., G. parvulum, and C. meneghiniana, all dominants in Chinese water bodies were chosen as representatives to investigate the effect of ammonia on photosystem and O2 evolving reaction of freshwater diatoms.
The purpose of this study was to compare sensitivity of three common freshwater diatoms (Cyclotella meneghiniana, Nitzschia sp. and Gomphonema parvulum) by measuring their sorption behavior, photosynthetic activity, and ultrastructure under different nitrogen sources (NO3 − and NH4 +). O2 evolving rate was employed as an indicator of the toxicity of ammonium on the photosystem of each diatom, and transmission electron microscopy (TEM) was further used to assess the ultrastructural chloroplast changes of each diatom under ammonia stress.
Experiments
Cultivation of algae
Nitzschia sp., Gomphonema parvulum, and Cyclotella meneghiniana were obtained from the Freshwater Algae Culture Collection of the Institute of Hydrobiology, the Chinese Academy of Sciences (Wuhan, Hubei, P. R. China). All experiments were carried out on batch cultures of Nitzschia sp., G. parvulum, and C. meneghiniana, grown in a modified form of D1 medium, containing 20.0 mg/L nitrogen in the form of either NaNO3 or NH4Cl, or 20.0 mg/L NaNO3 and 20.0 mg/L NH4Cl. For the control treatment, the medium was prepared with NaCl only. As the standard D1 medium contains 20.0 mg/L nitrogen in the form of NaNO3, this concentration was used to set up the experiments. All batch experiments were performed in 500 mL Erlenmeyer flasks in triplicate for each treatment. These three selected species were inoculated into the experimental flasks with 200 mL autoclaved diatom D1 medium and maintained at 22 °C under a photon flux density of 40 μmol photons/m2/s, with a cycle of 12 h light : 12 h dark. The initial cell concentration of each diatom in the batch cultures was about 5 × 104 cells/mL. Exponentially growing cultures in D1 medium, at a cell density about 50 × 106 cells/mL, were pelleted by centrifugation at 4,000 rpm for 15 min, washed twice with nitrogen-free medium, and then transferred to an equal volume of different nitrogen treated medium, respectively (Table 1).
Measurements of chlorophyll a and nitrogen concentrations
Samples for Chl-a measurements under various nitrogen sources were collected every 3 days from the batch cultures, with a study period of 15 days. An aliquot of 15 mL were filtered through 0.45 μm Whatman AH glass fiber filters, held frozen overnight prior to extraction using ethanol (Jespersen and Christoffersen, 1987). Chl-a was measured spectrophotometrically at 665 nm and 750 nm using a Shimadzu UV2450 spectrophotometer.
Samples for nitrogen (nitrate/nitrite/ammonia) measurements under various nitrogen sources were collected every 3 days from the batch cultures, with a study period of 15 days. An aliquot of 5 mL were filtered through 0.45 μm Whatman AH glass fiber filters, and the concentration of nitrate/nitrite/ammonia in the medium was then measured using a HACH DR890 analyzer (Ni et al. 2012).
Measurements of photosynthetic oxygen evolution
Effects on the photosynthetic activity of three typical freshwater diatoms were determined using a Clark-type oxygen electrode (Oxygraph, Hansatech, UK) at 22 °C with 40 μmol photons/m2/s of photosynthetically active radiation (Petit et al. 2010). Two mL of the fivefold concentrated algal suspension was transferred into an electrode unit, and oxygen evolution rate (nmol/mL/min) was determined according to the detected signal. Prior to each measurement, the samples were adapted for 2 min before the oxygen evolution rate reached a stable level, and then the increase in oxygen concentration was recorded during the next 10 min. For each measurement, the result was corrected for biomass of viable cells and expressed as μmol mg/(Chl-a)/h.
Characterization of ultrastructure of chloroplast
Double fixation
The specimen was first fixed with 2.5 % glutaraldehyde in phosphate buffer (0.1 M, pH 7.0) for more than 4 h then washed three times in the phosphate buffer for 15 min, respectively. The specimen was then postfixed with 1 % OsO4 in the phosphate buffer for 1 h and washed three times in the phosphate buffer for 15 min, respectively.
Dehydration
The specimen was first dehydrated by a graded series of mixed acetone and water (50, 70, 80, 90, 95 and 100 %) for about 15–20 min, respectively, then transferred to pure acetone for 20 min.
Infiltration: The specimen was placed in 1:1 mixture of absolute acetone and the final Spurr resin mixture for 1 h at room temperature then transferred to a 1:3 mixture of absolute acetone and the final resin mixture for 3 h and to the final Spurr resin mixture for overnight.
Embedding and ultrathin sectioning
The specimen was placed in eppendorf contained embedding and heated at 70 for about 9 h. The specimen sections were stained by uranyl acetate and alkaline lead citrate for 15 min, respectively, and observed in TEM of Model JEM-1230.
Data analysis
Three replicates were used in each experiment, and all the data were expressed as mean ± SD ANOVA was performed for each diatom to determine whether an overall significant response to different nitrogen sources was observed. If the response was significant, a multiple comparison using Turkey Highly Significant Difference test was performed for each diatom to determine which specific nitrogen source has significant effects on Chl-a and oxygen evolution rate. The data in the figures were presented with the mean ± SD.
Results
Diatom sensitivity to ammonium and nitrate
Cyclotella meneghiniana, Gomphonema parvulum, and Nitzschia sp. showed a little difference on the uptake preference to different nitrogen source (NH4 + and NO3 −). Nitrate concentration in the ammonium treatment and the control treatment showed no significant difference for both C. meneghiniana and G. parvulum (P > 0.05), and no significant difference on ammonia concentration was observed between the control treatment and D1 medium for all the treatments of C. meneghiniana, G. parvulum, and Nitzschia sp. (P > 0.05). But, nitrate concentration showed no significant difference between the nitrogen enriched treatments and in D1 medium of C. meneghiniana and Nitzschia sp. (P > 0.05) (Table 2), and ammonia concentration in the nitrogen enriched treatments of C. meneghiniana and Nitzschia sp. was statistically higher than that of the ammonium treatments (P < 0.01), suggesting that nitrate was the first nitrogen source for C. meneghiniana and Nitzschia sp., and nitrate had a stimulatory effect of ammonia uptake on both C. meneghiniana and Nitzschia sp.. Nitrate concentration in the nitrogen enriched treatments of G. parvulum was statistically higher than that of D1 medium (P < 0.01) (Table 2), and ammonia concentration in the nitrogen enriched treatments of G. parvulum was statistically higher than that of the ammonium treatments (P < 0.01), suggesting that nitrate was the first nitrogen source for G. parvulum, and ammonia had an inhibitory effect of nitrate uptake on G. parvulum.
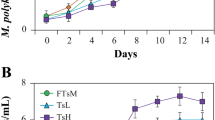
The growth of Nitzschia sp., G. parvulum and C. meneghiniana had a similar pattern under the nitrogen limited conditions. Chl-a concentration in D1 medium, the ammonium and nitrogen enriched treatments was statistically higher than that of the control (paired t test, p < 0.01). Ammonium did not support the growth of C. meneghiniana at concentrations higher than 20 mg/L, and even lowered the growth rate, when it was supplied in addition to growth-saturating nitrate concentrations. Chl-a concentration of C. meneghiniana in D1 medium was statistically higher than that of the nitrogen enriched and ammonium treatments. This seemed to be a combined effect of inhibition of nitrate uptake and direct ammonia stress on C. meneghiniana.
Chl-a concentrations of G. parvulum and Nitzschia sp. in D1 medium were statistically higher than those of the ammonium treatments (P < 0.05). Chl-a concentrations of G. parvulum and Nitzschia sp. showed no significant difference between the nitrogen enriched and ammonium treatments, and as well as between the nitrogen enriched treatments and D1 medium. It seemed that G. parvulum and Nitzschia sp. are more tolerant to ammonium than C. meneghiniana (Fig. 1).
Response of photosynthetic rate to ammonia stress
Nitzschia sp., G. parvulum, and C. meneghiniana had a little different pattern under ammonia stress. Oxygen evolution rate of C. meneghiniana in D1 medium was 79.57 μmol/mg/Chl-a/h, statistically higher than any other treatments in the experiments (P < 0.05) (Fig. 2a). Oxygen evolution rate of C. meneghiniana in the nitrogen enriched treatment was statistically higher than that of the ammonium treatment (P < 0.05), this seemed to be a direct ammonia stress on the photosynthetic activity of C. meneghiniana, and the effect can be alleviated by coexistence of nitrate in the nitrogen enriched treatment. Oxygen evolution rate of C. meneghiniana in the nitrogen enriched and ammonium treatments was significantly higher than that of the control (P < 0.01), but no difference was observed between the nitrogen enriched and ammonium treatments. This seemed to be a direct ammonia stress on the photosynthetic activity of C. meneghiniana, and the effect cannot be alleviated by coexistence of nitrate in the nitrogen enriched treatment.
Oxygen evolution rate of G. parvulum in D1 medium was 65.49 μmol/mg/Chl-a/h, significantly higher than any other treatments in the experiments (P < 0.01) (Fig. 2b). Oxygen evolution rate of G. parvulum in the nitrogen enriched and ammonium treatments was significantly higher than that of the control (P < 0.01), but no difference was observed between the nitrogen enriched and ammonium treatments. This seemed to be a direct ammonia stress on the photosynthetic activity of G. parvulum, and the effect cannot be alleviated by coexistence of nitrate in the nitrogen enriched treatment.
Oxygen evolution rate of Nitzschia sp. in D1 medium was 79.24 μmol/mg Chl-a/h, significantly higher than any other treatments in the experiments (P < 0.01) (Fig. 2c). Oxygen evolution rate of Nitzschia sp. in the nitrogen enriched and ammonium treatments was significantly higher than that of the control (P < 0.01), and oxygen evolution rate of Nitzschia sp. in the nitrogen enriched treatment was statistically higher than that of the ammonium treatment (P < 0.05), this seemed to be a direct ammonia stress on the photosynthetic activity of Nitzschia sp., and the effect can be alleviated by coexistence of nitrate in the nitrogen enriched treatment.
Ultrastructure changes of chloroplast to ammonia stress
Normal cell organelles of Nitzschia sp. were distinguishable except for the nucleus under TEM, and the cytoplasm became vacuolated (Fig. 3b). Figure 3c showed a crescent-moon shaped chloroplast after 15 days growth under D1 medium. Some undefined electron-dense granules were observed in the chloroplast (Fig. 3a). The Grana of Nitzschia sp. were compacted in the chloroplasts of all the treatments except the control (Fig. 4a–d), and lipid droplets were observed in most cells of the nitrogen enriched treatments.
Ultrastructural changes of the chloroplast in G. parvulum under the different treatments after 15 days were shown in Fig. 5. The cytoplasm of G. parvulum became vacuolated under TEM (Fig. 5a). The grana of G. parvulum were not compacted in the chloroplasts of the ammonium and nitrogen enriched treatment when compared to the chloroplasts in D1 medium (Fig. 5b, c, d).
Discussion
Sensitivity difference of typical freshwater diatoms on ammonia and nitrate
Freshwater diatoms showed different sensitivities on nitrogen uptake and ammonia stress. Gomphonema parvulum and Nitzschia sp. are more tolerant to ammonium than Cyclotella meneghiniana, as the Chl-a concentration of Gomphonema parvulum and Nitzschia sp. showed no significant difference between the nitrogen enriched treatments and D1 medium. Similar results reported by Kutka and Richards (1997) showed C. meneghiniana responded negatively and G. parvulum responded positively to ammonia diffusing pots in the field studies in a Minnesota river basin. Rushforth et al. (1981) also confirmed G. parvulum exhibited a better performance with a high level of ammonia, while C. meneghiniana was stimulated by a high level of nitrate.
The Chl-a concentration of C. meneghiniana in D1 medium was statistically higher than that of the nitrogen enriched and ammonium treatments (P < 0.05), but nitrate concentrations showed no significant difference between the nitrogen enriched treatment and in D1 medium of C. meneghiniana (P > 0.05) (Table 2). This suggested that nitrate was the first nitrogen source for C. meneghiniana, and ammonia turned out to have a direct stress on C. meneghiniana. Nitrate was also the first nitrogen source for G. parvulum, and ammonia has an inhibitory effect of nitrate uptake on G. parvulum, as the nitrate concentration in the nitrogen enriched treatment of G. parvulum was statistically higher than that of D1 medium (P < 0.01) (Table 2), and the ammonia concentration in the nitrogen enriched treatment of G. parvulum was statistically higher than that of the ammonium treatment (P < 0.01), but the Chl-a concentration of G.parvulum and Nitzschia sp. in D1 medium was statistically higher than that of the ammonium treatment (P < 0.05). Two distinct nitrate transport systems have been confirmed, one operating as a nitrate-proton symporter while the other as an energy-independent nitrate/nitrite antiporter (Wood et al. 2002). The mechanism of ammonia influencing nitrate uptake was either through the feedback regulation of nitrate metabolism (Zhuo et al. 1999; Orsel et al. 2002) or through the direct ion effect on the membrane potential (Ayling 1993). The cotransporter of nitrate and proton involves H+-ATPase, proton cross-membrane gradient, membrane potential difference, and other membrane characteristics (Colmer and Bloom 1998; Crawford and Glass 1998).
The pathway of nitrate assimilation in diatoms consists of two parts, and nitrate is taken up into the diatom cell first, reduced to nitrite by NADH-dependent nitrate reductase (NR) (Allent et al. 2005), nitrite then is transported into the chloroplast and further reduced to ammonium by nitrite reductase (NiR) (Bowler et al. 2010). Under the growth-saturating ammonia concentrations, NR in G. parvulum was still activated, as the nitrite concentration in the nitrogen enriched treatment was statistically higher than that of the ammonium treatment (P < 0.01) (Table 3).
Effects of ammonia stress on photosynthetic rate
In a natural environment, flux of nutrients from the water column to the sediment occur to an important extent by sinking of autotrophic cells, especially by diatoms (Stehfest et al. 2005). Meanwhile, considering the great contribution of diatoms to global carbon fixation (Hurtley 2009), the factors that may affect the diatom photosynthetic efficiency deserve much more attentions. In our study, the photosynthetic activities of typical freshwater diatoms showed a little different pattern under ammonia stress. Oxygen evolution rate of C. meneghiniana in D1 medium was statistically higher than that of any other treatments in the experiments (P < 0.05) (Fig. 2a). Oxygen evolution rate of G. parvulum and Nitzschia sp. in D1 medium was both significantly higher than that of any other treatments in the experiments (P < 0.01) (Fig. 2b, c). When ammonia was supplied in addition to growth-saturating nitrate concentrations for Nitzschia sp., the direct ammonia stress on photosynthetic activity can be alleviated by coexistence of nitrate in the nitrogen enriched treatment, as oxygen evolution rate of Nitzschia sp. in the nitrogen enriched treatment was statistically higher than that of the ammonium treatment (P < 0.05). But, G. parvulum or C. meneghiniana, showed no difference between the nitrogen enriched and ammonium treatments (P > 0.05), although oxygen evolution rate of both diatoms in the nitrogen enriched and ammonium treatments was significantly higher than that of the control (P < 0.01), indicating ammonia caused potentially more damage to G. parvulum or C. meneghiniana than Nitzschia sp.
Different species of diatoms respond differently to metal, organic pollution, eutrophication, ionic strength, and other environmental variables because of differences in their physiological tolerances (Gold et al. 2003). C. meneghiniana and G. parvulum have been described as high pollution-tolerant species in many studies (Morin et al. 2008; Duong et al. 2010), and when G. parvulum was transferred to unpolluted sites, its abundance declined (Rimet et al. 2009). G. parvulum was found to be tolerant to high levels of cadmium, and in contrast, C. meneghiniana was sensitive to cadmium pollution (Duong et al. 2008). However, in this study, Nitzschia sp. appeared to be more tolerant to ammonia stress than G. parvulum or C. meneghiniana. The different responses of pennate diatoms (G. parvulum and Nitzschia sp.) to ammonia stress may associate with their varied ultrastructure. C. meneghiniana is a small discoid diatom that becomes relatively elongate and cylindrical as the valve diameter decreases during vegetative division, and the chloroplasts are surrounded by a layer of chloroplast endoplasmic reticulum (Hoops and Floyd 1979).
The mechanism of ammonia as an inhibitory of O2 evolution on higher plants has recently been confirmed, by binding directly to the Mn site and replacing one of the bridging oxygen atoms on the core structure of Mn4CaO5 cluster in the Photosystem II. But, the mechanism of ammonia stress on the photosystem of diatoms remains unclear to our knowledge.
Effects of ammonia stress on ultrastructure of chloroplast
Normal diatom cells have similar morphological features. Chloroplasts are enveloped by chloroplast endoplasmic reticulum; pyrenoids are traversed by a single thylakoid; mitochondria contain tubular cristae (Hoops and Floyd 1979). Environmental stress may cause organelles disorganization and membrane disruption of cells (Lage-Pinto et al. 2008), and ultrastructural changes on chloroplast, such as deceased size accompanied by electron-dense stroma, thylakoids disorganization can be indicators for photosynthesis inhibition (Kivimaenpaa et al. 2004). Normal cell organelles of all three freshwater diatoms were distinguishable except for the nucleus under TEM, and the cytoplasm became vacuolated under nitrogen depletion treatments. Grana of G. parvulum were not compacted in the chloroplasts of the ammonium and nitrogen enriched treatments when compared to chloroplasts in D1 medium (Fig. 5b, c, d).
Crescent-moon shape chloroplast under D1 medium was showed in Fig. 5c, while some undefined electron-dense granules were observed in the chloroplast under the ammonia treatment of Nitzschia sp. (Fig. 3a). Grana of Nitzschia sp. were compacted in the chloroplasts of all the treatments except the control (Fig. 3a–d), and lipid droplets were observed in most cells of the nitrogen enriched treatment, but no thylakoids disorganization were recorded in all the treatments.
Conclusion
Gomphonema parvulum was more tolerant to ammonia stress than Cyclotella meneghiniana and Nitzschia sp. on growth. A combined effect of inhibition of nitrate uptake was found for C. meneghiniana and Nitzschia sp.. Direct ammonia stress on the photosynthetic activity of Nitzschia sp. can be alleviated by coexistence of nitrate in the nitrogen enriched treatment, which may be caused by different nitrate transporter system within algal cells. TEM results confirmed ultrastructural changes of Nitzschia sp. in chloroplast, with undefined electron-dense granules and lipid droplets, but the membrane integrity of cell was maintained, suggesting an adaptation of typical freshwater diatoms to adjustment to ammonia stress.
References
Allen AE, Ward BB, Song BK (2005) Characterization of diatom (Bacillariophyceae) nitrate reductase genes and their detection in marine phytoplankton communities. J Phycol 41:95–104. doi:10.1111/j.1529-8817.2005.04090.x
Ayling SM (1993) The effect of ammonium ions on membrane potential and anion flux in roots of barley and tomato. Plant, Cell Environ 16:297–303. doi:10.1111/j.1365-3040.1993.tb00872.x
Bennion H, Juggins S, Anderson NJ (1996) Predicting epilimnetic phosphorus concentrations using an improved diatom-based transfer function and its application to lake eutrophication management. Environ Sci Technol 30(6):2004–2007. doi:10.1021/es9508030
Bowler C, Vardi A, Allen AE (2010) Oceanographic and biogeochemical insights from diatom genomes. Annu Rev Mar Sci 2:333–365. doi:10.1146/annurev-marine-120308-081051
Chen WM, Zhang QM, Dai SG (2009) Effects of nitrate on intracellular nitrite and growth of Microcystis aeruginosa. J Appl Phycol 21:701–706. doi:10.1007/s10811-009-9405-1
Colmer TD, Bloom AJ (1998) A comparison of NH4 + and NO3 − net fluxes along roots of rice and maize. Plant, Cell Environ 21:240–246. doi:10.1046/j.1365-3040.1998.00261.x
Crawford NM, Glass ADM (1998) Molecular and physiological aspects of nitrate uptake in plants. Trends Plant Sci 3:389–395. doi:10.1016/S1360-1385(98)01311-9
Dai GZ, Shang JL, Qiu BS (2012) Ammonia may play an important role in the succession of cyanobacterial blooms and the distribution of common algal species in shallow freshwater lakes. Global Change Biol 18:1571–1581. doi:10.1111/j.1365-2486.2012.02638.x
Duong TT, Morin S, Herlory O, Feurtet-Mazel A, Coste M, Boudou A (2008) Seasonal effects of cadmium accumulation in periphytic diatom communities of freshwater biofilms. Aquat Toxicol 90:19–28. doi:10.1016/j.aquatox.2008.07.012
Duong TT, Morin S, Coste M, Herlory O, Feurtet-Mazei A, Boudou A (2010) Experimental toxicity and bioaccumulation of cadmium in freshwater periphytic diatoms in relation with biofilm maturity. Sci Total Environ 408:552–562. doi:10.1016/j.scitotenv.2009.10.015
Finlay BJ, Monaghan EB, Maberly SC (2002) Hypothesis: the rate and scale of dispersal of freshwater diatom species is a function of their global abundance. Protist 153(3):261–273. doi:10.1078/1434-4610-00103
Gold C, Feurte-Mazel A, Coste M, Boudou A (2003) Effects of cadmium stress on periphytic diatom communities in indoor artificial streams. Freshwater Biol 48:316–328. doi:10.1046/j.1365-2427.2003.00980.x
Guo L (2007) Doing battle with the green monster of Taihu Lake. Science 317:1116. doi:10.1126/science.317.5842.1166
Hoops HJ, Floyd GL (1979) Ultrastructure of the centric diatom, Cyclotella meneghiniana: vegetative cell and auxospore development. Phycologia 18(4):424–435. doi:10.2216/i0031-8884-18-4-424.1
Hou LH, Wu CM, Huang HH, Chu HA (2011) Effects of ammonia on the structure of the oxygen-evolving complex in photosystem II as revealed by light-induced FTIR difference spectroscopy. Biochemistry 50:9248–9254. doi:10.1021/bi200943q
Hurtley S (2009) Green for diatoms. Science 324:1614. doi:10.1126/science.324_1615d
Jespersen AM, Christofersen K (1987) Measurements of chlorophyll-a from phytoplankton using ethanol as extraction solvent. Fund Appl Limnol 109:445–454
Kivimaenpaa M, Jonsson AM, Stjernquist I, Sellden G, Sutinen S (2004) The use of light and electron microscopy to assess the impact of ozone on Norway spruce needles. Environ Pollut 127:441–453. doi:10.1016/j.envpol.2003.08.014
Kutka FJ, Richards C (1997) Short-term nutrient influences on algal assemblages in three rivers of the Minnesota River basin. J Freshwater Ecol 12(3):411–419. doi:10.1080/02705060.1997.9663551
Lage-Pinto F, Oliveira JG, Cunha MD, Souza CMM, Rezende CE, Azevedo RA, Vitoria AP (2008) Chlorophyll a fluorescence and ultrastructural changes in chloroplast of water hyacinth as indicators of environmental stress. Environmen Exp Bot 64:307–313. doi:10.1016/j.envexpbot.2008.07.007
Leland HV, Brown LR, Mueller DK (2001) Distribution of algae in the San Joaquin River, California, in relation to nutrient supply, salinity and other environmental factors. Freshwater Biol 46:1139–1167. doi:10.1046/j.1365-2427.2001.00740.x
Lenoci L, Camp PJ (2008) Diatom structures template by phase-separated fluids. Langmuir 24:217–223. doi:10.1021/la702278f
Morin S, Duong TT, Dabrin A, Coynel A, Herlory O, Baudrimont M, Delmas F, Durrieu G, Schafer J, Winterton P, Blanc G, Coste M (2008) Long-term survey of heavy metal pollution, biofilm contamination and diatom community structure in the Riou Mort watershed, South-West France. Environ Pollut 151:532–542. doi:10.1016/j.envpol.2007.04.023
Ni WM, Zhang JY, Ding TD, Stevenson RJ, Zhu YM (2012) Environmental factors regulating cyanobacteria dominance and microcystin production in a subtropical lake within the Taihu watershed, China. J Zhejiang Univ-Sci A (Appl Phys Eng) 13:311–322. doi:10.1631/jzus.A1100197
Orsel M, Filleur S, Fraisier V, Daniel-Vedele F (2002) Nitrate transport in plants: which gene and which control? J Exp Bot 53:825–833. doi:10.1093/jexbot/53.370.825
Pei GF, Liu GX, Hu ZY (2010) A comparative study of benthic algal colonization in shallow lakes of China. J Freshwater ecol 25(3):403–411. doi:10.1080/02705060.2010.9664383
Petit AN, Eullaffroy P, Debenest T, Gagne F (2010) Toxicity of PAMAM dendrimers to Chlamydomonas reinhardtii. Aquat Tox 100:187–193. doi:10.1016/j.aquatox.2010.01.019
Quinlan EL, Nietch CT, Blocksom K, Lazorchak JM, Batt AL, Griffiths R, Klemm DJ (2011) Temporal dynamics of periphyton exposed to tetracline in stream mesocosms. Environ Sci Technol 45(24):10684–10690. doi:10.1021/es202004k
Rimet F, Ector L, Cauchie HM, Hoffmann L (2009) Changes in diatom-dominated biofilms during simulated improvements in water quality: implications for diatom-based monitoring in rivers. Eur J Phycol 44(4):567–577. doi:10.1080/09670260903198521
Rushforth SR, Brotherson JD, Fungladda N, Evenson WE (1981) The effect of dissolved heavy metals on attached diatom populations in the Uintah Basin of Utah, USA. Hydrobiologia 83:313–323. doi:10.1007/BF00008282
Stehfest K, Toepel J, Wilhelm C (2005) The application of micro-FTIR spectroscopy to analyze nutrient stress-related changes in biomass composition of phytoplankton algae. Plant Physiol Biochem 43:717–726. doi:10.1016/j.plaphy.2005.07.001
Swift DM, Wheeler AP (1991) Some structural and functional properties of a possible organic matrix from the frustules of the freshwater diatom Cyclotella meneghiniana. Surface reactive peptides and polymers, chapter 24 (ACS symposium series, volume 444), pp 340–353 doi:10.1021/bk-1991-0444.ch024
Szczepocka E, Szulc B (2009) The use of benthic diatoms in estimating water quality of variously polluted rivers. Int J Oceanography Hydrobiol 38(1):17–26. doi:10.2478/v10009-009-0012-x
Thessen AE, Bowers HA, Stoecker DK (2009) Intra- and interspecies differences in growth and toxicity of Pseudo-nitzschia while using different nitrogen sources. Harmful Algae 8:792–810. doi:10.1016/j.hal.2009.01.003
Tsuno M, Suzuki H, Kondo T, Mino H, Noguchi T (2011) Interaction and inhibitory effect of ammonium cation in the oxygen evolving center of photosystem II. Biochemistry 2506–2514 doi:10.1021/bi101952g
Wang XL, Han JY, Xu LG, Zhang Q (2010) Spatial and seasonal variation of the contamination within water body of the Grand Canal, China. Environ Pollut 158:1513–1520. doi:10.1016/j.envpol.2009.12.018
Wood NJ, Alizadeh T, Richardson DJ, Ferguson SJ, Moir JWB (2002) Two domains of a dual-function NarK protein are required for nitrate uptake, the first step of denitrification in Paracoccus pantotrophus. Mol Microbiol 44:157–170 http://dx.doi.org/10.1046/j.1365-2958.2002.02859.x
Zhang M, Wang ZQ, Xu J, Liu Y, NI L, Cao T, Xie P (2011) Ammonium, microcystins, and hypoxia of blooms in eutrophic water cause oxidative stress and C-N inbalance in submersed and floating-leaved aquatic plants in Lake Taihu, China. Chemosphere 82:329–339. http://dx.doi.org/10.1016%2Fj.chemosphere.2010.10.038
Zhuo D, Okamoto M, Vidmar JJ, Glass ADM (1999) Regulation of a putative high-affinity nitrate transporter (Nrt2; 1At) in roots of Arabidopsis thaliana. Plant J 1999(17):563–568. doi:10.1046/j.1365-313X.1999.00396.x
Acknowledgments
The authors appreciate the generous financial support of this work by National Natural Science Foundation (No. 21177106). The authors would also like to thank Dr. R. Jan Stevenson (Michigan State University) for assistance in preparing this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Bas Ibelings.
Rights and permissions
About this article
Cite this article
Zhang, J.Y., Ni, W.M., Zhu, Y.M. et al. Effects of different nitrogen species on sensitivity and photosynthetic stress of three common freshwater diatoms. Aquat Ecol 47, 25–35 (2013). https://doi.org/10.1007/s10452-012-9422-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-012-9422-z