Abstract
This study presents a method that uses a standing surface acoustic wave (SSAW) to continuously separate particles in a size-gradient manner in a microchannel flow. The proposed method was applied to a colloidal suspension containing poly dispersed particles with three different sizes (1, 5, and 10 μm) but the same density and compressibility. Particle suspension was focused hydrodynamically at an entrance region, and particles were forced actively toward the side wall where SSAW-pressure nodes were generated by two interdigital transducers (IDTs) across the channel. The particles placed in the middle stream, in which the shear rate was minimized, were separated successfully in a size-gradient manner by acoustic force. In addition, this study further developed an analytical model to predict the displacement of particles in microchannel flow by considering viscous, acoustic, and diffusive forces. The predicted values of particle displacement showed excellent agreement with the experimental results, and diffusion was found to be important and not negligible. The advantage of this method is to minimize the shear rate on particles, which would be useful for potential applications of shear-dependent cells such as platelets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
A colloid is a multi-phase system in which micro- or nanometer-scale substances are dispersed in a medium; colloids are also called complex fluids (Jones 2002). Blood, ink, and milk are among the many familiar colloidal suspensions. The separation of particles in colloidal systems, such as those containing polymer beads, ceramics, cells, nanoparticles, or pharmaceutical emulsions, becomes an important issue in various applications, including industrial processing, environmental assessment, and biological or chemical research (Yamada et al. 2004). Such separation techniques are also essential for cell detection, tissue engineering, drug screening, and surgical operations (Lara et al. 2004; Johann 2006; Yang et al. 2006; Ye et al. 2007; Yung et al. 2009).
Various methods and techniques have been developed for separating colloidal particles, and these methods can be categorized as either immunological or physical methods (Kang and Park 2005). In immunological methods, the target particles are labeled fluorescently, and then separated by external forces that are applied selectively upon the observation of the label signal. Magnetic particles also can be used to label target particles, enabling separation according to differences in movement under an external magnetic field (Fu et al. 1999; Berger et al. 2001). Immunological methods show high selectivity but damage the samples during the laborious and time-consuming labeling step. The complicated processes associated with these methods also entail high-costs. In physical methods, by contrast, the target particles are separated by their natural physical properties, such as the size, density, or permittivity under force fields that are centrifugal (Pamme 2007; Seo et al. 2007), hydrodynamic (McCloskey et al. 2003; Qu et al. 2008), or electrokinetic/dielectrophoretic (Baret et al. 2009; Jellema et al. 2009). These methods are simple and fast, but have lower resolution and sample selectivity than immunological methods (Yang et al. 1999; Guillong et al. 2003; Kang and Park 2005; Yamada et al. 2004). A comprehensive review was reported recently for the most recent developments in microfluidic technology for cell and particle separation in continuous flow-based systems (Lenshof and Laurell 2010).
Acoustic forces based on Surface acoustic waves (SAWs) have emerged as a new technology for use in physical particle separation, particularly because they can be integrated easily into microfluidic circuits (Li et al. 2007; Tan et al. 2007; Shi et al. 2008a, b; Yeo and Friend 2009). Since a SAW-based technique features non-invasiveness, lack of contamination, and low power consumption, various basic researches and applications using SAW-based techniques have been reported increasingly. Typical applications are fluid-mixing (Tan et al. 2010; Saiki et al. 2010), pumping (Cecchini et al. 2008), concentration (Tan et al. 2007; Rogers et al. 2010), particle tweezing/patterning (Shi et al. 2009a), emulsion/particle sorting (Franke et al. 2010; Shi et al. 2009b) and particle manipulation (Tan et al. 2009). In addition, there is also a SBAW (Standing bulk acoustic wave)-based technique, which is slightly different from the SSAW (standing surface acoustic wave) technique (Petersson et al. 2007).
In fact, SAW-based particle separation methods are potentially important in biological analyses such as the detection of circulating tumor cells and malaria-infected cells. However, some biological applications require strict control of high-shear rates that may activate cells such as platelets and platelet-derived microparticles. Thus, it is necessary to develop a SSAW-based separation method while avoiding high-shear rates (Hellums 1993; Kroll et al. 1996). Also, there is a SSAW-less effective area near the wall due to the propagation characteristics of SAWs with an attack angle (Rayleigh angle) from a substrate surface to bulk fluid (Alvarez et al. 2008; Shilton et al. 2008). If particles are placed initially near the wall, SAWs may yield low operational efficiency for manipulating particles due to the Rayleigh angle. If it is necessary to place particles near the wall, a second sheath fluid is required to dislocate particles from the SSAW-less effective area (Shi et al. 2009b). The effect of the Rayleigh angle on the microchannel flow was described comprehensively and discussed in the section on the results and discussion.
Therefore, this article reports a simple but novel approach for standing surface acoustic wave (SSAW)-based particle separation that adopts hydrodynamic focusing. The use of hydrodynamic focusing is to not only minimize the shear rate but also locate particles in the SSAW-effective working area. In addition, this study extends analytical research by considering three primary forces, viz. the viscous force, the acoustic force, and the diffusive force, and providing comprehensive information on poly disperse particle behavior by comparison with experimental results.
2 Working principle and analytical models
2.1 Working principle
Figure 1 shows a schematic illustration of the SSAW-based microfluidic device. A pair of interdigital transducers (IDTs) was patterned onto a transparent piezoelectric substrate (LiNbO3), and a PDMS channel was positioned and bonded to the substrate between the IDT couple. Three inlet channels converged to a single straight channel and a main test section, followed by five dividing outlets. The inlet channels allowed the introduction of particle flow through the middle channel, with sheath-flow through the outer channels. The particles were focused hydrodynamically toward the central region of the channel downstream. Then, the particles were subject to the lateral acoustical forces of SSAWs. By adjusting the flow rate ratio between the sample and sheath fluids, the width of the focused sample stream was kept constant. The width of the focused stream typically ranged between 10 and 14 μm.
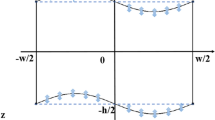
Diagrams of the microfluidic device and working principle. a The sample and sheath flows containing particles of three different diameter sizes (1, 5, and 10 μm) are illustrated. The sample flow was focused by the sheath-flow, and particles in the sample flow were separated within the SSAW field generated by the IDTs. The displacements of the particles were observed at two points, which were 3 and 5 mm beyond where the particles began to be exposed to the SSAW region. b–d Cross-sectional views of particles in the sample flow before focusing by the sheath-flow, after focusing, and under the SSAW field, respectively. e The positions of the pressure nodes along the channel (these are illustrated with respect to the channel dimensions shown in d); the pressure nodes were located at the walls, and the pressure anti-node(s) were located at the middle of the channel
The application of AC signals to the IDTs produced a pair of SAWs that propagated in opposing directions toward the main test-section channel. The interference between the two SAWs generated their cancellation and superposition, which resulted in a structured standing surface acoustic wave (SSAW). As the two SAWs having the same magnitude but opposite signs met in the microchannel, minimum pressure nodes were generated along the longitudinal direction and vice versa. These pressure fluctuations produced lateral acoustic radiation forces parallel to the y–z plane (see Fig. 1) on particles in the microchannel, forcing them to either pressure nodes (minimum pressure amplitude) or pressure anti-nodes (maximum pressure amplitude), depending on the relative density and compressibility of the particles and the medium (Yosioka and Kawasima 1955). Thus, particle behavior that is affected by SSAWs can be predicted in an analytical manner.
2.2 Analytical models
Prior to developing an analytical model for particle motion under SSAWs, it is necessary to define the flow condition and corresponding assumptions as follows: (1) the flow in the microchannel is steady, laminar, and fully developed; (2) the fluids are Newtonian; (3) the fluids and particles have the same density; (4) all channels have the same height; (5) the viscosities of all the fluids used in the experiment are identical; and (6) the lateral acoustic force driven by the SSAW is uniform throughout the test section. In fact, since the sample fluid consisted of a very low concentration of particles (0.5% vol.) that were suspended in the same sheath fluid, the particle suspension viscosity was almost the same as that of the sheath fluid. Thus, this assumption is reasonable, and there is no need to consider the effect of viscosity on hydrodynamic focusing in multiple fluid flows.
It is worth noting that since the applied SSAW is perpendicular to the axial flow direction, the pressure nodes would form in the two-dimensional plane along the axial flow (i.e., the y–z plane). The two-dimensional distribution has been manipulated further and patterned successfully in a one-dimensional dotted pattern by applying two perpendicular acoustic forces (Shi et al. 2009a). However, the two-dimensional particle distribution would be dependent on the channel height and aspect ratio of the microchannel, since the acoustic force initially propagates from the surface to fluid. Thus, flat rectangular channels would be more effective for applying SSAWs on particles.
The behavior of particles inside the microchannel flow under an SSAW can be predicted by considering various forces that impact upon particles: lateral acoustic radiant force, the viscous drag force (opposite direction of particles’ velocity relative to flow stream), diffusive force, gravity force, and buoyancy force. The gravity and buoyancy forces would cancel out each other, since the densities of both particles and the suspending medium are almost balanced. For a hydrodynamic focusing flow in a microchannel, it is necessary to consider three primary forces (viscous, diffusion, and acoustic radiation forces) to estimate the displacement of a particle from the center plane of the microfluidic channel. Each force already has been well developed for analytical models in previous research (Lee et al. 2006; Bruus 2008; Shi et al. 2009b), and detailed derivations can be found elsewhere.
First, since this study adopted hydrodynamic focusing, it is necessary to estimate the width of the focused flow to predict the maximum displacement of a particle from the center of a microchannel. The focused width in a rectangular microchannel was predicted by considering the mass conservation (Lee et al. 2006) under the assumption of identical viscosities and densities between the sample and sheath fluids. The width of the focused flow (w f) can be estimated by the volume flow rates of the inlet channel (Q i) and side channels (Q s), and the velocity ratio (γ), as follows:
where γ is the ratio of the average velocities \( \left( {\gamma = \bar{v}_{\text{f}} /\bar{v}_{\text{o}} } \right). \) Further, \( \bar{v} \) is the average velocity, and the subscripts of f, o, i, and s indicate focused, outlet, inlet, and side channel, respectively. For flow in parallel plates or flat rectangular channels (with low-aspect ratios), the velocity would be uniform in the lateral direction (x-direction) and assume a parabolic profile in the y-direction (across the channel height). Thus, the flow velocity ratio would be about unity for flow in a rectangular channel with a low-aspect ratio (Stiles et al. 2005). Since the present study used a low-aspect-ratio rectangular channel (AR = 0.2), the authors can assume the same velocity profiles as above. Then, the width of the focused stream (w f) can be easily predicted by the above equation, viz. (1). It is worth noting that this model did not include the direct effect of the viscosities of the sample and sheath fluids on the width of the focused flow. However, since the sample fluid exhibited almost the same viscosity as the sheath fluid in this study, it is not necessary to consider the viscosity effect on the width of the focused flow.
Secondly, the diffusive force also causes the displacement of a particle that moves by diffusion, which was comprehensively described by Bruus (2008) as follows:
While the particles experience acoustic forces, they diffuse toward the walls according to their diffusivities. In general, a small particle diffuses faster than a larger one (Weigl and Yager 1999). The displacement of particles by the diffusion effect was not considered in the previous study (Shi et al. 2009b), although it was found to be important for small-sized particles.
A surface acoustic wave also yields the displacement of a particle that is perpendicular to the flow under the force of the lateral acoustic radiation pressure. The acoustic radiation force can be described as follows (Yosioka and Kawasima 1955):
and
where p 0 is the pressure amplitude, V the volume, λ the ultrasonic wavelength, k the wave vector, x the distance from an anti-pressure node to a pressure node, ρ the density, and β the compressibility, respectively. The subscripts of p and m indicate particle and medium, respectively. With consideration of the viscous resistance and a given working time, t w, the displacement of a particle in a perpendicular direction to the flow can be approximately predicted, and a detailed derivation can be found in previous research (Shi et al. 2009b).
where \( c = t_{\text{w}} {\frac{{p_{ 0}^{2} V_{\text{p}} \beta_{\text{m}} \phi \left( {\rho ,\beta } \right)}}{{3\lambda^{2} \eta r/\pi }}} + {\text{In(tan(2}}\pi {\text{x}}_{ 1} /\lambda ) ). \) In the above, \( \eta \) is the viscosity of the medium, and r is the particle radius, respectively. The p 0, defined as (PΖ/A)1/2, is 1.927 × 105 (Pa) where P is the input power of AC signal, 320 mW, A is the SAW working area, 1.6 × 10−4 m2, and Z is the product of density and SAW velocity of the substrate (4650 kg/m3 × 4000 m/s) in this study (Pierce 1989; Wong 2002).
Therefore, the displacement of a particle, considering hydrodynamic focusing (d hydrof), diffusion (d diff), and the acoustic radiation force (d SSAW), can be integrated into an equation as follows:
In fact, the previous analytical model (Shi et al. 2009b) considered only two terms, namely, hydrodynamic focusing (d hydrof) and the acoustic force (d SSAW). However, in the preliminary experimental results, it was found that the diffusive term was not negligible. When small particles flowed in low flow rates, the diffusive force was dominant and should be considered in the analytical model. Thus, the proposed Eq. 6 in this study enables us to predict the particle displacement more accurately than previous models regardless of the size and flow rates of the particles.
3 Materials and methods
A thin film made of Au/Cr (100/10 nm) was deposited using an e-beam evaporator (Ulvac, Japan) and patterned by a conventional lithographic process, followed by a lift-off process to form IDTs on a piezoelectric material, LiNbO3 wafer (Y + 128° X-propagation; 500 μm thick; NEL Crystal Co, Fukushima) (Fig. 2a). Using a conventional soft-lithography process based on an SU-8 negative photoresist (MicroChem, MA), a PDMS channel was patterned (Fig. 2b). The PDMS channel was subsequently aligned with the IDT patterns, and bonded to the LiNbO3 substrate after pretreatment with air plasma at 250 W and 80 mTorr (CUTE, Femto Science Co., Korea) for 50 s. Beneath the LiNbO3 substrate, a transparent film with scale marks was attached to determine the exact measurement position in the microfluidic channel.
In general, the SAW working wavelength (λ) can be determined by the relation, f = c/λ, where c is the wave propagation speed of the substrate (~4000 m/s) and f is the applied signal frequency. Since the wave propagation speed, c, for LiNbO3 was about 4000 m/s and f was set to 19.5 MHz, the calculated SAW working wavelength was about 200 μm. To reflect a surface acoustic wave with a wavelength of λ, a large reflection rate can be obtained by aligning a number of reflection sources such as electrodes by a pitch of λ/2. Thus, the width and pitch of the IDTs were designed precisely to be 50 and 100 μm, respectively. In order to locate the first standing pressure nodes at the two side walls, the microchannel was fabricated with a width of 100 μm and height of 20 μm, and precisely placed on the line-marked substrate, as shown in Fig. 1e.
The IDTs were actuated by an RF signal generator (8657A, Hewlett Packard) with signal amplification by an amplifier (ZHL-1-2 W, mini-circuits) supplied by a DC power supply (E3634A, Agilent). The microparticles used in this study were plain polystyrene micro-beads (4000 series, Thermo Scientific, CA) with the same density and compressibility but different sizes (1, 5, and 10 μm), which were suspended in deionized water with a volume concentration of 0.5% and which flowed into the middle channel inlet. Deionized water was used as the sheath-flow in the outer inlets. Three glass syringes (500 μl; Hamilton, Nevada) were connected to each inlet and individually pushed by syringe pumps (KD100, KD Scientific). Particle displacements were imaged using an inverted microscope (IX71, Olympus, Japan) equipped with a high-speed camera (FASTCAM Ultima APX, Photron, Japan). The displacements were observed and recorded at several locations including the inlet and outlet of the straight channel and z = 3 and 5 mm. The origin of the z-axis is the point at which the particles entered the SSAW field. Then, the recorded particle displacements were analyzed.
4 Results and discussion
Figure 3 shows the separation of particles having three different sizes (1, 5, and 10 μm) under lateral acoustic radiation forces (because of SSAWs). The location of observation was 5 mm downstream from the onset of the SSAW field, and the sample flow rate was 0.22 μl/min. When SSAW is not activated, all particles keep flowing along the center region of the channel, even though the width of the focused particle flow is slightly increased due to diffusion. The effect of diffusion is discussed in relation to Fig. 4. When SSAW is activated, large particles (10 μm) show the maximum displacement toward the channel wall; mid-sized particles (5 μm) show intermediate displacements; and small particles (1 μm) exhibit a slight displacement from the centerline. The particle displacements are strongly dependent on the particle size when the particles are subject to acoustic force, as described in Eq. 3. However, the absolute magnitude of particle displacement is also affected by the flow rate, which is related directly to the SSAW-working time on particles. Thus, as the flow rate increases, the particle displacement will decrease. In this study, the sample flow rate ranged 0.03–0.3 μl/min, and the corresponding average velocity was 2–20 mm/sec based on the geometry of the channel.
Images of particles of three different sizes being separated within the SSAW field. The observation location was 5 mm downstream from the onset of the SSAW field, and the sample flow rate was 0.22 μl/min. w f is the focused width of the sample, and d1, d5, and d10 are the displacement distances of particles with sizes 1, 5, and 10 μm, respectively. a and c Show the focused colloidal flow in the absence of the SSAW field. b Shows the focused colloidal flow in the microchannel when the SSAW was activated. d–f Show the particle displacements as functions of the particle size
Image showing separation of two different particles by diameter, viz. 1 and 10 μm, among different sample medium flow rates (the flow rate ratio between the sample medium and the sheath medium is 1:3). a–c Show the particle flows when the SSAW is not activated. d–f Show the particle flows when the SSAW is activated. The particles in the circle are 10 μm in diameter
Separation of particles between 1 and 10 μm was also observed at the outlet area of the channel (Fig. 4). A comparison of SSAW off and on as shown in Fig. 4c and f reveals that large-sized particles were apparently moved near the wall and collected in outer channels, whereas small-sized particles remained in the middle stream and collected in the center channel. It is worth noting that as the flow rate decreased, the working time of SSAW on particles increased, and more displacement could be observed as shown in Fig. 4d and e. Thus, to separate poly disperse particles, it is necessary to find optimal conditions for the flow rate and the SSAW-active area. From an analysis of the captured images, at a fixed flow rate of 0.3 μl/min, regarding particles of 10 μm diameter, the efficiency of separation with reference to mixed particles of 1 and 10 μm diameters was found to reach 99.9%. The high efficiency is mainly because of the strong dependence of the acoustic force on the particle volumes. However, the flow rate of 0.3 μl/min was not optimal for the separation efficiency of particles with diameters of 5 and 10 μm. In Fig. 5, the lateral displacements of each particle were analyzed with respect to a varying flow rate.
a Particle separation as a function of the particle size, measured at 5 mm downstream from the onset of the SSAW field under various flow rates. b Particle separation as a function of the particle size at 3 mm downstream from the onset of the SSAW field under various flow rates. (In both parts a and b, the lines indicate the theoretical predictions of the particle displacements, and the marks indicate the experimental measurements). c The ratio of the displacement under an acoustic force to the total displacement of the particle as a function of the particle diameter. Here, each line indicates the duration of exposure of the particle to the SSAW region. d The total displacement of the particle as a function of the particle diameter under different durations of exposure to the SSAW. (The lines indicate the theoretical predictions of the particle displacements, and the marks indicate the experimental measurements)
The diffusion of particles was observed clearly at three different flow rates when SSAW was off. At a high-flow rate, as shown in Fig. 4c, the width of the focused particles was kept almost constant, and particles in the middle stream did not diffuse much. However, at a low flow rate (0.03 μl/min), as shown in Fig. 4a, the focused particles apparently were diffused, and the width of focused particle flow increased. Thus, it is reconfirmed that diffusion should be considered in developing an analytical model. In this experiment, even though the sample flow rate varied from 0.03 to 0.3 μl/min, the ratio of the sample flow rate to sheath flow was kept constant (1:3).
Particles were found to be distributed laterally within the microfluidic channel as a function of the particle size. Figure 5a and b show experimental data on the particle displacement at two different locations (z = 3 and 5 mm). The horizontal axis represents the sample flow rate, which is inversely proportional to the working time of SSAW on particles. The vertical axis represents the non-dimensional particle displacement (d/w ch). Here, d is the length of particle displacement, and w ch is the width of the channel. In Fig. 5a, the large particles (10 μm) were strongly affected by SSAW and displaced at the channel wall, i.e., (d/w ch = 0.5), for the entire range of the flow rate (0.02–0.3 μl/min). In contrast, the small particles (1 μm) were relatively less influenced by SSAW, and thus were displaced only slightly from the middle stream (d/w ch = 0.07–0.12). For the mid-sized particles (5 μm), an increasing flow rate caused a significant decrease in the displacement length since the working time of SSAW on particles was reduced. At flow rates between 0.2 and 0.3 μl/min, the three different groups of particles by size would be located at different lateral locations, and could be collected in dividing channels on that axial location. Thus, one can separate three different groups of particles by size by adjusting the SSAW parameters (power input, active area, etc.) and the flow rate. Similar results were also found at z = 3 mm in Fig. 5b.
Particle displacement was discussed by considering the diffusion effect, as can be seen from Fig. 4. Further analyses were done for the contribution ratio of the acoustic force with respect to the total displacement, viz. (d acoust/d total) and (d/w ch), for various particle sizes, as shown in Figs. 5c and d, respectively. The displacement ratio (d acoust/d total) was strongly dependent on the working time of SSAW as well as the particle size. If particles smaller than 1 μm in size were exposed for 0.25 s of SSAW, the contribution of the acoustic force was barely over 50%, which means that other effects such as diffusion cannot be neglected in an analytical model. This is reconfirmed in the inner graph of Fig. 5d: d/w ch for submicron particles was smaller than 0.05 for moderate SSAW working times. However, if either the particle size exceeded 2 μm or the SSAW working time exceeded 2.5 s, the effect of SSAW on particle displacement became dominant, which implies that other effects can be neglected. These results imply that the effective acoustic force applied on sub-microparticles is relatively much weaker than that for larger particles, and sub-microparticles can be separated from microparticles. These results would be useful information for the application of SSAW-based separation technology for platelet-derived microparticles (PDMPs, 0.1–1.0 μm) from other blood cells. PDMPs have become an important issue since they have been found to be involved in the pathophysiology of vascular disorders (Piccin et al. 2007).
As shown in Fig. 5, the analytical results for lateral particle displacements showed good agreement with the experimental results. Interestingly, all the experimental values were slightly less than the calculated ones. This discrepancy arose partially because the calculated values represent upper bounds. Overestimation in the theoretical predictions also could have resulted from the assumption of a uniform velocity (i.e., the average flow velocity) across the channel cross-section. The actual flow field in a microchannel is characterized by a parabolic flow profile in which particles at the center of the channel flow faster than the average velocity. This speculation can be answered by considering a full 3-D computational fluid dynamic analysis, which may be left for future study.
By way of demonstration, it was identified the optimum flow rates for separating particles of sizes 1, 5, and 10 μm: 0.22 and 0.66 μl/min for the sample flow rate and sheath-flow rate, respectively, at point 5 mm. A detailed movie clip is given in the supporting information. The input power for the IDTs was 320 mW. Under these conditions, the separated particles should optimally be collected 5 mm after applying a lateral acoustic radiation force to maximize selectivity among all the three different groups of particles by size. The developed model yielded a good theoretical estimation of particle movement, showing good agreement with the experimental results.
A lateral surface acoustic wave would not generate a uniform pressure distribution in the cross-section of a rectangular microchannel since the wave propagates to fluid with an attack angle from the bottom surface of the piezoelectric substrate. This was demonstrated by Friend’s research group (Alvarez et al. 2008; Shilton et al. 2008), namely, a primary internal recirculating flow was observed in droplets because of the direct leakage of SAW radiation from the substrate at the Rayleigh angle θR. Similarly, a single acoustic radiation force would cause acoustic streaming in the lateral direction of a microchannel with the Rayleigh angle. Subsequently, a pair of acoustic radiation force applied across a microchannel would be superposed and form a standing pressure distribution in the microchannel. Thus, the iso-pressure profile near the wall would not be parallel to that at the centerline of the flow. Some of the particles in the upper corners would be less driven than other particles by the acoustic radiation force and would remain at the corners. However, if the acoustic wave propagates from the side wall, e.g., bulk acoustic waves (BAW, Petersson et al. 2007), the pressure profiles would be parallel to each other and the side wall. For BAW, there would not be a less-effective area. However, the theoretical equation from past research describes a simplified model for the acoustic force under the assumption of the uniform propagation of a surface acoustic wave in the fluid (Yosioka and Kawasima 1955). The assumption in this study would not cause any significant error in the analytical model since all the particles were located in the middle stream at the entrance. Thus, in this study, the hydrodynamic focusing of the sample flow enables us to minimize any possible errors because of Rayleigh’s angle of SAW.
5 Conclusion
The authors have demonstrated the rapid and continuous separation of poly disperse particles in a size-dependent manner by applying SSAW technology in microfluidics. To avoid locating particles in the SSAW-less effective area, the sample flow was focused hydrodynamically, and all particles were placed initially in the middle stream at the entrance. Thus, SSAW can work efficiently on particles that are displaced easily toward the desired lateral positions. For the efficient separation of particles of three different sizes, it is also necessary to control the sample flow rate precisely according to the given particle sizes and SSAW-working area. However, for sub-microparticles (0.1–1 μm), SSAW was not so effective for manipulating particles, and diffusion should be considered in analytical study. Thus, the findings of this analytical model considering three primary forces (acoustic, viscous, and diffuse forces) showed good agreement with the experimental results. Since recent studies on blood have upheld the importance of platelet-derived microparticles (PDMPs) that are smaller than 1 μm, the separation of sub-microparticles is increasingly important. Furthermore, as some of the PDMPs were found to be sensitive to the shear rate, the flow control of a low shear rate through the entire separation process is highly required. As a proof of concept, this study has demonstrated the separation of microparticles that are kept in the region of low shear rate. These features of SSAW-based particle separation with hydrodynamic focusing would be used widely in various biomedical applications.
References
Alvarez M, Friend JR, Yeo LY (2008) Surface vibration induced spatial ordering of periodic polymer patterns on a substrate. Langmuir 24:10629–10632
Baret JC, Miller OJ, Taly V, Ryckelynck M, El-Harrak A, Frenz L, Rick C, Samuels ML, Hutchison JB, Agresti JJ, Link DR, Weitz DA, Griffiths AD (2009) Fluorescence-activated droplet sorting (FADS): efficient microfluidic cell sorting based on enzymatic activity. Lab Chip 9:1850–1858
Berger M, Castelino J, Huang R, Shah M, Austin RH (2001) Design of a microfabricated magnetic cell separator. Electrophoresis 22:3883–3892
Bruus H (2008) Theoretical microfluidics. Oxford University, Oxford
Cecchini M, Girardo S, Pisignano D, Cingolani R, Beltram F (2008) Acoustic-counterflow microfluidics by surface acoustic waves. Appl Phys Lett 92:104103
Franke T, Braunmuller S, Schmid L, Wixforth A, Weitz DA (2010) Surface acoustic wave actuated cell sorting (SAWACS). Lab Chip 10:789–794
Fu AY, Spence C, Scherer A, Arnold FH, Quake SR (1999) A microfabricated fluorescence-activated cell sorter. Nat Biotechnol 17:1109–1111
Guillong M, Kuhn HR, Gunther D (2003) Application of a particle separation device to reduce inductively coupled plasma-enhanced elemental fractionation in laser ablation-inductively coupled plasma-mass spectrometry. Spectrochimica Acta Part B 58:211–220
Hellums JD (1993) Whitaker lecture: biorheology in thrombosis research. Ann Biomed Eng 22:445–455
Jellema LC, Mey T, Koster S, Verpoorte E (2009) Charge-based particle separation in microfluidic devices using combined hydrodynamic and electrokinetic effects. Lab Chip 9:1914–1925
Johann RM (2006) Cell trapping in microfluidic chips. Anal Bioanal Chem 385:408–412
Jones RAL (2002) Soft condensed matter. Oxford University, Oxford
Kang JH, Park JK (2005) Technical paper on microfluidic devices-cell separation technology. Asia Pacific Biotech News 9(21):1135–1146
Kroll MH, Hellums JD, McIntire LV, Schafer AI, Moake JL (1996) Platelets and shear stress. Blood 88:1525–1541
Lara O, Tong XD, Zborowski M, Chalmers JJ (2004) Enrichment of rare cancer cells through depletion of normal cells using density and flow-through, immunomagnetic cell separation. Exp Hematol 32:891–904
Lee GB, Chang CC, Huang SB, Yang RJ (2006) The hydrodynamic focusing effect inside rectangular microchannels. J Micromech Microeng 16:1024–1032
Lenshof A, Laurell T (2010) Continuous separation of cells and particles in microfluidic systems. Chem Soc Rev 39:1203–1217
Li H, Friend JR, Yeo LY (2007) Surface acoustic wave concentration of particle and bioparticle suspensions. Biomed Microdev 9:647–656
McCloskey KE, Chalmers JJ, Zborowski M (2003) Magnetic cell separation: characterization of magnetophoretic mobility. Anal Chem 75:6868–6874
Pamme N (2007) Continuous flow separations in microfluidic devices. Lab Chip 7:1644–1659
Petersson F, Åberg L, Swärd-Nilsson AM, Laurell T (2007) Free flow acoustophoresis: microfluidic-based mode of particle and cell separation. Anal Chem 79:5117–5123
Piccin A, Murphy WG, Smith OP (2007) Circulating microparticles: pathophysiology and clinical implications. Blood Rev 21:157–171
Pierce AD (1989) Acoustics-an introduction to its physical principles and applications. Acoustical Society of America, New York
Qu BY, Wu ZY, Fang F, Bai ZM, Yang DZ, Xu SK (2008) A glass microfluidic chip for continuous blood cell sorting by a magnetic gradient without labeling. Anal Bioanal Chem 392:1317–1324
Rogers PR, Friend JR, Yeo LY (2010) Exploitation of surface acoustic waves to drive size-dependent microparticle concentration within a droplet. Lab Chip 10:2979–2985
Saiki T, Okada K, Utsumi Y (2010) Micro liquid rotor operated by surface-acoustic-wave. Microsyst Technol 16:1589–1594
Seo J, Lean MH, Kole A (2007) Membrane-free microfiltration by asymmetric inertial migration. Appl Phys Lett 91:033901
Shi JJ, Ahmed D, Mao X, Huang TJ (2008a) Surface acoustic wave (SAW) induced patterning of micro beads in microfluidic channels. In: MEMS 2008: 21st IEEE international conference on micro electro mechanical systems, technical digest, Tucson, 2008, pp 26–29
Shi JJ, Mao X, Ahmed D, Colletti A, Huang TJ (2008b) Focusing microparticles in a microfluidic channel with standing surface acoustic waves (SSAW). Lab Chip 8:221–223
Shi JJ, Ahmed D, Mao X, Lin SCS, Lawit A, Huang TJ (2009a) Acoustic tweezers: patterning cells and microparticles using standing surface acoustic waves (SSAW). Lab Chip 9:2890–2895
Shi JJ, Huang H, Stratton Z, Huang YP, Huang TJ (2009b) Continuous particle separation in a microfluidic channel via standing surface acoustic waves (SSAW). Lab Chip 9:3354–3359
Shilton R, Tan MK, Yeo LY, Friend JR (2008) Particle concentration and mixing in microdrops driven by focused surface acoustic waves. J Applied Physics 104:1–9
Stiles T, Fallon R, Vestad T, Oakey J, Marr DWM, Squier J, Jimenez R (2005) Hydrodynamic focusing for vaccum-pumped microfluidics. Microfluid Nanofluid 1:280–283
Tan MK, Friend JR, Yeo LY (2007) Microparticle collection and concentration via a miniature surface acoustic wave device. Lab Chip 7:618–625
Tan MK, Tjeung R, Ervin H, Yeo LY, Friend JR (2009) Double aperture focusing transducer for controlling microparticle motions in trapezoidal microchannels with surface acoustic waves. Appl Phys Lett 95:134101
Tan MK, Yeo LY, Friend JR (2010) Unique flow transitions and particle collection switching phenomena in a microchannel induced by surface acoustic waves. Appl Phys Lett 97:234106
Weigl BH, Yager P (1999) Microfluidic diffusion-based separation and detection. Science 283(5400):346–347
Wong KK (2002) Properties of lithium niobate. INSPEC, London
Yamada M, Nakashima M, Seki M (2004) Pinched flow fractionation: continuous size separation of particles utilizing a laminar flow profile in a pinched microchannel. Anal Chem 71:5465–5471
Yang J, Huang Y, Wang XB, Becker FF, Gascoyne PRC (1999) Cell separation on microfabricated electrodes using dielectrophoretic/gravitational field flow fractionation. Anal Chem 71:911–918
Yang S, Undar A, Zahn JD (2006) A microfluidic device for continuous, real time blood plasma separation. Lab Chip 6:871–880
Ye NN, Qin JH, Shi WW, Liu X, Lin BC (2007) Cell–based high content screening using an integrated microfluidic device. Lab Chip 7:1696–1704
Yeo LY, Friend JR (2009) Ultrafast microfluidics using surface acoustic waves. BIomicrofluidics 3:012002
Yosioka K, Kawasima Y (1955) Acoustic radiation pressure on a compressible sphere. Acustica 5:167–173
Yung CW, Fiering J, Mueller AJ, Ingber DE (2009) Micromagnetic–microfluidic blood cleansing device. Lab Chip 9:1171–1177
Acknowledgments
This study was supported by a National Research Foundation of Korea (NRF) grant funded by the Korea government (MEST) (no. 2009-0080636).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nam, J., Lee, Y. & Shin, S. Size-dependent microparticles separation through standing surface acoustic waves. Microfluid Nanofluid 11, 317–326 (2011). https://doi.org/10.1007/s10404-011-0798-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10404-011-0798-1