Abstract
Purpose
Sterile ulceration is frequently observed in the cornea following persistent corneal epithelial damage. We examined the effect of alarmins released by necrotic corneal epithelial cells (HCE) on the production of matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs) by corneal fibroblasts.
Methods
IL-1α and high-mobility group box 1 protein (HMGB1) released into the supernatant derived from necrotic HCE cells were measured with enzyme-linked immunosorbent assay (ELISA). MMPs and TIMPs produced by corneal fibroblasts, stimulated with the supernatant from necrotic HCE cells, were analyzed and measured with protein array and ELISA. To investigate dynamic expression of alarmins in the corneal epithelium, we used immunohistochemistry to observe the expression of human IL-1α in the corneal epithelium of human IL-1α Tg mice with or without cryopexy. We also investigated the expression of MMPs in corneal stroma of the mice treated with cryopexy, using RT-PCR.
Results
We detected IL-1α and HMGB-1 in the supernatant of necrotic HCE cells. These supernatants increased the expression of MMP-3 and MMP-1, and decreased that of TIMP-1 and TIMP-2 in human corneal fibroblasts. Almost always these were inhibited by IL-1 receptor antagonist. Recombinant IL-1α increased the production MMP-3 and MMP-1 in corneal fibroblasts. After cryopexy of the epithelium of human IL-1α Tg mice, the expression of human IL-1α was recognized in the cytoplasm but not nucleus of epithelial cells. The level of MMP-3 and MMP-1 mRNAs was elevated in the corneal stroma in mice treated with cryopexy.
Conclusion
Alarmins, especially IL-1α, released from necrotic HCE cells may play an important role in the expression of MMPs and TIMPs by corneal fibroblast, resulting in sterile ulceration.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Various endogenous molecules released by necrotic cells are known as alarmins as they signal possible danger to surrounding tissue [1,2,3]. These molecules, which include high-mobility group box 1 protein (HMGB1), IL-1α, IL-33, heat shock protein 60, uric acid, S100 proteins, DNA, ATP and β-defensin2, induce inflammatory responses characterized by the rapid migration of inflammatory cells into the injured tissues. We have shown that alarmins released by necrotic corneal epithelial cells play an important role in corneal sterile inflammation, wound healing and allergic inflammation [4, 5]. However, the mechanism responsible for triggering sterile corneal ulceration in response to the injury of the corneal epithelium is unclear. Given that in individuals with sterile corneal ulceration the corneal epithelium is often damaged, we have examined whether alarmins released from necrotic corneal epithelial cells might contribute to sterile corneal ulceration. The necrosis of corneal epithelium can lead to corneal ulceration, which itself results from the destruction of collagen fibrils and proteoglycans in the stroma by proteolytic enzymes. Matrix metalloproteinases (MMPs) are proteolytic enzymes known to participate in degradation and remodeling of components of the extracellular matrix (ECM) and have been implicated in tissue injury and repair. There are at least 25 members in the MMP family [6]. Under normal conditions, MMP activity is required for tissue remodeling, but altered MMP activity is reported in association with several diseases such as tumor metastasis, heart failure, neurodegenerative diseases and rheumatoid arthritis [7,8,9,10,11,12,13,14,15]. MMPs are thought to play an important role in corneal ulceration and have also been identified in ulcerated corneal tissue. Differential expressions of MMPs in cornea were detected in patients with sterile ulceration associated with rheumatoid arthritis [16,17,18], sjögren’s syndrome [19], and in patients after photorefractive keratectomy with perioperative diclofenac use [20, 21]. Of the various MMPs, MMP-3, also known as stromelysin-1, is associated with pathogenesis of sterile ulceration of the cornea [16,17,18,19,20,21]. MMP-3 has broad substrate specificity and can degrade fibronectin, laminin, elastin, collagen I, II and proteoglycans of the ECM. MMP activity can be regulated by the formation of complexes with the naturally occurring endogenous inhibitor proteins called tissue inhibitors of metalloproteinase (TIMPs). There are four TIMPs known thus far, and among them, interaction of TIMP-1 with MMP-3 is best known. TIMP-1 binds non-covalently with MMP-3 at a molar ratio of 1:1 [22]. It is reported that the active form of MMP-3, not pro MMP-3, binds with TIMP-1 [23]. Therefore, the balance between TIMP-1 and MMP-3 would be critical in determining the cellular activity of MMP-3.
Epithelial-stromal interactions may play an important role in sterile corneal ulceration. The corneal epithelial cells also contribute either directly or indirectly to the production of MMPs and TIMPs. On the other hands, MMP-3 and MMP-1 are released at sites of corneal stromal wounds and contribute to remodeling or degradation of collagen fibrils. Several reports reveal that corneal fibroblasts can produce MMP-3, MMP-1 and MMP-9 [24,25,26,27]. Therefore we have now investigated the effects of alarmins released from necrotic cornea epithelial cells on the production of MMPs and TIMPs in corneal fibroblasts in vitro and in vivo.
Materials and methods
The use of cultured human cells and genetically modified mice was approved by the Ethical Committee of Juntendo Graduate school of Medicine. The Declaration of Helsinki protocols were followed. All animal experiments were performed in accordance with the guidelines in the ARVO statement for the Use of Animals in Ophthalmic and Vision Research.
Antibodies and ligands
A recombinant human IL-1α, HMGB-1 and TGF-β1 was obtained from PeproTech Inc. (Rocky Hill, NJ, USA). Recombinant human IL-1 receptor antagonist was purchased from ProSpec-TechnoGene Ltd. (Rehovot Science Park, Rehovot, Israel). Goat anti-human IL-1α polyclonal Ab (pAb) (R&D Systems, Minneapolis, MN, USA) was used in primary antibodies for immunohistochemistry. And biotinylated goat anti-rabbit Ab (DAKO, Glostrup, Denmark) was used in second antibody.
Cell culture
In this study we used cultured corneal epithelial cells and fibroblasts at second or third passages (ScienCell Research Laboratories, Carlsbad, CA, USA). Corneal fibroblasts were primarily cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal calf serum (FCS). The keratocytes showed transdifferentiation into corneal fibroblasts. Corneal epithelial cells grown in FCS-free Epi-medium (ScienCell Research Laboratories) were also used in primary culture.
Inducing necrosis of corneal epithelial cells
Following the procedure in previous reports [4, 5], necrosis was induced by three cycles of freezing and thawing. Corneal epithelial cells cultured with serum-free DMEM were frozen (− 80 °C) for 20 min and thawed (37 °C) for 20 min over three cycles. The supernatant obtained from necrotic corneal epithelial cells was used in this study.
Antibody array
Culture supernatant obtained from corneal fibroblasts was analyzed with an antibody array (RayBio; Human Matrix Metalloproteinase Antibody I kit; RayBiotech Inc., Norcross, CA, USA) according to the manufacturer’s instructions. Corneal fibroblasts were grown to subconfluence in DMEM containing 10% FCS, washed twice with PBS, and then incubated in serum-free DMEM for 24 h with or without the supernatant of necrotic corneal epithelial cells (final concentration: 20%) or recombinant IL-1α (30 ng/mL) or TGF-β1(30 ng/mL) or HMGB-I (100 μg/mL). The culture supernatant was then harvested for antibody array analysis. To estimate the effect of IL-1 receptor antagonist, corneal fibroblasts were incubated in serum-free DMEM for 24 h with the supernatant of necrotic corneal epithelial cells and IL-1 receptor antagonist (100 ng/mL), after which the culture supernatant was harvested for antibody array analysis. The mean optical intensity of positive spots was estimated by densitometry with analyzer software (Gel-Pro; Media Cybemetics, Inc, Silver Spring, MD, USA).
Enzyme-linked immunosorbent assay (ELISA)
To detect IL-1α, and HMGB-I in the supernatant of necrotic corneal epithelial cells, we used ELISA kits (R&D systems) according to the manufacturer’s instructions.
To detect MMP-3 in the supernatant of corneal fibroblasts, we used MMP-3 ELISA kits according to the manufacturer’s instructions (Quantikine; R&D Systems). Corneal fibroblasts were grown to subconfluence and then incubated in serum-free DMEM for 24 h with or without exposure to the supernatant of necrotic corneal epithelial cells or recombinant IL-1α. The supernatant was then harvested for ELISA.
Animals
Female C57BL/6N mice (10 weeks old) and human IL-1α Tg mice [28,29,30] were used in this experiment. IL-1α Tg mice were gifted by Norihiro Tada (Atopy Research Center, Juntendo Graduate School of Medicine, Tokyo, Japan). The mice were given an intraperitoneal (IP) injection of pentobarbital (40–50 mg/Kg) for deep anesthesia and a drop of 1% pro-paracaine for local anesthesia before creating corneal injury. Corneal injury was created by cryopexy while viewing the eye through an operating microscope. A cryoprobe was cooled to − 80 °C and placed on corneas for 3 s. Repetitive freezing-and-thawing procedures were applied to corneas. Samples were evaluated through hematoxylin–eosin staining. Twenty-four and 72 h after cryopexy, the animals were euthanized with an overdose of pentobarbital (100 mg/kg, administered IP), after which the eyes were removed and prepared for immunohistochemical and molecular analysis.
Immunohistochemical staining
The excised mouse eyes were fixed in 20% formalin in phosphatebuffered saline (PBS) at 4 °C overnight and then embedded in paraffin. Next, 3 μm thick sections of the eyes were mounted on microslides (New Silane; Muto-Glass, Tokyo, Japan). Deparaffinized sections were washed in PBS and rehydrated with 100% ethanol. Antigen retrieval was performed by boiling the sections in 0.01 M citrate buffer (pH 6) for 10 min. The slides were washed with PBS and blocked with 4% normal serum of second antibody animals and 0.3% bovine serum albumin for 30 min at room temperature. Subsequently, the slides were incubated with primary antibody (antihuman IL-1α pAb) overnight at 4 °C, followed by three washes in PBS and incubation for 40 min with biotinylated goat anti-rabbit Ab (1:300 dilution; DAKO, Glostrup, Denmark) at room temperature. After the slides were washed three times in PBS, they were incubated for 40 min with streptavidin-Alexa Fluor 488 (1:200 dilution; Molecular Probe). Counterstaining was done with DAPI (H-1200; Vector Laboratories, Inc., Burlingame, CA, USA). All sections were examined with a fluorescence microscope (AxioVision 3.1; Carl Zeiss Meditec, Inc. Dublin, CA, USA) and a laser confocal microscopy (TCS-SP5/TIRF; Leica Microsystems AG, Solms, Germany).
Preparation of RNA and real-time PCR
Total RNA was isolated from the removed corneas of h IL-1α Tg mouse, using NucleoSpin® RNA II (Macherey–Nagel GmbH, Duren, Germany). The steps were performed according to the manufacturer’s instructions. The concentration of RNA were assessed using a NanoDrop 1000 spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, USA). Complementary DNA was generated from 0.5 μg total RNA (ReverTra Ace®; Toyobo Co., Ltd., Osaka, Japan). Real-time PCR was performed with SYBR green master mix (Fast SYBR® Green Master Mix; Applied Biosystems Inc. Foster City, CA, USA) on a commercial system (7500 Fast Real-Time PCR System; ABI). Primers sequences are listed in Table 1. 10 μL of SYBR Green master mix was added to 2 μL of cDNA (corresponding to 50 ng of total RNA input) and 250 nM forward and reverse primers in water. Plates were heated at 95 °C for 20 s. Subsequently, 40 PCR cycles consisting of 3 s at 95 °C and 30 s at 60 °C were applied. Relative transcript levels were calculated using the delta–delta Ct method, normalized to GAPDH expression according to the following equation.
Statistical analysis
Results are expressed as the mean ± SE. Differences were evaluated by Student’s t-test (Figs. 1, 3) using analytical software (Excel; Microsoft, Redmond, WA, USA). Statistical evaluations (Figs. 2, 4, 5 and 7) were performed with the two tailed unpaired Mann–Whitney U test. P < 0.05 was considered statistically significant.
Antibody array of cultured supernatants from corneal fibroblasts stimulated by the supernatant of necrotic corneal epithelial cells. Cultured supernatants from corneal fibroblasts was analyzed with an antibody array. Representative arrays analyzing culture supernatant obtained from corneal fibroblasts are shown: a cells cultured with serum-free medium, b cells cultured with serum-free medium containing the supernatant of necrotic corneal epithelial cells, c: cells cultured with serum-free medium containing the supernatant of necrotic corneal epithelial cells and human/IL-1 receptor antagonist (IL-1RA: 100 ng/mL). Estimation of the mean optical intensity of positive spots from the culture supernatants. Corneal fibroblasts constitutively produced MMP-3, MMP-1, TIMP-1 and TIMP-2. a Blue square. When cells were cultured with the supernatant of necrotic corneal epithelial cells, the production of MMP-3 and MMP-1 was enhanced. b Red square. On the other hand, the production of TIMP-1 and TIMP-2 was decreased. a Green square. Treatment with an IL-1RA almost completely inhibited the increased production of MMP-3 and MMP-1. c Blue square
Results
IL-1α and HMGB1 in the supernatant of necrotic corneal epithelial cells
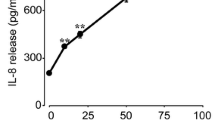
Using ELISA, we detected two kinds of alarmins: HMGB-1 and IL-1α in the supernatant of necrotic corneal epithelial cells (Fig. 1).
Antibody array of culture supernatants from corneal fibroblasts stimulated by the supernatant of necrotic corneal epithelial cells
Culture supernatant from corneal fibroblasts was analyzed with an antibody array.
The mean optical intensity of positive spots from the culture supernatants was estimated. Corneal fibroblasts constitutively produced MMP-3, MMP-1, TIMP-1 and TIMP-2 (Fig. 2a). When cells were cultured with the supernatant of necrotic corneal epithelial cells, the production of MMP-3 and MMP-1 was enhanced, whereas the production of TIMP-1 and TIMP-2 was decreased (Fig. 2b). Treatment with an IL-1RA almost completely inhibited the production of MMP-3 and MMP-1 (Fig. 2c).
The production of MMP-3 by corneal fibroblasts was examined by ELISA
The supernatant of normal corneal epithelial cells slightly induced the production of MMP-3. The supernatant of necrotic corneal epithelial cells or recombinant IL-1α (30 ng/mL) significantly enhanced the production of MMP-3 (Fig. 3).
Antibody array of culture supernatants from corneal fibroblasts stimulated by recombinant IL-1α and/or TGF-β1
Culture supernatant from corneal fibroblasts treated with recombinant human IL-1α (30 ng/mL) and/or TGF-β1 (30 ng/mL) was analyzed with antibody array. The mean optical intensity of positive spots was estimated from the culture supernatants. Corneal fibroblasts constitutively produced MMP-3, MMP-1, TIMP-1 and TIMP-2 strongly (Fig. 4a). Small amounts of other MMPs were also produced. When cells were treated with TGF-β1, the production of MMP-3 was slightly enhanced (Fig. 4b). When cells were treated with IL-1α, the production of MMP-3 was mildly enhanced (Fig. 4c). When cells were treated with TGF-β1 and IL-1α, the production of MMP-3 was strongly enhanced (Fig. 4d).
Antibody array of cultured supernatants from corneal fibroblasts stimulated by recombinant IL-1α and/or TGF-β1. Cultured supernatants from corneal fibroblasts treated with recombinant human IL-1α (30 ng/mL) and/or TGF-β1 (30 ng/mL) were analyzed with antibody array. Representative arrays analyzing cultured supernatants are shown: a Cells cultured with serum free medium. b Cells cultured with serum free medium with recombinant TGF-β1. c Cells cultured with serum free medium with recombinant IL-1α. d Cells cultured with serum free medium with recombinant TGF-β1 and IL-1α. The mean optical intensity of positive spots was estimated from the culture supernatants. Corneal fibroblasts constitutively produced MMP-3, MMP-1, TIMP-1 and TIMP-2 strongly. a Red square, green square. Other MMPs were produced slightly. When cells were treated with TGF-β1, the production of MMP-3 was slightly enhanced. b Red square. When cells were treated with IL-1α, the production of MMP-3 was mild enhanced. c red square. When cells were treated with TGF-β1 and IL-1α, the production of MMP-3 was strongly enhanced. d Red square
Antibody array of culture supernatants from corneal fibroblasts stimulate by recombinant HMGB-1
Cell culture supernatant from corneal fibroblasts treated with recombinant HMGB-1 (100 ng/mL) was analyzed with antibody array. The mean optical intensity of positive spots was estimated from the cell culture supernatants. HMGB-1 did not induce the production of MMP-3 and MMP-1 (Fig. 5a, b).
Antibody array of cultured supernatants from corneal fibroblasts stimulated by recombinant HMGB-1 (100 ng/mL). Representative arrays analyzing cultured supernatants are shown: a cells cultured with serum free medium. b Cells cultured with serum free medium with recombinant HMGB-1. The mean optical intensity of positive spots was estimated from the cultured supernatants. HMGB-1 did not induce the production of MMP-3 and MMP-1. b Blue square
The expression of human IL-1α in the cornea of human IL-1α Tg mice by immunohistochemistry
The expression of human IL-1α was limited in the nuclei of corneal epithelial cells of human IL-1α Tg mice (Fig. 6a, red stain). The expression of human IL-1α was enlarged to cytoplasm of corneal epithelial cells of human IL-1α Tg mice after cryopexy (Fig. 6b, red stain). It was observed that corneal endothelial cells were lost following transcorneal cryoinjury, and Descemet’s membrane was denuded where the cryoprobe was applied. In contrast, corneal epithelial cells showed no cryo-induced necrosis. It is well known that alarmins are released by not only necrotic cells, but also damaged cells. Therefore, corneal epithelial cells treated with cryoprobe released IL-1α from nucleus to cytoplasm.
The expression of human IL-1α in the cornea of human IL-1α Tg mice by immunohistochemistry. a Corneas of human IL-1α Tg mice with no treatment. b Corneas of human IL-1α Tg mice treated with cryopexy (after 24 h). a The expression of h IL-1α was limited in nucleus of corneal epithelial cells of human IL-1α Tg mice with no treatment. b The expression of human IL-1α was enlarged to cytoplasm of corneal epithelial cells of human IL-1α Tg mice treatment with cryopexy. (bar = 100 µM)
The expression of MMP-3·1 m-RNA in the corneal stroma by real-time PCR
Relative transcript levels were calculated using the delta–delta Ct methods. m-RNA of MMP-3 (Fig. 7a) and MMP-1 (Fig. 7b were elevated in corneal stroma after cryopexy at 24 or 72 h.
Discussion
In this study we detected alarmins such as IL-1α and HMGB1 in the supernatant of necrotic corneal epithelial cells. These supernatants increased the expression of MMP-3 and MMP-1, and decreased that of TIMP-1 and TIMP-2 in cultured human corneal fibroblasts at protein levels. IL-1R antagonist almost entirely inhibited this production. Furthermore, recombinant IL-1α enhanced the expression of MMP-3. Costimulation with IL-1α and TGF-β increased MMP-3 expression synergistically. HMGB-1, on the other hand, did not influence this production. These results demonstrate that IL-1α derived from necrotic corneal epithelial cells stimulated corneal fibroblasts via IL-1R, resulting in the upregulation of the production of MMP-3 and MMP-1, and down regulation of that of TIMP-1 and TIMP-2.
IL-1 molecules are encoded by two distinct genes, IL-1α and IL-1β. Both initially produce precursor polypeptides with a predicted Mr of 31 kDa. IL-1α precursor is biologically fully active, whereas IL-1β precursor displays no biological activity until it has been processed to form the 17-kDa mature form. Unlike other secreted proteins, IL-1α precursor lacks a hydrophobic leader sequence and is never found in organelles involved in classical secretory pathways. Wessendorf et al. reveal that IL-1α precursor protein contains a functional nuclear localization sequence within the structure of the precursor domain and exists in the nucleus [31]. Recently IL-1α became known as one alarmin, an endogenous danger signal released from necrotic cells [32].
Among the many alarmins, IL-1α was investigated in corneal sterile inflammation. Wilson et al. reveal that IL-1α is expressed by unwounded corneal epithelial cells and released corneal stroma after mechanical injury [33,34,35]. Hong et al. reveal that IL-1α had a crucial role in inflammatory cell infiltration into the cornea after corneal scrape injury in rabbits [36]. Stapleton examined the effect of topical soluble IL-1R antagonist on bone marrow-derived cell influx following corneal epithelial scrape injury in a mouse model and demonstrated that topical IL-1R antagonist markedly down regulates cell infiltration in corneal stroma [37]. Sotozono et al. show that in alkali-burned mouse corneas, IL-1α levels are dramatically elevated in the regenerated epithelium during early stages of alkali burn [38]. Recently, we revealed that IL-1α derived from necrotic corneal epithelial cells induces the production of IL-6 by corneal fibroblasts and has an important role in corneal sterile inflammation and wound healing [4]. Furthermore, we also revealed that alarmins including IL-1 are released from necrotic corneal epithelial cells and cooperate with Th2 cytokines to induce CCL 11 production and VCAM-1 expression in corneal fibroblasts [5]. Although IL-1α may be the master regulator of corneal sterile inflammation. However, the contribution of epithelial cell damage to corneal sterile ulceration does not provide sufficient information to merit a detailed investigation.
In this study, we revealed that IL-1α derived by damaged corneal epithelial cells induced the production of MMP-3 and MMP-1, and may thereby play an important role in ulceration associated with damages of corneal epithelium.
In this study, HMGB-1 did not induce corneal fibroblasts to produce MMP-3 and MMP-1. The exact mechanism that prevents corneal fibroblasts from reacting to the stimulation of HMGB-1 is still unknown. Recently, Park et al. revealed that HMGB-1 could interact with both Toll-like receptor2 (TLR2) and TLR4 and induce cellular activation and generate inflammatory responses that are similar to those initiate by LPS [39]. Our previous study revealed that corneal fibroblasts expressed TLR2 and 4 mRNA. However, the surface expression of TLR2 and 4 at protein levels can not be established in flow cytemetry analysis [40]. Therefore, the reason that corneal fibroblasts do not respond to HMGB1 may depend on the surface expression of TLR2 and 4. Furthermore, Chen et al. reveal that when they injected necrotic cells into TLR2/TLR4 double-deficient mice, they found a small reduction in neutrophil infiltration. On the other hand, when injected into IL-1 receptor mutant mice, they observed significant reduction in the neutrophil infiltration to necrotic cells [41]. Therefore, in this study, the reason that HMGB-1 did not influence the production of MMPs and TIMPs may depend on the expression and function of TLRs in corneal fibroblasts.
It is known that sterile ulceration of the cornea is observed in rheumatoid arthritic (RA) patients [16,17,18]. The exact mechanism of sterile ulceration in RA is not well known. Okamoto et al. reveal that IL-1 was highly expressed in the epithelium of MRL/l pr mice [42]. These mice spontaneously developed polyarthritis resembling RA, and also corneal involvement such as keratopathy and scleritis, which is a major complication in RA patients. Recently, Tada et al. generated transgenic (Tg) mice overexpressing the human IL-1α gene [28,29,30]. Macroscopic findings of Tg mice included hair loss, body weight loss, and bilaterally symmetrical polyarthritis. These mice spontaneously developed polyarthritis resembling RA. Tada et al. also examined the biological activity of transgene derived human IL-1α because the transgene was of human origin. The transgene-derived human IL-1α was biologically active [28,29,30]. In this study, we detected the expression of human IL-1α in the nucleus of corneal epithelial cells of these mice at protein level. When corneal epithelial cells were induced to damage by cryopexy, the expression of human IL-1α was enlarged and observed in the cytoplasm of corneal epithelial cells. Furthermore, the expression of MMP-3 and MMP-1 m-RNAs were increased in the cornea. These results may indicate that cryopexy induces damaged corneal epithelial cells and IL-1α in the nucleus is released to cytoplasm. IL-1α released from corneal epithelial cells may stimulate corneal fibroblasts to express MMP-3 and MMP-1 m-RNAs. Thus, IL-1α derived from damaged corneal epithelial cells may enhance the expression of MMP-3 and MMP-1 in corneal fibroblasts, resulting in sterile corneal ulceration. In conclusion, IL-1α derived from damaged corneal epithelial cells increased the production of MMP-3, MMP-1, and decreased the production of TIMP-1 and TIMP-2 in corneal fibroblasts. The interaction between corneal epithelial cells and fibroblasts may be essential to sterile ulceration in cornea. In future, topical IL-1α receptor antagonists or anti-IL-1α antibodies may become effective in the treatment for sterile ulceration with epithelial damage.
References
Matzinger P. The danger model: a renewed sense of self. Science. 2002;296:301–5.
Oppenheim JJ, Yang D. Alarmins: chemotactic activators of immune responses. Curr Opin Immunol. 2005;17:359–65.
Bianchi ME. DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol. 2007;81:1–5.
Ebihara N, Matsuda A, Nakamura S, Matsuda H, Murakami A. Role of the IL-6 classic- and trans-signaling pathways in corneal sterile inflammation and wound healing. Investig Ophthalmol Vis Sci. 2011;52:8549–57.
Fukuda K, Ishida W, Tanaka H, Harada Y, Matsuda A, Ebihara N, et al. Alarmins from corneal epithelial cells upregulate CCL11 and VCAM-1 in corneal fibroblasts. Invest Ophthalmol Vis Sci. 2013;54:5817–23.
Overall CM, López-Otín C. Strategies for MMP inhibition in cancer: innovations for the post-trial era. Nat Rev Cancer. 2002;2:657–72 (review).
Deryugina EI, Quigley JP. Matrix metalloproteinases and tumor metastasis. Cancer Metastasis Rev. 2006;25:9–34.
Martin MD, Matrisian LM. The other side of MMPs: protective roles in tumor progression. Cancer Metastasis Rev. 2007;26:717–24.
Graham HK, Horn M, Trafford AW. Extracellular matrix profiles in the progression to heart failure. European Young Physiologists Symposium Keynote Lecture-Bratislava 2007. Acta Physiol (Oxf). 2008;194:3–21.
Cauwe B, Van den Steen PE, Opdenakker G. The biochemical, biological, and pathological kaleidoscope of cell surface substrates processed by matrix metalloproteinases. Crit Rev Biochem Mol Biol. 2007;42:113–85.
Gueders MM, Foidart JM, Noel A, Cataldo DD. Matrix metalloproteinases (MMPs) and tissue inhibitors of MMPs in the respiratory tract: potential implications in asthma and other lung diseases. Eur J Pharmacol. 2006;533:133–44.
Sapolsky AI, Keiser H, Howell DS, Woessner JF Jr. Metalloproteases of human articular cartilage that digest cartilage proteoglycan at neutral and acid pH. J Clin Invest. 1976;58:1030–41.
Dean DD, Martel-Pelletier J, Pelletier JP, Howell DS, Woessner JF Jr. Evidence for metalloproteinase and metalloproteinase inhibitor imbalance in human osteoarthritic cartilage. J Clin Invest. 1989;84:678–85.
Arend WP, Deter JM. Cytokines and cytokine inhibitors or antagonists in rheumatoid arthritis. Arthritis Rheumatol. 1990;33:305–15.
Martel PJ, Zafarullah M, Kodama S, Pelletier JP. In vitro effects of interleukin 1 on the synthesis of metalloproteases, TIMP, plasminogen activators and inhibitors in human articular cartilage. J Rheumatol Suppl. 1991;27:80–4.
Riley GP, Harrall RL, Watson PG, Cawston TE, Hazleman BL. Collagenase (MMP-1) and TIMP-1 in destructive corneal disease associated with rheumatoid arthritis. Eye. 1995;9:703–18.
Okada Y, Nagase H, Harris ED Jr. Matrix metalloproteinases 1, 2, and 3 from rheumatoid synovial cells are sufficient to destroy joints. J Rheumatol. 1987;14:41–2.
Okada Y, Harris ED Jr, Nagase H. The precursor of a metalloendopeptidase from human rheumatoid synovial fibroblasts. Purification and mechanisms of activation by endopeptidases and 4-aminophenylmercuric acetate. Biochem J. 1988;254:731–41.
Brejchova K, Liskova P, Hrdlickova E, Filipec M, Jirsova K. Matrix metalloproteinases in recurrent corneal melting associated with primary Sjörgen’s syndrome. Mol Vis. 2009;15:2364–72.
Gabison EE, Chastang P, Menashi S, Mourah S, Doan S, Oster M, et al. Late corneal perforation after photorefractive keratectomy associated with topical diclofenac: involvement of matrix metalloproteinases. Ophthalmology. 2003;110:1626–31.
O’Brien TP, Li QJ, Sauerburger F, Reviglio VE, Rana T, Ashraf MF. The role of matrix metalloproteinases in ulcerative keratolysis associated with perioperative diclofenac use. Ophthalmology. 2001;108:656–9.
Okada Y, Watanabe S, Nakanishi I, Kishi J, Hayakawa T, Watorek W, et al. Inactivation of tissue inhibitor of metalloproteinases by neutrophil elastase and other serine proteinases. FEBS Lett. 1988;229:157–60.
Hornebeck W, Lambert E, Petitfrere E. Bernard P. Beneficial and detrimental influences of tissue inhibitor of metalloproteinase-1 (TIMP-1) in tumor progression. Biochimie. 2005;87:377–83.
Fini ME, Girard MT, Matsubara M, Bartlett JD. Unique regulation of the matrix metalloproteinase, gelatinase B. Invest Ophthalmol Vis Sci. 1995;36:622–33.
Sivak JM, Fini ME. MMPs in the eye: emerging roles for matrix metalloproteinases in ocular physiology. Prog Retin Eye Res. 2002;21:1–14.
Hao JL, Nagano T, Nakamura M, Kumagai N, Mishima H, Nishida T. Galardin inhibits collagen degradation by rabbit keratocytes by inhibiting the activation of pro-matrix metalloproteinases. Exp Eye Res. 1999;68:565–72.
Kimura K, Orita T, Kondo Y, Zhou H, Nishida T. Upregulation of matrix metalloproteinase expression by poly(I:C) in corneal fibroblasts: role of NF-κB and interleukin-1ß. Invest Ophthalmol Vis Sci. 2010;51:5012–8.
Niki Y, Yamada H, Sek S, Kikuchi T, Takaishi H, Toyama Y, et al. Macrophage- and neutrophil-dominant arthritis in human IL-1 alpha transgenic mice. J Clin Invest. 2001;107:1127–35.
Niki Y, Yamada H, Kikuchi T, Toyama Y, Matsumoto H, Fujikawa K, et al. Membrane-associated IL-1 contributes to chronic synovitis and cartilage destruction in human IL-1 alpha transgenic mice. J Immunol. 2004;172:577–84.
Niki Y, Takaishi H, Takito J, Miyamoto T, Kosaki N, Matsumoto H, et al. Administration of cyclooxygenase-2 inhibitor reduces joint inflammation but exacerbates osteopenia in IL-1 alpha transgenic mice due to GM-CSF overproduction. J Immunol. 2007;179:639–46.
Wessendorf JH, Garfinkel S, Zhan X, Brown S, Maciag T. Identification of a nuclear localization sequence within the structure of the human interleukin-1 alpha precursor. J Biol Chem. 1993;268:22100–4.
Eigenbrod T, Park JH, Harder J, Iwakura Y, Núñez G. Cutting edge: critical role for mesothelial cells in necrosis-induced inflammation through the recognition of IL-1 alpha released from dying cells. J Immunol. 2008;181:8194–8.
Wilson SE, He YG, Weng J, Li Q, McDowall AW, Vital M, et al. Epithelial injury induces keratocyte apoptosis: hypothesized role for the interleukin-1 system in the modulation of corneal tissue organization and wound healing. Exp Eye Res. 1996;62:325–7.
Wilson SE, Liu JJ, Mohan RR. Stromal–epithelial interactions in the cornea. Prog Retin Eye Res. 1999;18:293–309.
Wilson SE, Esposito A. Focus on molecules: interleukin-1: a master regulator of the corneal response to injury. Exp Eye Res. 2009;89:124–5.
Hong JW, Liu JJ, Lee JS, Mohan RR, Mohan RR, Woods DJ, et al. Proinflammatory chemokine induction in keratocytes and inflammatory cell infiltration into the cornea. Invest Ophthalmol Vis Sci. 2001;42:2795–803.
Stapleton WM, Chaurasia SS, Medeiros FW, Mohan RR, Sinha S, Wilson SE. Topical interleukin-1 receptor antagonist inhibits inflammatory cell infiltration into the cornea. Exp Eye Res. 2008;86:753–7.
Sotozono C, He J, Matsumoto Y, Kita M, Imanishi J, Kinoshita S. Cytokine expression in the alkali-burned cornea. Curr Eye Res. 1997;16:670–6.
Park JS, Svetkauskaite D, He Q, Kim JY, Strassheim D, Ishizaka A, et al. Involvement of toll-like receptors 2 and 4 in cellular activation by high mobility group box 1 protein. J Biol Chem. 2004;279:7370–7.
Ebihara N, Yamagami S, Chen L, Tokura T, Iwatsu M, Ushio H, et al. Expression and function of toll-like receptor-3 and -9 in human corneal myofibroblasts. Invest Ophthalmol Vis Sci. 2007;48:3069–76.
Chen CJ, Kono H, Golenbock D, Reed G, Akira S, Rock KL. Identification of a key pathway required for the sterile inflammatory response triggered by dying cells. Nat Med. 2007;13:851–6.
Okamoto M, Takagi M, Kutsuna M, Hara Y, Nishihara M, Zhang MC. High expression of interleukin-1beta in the corneal epithelium of MRL/lpr mice is under the control of their genetic background. Clin Exp Immunol. 2004;136:239–44.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
A. Iwatake, None; A. Murakami, None; N. Ebihara, None.
About this article
Cite this article
Iwatake, A., Murakami, A. & Ebihara, N. The expression of matrix metalloproteinases and their inhibitors in corneal fibroblasts by alarmins from necrotic corneal epithelial cells. Jpn J Ophthalmol 62, 92–100 (2018). https://doi.org/10.1007/s10384-017-0541-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-017-0541-x