Summary
Background
Complete resection is the standard of care for thymic malignancies. There is still a debate about the optimal surgical approach for thymic tumors, particularly regarding long-term oncological outcome. This study aimed to compare videothoracoscopic surgery (VTS) with open surgery for thymic malignancies, regarding perioperative and long-term oncological outcomes.
Methods
A prospective study ran from 2010 to 2019. Patients with thymoma or thymic carcinoma underwent complete thymectomy via VTS or open surgery and were followed up. The long-term oncological outcome was disease recurrence.
Results
There were 29 patients in the VTS group and 35 patients in the open group with an average follow-up period of 80 months. The VTS approach significantly shortened operative duration (89.7 versus 116.9 min), caused less blood loss (56.9 versus 176.3 ml), reduced pain score (4.8 versus 6.7), and shortened chest drainage duration (2.1 versus 3.1 days) as well as hospital stay (5.1 versus 7.7 days). The two groups were comparable in long-term oncological outcome (two recurrences in the VTS group and one in the open group).
Conclusions
Compared to open surgery, VTS leads to superior perioperative outcomes and a comparable long-term oncological outcome. The authors advocate the VTS approach as a routine option for the surgery of thymic malignancies.
Main novel aspects
Videothoracoscopic surgery is less traumatic and has faster recovery times than open surgery, with a comparable long-term oncological outcome.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Thymic tumors account for 0.2–1.5% of all malignancies with a reported incidence from 0.13 to 0.17 per 100,000 person-years [1,2,3]. Although rare, they are still the most common type of mediastinal tumors reported in adults [4]. Thymomas and thymic carcinomas account for the majority of thymic malignancies. Surgical treatment with complete resection has been found an important factor for overall survival of patients. Therefore, this procedure is the standard of care in the management of thymic malignancies [5, 6]. Traditionally, this has been achieved with open procedures via median sternotomy or thoracotomy. The open procedures have been proved to be successful in obtaining optimal outcomes in patients with thymic tumors [5, 7]. Recently, with the widespread use of minimally invasive techniques in thoracic surgery, videothoracoscopic surgery (VTS) has been widely used in the treatment of thymic malignancies. A number of studies in the last decade were carried out to compare the open procedures with the VTS in clinical outcomes of patients with thymic tumors. Unfortunately, there was no randomized controlled trial in this area due to the infrequence of this disease, and there is still a debate about which technique is optimal for the treatment of thymic malignancies, particularly regarding long-term oncological outcome [8,9,10,11,12,13,14,15]. Therefore, the aim of the study was to investigate whether the VTS approach was better than open procedures in the treatment of thymic malignancies, regarding perioperative outcomes and long-term oncological outcomes in the authors’ institution.
Methods
Patients and study design
This was a subgroup follow-up of a prospective single-center study comparing clinical outcomes between patients with mediastinal tumors that underwent VTS resection (group 1) versus patients that underwent open resection (group 2) via thoracotomy or median sternotomy. Patients were recruited between 2010 and 2013 in a tertiary referral center in Ho Chi Minh City, Vietnam. The study protocol was approved by the local institutional ethics committee. Briefly, patients with radiological evidence of mediastinal tumor or cyst on computed tomography (CT), either in the anterior, middle, or posterior mediastinum were consecutively enrolled to the study at the time the radiological diagnosis of primary mediastinal tumor became available. Eligible patients were excluded if: (1) there was any evidence of invasion to the heart, lungs, large blood vessels, trachea, and esophagus on CT images; (2) patients had comorbidity of myasthenia gravis; and (3) postoperative pathologies were tumors of the esophagus or trachea, secondary tumors, lymph nodes due to the metastasis of other cancers, or pulmonary tuberculosis. Written informed consent was obtained from all patients and relatives before enrollment. Enrolled patients were divided into the two treatment arms (VTS and open surgery) based on the consultation of surgeons, radiologists, and patients. All enrolled patients were operated and managed by the same surgical team. At the authors’ center, before performing this study, VTS services had been started in 1998 and performed in cases of lung tumors, recurrent pneumothorax due to subpleural blebs or bullae, pleural effusion, and mediastinal tumors. All patients with the postoperative pathological finding of thymic malignancies (thymoma or thymic carcinoma) were then included in this study and followed-up until 2019 to compare the long-term oncological outcome of the two treatment arms.
Operative techniques
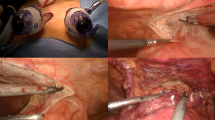
Complete thymectomy was performed in all cases in both groups. In the VTS group, the procedure was conducted with patients in the lateral position. General anesthesia was established in all patients with single lung ventilation via double-lumen endotracheal intubation. Three ports were inserted using the triangle method: one port for a 10-mm 30° thoracoscope located at the mid-axillary line in the forth to eighth intercostal spaces, and two 10-mm ports for instrument manipulation. The procedures were carefully performed to avoid injury to the phrenic nerve and other major organs. The thymic vein and other large vessels were clamped with endoclippers or sealed with ultrasonic scalpel. An endobag was used to place all resected specimens before retrieving them. If a mass was too large to be removed with a retrieval bag, it was cut into pieces and removed in a double-layered bag. In the case of difficulty in the dissection or removal of the tumor via the VTS procedure, one of the three ports was converted to a 4-cm transaxillary incision to facilitate the procedure. One 32‑F chest tube was routinely retained after the procedure. Figure 1 outlines the most important steps in performing VTS in a case of thymoma. In the open surgery group, thymic tumors were removed through a conventional thoracotomy or median sternotomy, depending on the location of the tumor. Postoperative treatment was according to the routine approach for thoracotomy or sternotomy. All patients with postoperative pathological findings of thymic carcinoma or Masaoka-Koga stage III–IV thymoma then received a medical oncology consultation to receive adjuvant chemotherapy or radiotherapy.
Major steps in videothoracoscopic surgery for thymic tumor. a Preoperative computed tomographic image. b Identification of the thymic tumor and major organs. c, d, e Dissection of the thymic tumor. f The thymic vein was dissected and clamped with endoclippers. g Removal of the thymic tumor without any injury to the phrenic nerve and other major organs. h Histopathology image of thymoma. Asterisk thymic tumor; hashtag superior vena cava; arrow phrenic nerve; circle heart; square lung; triangle innominate vein
Study outcomes
Perioperative outcomes included the duration of surgery, the amount of blood loss during the operation, the period of time required for chest tube drainage, the duration of postoperative hospitalization, postoperative pain score measured by the visual analog scale (VAS), and postoperative complications. The criteria for chest tube removal included: (1) no blood or chyle in the output; (2) no air leakage; (3) less than 200 ml of output within 24 h; and (4) fully expanded lungs on chest X‑ray. The criteria for discharge from hospital included: (1) chest tube removal, (2) oral intake, and (3) no complications.
Clinical and chest X‑ray examinations were performed for all patients after discharge 1 week, 1, 3, 6, and 12 months, as well as every year thereafter. During the follow-up period, if patients had signs or symptoms suggestive of recurrence of mediastinal tumor, they were evaluated by chest CT scan. The long-term oncological outcome was the evidence of recurrence of thymic tumor.
Statistical analysis
All baseline characteristics and perioperative outcomes were compared between the two groups using two-sided Fisher’s exact test for categorical variables, and Mann-Whitney-U test for numeric variables, when appropriate. Regarding the long-term outcome, the cumulative probability of recurrence-free survival was calculated by a Kaplan-Meier estimate and illustrated by the Kaplan-Meier curves with the number of patients at risk indicated below the plot at specific time points. The two-sided log-rank test was used to compare the recurrence rate between the two treatment arms. In addition, the recurrence rate was estimated in each subgroup of age, gender, tumor size, tumor location, invasive property on imaging, pathological finding, pathological Masaoka-Koga stage, surgical technique, and adjuvant therapy to find potential factors related to the long-term outcome. The differences in the recurrence rate between these subgroups were tested using the two-sided log-rank test. Statistical significance was defined when the P-value was less than 0.05. Statistical analyses were performed with R statistical software version 3.4.4 (Austria) [16].
Results
Patient enrollment
During the study recruitment period from 2010 to 2013, 246 patients were screened for eligibility at the authors’ institution, of which 182 patients were excluded and 64 patients with the postoperative pathological diagnosis of thymic malignancies were followed-up (29 cases in the VTS group, and 35 cases in the open group) (Fig. 2). In the VTS group, five cases required a 4-cm transaxillary incision to facilitate the procedure. In the open group, thoracotomy was performed in 30 cases, the other five cases underwent sternotomy. There were six patients lost to follow-up after an outpatient visit at 3 months; among these, two patients were in the VTS group, and four patients were in the open group.
Baseline characteristics
Baseline characteristics of included patients in each group are shown in Table 1. The two groups did not significantly differ in terms of age, gender, American Society of Anesthesia (ASA) physical status classification, and comorbidities. The tumor size was slightly lower in the VTS group compared to the open group (7.5 ± 2.3 vs. 8.2 ± 2.4 cm). The majority of thymic tumors were located in the anterior mediastinum (93.1% in the VTS group, and 94.3% in the open group). The proportion of tumor invasion to the capsule or surrounding tissues was lower in the VTS group compared to the open group (20.7% vs. 31.5%), but the difference was not significant. There were 55 cases with thymoma, which were equally divided into the two groups (26 cases in the VTS group and 27 cases in the open group). The number of cases with thymic carcinoma was 11 (three cases in the VTS group and eight cases in the open group). There were more patients in the early stage (stage I) according to the pathological Masaoka-Koga classification in the VTS group compared to the open group (79.3% vs. 57.1%). Due to higher proportions of thymic carcinoma and the later Masaoka-Koga stages, the open group had a significantly higher proportion of adjuvant therapy (31.5% vs. 6.9%). The average follow-up duration was approximately 80 months in both groups.
Perioperative outcomes
In the VTS group, no case required conversion to an open procedure. The VTS group was significantly better than the open group regarding most of the perioperative outcomes, which are shown in Table 2. Compared to the open group, the VTS group required shorter duration of surgery (89.7 ± 43.8 vs. 116.9 ± 45.2 min), caused less intraoperative blood loss (56.9 ± 59.2 vs. 176.3 ± 203.1 ml), and had lower postoperative pain scores (4.8 ± 1.5 vs. 6.7 ± 1.4). The postoperative chest tube drainage and hospitalization durations were also significantly shorter in the VTS group (chest drainage duration: 2.1 ± 0.3 vs. 3.1 ± 0.7 days; postoperative hospital length of stay: 5.1 ± 1.4 vs. 7.7 ± 1.9 days). Regarding perioperative complications, two patients had bleeding and one patient had postoperative atelectasis in the open group, while no complications were observed in the VTS group.
Long-term oncological outcome
No mortality was observed within the two groups during the follow-up period. There were three cases (4.7%) with recurrence of thymic malignancies (two cases in the VTS group and one case in the open group). In the VTS group, one recurrence was detected in a thymoma case with Masaoka-Koga stage II at 6 months of follow-up. The other recurrence was detected at 12 months of follow-up in a thymic carcinoma case with Masaoka-Koga stage III. In the open group, the recurrence occurred in a thymic carcinoma case with Masaoka-Koga stage III at 12 months of follow-up. None of the recurrence cases received adjuvant therapy after their first operations due to their decisions. The thymoma case received open surgery and chemotherapy at the time of recurrence. The two thymic carcinoma cases received only chemotherapy for their recurrence treatment. These cases did not have any later recurrence during the follow-up period.
Figure 3 shows the Kaplan-Meier estimates for the probability of recurrence-free survival across treatment groups. There was no significant difference between the two groups regarding the long-term oncological outcome. The recurrence rate for each category of pre- and postoperative characteristics is shown in Table 3. The authors found that four factors significantly increased the recurrence rate: tumor size of larger than 9 cm; tumor with capsular invasion or invasion in surrounding tissues on imaging; thymic carcinoma (compared to thymoma); and tumor with more advanced Masaoka-Koga stage.
Discussion
Results from this study suggest that VTS is feasible and safe in the treatment of thymic malignancies. Compared to open surgical approaches, VTS required significantly shorter duration of surgery, postoperative chest tube drainage, and postoperative hospital stay, and reduced postoperative pain in patients with thymic malignancies. Regarding long-term oncological outcome, no mortality and a low probability of recurrence (3/64 cases, 4.7%) were observed over an average follow-up period of 80 months. Recurrence was not associated with the surgical techniques, but other factors were found that related to the recurrence outcome, including tumor size, tumor invasion on imaging, histology of thymic malignancy, and Masaoka-Koga stage.
With the advantages of minimal invasiveness, minimally invasive surgery has been gaining popularity in recent years for thymectomy worldwide. Several large cohort retrospective studies showed a noticeable increase in the VTS approach in the treatment of thymic malignancies, particularly in Asian countries [10,11,12,13]. Previous studies have shown superior perioperative results of VTS in terms of reduced blood loss, less trauma to the chest wall, shortened operative time, fast healing, shortened postoperative hospital stay, and early return to normal activities. Due to reduced tissue damage, VTS also decreases postoperative complications and pain compared to open approaches [17, 18]. These advantages of VTS were confirmed in the present study. With well-trained surgeons, VTS obviously shortened operative duration, reduced the amount of perioperative blood loss, postoperative pain, the duration of chest tube drainage, and postoperative hospital stay. There was no major complication and no mortality in the VTS group. Another consequence of the rapid healing time in VTS is that this permitted earlier administration of adjuvant therapy in cases with advanced Masaoka-Koga stage. However, VTS has some technical difficulties due to large tumor size or adhesion with other organs, e.g., the esophagus, aorta, vena cava, heart, or phrenic nerve. The injury to these organs should be strictly avoided when performing the procedure. In addition, some authors stated that VTS was contraindicated for large thymic tumors, especially for tumors larger than 10 cm [19, 20]. In this study, the average tumor size was 7.5 ± 2.3 cm in the VTS group. There were two cases with tumors larger than 10 cm and one case required a transaxillary incision to facilitate the procedure. No case in the VTS group required conversion to an open procedure. Therefore, VTS is feasible and safe in the surgery of thymic malignancies, given well-trained surgeons and careful evaluation before surgery.
Currently, minimally invasive procedures have not been routinely recommended by the guidelines for the treatment of thymic malignancies due to lack of evidence for oncological efficacy. Several non-randomized retrospective studies reported similar overall survivals, disease-free survivals, and recurrence rates between VTS and open surgery [10,11,12,13, 20,21,22]. The results of this study consolidated this finding. With an approximately 9‑year follow-up period, no mortality was observed, and recurrence occurred in less than 5% of the study population and did not differ between the VTS and open groups. This might be due to the fact that complete thymectomy was performed in all cases. Complete tumor resection has been shown to be an important indicator for long-term oncological outcomes in the treatment of thymic malignancies [5, 6, 23]. Therefore, whichever minimally invasive technique or open technique is performed, complete thymectomy should be achieved to gain better long-term survival and recurrence-free outcomes. Several studies in early-stage thymic tumors found that VTS and open surgery were comparable in terms of the R0 resection rate [8, 13, 24]. Thus, it is reasonable to assume that thymic malignancies in stage I–II could be managed via VTS due to its feasibility to completely remove the tumor and to achieve satisfactory long-term oncological outcomes.
Several prognostic factors for the treatment of thymic malignancies have been found, whereby tumor stage, histology, and resection status have been considered the three most important factors [12, 13, 25,26,27]. It was not possible to make a robust finding for prognostic factors in this study due to limited sample size and the small number of recurrence events. However, the univariable analysis showed similar results to the literature, in which tumor stage and histology were significantly associated with the long-term recurrence outcome. In fact, early-stage thymic tumors could be completely resected by either VTS or open procedure. Given the feasibility of VTS in the resection of thymic tumors, the long-term prognosis of patients would depend on the nature of the disease, including tumor histology and stage of disease. It would be unusual for the surgical approach to be an important prognostic factor as long as the tumor could be completely removed by either minimally invasive or traditional open techniques.
A major limitation of our study is the limited sample size due to the rarity of the disease in a single center. There should be a multi-center database in our country, as in Europe or China, so that more robust results could be reported. Another limitation is the lack of randomization, which might lead to the none-equivalence between the two groups. This is similar to many studies in this area, as a powerful randomized controlled trial could not be feasible due to the infrequence of this disease. However, with the main strength of a prospective design and long-term follow-up period with a low percentage lost to follow-up, the results support VTS in the treatment of thymic malignancies.
Conclusion
VTS for resection of thymic malignancies is feasible, effective, and safe. Compared to traditional open surgery, VTS is superior regarding perioperative results by reducing intraoperative blood loss, postoperative pain, and shortening the duration of surgery, postoperative chest drainage, and hospital stay. Given the comparable long-term oncological and survival outcomes with open surgery, VTS could be considered as a routine approach for the surgical management of thymic malignancies.
References
Engels EA, Pfeiffer RM. Malignant thymoma in the United States: Demographic patterns in incidence and associations with subsequent malignancies. Int J Cancer. 2003;105(4):546–51.
Engels EA. Epidemiology of thymoma and associated malignancies. J Thorac Oncol. 2010;5(10):S260–S5.
Siesling S, Van Der Zwan JM, Izarzugaza I, Jaal J, Treasure T, Foschi R, et al. Rare thoracic cancers, including peritoneum mesothelioma. Eur J Cancer. 2012;48(7):949–60.
Strollo DC, Rosado de Christenson ML, Jett JR. Primary mediastinal tumors. Part 1: Tumors of the anterior mediastinum. Chest. 1997;112(2):511–22.
Detterbeck F, Youssef S, Ruffini E, Okumura M. A review of prognostic factors in thymic malignancies. J Thorac Oncol. 2011;6(7):S1698–S704.
Scorsetti M, Leo F, Trama A, D’Angelillo R, Serpico D, Macerelli M, et al. Thymoma and thymic carcinomas. Crit Rev Oncol Hematol. 2016;99:332–50.
Kondo K, Monden Y. Therapy for thymic epithelial tumors: A clinical study of 1,320 patients from Japan. Ann Thorac Surg. 2003;76(3):878–84.
Friedant AJ, Handorf EA, Su S, Scott WJ. Minimally invasive versus open thymectomy for thymic malignancies: Systematic review and meta-analysis. J Thorac Oncol. 2016;11(1):30–8.
Hess NR, Sarkaria IS, Pennathur A, Levy RM, Christie NA, Luketich JD. Minimally invasive versus open thymectomy: A systematic review of surgical techniques, patient demographics, and perioperative outcomes. Ann Cardiothorac Surg. 2016;5(1):1.
Wang H, Gu Z, Ding J, Tan L, Fu J, Shen Y, et al. Perioperative outcomes and long-term survival in clinically early-stage thymic malignancies: Video-assisted thoracoscopic thymectomy versus open approaches. J Thorac Dis. 2016;8(4):673.
Agatsuma H, Yoshida K, Yoshino I, Okumura M, Higashiyama M, Suzuki K, et al. Video-assisted thoracic surgery thymectomy versus sternotomy thymectomy in patients with thymoma. Ann Thorac Surg. 2017;104(3):1047–53.
Fang W, Yao X, Antonicelli A, Gu Z, Detterbeck F, Vallières E, et al. Comparison of surgical approach and extent of resection for Masaoka-Koga stage I and II thymic tumours in Europe, North America and Asia: An International Thymic Malignancy Interest Group retrospective database analysis. Eur J Cardiothorac Surg. 2017;52(1):26–32.
Gu Z, Chen C, Wang Y, Wei Y, Fu J, Zhang P, et al. Video-assisted thoracoscopic surgery versus open surgery for stage I thymic epithelial tumours: A propensity score-matched study. Eur J Cardiothorac Surg. 2018;54(6):1037–44.
Ruffini E, Filosso PL, Guerrera F, Lausi P, Lyberis P, Oliaro A. Optimal surgical approach to thymic malignancies: New trends challenging old dogmas. Lung Cancer. 2018;118:161–70.
Zhang X, Gu Z, Fang W. Minimally invasive surgery in thymic malignances: The new standard of care. J Thorac Dis. 2018;10(Suppl 14):S1666.
R Core Team. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2018. Available from: https://www.R-project.org.
Qi K, Wang B, Wang B, Zhang LB, Chu XY. Video-assisted thoracoscopic surgery thymectomy versus open thymectomy in patients with myasthenia gravis: A meta-analysis. Acta Chir Belg. 2016;116(5):282–8.
Yang Y, Dong J, Huang Y. Thoracoscopic thymectomy versus open thymectomy for the treatment of thymoma: A meta-analysis. Eur J Surg Oncol. 2016;42(11):1720–8.
Girard N, Mornex F, Van Houtte P, Cordier JF, van Schil P. Thymoma: A focus on current therapeutic management. J Thorac Oncol. 2009;4(1):119–26.
Youssef SJ, Louie BE, Farivar AS, Blitz M, Aye RW, Vallieres E. Comparison of open and minimally invasive thymectomies at a single institution. Am J Surg. 2010;199(5):589–93.
Pennathur A, Qureshi I, Schuchert MJ, Dhupar R, Ferson PF, Gooding WE, et al. Comparison of surgical techniques for early-stage thymoma: Feasibility of minimally invasive thymectomy and comparison with open resection. J Thorac Cardiovasc Surg. 2011;141(3):694–701.
Jurado J, Javidfar J, Newmark A, Lavelle M, Bacchetta M, Gorenstein L, et al. Minimally invasive thymectomy and open thymectomy: Outcome analysis of 263 patients. Ann Thorac Surg. 2012;94(3):974–82.
Fang W, Chen W, Chen G, Jiang Y. Surgical management of thymic epithelial tumors: A retrospective review of 204 cases. Ann Thorac Surg. 2005;80(6):2002–7.
Burt BM, Yao X, Shrager J, Antonicelli A, Padda S, Reiss J, et al. Determinants of complete resection of thymoma by minimally invasive and open thymectomy: Analysis of an international registry. J Thorac Oncol. 2017;12(1):129–36.
Fang W, Fu J, Shen Y, Wei Y, Tan L, Zhang P, et al. Management of thymic tumors—Consensus based on the Chinese Alliance for Research in Thymomas Multi-institutional retrospective studies. J Thorac Dis. 2016;8(4):641.
Tseng Y‑C, Tseng Y‑H, Kao H‑L, Hsieh C‑C, Chou T‑Y, Goan Y‑G, et al. Long term oncological outcome of thymoma and thymic carcinoma—An analysis of 235 cases from a single institution. PLoS ONE. 2017;12(6):e179527.
Li J‑F, Hui B‑G, Li X, Xiao R‑X, Jiang G‑C, Liu J, et al. Video-assisted thoracic surgery for thymoma: Long-term follow-up results and prognostic factors—Single-center experience of 150 cases. J Thorac Dis. 2018;10(1):291.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation as well as data collection and analysis were performed by Huynh Quang Khanh, Nguyen Viet Dang Quang, and Nguyen Lam Vuong. The first draft of the manuscript was written by Huynh Quang Khanh and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
H.Q. Khanh, N.V.D. Quang, T.Q. Tien, and N.L. Vuong declare that they have no competing interests.
Ethical standards
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration. Informed consent was obtained from all patients for being included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khanh, H.Q., Quang, N.V.D., Tien, T.Q. et al. Long-term oncological outcome in thymic malignancies: videothoracoscopic versus open thymectomy. Eur Surg 51, 295–302 (2019). https://doi.org/10.1007/s10353-019-00610-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-019-00610-6