Abstract
The population decline of the European hare (Lepus europaeus) in Switzerland is generally attributed to low leveret survival. A significant intensification of agricultural practices led to a landscape transformation that reduced leveret survival by increasing negative factors such as predation pressure. Habitat improvement by means of wildflower strips has yielded some positive effects on European hare population trends, probably by improving food supply and providing year-round cover from predation. For this study, remote cameras were used to examine relationships between landscape and wildflower strip variables and the frequency of predator visits to wildflower strips as well as the probability of them visiting core areas of the strips. Of a total of 1586 visits of potential predators to wildflower strips, 91% were mammals and 9% were birds. Predators were more frequently observed at the edges of the wildflower strips than in their cores (72% of visits by mammalian predators and 76% by avian predators were at the edge). The results revealed that the frequency of observing predators was negatively correlated with adjacent crop height and the distance of the wildflower strip from settlements, roads and forests or hedgerows. The probability of a predator penetrating the core of the wildflower strip was negatively correlated with the vegetation cover, especially with the cover of wood, herbaceous plant species and teasel (Dipsacus fullonum). Appropriate management of wildflower strips by considering their spatial placement, creating low margin to surface area ratios and promoting heterogeneous wildflower structure can thus lead to reduced predator pressure on leverets as well as on ground-nesting birds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Agri-environment schemes (AES) have been established throughout Europe to halt and reverse biodiversity declines in farmland. The benefits of AES differ between different taxa (Kleijn et al. 2006). For the European hare (Lepus europaeus), AES indicate some positive effects on population trends, although the quantity and quality of habitat improvements are still not sufficient for an effective and sustainable enhancement (Tapper 2001; Zellweger-Fischer et al. 2011; Langhammer et al. 2017). Adult European hares select AES elements for feeding and resting (Petrovan et al. 2013; Schai-Braun et al. 2013), but it remains unclear to which extent these elements contribute to the fertility or survival of hares (see Discussion in Petrovan et al. 2013).
The decline of European hare populations is thought to be caused by the interaction of multiple factors like climate, habitat and predation, which influence resource availability, reproduction and survival (Hoffmann 2003; Smith et al. 2004; Olesen and Asferg 2006; Van Wieren et al. 2006). As a typical species of the open country, the hare is greatly affected by agricultural activities (Tapper and Yalden 2010). Therefore, significant land-use changes due to the intensification of agricultural practices, especially those involving increased mechanisation, habitat fragmentation and urbanisation, have led to a loss of habitat diversity and are thought to be the main cause of the decline (Smith et al. 2004, 2005; Pépin and Angibault 2007; Schai-Braun et al. 2015).
Important mortality factors for young hares or leverets include climatic influences, especially high precipitation and cold temperatures during the most vulnerable phase of reproduction in spring (Hoffmann 2003; Hackländer 2005; Tapper and Yalden 2010), and losses due to intensive farming practices (Voigt 2010). However, the primary cause of death for leverets seems to be predation, which may comprise more than 80% of the annual losses (Voigt 2010). The number of hares eaten by predators can easily exceed their breeding density and may amount to 100% of their annual production (Reynolds and Tapper 1995). Red foxes (Vulpes vulpes) are often considered to be the most important hare predators (Hoffmann 2003; Schmidt et al. 2004; Tapper and Yalden 2010) but almost all carnivorous species like badgers (Meles meles), wild boars (Sus scrofa) or various bird species prey upon leverets if they come across them by chance (Averianov et al. 2003).
The influence of predation on the decline of the European hare population has been the subject of controversy. Population densities of predators, particularly red foxes, are often found to be negatively correlated with hare densities, and the implementation of efficient predator control leads to an increase in hare numbers (Lindström et al. 1994; Hoffmann 2003; Schmidt et al. 2004; Panek et al. 2006; Maas 2010; Reynolds et al. 2010). But fox control is usually ineffective if executed on a local scale, due to compensatory immigration (Lieury et al. 2015), and many researchers conclude that, as the two species have coexisted for thousands of years, the influence of the predator on populations of its prey is mainly dependent on landscape structure (Schneider 2001; Olesen and Asferg 2006). Therefore, habitat improvements providing year-round cover for hares may reduce the probability of them being detected by predators, thus decreasing predation pressure (Vaughan et al. 2003; Panek 2009).
Habitat selection analyses have shown that the European hare prefers wildflower strips or agri-environment field margins and their immediate surroundings for both foraging and resting (Baumann 2003; Vaughan et al. 2003; Smith et al. 2004; Tillmann et al. 2007; Kinser et al. 2010; Petrovan et al. 2013, Wagner et al. 2014). Habitat improvement by means of creating wildflower strips has had some positive effects on European hare population trends, especially on arable land (Hutchings and Harris 1996; Holzgang et al. 2005; Perron 2013; Meichtry-Stier et al. 2014; Langhammer et al. 2017). Such heterogeneous habitat structures provide year-round cover, which protects against predation, adverse weather conditions and agricultural practices, and may enhance the survival probability of leverets (Hackländer et al. 2002a; Smith et al. 2004; Jennings et al. 2006), but studies on the survival of leverets in wildflower strips have not yet been published. At our study site in Switzerland, we regularly find leverets in wildflower strips, but we do not know whether female hares select these places to give birth (Weber 2017). Furthermore, wildflower strips offer food with a high fat content, which enhances milk quality and hence the growth rate of leverets, making them less prone to predation pressure (Hackländer et al. 2002b; Schai-Braun et al. 2015).
The risk to a leveret of being killed by predators is not only dependent on the density of predators and of cover, but it may also be influenced by whether predators decide to forage in the near surroundings of the leverets’ hiding place. Like the eggs and chicks of ground-nesting birds, including lapwings (Seymour et al. 2003), other waders (Meisner et al. 2014) and skylarks (Yanes and Suarez 1996), leverets may not be prime targets of foxes and other predators, but killed as by-catch whenever encountered by predators whose foraging behaviour is optimised for more abundant prey such as rodents.
Linear strips dominated by long-stemmed grasses, often with shrubs growing within them, are more likely to be visited by carnivores than the surrounding farmland (Šálek et al. 2009); their use of such corridors is not influenced by the amount of shrub cover. Carnivores may visit the strips because of the elevated abundance of potential prey in them and because they function as ‘movement corridors’ (Šálek et al. 2009). The fine-scale spatial preference of predators for edge structures increases the predation risk to leverets and to ground-nesting birds in such places. Wildflower strips are linear elements providing an attractive combination of cover and high densities of rodents (Briner et al. 2005) for carnivores, and they could therefore act as an ecological trap (Šálek et al. 2010; Červinka et al. 2011; Svobodová et al. 2011) for leverets and ground-nesting birds. A field experiment with lures has shown that carnivores concentrated their foraging activity at the edge of wildflower strips, while leverets in the interior were comparatively well protected (Fernex et al. 2011). This finding suggests that, in order to protect leverets (and ground-nesting birds), wildflower strips with a given surface area should preferably be wide and short, rather than narrow and long, to minimise the margin to surface area ratio.
The general goal of this study was to verify the results of Fernex et al. (2011) by using remote cameras to avoid the impact of lures, and, based on our findings, to identify the factors that may influence the frequency with which predators visit wildflower strips, and the probability of predators penetrating the strips. Specifically, we investigated whether there is any difference in the visit frequency between the wildflower strip edge or margin and the core area of the strip. We hypothesised (H1) that the wildflower strip edges are more attractive to visiting predators, and thus are more intensely foraged, than the core areas. Furthermore, we investigated whether any local landscape variables attract predators to a wildflower strip. We hypothesised (H2) that the frequency with which predators visit a wildflower strip is influenced by its distance to permanent landscape elements that are important for predators, such as forests, hedges, roads and settlements. Finally, we investigated whether any variables of wildflower strips or their immediate surroundings induce predators visiting the strip to penetrate into its interior or to remain along its edge. We hypothesised (H3) that the probability of mammalian predators foraging in the core area of a wildflower strip depends on the structure of the wildflower strip and the adjacent agricultural crops.
Materials and methods
Study area
The study was conducted in north-western Switzerland in the canton of Basel-Landschaft (518 km2, WGS84 47° 27′ N, 7° 45′ E). Elevation ranges from 250 to 1170 m above sea level; the yearly precipitation is approximately 900 mm and the average yearly temperature lies around 10 °C (MeteoSchweiz 2014). Plants in the study area are mostly dormant from October to late March/early April (MeteoSchweiz 2014). The region is comprised of a small-scale mosaic of forests, farmlands and settlements. The canton has a high population density (547 people/km2) leading to a high degree of landscape fragmentation (Statistisches Amt 2016). Domestic cat (Felis silvestris catus) density in Switzerland is high (0.4 cats/household; Statista 2016), and fragmented settlement boundaries are known to increase the influence of domestic cats in the surrounding agricultural landscape (Schneider 2001). Urban area covers 17%, forests 38% and agriculture 41% of the territory. Agricultural production is dominated by grassland (69%), followed by arable land (28%) with crop rotation; wildflower strips comprise only 0.2% of the cultivated area (Statistisches Amt 2016). Wildflower strips in the canton of Basel-Landschaft must be sown with a specified seed mixture comprising 35 indigenous plant species (arable weeds and species of ruderal sites and areas), having a minimum area of 20 ares (0.2 ha) and a minimum width of 9 m, and must be cultivated for at least six consecutive years in the same place (Kaufmann et al. 2015). The maintenance of the wildflower strips consists of selective, mechanical control of problem plants and, beginning with the second year, up to half of the strip may be mown between October and mid-March if problems arise (problematic plant species, overgrowing with grass). During the third and fourth winters, the mown strip area may be harrowed and milled (Kaufmann et al. 2015). At the beginning of 2016, farmers in the canton cultivated 118 wildflower strips. Of these, 64 were selected for this study.
Compared to other European countries, European hare population densities in Switzerland are low. Population estimates based on spotlight counts in the canton of Basel-Landschaft in selected reference areas range between 0.2 and 4.6 individuals/100 ha (Zellweger-Fischer 2015).
For this study, all mammalian and avian species that prey upon voles (Arvicolinae) and larger prey species were considered to be potential predators of leverets. Within the study area, this assumption applies to the following regularly occurring species (Fernex et al. 2011): mammalian predators: domestic dog (Canis lupus familiaris), domestic cat, wildcat (Felis silvestris silvestris), stone marten (Martes foina), pine marten (Martes martes), badger, European polecat (Mustela putorius), wild boar and red fox; avian predators: grey heron (Ardea cinerea), long-eared owl (Asio otus), Eurasian eagle owl (Bubo bubo), common buzzard (Buteo buteo), white stork (Ciconia ciconia), common raven (Corvus corax), carrion crow (Corvus corone), rook (Corvus frugilegus), common kestrel (Falco tinnunculus), black kite (Milvus migrans), red kite (Milvus milvus) and tawny owl (Strix aluco).
Data collection
At the beginning of 2016, 118 wildflower strips that could be used for the survey were identified. In the following month, the available number of wildflower strips was reduced by approximately 20 to 30 (due to new seeding after damage, loss of floristic diversity, dominance of weeds or neophytes or expiring contracts). Freshly seeded wildflower strips were not included in the survey. Wildflower strips were chosen systematically from the remaining dataset to enable the equal distribution of the variables area and age. We characterised wildflower strips by defining categories of area (≤ 0.25, 0.26–0.5, 0.51–0.75, > 0.75 ha) and of age (≤ 1, 2, 3, 4, 5, 6, > 6 years).
Between 6 February and 27 May 2016, a total of 64 wildflower strips were surveyed. Each wildflower strip was equipped with a pair of remote cameras of the same camera type, to keep constant within sites unknown biases, such as animal responses to camera traps (Meek et al. 2016). The positions of the cameras were chosen randomly by dividing the perimeter of the strip into 10-m segments, excluding sections along roads. The first remote camera was placed on the boundary line, between the wildflower strip and the adjacent agricultural field; the second was positioned facing parallel to the first one, at a distance of 6 m into the interior of the wildflower strip (this allowed us to avoid placing the interior camera too close to the opposite boundary line in narrow strips). The camera direction was always chosen to point away from the nearest road. The cameras were mounted on wooden poles 1.5 m tall. The top of each pole was equipped with four thin wooden sticks to make it impossible for birds of prey to perch on the poles and use them as lookout. Vegetation was cut back in an area of 1 m width and 4 m length in the direction of the camera’s field of view, to ensure an unobstructed trigger range and to enhance detection probability. The far end of this cut area was marked with two unobtrusive bamboo sticks. The camera lens was installed at 1 m above the ground and the mounting angle of the camera was 20°. Due to the equal installation height and mounting angle of the cameras and the reduced vegetation height within their trigger range, the detection areas were identical for all cameras and were restricted to a maximum distance of 4 m, which is the effective detection distance for the predators (Hofmeester et al. 2016). In total, 78 remote cameras of different types were used for this study, but not all were available for the entire study period. As far as camera type allowed, infrared (IR) flash was used (as it reduced the visibility of the cameras for animals and potential thieves). The camera types and numbers of each used were as follows: × 24 Cuddeback Capture Model 1125 colour flash, × 7 Cuddeback Ambush Model 1170 colour flash, × 21 Cuddeback C multiple flash IR (Cuddeback models: Green Bay, WI, USA), × 22 Reconyx HC600 Hyperfire IR, × 2 Reconyx XR6 Ultrafire IR and × 2 Reconyx RC60 Rapidfire IR (Reconyx inc., Holmen, WI, USA).
Cameras were set on photograph mode (single shot per trigger) and the recovery period was set to be as short as possible (30 s for Cuddeback Capture, 5 s for Cuddeback Ambush and fast-as-possible mode [0 s.] for Cuddeback C and all Reconyx models). After setting up the cameras and when each camera was checked, stations were approached and left by walking across the camera trigger range to check for proper operation. If one camera failed (due to damage or battery failure), images from the other camera in the same strip for the same time period were not analysed. The survey interval was one to 2 weeks. Several individual predators of the same species on the same picture were counted as one event. If an individual of the same species was photographed several times within a period of 30 min, this was counted as a single event unless several individuals could be distinguished (by special identification features, such as mange in foxes or distinctive coat patterns in domestic cats). Detections of domestic cats and wildcats were pooled.
The focus was on red foxes and badgers, to ensure that these naturally occurring predators were well represented in the data set. The aim was to survey as many wildflower strips as possible, given the available time and materials. A pair of cameras was relocated to a new wildflower strip once the required data were available (ten photos of red foxes or badgers or 20 photos of the two species together), or if it seemed unlikely that the site would generate the necessary data within a reasonable time-frame (after five consecutive weeks with no more than one photo of red foxes and/or badgers).
To characterise the wildflower strips, 22 landscape and wildflower strip variables were used. Permanent landscape variables such as distances to settlements, roads, forests and hedges and the length of boundary line between the road and the wildflower strip were measured in the GeoView BL (GIS-Fachstelle BL 2016). Wildflower strip data such as age, area and perimeter were provided by the Landwirtschaftliches Zentrum Ebenrain. The number of game tracks crossing the wildflower strip in sight of the interior camera and corridor availability (whether or not there was a gap between wildflower strip and adjacent crop) was determined on-site. The factors vegetation height, species assemblage, percent cover (wood, teasel [Dipsacus fullonum], grass, herbaceous plants) and the proportion of vegetation gaps were estimated or measured along a transect line of 10 m. The transect started 5 m behind the camera at the edge of the strip and led to the strip interior, on a line parallel to the line between the two cameras, so as not to trample the vegetation layer in the detection area. To characterise small mammal abundance, the number of small mammal burrows within 4 m2 was counted in front of the cameras. The transect line described above was extended 10 m into the adjacent field to measure crop height and to determine the type of crop (see Appendix, Tables 5 and 6 for a detailed description of all variables collected for the study).
Most of the survey took place during the winter, when plants were dormant (MeteoSchweiz 2014), so cover estimates of plants were mainly based on plant remains from the previous year (dry stems and leaves). Some of these were abundant in most wildflower strips, as reflected in the two variables for teasel and herbaceous plants. All plant species that are referred to as herbaceous plants are listed in Appendix, Table 7 .
Data analyses
Each predator photo was combined with the associated landscape and wildflower strip variables (Appendix, Table 5). Statistical analyses were performed in R (version 3.3.1) (R Core Team 2016) using the lme4 package (Bates et al. 2015). Variables were tested for correlation and only unconfounded variables were used (Spearman’s correlation coefficient, r < 0.7) to select an independent set of predictors. The width was highly correlated with area (ρ = 0.83, p < 0.0001). As the width of each strip was calculated from its area and perimeter (Appendix, Tables 5 and 6), the area was a more straightforward variable and was thus used for further analysis.
To test if strip edges were more attractive to visiting predators than core areas (H1), we used a two-tailed exact binomial test (null hypothesis: a 50:50% expected distribution of edge and interior photos). The test was performed for potential predators and also for adult hares.
We applied generalised linear mixed-effects models (GLMM) to test if the predator visit frequency in a wildflower strip was influenced by its distance to landscape elements (H2) and if the probability of mammalian predators foraging in the core area of a wildflower strip depended on the structure of the wildflower strip and adjacent agricultural crops (H3). The GLMM to test H2 was performed using a Poisson distribution and the dependent variable visit frequency (number of predators per wildflower strip within 30 days), with the associated landscape variables (Appendix, Table 5) as independent variables. The GLMM to test H3 was conducted with a binomial distribution and the dependent variable predator detection position (observed at the edge or in the interior of the wildflower strip), with the associated wildflower strip variables (Appendix, Table 5) as independent variables.
To improve GLMM performance, all numerical independent variables were converted with a z-transformation (standard score). Camera type and wildflower strip identification number were added to the models as random effects. All fixed effects were tested for a linear relationship, apart from the variable age of the wildflower strip, which was additionally tested for a non-linear relationship. We generated sets of models with combinations (subsets) of fixed effect terms in the global model, using the dredge function from the MuMin package (Barton 2017). We used Akaike’s information criterion adjusted for small sample size (AICc) to rank models (Crawley 2012). We considered all models with a difference in ∆AICc ≤ 2 units from the highest-ranking model to have strong support and computed model-averaged parameter estimates of all these models. Additionally, we calculated cumulative AICc weights (0 ≤ ∑ ω i ≤ 1), to evaluate the relative variable importance (Arnold 2010).
To look at spatial patterns in our data sets, we applied geostatistics on the models to test for spatial autocorrelation of the residuals. For this purpose, we used the variogram function from the gstat package (Korner-Nievergelt et al. 2015). All calculated variograms showed no clear spatial patterns, indicating, that the variation in our predator detections has no spatial component.
Results
The mean length of the survey period per wildflower strip was 50.4 days (95% CI = 5.3). In total, the study consisted of 6454 camera station nights. Overall, a total of 1586 potential predators of leverets, with an average visit frequency of 15.3 (95% CI = ± 4.2) predators per wildflower strip per month (30 days), were detected (cat [Felis silvestris ssp.]: n = 696, red fox: n = 373, badger: n = 225, domestic dog: n = 80, marten [Martes sp.]: n = 58, wild boar: n = 11, corvids [Corvus sp.]: n = 112, grey heron: n = 23, common buzzard: n = 8). Ninety-one percent of the recorded predators were mammals; the most frequent predators visiting wildflower strips were cats (44%), followed by red foxes (24%) and badgers (14%). These three species made up 82% of all detections. The total number of adult European hares recorded was 166.
Visit frequency in the core and at the edge of strips
Of the 1443 photos of mammalian predators, 28% were taken in the cores of the wildflower strips, as opposed to 72% at their edges (test against 50:50% expected distribution, p < 0.001; Fig. 1). All mammal species were observed more frequently at the edge of the wildflower strips than in their interiors (red fox 64%, p = 0.002, n = 373; badger 77%, p < 0.001, n = 225; cat 73%, p < 0.001, n = 696; Fig. 1). The same was true for avian predator species (76%, p < 0.001, n = 143; Fig. 1). The probability of detecting adult European hares did not differ between the interior and the edge of the wildflower strips (50%, p = 1, n = 166; Fig. 1).
Relative frequency of visitors per unit area at the wildflower strip (WFS) edge and 95% confidence intervals. WFS visit frequency by mammalian predators (red foxes, badgers, cats, domestic dogs, martens, wild boar), avian predators (grey heron, common buzzard, corvids) and separately red fox, badger, cat and European hare
Visit frequency and landscape variables
Mammalian predators
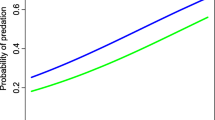
According to the model-averaged parameters of all models ∆AIC ≤ 2, the number of mammalian predators observed per time interval was negatively correlated with increasing distance to human settlements (∑ ω i = 0.61), forests/hedgerows (∑ ω i = 1.00) and the availability of an interspace between road and WFS (∑ ω i = 1.00; Table 1, Fig. 2a–c). The variables area, perimeter, boundary line, type of crop, crop height and distance to forests or hedgerows, when analysed separately, provided only little information on mammalian predators’ visit frequency at the wildflower strips.
Number of mammalian predators (MPs, per 30 days) visiting the wildflower strip (WFS), in relation to the landscape variables distance to settlement (a), distance to forest edges or hedgerows (b, trend line and 80% prediction intervals) and road interspace availability (c, boxplot with median, lower and upper quartile; lowest and highest value and outliers)
Avian predators
According to the model-averaged parameters of all models ∆AIC ≤ 2, the number of avian predators observed per time interval was negatively correlated with increasing crop height (∑ ω i = 1.00). Visit frequency was lower if the adjacent type of crop (∑ ω i = 1.00) was cereal and higher if the adjacent crop was ploughed field, vegetable or rapeseed (others; Table 2). The variables area, perimeter and distance to settlement, road interspace, forest, hedgerow and forest/hedgerow, as well as the length of the boundary line and grassland, provided only little information on avian predators’ visit frequency at the wildflower strips.
Penetration probability and wildflower strip variables
Mammalian predators
According to the model-averaged parameters of all models ∆AIC ≤ 2, the probability of mammalian predators penetrating into wildflower strip cores rather than remaining along the edges was positively correlated with increasing age of the strips (∑ ω i = 1.00; Table 3, Fig. 3a) and the number of game tracks (∑ ω i = 1.00) and negatively correlated with increasing area and cover of wood, teasel, herbaceous plants and grass (∑ ω i = 1.00; Table 3, Fig. 3b–c). The probability of mammalian predators visiting the wildflower strip interior was the highest when the adjacent crop was grassland (Table 3). The variables plant height, vegetation gaps, low vegetation, small mammal burrows, corridor availability, crop height and the crop types cereal, rapeseed, vegetable and ploughed field provided only little information on the probability of mammalian predators penetrating the wildflower strip core area rather than remaining along its edge.
Avian predators
According to the model-averaged parameters of all models ∆AIC ≤ 2, the probability of avian predators being photographed within the wildflower strip cores as opposed to along the edges was positively correlated with increasing cover of grass (∑ ω i = 0.57) and corridor availability (∑ ω i = 0.53) and negatively correlated with increasing cover of herbaceous plants (∑ ω i = 0.88; Table 4). The variables area, plant height, low vegetation, vegetation gaps, small mammal burrows, type of crop, crop height, number of game tracks and cover of wood and teasel provided only little information on the probability of avian predators entering the wildflower strip core area rather than being observed along its edge.
Discussion
Wildflower strips as a safe refuge for leverets
Most wildflower strips in the study area were regularly visited by various predator species. The most frequent predator visitors were cats, red foxes and badgers, which together comprised 82% of all visits.
Various studies suggest that narrow, linear strips of unmown vegetation with or without shrubs are more likely to be foraged by predators than the surrounding agricultural environment (Šálek et al. 2009, 2010; Červinka et al. 2011). A crucial factor for corridor use is the availability of the principal prey (Červinka et al. 2013). Wildflower strips are known to be a high-quality habitat for small mammals and to sustain higher population densities than agricultural crops (Briner et al. 2005; Aschwanden et al. 2007). Even if hares, which are present at low densities, are not the target of the predators, they might incidentally be found by predators searching for more abundant prey such as rodents. To sum up, wildflower strips are attractive places for potential predators of leverets, and predators may threaten leverets hiding within the wildflower strips.
On the other hand, several researchers found positive effects of wildflower strips on European hare population trends (Hutchings and Harris 1996; Holzgang et al. 2005; Perron 2013; Meichtry-Stier et al. 2014; Langhammer et al. 2017). Population decline is mainly attributed to low leveret survival (Voigt 2010; Weber 2010), and leverets in wildflower strips are probably relatively safe from agricultural machines. Our study shows that the risk of a leveret being approached by a predator in a wildflower strip depends on the spatial placement of the wildflower strip within the landscape mosaic, on its perimeter-area ratio and on its plant structure. In other words, wildflower strips can be more or less dangerous for leverets and the degree of risk is predictable. This leads us to practical recommendations for leveret-friendly (predator-unfriendly) wildflower strips, based on the assumption that leverets survive better where potential predators are less likely to be present.
Locations of wildflower strips to benefit leveret survival
An important factor influencing predator activity was the distance to hedgerows or forests: mammalian predators’ visits were lower when the distance between these features and the wildflower strip was greater. Badgers and red foxes are known to show a preference for forests and habitats that offer the shelter they need for their breeding setts and dens (Keuling et al. 2011). In an agricultural region such as the study area, forests and hedgerows are most likely to fulfil their requirements. Therefore, setts and dens are often located in these tree-sheltered habitats and form the core area of these animals’ home ranges; the highest level of predator activity is in their immediate vicinity. Forest-farmland edge habitats are also highly preferred by mammalian predators for foraging, so the probability of occurrence of these predators decreases considerably with increasing distance from forest edges (Červinka et al. 2011; Šálek et al. 2014).
Settlements and their surroundings offer abundant anthropogenic food sources that make them attractive to many predator species, especially red foxes (Contesse et al. 2004). Another important predator species is the domestic cat, which is kept and fed by humans and disperses from settlements to adjacent agricultural areas for hunting. Especially in suburban landscapes, where human settlements cover large portions of such fragmented landscapes, cats can have a pronounced detrimental effect on prey populations (Schneider 2001; Spotte 2014). Not surprisingly, the further away from settlements a wildflower strip was located, the lower the mammalian predator activity was. Roads, also anthropogenic habitat features, influenced mammalian predator activity at wildflower strips accordingly. Predator numbers were lower in wildflower strips not placed adjacent to roads. Roads can facilitate territorial patrolling and serve to increase travel distance and thus increase the hunting ground covered by a predator during night (Frey and Conover 2006). Especially if wildflower strips consist of linear narrow habitat strips along roads, there is an increased hazard of predators locating leverets or ground-nesting birds while foraging. Moreover, in wildflower strips completely surrounded by crops, hares are somewhat protected from the constant disturbance caused by vehicles, walkers and their dogs (Roedenbeck and Voser 2008).
The height of the crop adjacent to the wildflower strip influenced the frequency of visits by avian predators. Lower crop height may ease locomotion and increase travel speed, making such areas attractive to walk across and also improving the visibility, thus facilitating searching for prey. Additionally, birds may be attracted to harvested fields and vegetable cultivation areas because the open ground allows for efficient movement and detection of abundant food sources such as insects, worms and voles, whereas the dense seed row spacing in cereal crops prevents free locomotion and restricts visibility. But the type and height of crop adjacent to wildflower strips cannot be influenced just to fulfil European hare conservation needs. Crop rotation practices lead to constant changes in the adjacent fields during the contractual period of a wildflower strip and crops itself are subject to strong changes during the long hare breeding season (for example from tall cereals during summer to short stubble fields after harvest in September).
Wildflower strip variables beneficial for leveret survival
The most important practical finding of our study is that leverets appear to be relatively well protected from predation by both birds and mammals in the core areas of wildflower strips. Our data show that predators concentrate their activity at a fine scale at the very edge of the wildflower strips; only 6 m inside, we found reduced activity. This finding is in agreement with the results of Fernex et al. (2011). If the width of a wildflower strip is increased, the relationship between edge and core area of the wildflower strip is improved. Thus, leverets that hide within the wildflower strip’s core likely have a higher chance of escaping predator detection, because, in broader and larger wildflower strips, the increased edge-to-edge distance is expected to decrease foraging intensity of predators. There is no minimum width of wildflower strips in Switzerland, and some are only 3 m wide. Farmers should be encouraged to cultivate wider wildflower strips.
We do not know why predators prefer to remain along the edges of wildflower strips rather than to penetrate their cores, which would provide rodents and cover. Herbaceous plants with rigid stems, dense foliage and thorny leaves and grass growing in clumps may simply hinder predators from walking and foraging in the strips, as suggested by the reduced mammalian and avian predator activity in strips with a higher cover of wood, teasel, herbaceous plant species and grasses. It is also known that enhanced vegetation heterogeneity and complexity along edge structures can reduce the search efficiency of predators (Červinka et al. 2011; Gorini et al. 2012), but wildflower strips often lose their heterogeneous wildflower structure with increasing age if they are not maintained regularly.
Implications and conclusions
Wildflower strips are a precious instrument in agri-environmental schemes, beneficial for a variety of species dwelling in open landscapes, including insects, ground-nesting birds and small mammals (Aschwanden et al. 2007; Haaland et al. 2011; Meichtry-Stier et al. 2014). For the European hare, wildflower strips provide not only shelter from farm machinery, but also more herbs and grasses with a higher fat content than the surrounding intensive agricultural area (Reichlin et al. 2006; Schai-Braun et al. 2015). Plants with a high fat content are important in the European hare’s diet, especially for lactating females, as a high-quality diet for the mother enhances leveret survival (Hackländer et al. 2002a, b). Wildflower strips might also be attractive for predators as parts of a network of moving corridors (Šálek et al. 2009, 2010; Červinka et al. 2011) and as foraging areas with high densities of rodents (Briner et al. 2005; Aschwanden et al. 2007).
Optimising wildflower strips as instruments to favour the survival of leverets and ground-nesting birds means making them unattractive for predators. According to our findings, both foraging and visit frequency of predators can be managed by correct spatial placement of wildflower strips, by low perimeter-area ratio in strips and by promoting a heterogeneous wildflower structure. We recommend the following, for leveret-friendly (predator-unfriendly) wildflower strips:
-
1.
Place wildflower strips at least 150 m away from forests, hedgerows and settlements.
-
2.
Do not place wildflower strips alongside roads.
-
3.
Forget the notion of a ‘strip’: wildflower areas should be wide (> 10 m) and short, rather than long and narrow (< 10 m).
-
4.
Favour strips with a high vegetative cover and heterogeneous wildflower structure.
References
Arnold TW (2010) Uninformative Parameters and Model Selection Using Akaike’s Information Criterion. J Wildl Manag 74:1175–1178. https://doi.org/10.2193/2009-367
Aschwanden J, Holzgang O, Jenni L (2007) Importance of ecological compensation areas for small mammals in intensively farmed areas. Wildlife Biol 13(2):150–158. https://doi.org/10.2981/0909-6396(2007)13[150:IOECAF]2.0.CO;2
Averianov A, Niethammer J, Pegel JM (2003) Lepus europaeus Pallas, 1778—Feldhase. In: Krapp F (ed) Handbuch der Säugetiere Europas. Aula-Verlag, Wiebelsheim, pp 35–104
Barton K (2017) Model selection and model averaging based on information criteria (AICc and alike). https://cran.r-project.org/web/packages/MuMIn/MuMIn.pdf Accessed 7 Nov 2017
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. 10.18637/jss.v067.i01
Baumann M (2003) “Pflugfurchenprojekt” zur Förderung des Feldhasen in der Solothurner Witi. Bericht 2001–2003. Jagd & Fischerei Kanton Solothurn
Briner T, Nentwig W, Airoldi J-P (2005) Habitat quality of wildflower strips for common voles (Microtus arvalis) and its relevance for agriculture. Agric Ecosyst Environ 105(1-2):173–179. https://doi.org/10.1016/j.agee.2004.04.007
Červinka J, Šálek M, Pavluvčík P, Kreisinger J (2011) The fine-scale utilization of forest edges by mammalian mesopredators related to patch size and conservation issues in Central European farmland. Biodivers Conserv 20(14):3459–3475. https://doi.org/10.1007/s10531-011-0135-8
Červinka J, Šálek M, Padyšáková E, Šmilauer P (2013) The effects of local and landscape-scale habitat characteristics and prey availability on corridor use by carnivores: a comparison of two contrasting farmlands. J Nat Conserv 21(2):105–113. https://doi.org/10.1016/j.jnc.2012.11.004
Contesse P, Hegglin D, Gloor S, Bontadina F, Deplazes P (2004) The diet of urban foxes (Vulpes vulpes) and the availability of anthropogenic food in the city of Zurich, Switzerland. Mamm Biol 69(2):81–95. https://doi.org/10.1078/1616-5047-00123
Crawley MJ (2012) The R book, 2nd edn. John Wiley & Sons, Ltd, Chichester, UK. https://doi.org/10.1002/9781118448908.ch3
Fernex A, Nagel P, Weber D (2011) Sites with reduced predation risk to young hares within an agricultural landscape. Mammalia 75(4):395–397. https://doi.org/10.1515/MAMM.2011.053
Frey SN, Conover MR (2006) Habitat use by meso-predators in a corridor environment. J Wildl Manag 70(4):1111–1118. https://doi.org/10.2193/0022-541X(2006)70[1111:HUBMIA]2.0.CO;2
GIS-Fachstelle BL (2016) GeoView BL. http://geoview.bl.ch/. Accessed 11 Oct 2016
Gorini L, Linnell JDC, May R, Panzacchi M, Boitani L, Odden M, Nilsen EB (2012) Habitat heterogeneity and mammalian predator–prey interactions. Mamm Rev 42(1):55–77. https://doi.org/10.1111/j.1365-2907.2011.00189.x
Haaland C, Naisbit RE, Bersier L-F (2011) Sown wildflower strips for insect conservation: a review. Insect Conserv Divers 4(1):60–80. https://doi.org/10.1111/j.1752-4598.2010.00098.x
Hackländer K (2005) Was ist mit dem Feldhasen los? Vom Fruchtbarkeitssymbol zur “Rote-Liste-Art”. Wild 4:1–8
Hackländer K, Arnold W, Ruf T (2002a) Postnatal development and thermoregulation in the precocial European hare (Lepus europaeus). J Comp Physiol B 172(2):183–190. https://doi.org/10.1007/s00360-001-0243-y
Hackländer K, Tataruch F, Ruf T (2002b) The effect of dietary fat content on lactation energetics in the European hare (Lepus europaeus). Physiol Biochem Zool 75(1):19–28. https://doi.org/10.1086/324770
Hoffmann D (2003) Populationsdynamik und–entwicklung des Feldhasen in Schleswig-Holstein im Beziehungsgefüge von Klima, Prädation und Lebensraum. Dissertation. Universität Trier
Hofmeester TR, Rowcliffe M, Jansen PA (2016) A simple method for estimating the effective detection distance of camera traps. Remote Sens Ecol Conserv 1–9. doi: https://doi.org/10.1002/rse2.25
Holzgang O, Heynen D, Kéry M (2005) Rückkehr des Feldhasen dank ökologischem Ausgleich? Schriftenr der FAL 56:150–160
Hutchings MR, Harris S (1996) The current status of the brown hare (Lepus europaeus) in Britain. Joint Nature Conservation Commitee, Peterborough
Jennings N, Smith RK, Hackländer K, Harris S, White PCL (2006) Variation in demography, condition and dietary quality of hares (Lepus europaeus) from high-density and low-density populations. Wildlife Biol 12(2):179–189. https://doi.org/10.2981/0909-6396(2006)12[179:VIDCAD]2.0.CO;2
Kaufmann S, Sattler M, Wenger S (2015) Weisungen. Kantonale Biodiversitätsbeiträge 2015. Landwirtschaftliches Zentrum Ebenrain, Sissach
Keuling O, Greiser G, Grauer A, Strauß E, Bartel-Steinbach M, Klein R, Wenzelides L, Winter A (2011) The German wildlife information system (WILD): population densities and den use of red foxes (Vulpes vulpes) and badgers (Meles meles) during 2003–2007 in Germany. Eur J Wildl Res 57(1):95–105. https://doi.org/10.1007/s10344-010-0403-z
Kinser A, Strauss E, Herzog S (2010) Lebensraumnutzung von Feldhasen während der Nacht—Ergebnisse aus dem Projekt “Lebensraum Brache”. In: Lang J, Godt J, Rosenthal G (eds) Fachtagung Feldhase. Ergebnisse der “Fachtagung Feldhase—Der aktuelle Stand der Hasenforschung” 19–20. März in Kassel. lutra Verlags- und Vertriebsgesellschaft, Tauer, pp 73–82
Kleijn D, Baquero RA, Clough Y, Díaz M, De Esteban J, Fernández F, Gabriel D, Herzog F, Holzschuh A, Jöhl R, Knop E, Kruess A, Marshall EJP, Steffan-Dewenter I, Tscharntke T, Verhulst J, West TM, Yela JL (2006) Mixed biodiversity benefits of agri-environment schemes in five European countries. Ecol Lett 9(3):243–254. https://doi.org/10.1111/j.1461-0248.2005.00869.x
Korner-Nievergelt F, Roth T, Von Felten S, Guélat J, Almasi B, Korner Nievergelt P (2015) Bayesian data analysis in ecology using linear models with R, BUGS, and Stan. Elsevier Ltd, Oxford
Langhammer M, Grimm V, Pütz S, Topping CJ (2017) A modelling approach to evaluating the effectiveness of ecological focus areas: the case of the European brown hare. Land Use Policy 61:63–79. https://doi.org/10.1016/j.landusepol.2016.11.004
Lieury N, Ruette S, Devillard S, Albaret M, Drouyer F, Baudoux B, Millon A (2015) Compensatory immigration challenges predator control: an experimental evidence-based approach improves management. J Wildl Manag 79(3):425–434. https://doi.org/10.1002/jwmg.850
Lindström ER, Andrén H, Angelstam P, Cederlund G, Hörnfeldt B, Jäderberg L, Lemnell P-A, Martinsson B, Sköld K, Swenson JE (1994) Disease reveals the predator: sarcoptic mange, red fox predation, and prey populations. Ecology 75(4):1042–1049. https://doi.org/10.2307/1939428
Maas A (2010) Entwicklung der Hasenpopulation und-strecke im Jagdrevier Störmede-Ost von 1973 bis 2009—Erfahrungen eines Praktikers. In: Lang J, Godt J, Rosenthal G (eds) Fachtagung Feldhase. Ergebnisse der “Fachtagung Feldhase-Der aktuelle Stand der Hasenforschung” 19–20. März in Kassel. lutra Verlags- und Vertriebsgesellschaft, Tauer, pp 47–56
Meek P, Ballard G, Fleming P, Falzon G (2016) Are we getting the full picture? Animal responses to camera traps and implications for predator studies. Ecol Evol 6(10):3216–3225. https://doi.org/10.1002/ece3.2111
Meichtry-Stier KS, Jenny M, Zellweger-Fischer J, Birrer S (2014) Impact of landscape improvement by agri-environment scheme options on densities of characteristic farmland bird species and brown hare (Lepus europaeus). Agric Ecosyst Environ 189:101–109. https://doi.org/10.1016/j.agee.2014.02.038
Meisner K, Sunde P, Kuhlmann Clausen K, Clausen P, Fælled CC, Hoelgaard M (2014) Foraging ecology and spatial behaviour of the red fox (Vulpes vulpes) in a wet grassland ecosystem. Acta Theriol 59(3):377–389. https://doi.org/10.1007/s13364-014-0178-9
MeteoSchweiz (2014) Klimadiagramme und Normwerte pro Station. Normperiode 1981–2010. http://www.meteoschweiz.admin.ch/home/klima/vergangenheit/klima-normwerte/klimadia gramme-und-normwerte-pro-station.html. Accessed 9 Oct 2016
Olesen CR, Asferg T (2006) Assessing potential causes for the population decline of European brown hare in the agricultural landscape of Europe - a review of the current knowledge. Natl Environ Res Institute, Denmark, 32 p. NERI Technical report No. 600. http://www.dmu.dk/Pub/FR600.pdf. Accessed 30 Nov 2017
Panek M (2009) Factors affecting predation of red foxes (Vulpes vulpes) on brown hares (Lepus europaeus) during the breeding season in Poland. Wildlife Biol 15(3):345–349. https://doi.org/10.2981/07-042
Panek M, Kamieniarz R, Bresiński W (2006) The effect of experimental removal of red foxes Vulpes vulpes on spring density of brown hares Lepus europaeus in western Poland. Acta Theriol (Warsz) 51(2):187–193. https://doi.org/10.1007/BF03192670
Pépin D, Angibault JM (2007) Selection of resting sites by the European hare as related to habitat characteristics during agricultural changes. Eur J Wildl Res 53(3):183–189. https://doi.org/10.1007/s10344-007-0087-1
Perron M (2013) Der Feldhase benötigt geeignete Brachen zum Überleben. Umwelt Aargau 59:43–46
Petrovan SO, Ward AI, Wheeler PM (2013) Habitat selection guiding agri-environment schemes for a farmland specialist, the brown hare. Anim Conserv 16(3):344–352. https://doi.org/10.1111/acv.12002
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.r-project.org/. Accessed 16 Oct 2016
Reichlin T, Klansek E, Hackländer K (2006) Diet selection by hares (Lepus europaeus) in arable land and its implications for habitat management. Eur J Wildl Res 52(2):109–118. https://doi.org/10.1007/s10344-005-0013-3
Reynolds JC, Tapper SC (1995) Predation by foxes (Vulpes vulpes) on brown hares (Lepus europaeus) in central southern England, and its potential impact on annual population growth. Wildlife Biol 1:145–158
Reynolds JC, Stoate C, Brockless MH, Aebischer NJ, Tapper SC (2010) The consequences of predator control for brown hares (Lepus europaeus) on UK farmland. Eur J Wildl Res 56(4):541–549. https://doi.org/10.1007/s10344-009-0355-3
Roedenbeck IA, Voser P (2008) Effects of roads on spatial distribution, abundance and mortality of brown hare (Lepus europaeus) in Switzerland. Eur J Wildl Res 54(3):425–437. https://doi.org/10.1007/s10344-007-0166-3
Šálek M, Kreisinger J, Sedláček F, Albrecht T (2009) Corridor vs. hayfield matrix use by mammalian predators in an agricultural landscape. Agric Ecosyst Environ 134(1-2):8–13. https://doi.org/10.1016/j.agee.2009.06.018
Šálek M, Kreisinger J, Sedláček F, Albrecht T (2010) Do prey densities determine preferences of mammalian predators for habitat edges in an agricultural landscape? Landsc Urban Plan 98(2):86–91. https://doi.org/10.1016/j.landurbplan.2010.07.013
Šálek M, Červinka J, Pavluvčík P, Poláková S, Tkadlec E (2014) Forest-edge utilization by carnivores in relation to local and landscape habitat characteristics in central European farmland. Mamm Biol 79(3):176–182. https://doi.org/10.1016/j.mambio.2013.08.009
Schai-Braun SC, Weber D, Hackländer K (2013) Spring and autumn habitat preferences of active European hares (Lepus europaeus) in an agricultural area with low hare density. Eur J of Wildl Res 59(3):387–397. https://doi.org/10.1007/s10344-012-0684-5
Schai-Braun SC, Reichlin TS, Ruf T, Klansek E, Tataruch F, Arnold W, Hackländer K (2015) The European hare (Lepus europaeus): a picky herbivore searching for plant parts rich in fat. PLoS One 10(7):e0134278. https://doi.org/10.1371/journal.pone.0134278
Schmidt NM, Asferg T, Forchhammer MC (2004) Long-term patterns in European brown hare population dynamics in Denmark: effects of agriculture, predation and climate. BMC Ecol 4(1):15. https://doi.org/10.1186/1472-6785-4-15
Schneider MF (2001) Habitat loss, fragmentation and predator impact: spatial implications for prey conservation. J Appl Ecol 38(4):720–735. https://doi.org/10.1046/j.1365-2664.2001.00642.x
Seymour AS, Harris S, Ralston C, White PCL (2003) Factors influencing the nesting success of lapwings (Vanellus vanellus) and behaviour of red fox (Vulpes vulpes) in lapwing nesting sites. Bird Stud 50(1):39–46. https://doi.org/10.1080/00063650309461288
Smith RK, Jennings NV, Robinson A, Harris S (2004) Conservation of European hares Lepus europaeus in Britain: is increasing habitat heterogeneity the answer? J Appl Ecol 41(6):1092–1102. https://doi.org/10.1111/j.0021-8901.2004.00976.x
Smith RK, Harris S, Smith RK, Jennings NV (2005) A quantitative analysis of the abundance and demography of European hares Lepus europaeus in relation to habitat type, intensity of agriculture and climate. Mamm Rev 35(1):1–24. https://doi.org/10.1111/j.1365-2907.2005.00057.x
Spotte S (2014) Free-ranging cats: behavior, ecology, management. John Wiley & Sons Ltd, Chichester. https://doi.org/10.1002/9781118884058
Statista (2016) Das Statistik-Portal. www.de.statista.com. Accessed 3 Dec 2016
Statistisches Amt (2016) Statistik Basel-Landschaft. https://www.baselland.ch/politik-und-behorden/direktionen/finanz-und-kirchendirektion/statistisches-amt//. Accessed 9 Oct 2016
Svobodová J, Kreisinger J, Šálek M, Koubová M, Albrecht T (2011) Testing mechanistic explanations for mammalian predator responses to habitat edges. Eur J Wildl Res 57(3):467–474. https://doi.org/10.1007/s10344-010-0455-0
Tapper SC (2001) Technical annex VII brown hare. Ecological evaluation of the arable stewardship pilot scheme, 1998–2000, 1–16. Game Conservancy Trust, Fordingbridge
Tapper S, Yalden D (2010) The brown hare. The Mammal Society, Southampton
Tillmann JE, Kinser A, Strauss E (2007) Baustein “Wissenschaft & Monitoring”-Wildtierökologische Untersuchungen. In: Börner M (ed) Projekt: “Lebensraum Brache”-Wildtierfreundliche Massnahmen im Agrarbereich-AZ20271. Deutsche Wildtierstiftung, Hamburg, pp 20–30
Van Wieren SE, Wiersma M, Prins HHT (2006) Climatic factors affecting a brown hare (Lepus europaeus) population. Lutra 49:103–110
Vaughan N, Lucas EA, Harris S, White PCL (2003) Habitat associations of European hares (Lepus europaeus) in England and Wales: implications for farmland management. J Appl Ecol 40(1):163–175. https://doi.org/10.1046/j.1365-2664.2003.00784.x
Voigt U (2010) Zur Raumnutzung und Mortalitätsursachen bei Junghasen (Lepus europaeus). In: Lang J, Godt J, Rosenthal G (eds) Fachtagung Feldhase. Ergebnisse der “Fachtagung Feldhase—Der aktuelle Stand der Hasenforschung” 19.–20. März in Kassel. lutra Verlags- und Vertriebsgesellschaft, Tauer, pp 83–92
Wagner C, Karl N, Schönfeld F (2014) Blühflächen als Habitat für Niederwild. In: Wagner C, Bachl-Staudinger M, Baumholzer S, Burmeister J, Fischer C, Karl N, Köppl A, Volz H, Walter R, Wieland P (eds) Faunistische Evaluierung von Blühflächen, Schriftenreihe der Bayerischen Landesanstalt für Landwirtschaft (LfL) 1/2014. pp 117–126.https://www.lfl.bayern.de/mam/cms07/publikationen/daten/schriftenreihe/faunistischeevaluierung_bluehflaechen_lfl-schriftenreihe-1-2014.pdf. Accessed 30 Nov 2017
Weber D (2010) Adaptives Feldhasenmanagement-Das Projekt “HOPP HASE” im Kanton Baselland. In: Lang J, Godt J, Rosenthal G (eds) Fachtagung Feldhase. Ergebnisse der “Fachtagung Feldhase—Der aktuelle Stand der Hasenforschung” 19–20. März in Kassel. lutra Verlags- und Vertriebsgesellschaft, Tauer, pp 113–120
Weber D (2017) Feldhasen fördern funktioniert! Bristol-Stiftung, Zürich and Haupt, Bern
Yanes M, Suarez F (1996) Incidental nest predation and lark conservation in an Iberian semiarid shrubsteppe. Conserv Biol 10(3):881–887. https://doi.org/10.1046/j.1523-1739.1996.10030881.x
Zellweger-Fischer J (2015) Schweizer Feldhasenmonitoring 2015. Schweizerische Vogelwarte, Sempach
Zellweger-Fischer J, Kéry M, Pasinelli G (2011) Population trends of brown hares in Switzerland: the role of land-use and ecological compensation areas. Biol Conserv 144(5):1364–1373. https://doi.org/10.1016/j.biocon.2010.11.021
Acknowledgments
We would like to thank Philipp Franke for his support and for providing data on the wildflower strips of the canton of Basel-Landschaft and Tobias Roth for his help with the statistical analysis. We are grateful to OEKO-LOG, the HOPP HASE association and the hunting administration of the canton of Aargau for providing the remote cameras. We thank all landowners, farmers and hunters who gave us access to their property. The study was funded by the HOPP HASE association.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Hummel, S., Meyer, L., Hackländer, K. et al. Activity of potential predators of European hare (Lepus europaeus) leverets and ground-nesting birds in wildflower strips. Eur J Wildl Res 63, 102 (2017). https://doi.org/10.1007/s10344-017-1158-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-017-1158-6