Abstract
Whole-tree harvesting (WTH), where logging residues are removed in addition to stems, is widely practised in Fennoscandian boreal forests. WTH increases the export of nutrients from forest ecosystems. The extent of nutrient removals may depend on tree species, harvesting method, and the intensity of harvesting. We developed generalized nutrient equations for Scots pine (Pinus sylvestris L.), Norway spruce (Picea abies Karsten), and birch (Betula pendula Roth and Betula pubescens Ehrh.) stands to be able to calculate the amounts of nitrogen, phosphorus, potassium, and calcium in stems and above-ground biomass (stem and crown) as a function of stand volume. The equations were based on Fennoscandian literature data from 34 pine, 26 spruce, and 5 birch stands, and they explained, depending on the tree species and nutrient, 61–99% and 56–87% of the variation in the nutrient amounts of stems and above-ground biomass, respectively. The calculations based on the equations showed that nutrient removals caused by stem-only harvesting (SOH) and WTH per harvested stem m3 were smaller in pine than in spruce and birch stands. If the same volume of stem is harvested, nutrient removals are, in general, nearly equal at thinnings and final cuttings in SOH, but larger in thinnings than final cuttings in WTH. If the principal aim is to minimize the nutrient removals per harvested stem m3, the harvesting should be done at mature pine stands. The effect of biomass removal on overall site nutrient status depends on site-specific factors such as atmospheric deposition, weathering of minerals, and the size of the nutrient pools in the soil.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The substitution of fossil fuels with renewable energy sources increases the use of forest biomass for energy production (European Commission 1997). Finland and Sweden have become leading countries in Europe regarding the utilization of wood-based energy in 2000s (Asikainen et al. 2008; Röser et al. 2008), and the use of forest energy is expected to further increase in the next decades (Björheden 2006; Hakkila 2006). In Finland, for example, the annual consumption of forest chips has increased from 1 million solid m3 to 5 million solid m3 during the past 10 years (Peltola 2009), and the objective of Finland’s National Forest Programme 2015 is to increase the annual use of forest chips to 13 million solid m3 by 2015. Energy wood is harvested at all stages of forest development, in final fellings, thinnings, and tending of seedling stands. Forest chips are produced from logging residues, roundwood, stumps, and small diameter trees. Currently, the logging residues from clear-cut areas are the largest source for forest chips (Peltola 2009).
Intensive biomass harvesting increases the export of nutrients from forests, as a result soil nutrient pools (Olsson et al. 1996a, b; Saarsalmi et al. 2010) and later on also tree growth (Jacobson et al. 2000; Egnell and Valinger 2003) may decrease. Particularly whole-tree harvesting (WTH), where all the above-ground biomass (stem and crown) is removed, can substantially increase nutrient losses compared with stem-only harvesting (SOH) where only the stem is removed (Mälkönen 1972; Nykvist 1974; Björkroth and Rosén 1977; Finér et al. 2003). Nutrient balance calculations indicate that in many cases, nutrient removals by WTH exceed the replenishment rate of plant-available nutrient pools in soil by mineral weathering and atmospheric deposition (Olsson et al. 1993; Sverdrup and Rosen 1998; Joki-Heiskala et al. 2003; Akselsson et al. 2007; Raulund-Rasmussen et al. 2008). This contravenes the principles of sustainable forest management where the productive capacity of forest ecosystems has to be maintained (Raulund-Rasmussen et al. 2008). In Fennoscandian boreal forests on upland mineral soils, WTH affects first nitrogen (N) supply and on peatlands, the phosphorus (P) and potassium (K) supply since upland forests are mostly N limited (e.g., Nohrstedt 2001) and those of peatlands P and K limited (e.g., Paarlahti et al. 1971). In addition, nutrient balance calculations also suggest that in many parts of Fennoscandia, WTH can result in net losses of base cations (K and Ca) (Sverdrup and Rosen 1998; Joki-Heiskala et al. 2003; Akselsson et al. 2007).
The magnitude of nutrient removals depends on the harvesting method, the developmental stage of the stand, and the tree species. WTH causes greater nutrient removals than SOH because nutrient concentrations are generally higher in foliage and branches than in stems (Mälkönen 1974; Mälkönen and Saarsalmi 1982; Kubin 1983; Finér 1989). The developmental stage of the stand, in turn, affects biomass allocation and nutrient contents (Mälkönen 1974; Helmisaari 1995; Augusto et al. 2000). The proportion of nutrient-rich crown and bark out of the total biomass decreases as a stand ages (Albrektson 1980; Hakkila 1989; Vanninen et al. 1996) and accordingly also the nutrient amount per unit of stem and total biomass (Augusto et al. 2000). Nutrient concentrations (Finér 1989; Alriksson and Eriksson 1998) and the patterns of biomass production and allocation also differ between tree species (Björkroth and Rosén 1977; Hakkila 1989; Finér et al. 2003). Scots pine (Pinus sylvestris L.), Norway spruce (Picea abies Karsten), and birch (Betula pendula Roth and Betula pubescens Ehrh.) are the dominant tree species in Fennoscandian forests. Birch generally has higher nutrient concentrations than pine or spruce (Finér 1989; Alriksson and Eriksson 1998). Spruce, in turn, allocates a greater proportion of biomass to its crown than pine or birch (Hakkila 1989; Kärkkäinen 2005; Repola 2009).
The amount of nutrients bound in tree biomass has been estimated by calculating the biomass of different tree fractions with biomass equations and multiplying them by the corresponding nutrient concentrations (Nihlgård 1972; Mälkönen 1974; Mälkönen and Saarsalmi 1982; Finér 1991; Brække and Håland 1995). These calculations have first been done on the tree level, and thereafter, the stand-level calculations have been done by summing up the nutrient content of the individual trees. The determination of the amounts of nutrients bound in tree biomass in such a way is time consuming and expensive. An easier and less laborious way to estimate accumulated nutrient amounts is to use nutrient equations, in which nutrient content of trees is predicted by tree height, breast height diameter, or tree biomass (Jokela et al. 1981; Laiho 1997; Ingerslev and Hallbäcken 1999; Augusto et al. 2000; Ponette et al. 2001; Raulund-Rasmussen et al. 2008; Stupak et al. 2008). So far, most of the existing nutrient equations have been developed for temperate forests; they are site specific or applicable only for single trees (Jokela et al. 1981; Ingerslev and Hallbäcken 1999; Ponette et al. 2001; Stupak et al. 2008). Since the utilization of forest biomass is increasing in Fennoscandian boreal forests, there is a need for generalized easy to use stand-level nutrient equations for estimating the nutrient removals in different harvesting operations. Nutrient equations form a base for nutrient balance calculations, and they are useful for forest managers in adjusting forest management strategies to the resiliency of the site concerned and in evaluating the need for fertilization. Nutrient equations based on the relationships between stand volume, which is a commonly estimated stand characteristic, and the nutrient content in stems and the whole above-ground stand biomass would provide a good tool for estimating nutrient removals in SOH or WTH. Data from several individual studies in boreal forests can be utilized for making such nutrient equations (e.g., Holmén 1964; Nihlgård 1972; Mälkönen 1974; Nykvist 1974; Lehtonen 1978; Mälkönen and Saarsalmi 1982; Finér 1991; Brække and Håland 1995; Ukonmaanaho et al. 2008).
The aim of this study was to develop, based on the literature data from Fennoscandian Scots pine, Norway spruce, and birch forests, generalized stand-level equations for the N, P, K, and Ca concentration and content of stems and total above-ground biomass with stand volume as an independent variable. The data and the developed equations were used for evaluating the effects of different tree species and harvesting intensities on nutrient removals.
Materials and methods
Material
We collected literature data of biomass and nutrient content in the above-ground parts of trees from pure Fennoscandian Scots pine, Norway spruce, and birch forests (Tables 1, 2, 3). Data from fertilized stands were not included. The data consisted of 34 pine, 26 spruce, and 5 birch stands. They covered a wide range of variation in climatic conditions, site types, and stand characteristics. The southernmost pine and spruce stands were situated in southern Sweden (55–57°N) and the northernmost ones near the Arctic Circle (66–67°N). Birch stands were situated at latitudes between 61°N and 64°N. The mean annual temperatures varied between −0.4 and +6°C and the annual precipitation between 480 and 1,040 mm. The stand volumes were in the range of 6–359, 22–802, and 39–169 m3 ha−1 in pine, spruce, and birch stands, respectively. The ages of stands ranged from 15 to 150 years in pine stands, from 18 to 250 years in spruce stands, and from 20 to 60 years in birch stands.
Biomass and N, P, K, and Ca contents in stems (stem wood and stem bark) and the total above-ground biomass (stems, live and dead branches and foliage) of the forest stands were collected from the literature. The studies used sample trees for analyzing the nutrient concentrations and for formulating biomass equations between different stem or crown fractions (dead and living branches and foliage) and easily measurable stem variables (e.g., breast height diameter, height). The sample trees were harvested at the end of the growing season. Nitrogen concentrations were analyzed by the Kjeldahl method. The determinations of P, K, and Ca concentrations were made from wet acid digested samples. In the most recent studies, the concentrations of P, K, and Ca were determined with inductively coupled plasma atomic emission spectrophotometer (ICP), whereas in older studies (before the 1990s), P was usually determined with colorimetric methods, K with flame photometer or atomic absorption spectrophotometer (AAS), and Ca with AAS. The methodologies for analyzing nutrient concentrations varied between studies, but we think that they are well comparable, since different nutrient analysis methods have generally shown good agreement (Handson and Shelley 1993). The biomass equations and the nutrient concentrations were then used to calculate the nutrient content for all trees in the stand.
Statistical analyses
Nutrient concentrations in stems, crowns, and total above-ground biomass were calculated by dividing the nutrient content by the corresponding biomass. Differences in nutrient concentrations between tree species and biomass fractions (stem and crown) were analyzed using one-way ANOVA followed by either Bonferroni (equal variances) or Dunnett T3 (unequal variances) post hoc tests. The homogeneity of variances was tested using the Levene’s test. Differences in the nutrient content of stem and total above-ground biomass between tree species were tested with a general linear model by using stand volume as a covariate. All statistical tests were conducted using SPSS software (PASW Statistics 17.0, SPSS Inc., Chicago, USA).
Derivation of equations and calculations
Regression equations were formulated between stem biomass or total above-ground biomass (kg ha−1) and stand volume (m3 ha−1) and the N, P, K, and Ca content (kg ha−1) in stems or total above-ground biomass and stand volume (m3 ha−1). The parameters were estimated by the Levenberg–Marquardt algorithm (PASW Statistics 17.0, SPSS Inc., Chicago, USA). The following model was used:
where Y is the amount of biomass or nutrient (kg ha−1), X is stand volume (m3 ha−1), and a and b are parameters. The models were converted to linear form using the natural log transformation:
Logarithmically transformed values were used in the analyses. In order to correct the underestimate of the regression estimate caused by this transformation, a condensed form (Madgwick and Satoo 1975) of the correction term (Finney 1941) was used for calculating the results:
k = correction term, S 2 e = residual variance.
Since stand age was available from most stands and it has been shown that nutrient concentrations in stand biomass are related to stand age (Augusto et al. 2000), we also attempted to formulate regression equations for N, P, K, and Ca content in stems or above-ground biomass with stand age as an independent variable. In addition, we tried to formulate equations for N, P, K, and Ca concentrations in stems or total above-ground biomass with stand volume as an independent variable, since its has been shown that nutrient concentrations of trees are related to tree size (Augusto et al. 2008). None of these equations were statistically significant (P > 0.05), thus they are not presented here. The fact that the nutrient content of stems and above-ground biomass could not be explained by stand age could be related to the correlation between the geographical location and the fertility of the site and stand age. Tree stands of a particular volume are older on infertile or northern sites than the tree stands of the same volume growing on fertile or southern sites (Kärkkäinen 2005). Thus, when the range of site fertilities or geographical locations is large, the correlation between age and tree size is weakened. The age of the stands in this study were also at the range where the relationship between nutrient concentrations of biomass and stand age is stable (Augusto et al. 2000). The weak correlation between stand volume and the nutrient concentrations of stems and above-ground biomass, in turn, may be related to the variation in stand density and accordingly to the mean tree size between the stands of the same volume in this study. Since the nutrient concentrations are related to tree size (Augusto et al. 2008), the relationships between volume and nutrient concentrations were not observed at stand level in this small dataset.
The derived equations were used to evaluate the effects of different harvesting intensities on nutrient removals and to give an example of the amounts of nutrients removed in Scots pine, Norway spruce, and birch stands at thinning and final cutting. In this study, nutrient removals by harvesting were assumed to be equal to the accumulated nutrient amounts. The example calculations were made only for one site type, one region in Finland and one management option (number of seedlings at stand establishment, number of thinnings, thinning regime, and rotation age) that was based on current Finnish forest management practices. We used the MOTTI stand simulator (Hynynen et al. 2005; Salminen et al. 2005) to simulate the stand volume development over the whole rotation on medium-rich Myrtillus type (MT) forest in Central Finland (Table 4). The MT site type was chosen because it is the most common site type in Finland, and all these three tree species are grown on MT sites. The stand development was started by planting (2,000 seedlings ha−1), and the stands were thinned two times before the end of the rotation. The total yield of stem wood during the whole rotation was 425, 430, and 358 m3 ha−1 in the pine, spruce, and birch stands, respectively.
Results
Nutrient concentrations and contents
The concentrations of all studied nutrients were lower in stems than in the crowns of pine, spruce, and birch (Table 5). Nitrogen concentrations did not differ in the stems (stem wood and stem bark combined) of the three tree species. Phosphorus concentrations were higher in birch stems than in pine stems. Potassium concentrations were higher in spruce stems than in pine stems, and Ca concentrations were higher in spruce stems than in the stems of pine and birch. In the crowns (including branches and foliage), N concentrations were highest in birch crowns. Phosphorus and Ca concentrations were lowest in pine crowns. The K concentrations of crowns did not differ significantly between the tree species. Nitrogen, P, and K concentrations in the total above-ground biomass did not differ between the tree species, and Ca concentrations were lowest in pine.
The N and P content of stems were higher in birch than in pine (Table 6). The K content of stems did not differ significantly between the tree species. The Ca content of stems was higher in spruce than in pine. The N, P, K, and Ca content of total above-ground biomass were higher in spruce than in pine. Spruce also had higher Ca content in total above-ground biomass than birch.
Nutrient equations
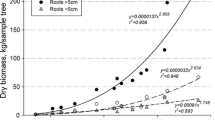
There was a clear relationship both between stem or total above-ground biomass and stem volume and the content of different nutrients in stem or total above-ground biomass and stand volume (Figs. 1, 2, 3, 4, 5). All of the equations for pine and spruce were statistically highly significant (P < 0.001), whereas the significance levels of birch equations were generally somewhat lower (Table 7). The equations explained, depending on the nutrient, 61–89, 60–87, and 78–99% of the variation in stem nutrient content in pine, spruce, and birch stands, respectively. The equations also accounted for most of the variation in the nutrient content of total above-ground biomass, the R 2 values ranging from 0.58 to 0.80 in pine stands, from 0.56 to 0.84 in spruce stands, and from 0.56 to 0.87 in birch stands. The crown represented a small proportion of the total above-ground biomass, but its nutrient content was high. The relative proportion of the crown out of total above-ground biomass was considerably larger in spruce than in pine (Fig. 1). The form of the curves indicated that the proportion of crown out of the total above-ground nutrient content generally decreases with the increasing stand volume. WTH thus removes more nutrients per harvested stem m3 in small volume stands.
Stem (stem wood and stem bark) and total above-ground (stem and crown) biomass (kg ha−1) in Scots pine, Norway spruce and birch stands as a function of stem volume (m3 ha−1). Solid lines are predictions of Eq. 1 for stems and dashed lines are predictions of Eq. 1 for above-ground biomass (Table 7). The correction terms (Eq. 2, Table 7) were included into the equations when calculating predicted values. Note the different scales of the figures. The figure at lower right shows differences between tree species in the predicted stem (lower line) and above-ground biomass (upper line)
The amount of nitrogen (kg ha−1) in Scots pine, Norway spruce and birch stems (stem wood and stem bark) and above-ground biomass (stem and crown) as a function of stem volume (m3 ha−1). Solid lines are predictions of Eq. 1 for stems and dashed lines are predictions of Eq. 1 for above-ground biomass (Table 7). The correction terms (Eq. 2, Table 7) were included into the equations when calculating predicted values. Note the different scales of the figures. The figure at lower right shows differences between tree species in the predicted amount of nitrogen in stems (lower line) and above-ground biomass (upper line)
The amount of phosphorus (kg ha−1) in Scots pine, Norway spruce and birch stems (stem wood and stem bark) and above-ground biomass (stem and crown) as a function of stem volume (m3 ha−1). Solid lines are predictions of Eq. 1 for stems and dashed lines are predictions of Eq. 1 for above-ground biomass (Table 7). The correction terms (Eq. 2, Table 7) were included into the equations when calculating predicted values. Note the different scales of the figures. The figure at lower right shows differences between tree species in the predicted amount of phosphorus in stems (lower line) and above-ground biomass (upper line)
The amount of potassium (kg ha−1) in Scots pine, Norway spruce and birch stems (stem wood and stem bark) and above-ground biomass (stem and crown) as a function of stem volume (m3 ha−1). Solid lines are predictions of Eq. 1 for stems and dashed lines are predictions of Eq. 1 for above-ground biomass (Table 7). The correction terms (Eq. 2, Table 7) were included into the equations when calculating predicted values. Note the different scales of the figures. The figure at lower right shows differences between tree species in the predicted amount of potassium in stems (lower line) and above-ground biomass (upper line)
The amount of calcium (kg ha−1) in Scots pine, Norway spruce and birch stems (stem wood and stem bark) and above-ground biomass (stem and crown) as a function of stem volume (m3 ha−1). Solid lines are predictions of Eq. 1 for stems and dashed lines are predictions of Eq. 1 for above-ground biomass (Table 7). The correction terms (Eq. 2, Table 7) were included into the equations when calculating predicted values. Note the different scales of the figures. The figure at lower right shows differences between tree species in the predicted amount of calcium in stems (lower line) and above-ground biomass (upper line)
The results indicate that WTH substantially increases nutrient removals compared with SOH, and the relative increase in nutrient removals are generally greater the smaller the stand volume is. On average, WTH increased the removals of N, P, K, and Ca by 2.9 (range 1.7–5.1), 3.9 (range 2.4–11.2), 2.7 (range 1.8–5.3), and 1.9 (range 1.3–3.3) times, respectively, compared with SOH in pine stands. In spruce stands, WTH removed on average 3.7 (range 2.1–8.2), 5.0 (range 2.4–11.7), 2.9 (range 1.8–5.9), and 2.5 (range 1.4–9.8) times more N, P, K, and Ca, respectively, than did SOH. Corresponding values for birch stands were 2.4 (range 1.9–2.9), 2.7 (range 1.9–3.1), 2.2 (range 1.6–2.9), and 2.1 (range 1.6–3.0).
According to the equations, about two times more N and P are removed in birch than in pine and spruce stands with equal stand volumes in SOH. According to the equations, the amounts of K and Ca in stems are the highest in spruce at small stand volumes but after stand volume exceeds 98 m3 ha−1, the amounts of K are the highest in birch stands (Fig. 4). The amount of Ca in birch stems becomes higher than in spruce stems when stand volume exceeds 154 m3 ha−1 (Fig. 5). Nutrient removal in WTH is smallest in pine stands. Pine has about half the amount of nutrients in above-ground biomass as spruce. The equations predict that the removals of N, P, and K are greatest in the WTH of spruce stands if stand volume is ≤256, ≤220, and ≤228 m3 ha−1, respectively (Figs. 2, 3, 4). For stand volumes greater than that, the removals of N, P, and K were largest in birch stands. The removal of Ca in WTH would always be greatest in spruce stands (Fig. 5).
Example calculation over the whole rotation period
The amounts of nutrients removed during the whole rotation of typical pine, spruce, and birch stands growing in southern Finland were calculated for SOH and WTH intensities by using the developed nutrient equations (Fig. 6). During the rotation period almost equal amounts, 425, 430, and 358 m3 ha−1 of stems were harvested in pine, spruce, and birch stands, respectively, corresponding to 174,468, 177,736, and 195,672 kg ha−1 of biomass in SOH and 194,876, 230,332, and 216,712 kg ha−1 in WTH in pine, spruce, and birch stands, respectively (Table 4). Results show that WTH increases nutrient removals by 2- to 3-fold in pine stands and by 2- to 4-fold in spruce stands as compared to SOH. In birch stands, the increase was 2-fold. During the rotation period, the nutrient removals in SOH were the largest in birch stands and the smallest in pine stands. In WTH, the nutrient removals were the lowest in pine stands and somewhat higher in spruce than in birch stands. When considered per unit volume of harvested stem, nutrient removals in SOH were the largest in birch stands and the smallest in pine stands. In WTH, nutrient removals per m3 of harvested stem were the lowest in pine stands. The most N, P, and K per harvested stem m3, in turn, were removed in the harvesting of birch stands, and the removals of Ca were largest in spruce stands.
We also calculated the amounts of nutrients removed at the first thinning and final cutting when the same amount of stem (100 m3) is harvested. The calculations show that almost equal amounts of nutrients are removed at first thinning and final cutting per unit of harvested stem in pine, spruce, and birch stands in SOH, whereas in WTH, nutrient removals at first thinning are considerably greater than at final cutting especially in pine and spruce stands (Fig. 7). In SOH, both at first thinning and final cutting nutrient removals are the largest from birch stands and the smallest from pine stands. When harvesting the stem wood in WTH at first thinning, the nutrient removals are the smallest from pine and the largest from spruce stands. Likewise at final cutting, the nutrient removals are the smallest in pine stands, but the difference between spruce and birch is small.
Discussion
We succeeded in formulating regression equations with high degrees of determination for N, P, K, and Ca content in stem wood or total above-ground biomass and stem volume as the independent variable for pine, spruce, and birch stands. We used these equations for showing the differences in harvesting intensities, tree species, and stand developmental stages on nutrients removals from forest stands. The nutrient removals will be compared with the other nutrient fluxes and soil nutrient pools to enable the evaluation of their importance on the maintaining nutrient balance in Fennoscandian boreal forest stands.
The formulated nutrient equations were based on data representing relatively well the variation in stand age, fertility, and geographical location in pure Scots pine and Norway spruce stands in boreal region of Fennoscandia, whereas the equations for birch were based on a very limited number of stands. The equations may not be applicable for mixed stands, e.g., because the nutrient concentrations of different tree fractions may differ between pure and mixed stands (Thelin et al. 2002). The validity of the equations depends not only on the number of stands but also on the level of precision of individual studies in determining stand nutrient content. It has been pointed out that the sample tree material has to be large enough in order to determine stand nutrient content precisely (Comerford and Leaf 1982a, b).
Even though the degree of the determination was high in most nutrient equations, the error terms were relatively high. Several factors, like site fertility (Kellomäki and Väisänen 1986; Vanninen et al. 1996; Kärkkäinen 2005), geographical location (Kärkkäinen 2005), stand density (Kellomäki and Väisänen 1986; Nilsson and Gemmel 1993; Mäkelä and Vanninen 1998; Ilomäki et al. 2003), and stand age (Albrektson 1980; Hakkila 1989; Helmisaari 1995; Vanninen et al. 1996; Lehtonen et al. 2004; Kantola and Mäkelä 2006) affect the allocation of biomass between stems and crowns in Fennoscandian boreal forests. In addition, the nutrient concentrations of tree stands are affected by geographical location (Ericsson et al. 1993; Oleksyn et al. 2003), stand age (Augusto et al. 2000), and to small extent also site fertility (Reinikainen et al. 1998; Augusto et al. 2000). These factors are among the ones, which could possibly improve the nutrient equations, if they are included in the equations. The inclusion of these factors as independent variables is problematic since many of the variables are confounded with each other, and the more there are independent variables the more data are needed for deriving the equations. In all cases, more data are needed to improve and to independently validate the equations.
We found that WTH increases nutrient removals 2- to 4-fold compared with SOH in pine, spruce, and birch stands (Fig. 6) and was attributed to the fact that the proportion of crown out of the total above-ground nutrient amount decreases with increasing stand volume. This finding is comparable to the estimates based on the previous experimental (Mälkönen 1972; Kubin 1977; Mälkönen and Saarsalmi 1982) and modeling (Raulund-Rasmussen et al. 2008) studies in single tree species stands, which indicate WTH to remove from 2 to 7 times more nutrients than SOH in Scots pine, Norway spruce, and birch stands. So far, little attention has been paid on how the developmental stage of the stand affects nutrient removals. We found that the nutrient removals are the same in thinnings and final cuttings in SOH but considerably larger in thinnings than final cuttings in WTH, especially in pine and spruce stands when the same volume of stem is harvested.
It has also been shown earlier that the nutrient removals in SOH are higher in birch stands compared with pine or spruce stands (Finér 1989; Hakkila 1989). This is probably due to that stem biomass is higher in birch stands compared with pine and spruce stands with similar stem volume (Fig. 1). The results on the effects of nutrient removals could be somewhat biased for birch, since the data originated from younger stands with smaller trees than those for pine and spruce. Tree nutrient concentrations decrease with the size and age of the trees (Augusto et al. 2000, 2008). In WTH, nutrient removals were found to be smallest in pine stands, which is probably because the nutrient concentrations were lowest in pine stems and crowns (Table 5). Considerably smaller nutrient removals in pine than in spruce WTH can be also explained by the fact that the biomass of crown is two times smaller in pine than in similar size spruce trees (Repola 2009).
The intensification of the utilization of forest biomass has increased the concern about its impact on the nutritional status of forests. In this study, nutrient removals by harvesting were assumed to be equal to the accumulated nutrient amounts. In reality, the nutrient removals are somewhat smaller because, in practice, part of the logging residues will remain on the site in WTH (Hakkila 2002). It should also be noted that nutrient removals are smaller in birch stands if WTH is done during the leafless period. About 20–30% of the above-ground tree nutrients are in the leaves in birch stands (Tamm 1969; Mälkönen 1977). A part of the nutrients contained in the leaves are, however, translocated to storage tissues during leaf senescence. In addition to harvesting, nutrients are lost from the forest ecosystem through leaching which is increased after final cutting (Table 8). Nutrients removed in harvesting operations can be compensated by inputs from atmospheric deposition and in mineral soils also by weathering. Nutrient deposition is highest in southern Fennoscandia and decreases toward the north (e.g., Ruoho-Airola et al. 2003; Akselsson et al. 2010). In a typical tree stand in southern Finland (Table 4), the removals of N caused by SOH (Fig. 6) are smaller in pine, spruce, and birch stands than the inputs by N deposition during the rotation (Table 8). Deposition and weathering together can compensate P and K losses caused by SOH during the rotation in pine and spruce stands but in birch stands, more P (35 kg P ha−1) and K (156 kg K ha−1) are removed in SOH than are received through weathering and deposition (17 kg P ha−1 and 102 kg K ha−1 at maximum). If we take into account the leaching of P and K, the balance can come negative also in pine and spruces stands. Deposition and weathering were sufficient to compensate for the Ca losses caused by SOH during the rotation in all studied tree species. On stands where the Ca leaching is high, the balance can be negative. If all biomass is removed, the nutrient inputs cannot compensate the nutrients removed from the forest. Even if WTH is done only at final felling, the removals of N are greater than N deposition in spruce and birch stands, and the removals of P exceed the inputs through deposition and weathering in all tree species. Deposition and weathering are also insufficient to compensate for the losses of K in spruce and birch stands and the losses of Ca in spruce stands. These nutrient input–output comparisons suggest that in most cases, nutrient balance is negative if WTH is practised, and especially in birch stands, the balances of P and K can be negative even in SOH. These results are consistent with nutrient balance studies, which have demonstrated that there is a depletion of N and base cations in many parts of Fennoscandia in the long-term if WTH is practised (Sverdrup and Rosen 1998; Joki-Heiskala et al. 2003; Akselsson et al. 2007). The N balance has been found to be positive in WTH only in southern Sweden where N deposition is high (Akselsson et al. 2007). Some uncertainties are always involved in these nutrient balance calculations because nutrient fluxes vary temporally and spatially and it is technically difficult to determine some of them (Raulund-Rasmussen et al. 2008). The estimates of weathering rates are probably most uncertain because there are only a very few studies on boreal forest soils, and the estimates have varied greatly depending on the method of investigation (Jacks 1990; Starr et al. 1998). Nitrogen may also be lost though denitrification and gained through N-fixation (Rosén and Lindberg 1980; DeLuca et al. 2002; Maljanen et al. 2006). These fluxes are, however, small compared with the nutrient removals caused by harvesting (Rosén and Lindberg 1980; DeLuca et al. 2002; Maljanen et al. 2006).
The capacity of forest soil to sustain the tree production in the long term can also be assessed by comparing nutrient removals associated with harvesting to total and plant-available nutrient pools in the soil. In cases where the pools are small compared the removals, the risk for unbalanced tree nutrition is high and there is a need for compensating fertilization. The amounts of N removed with harvested biomass during the rotation were relatively large compared with the total soil N pools in mineral soil sites, and the removed P and K amounts were relatively large compared with the total soil P and K pools in peatland sites particularly in WTH (Fig. 6, Table 8). The removals of Ca with harvested biomass were relatively large in both mineral soil and peatlands. Other studies have also suggested that wood production and especially WTH may be a threat for the sufficiency of K in peatlands because the K pools in trees are of a similar order of magnitude as total K pools in the surface peat (Holmén 1964; Kaunisto and Paavilainen 1988; Finér 1989; Laiho 1997). Only a small part of soil nutrients are in a form available for trees (Table 8). The amounts of nutrients removed during the rotation are considerable compared with the plant-available nutrient pools in the rooting zone. Although nutrient budget calculations can be imprecise, comparisons with the size of soil nutrient pools confirm that WTH causes considerable nutrient losses in north European boreal forests, and if forests are not fertilized, the soil nutrient pools might be depleted and the growth of trees reduced as has already been shown in a few studies (Olsson et al. 1996a, b; Jacobson et al. 2000; Egnell and Valinger 2003; Saarsalmi et al. 2010). Tree stands are probably the most sensitive to nutrient removals caused by WTH at the thinning stage (Jacobson et al. 2000) when the nutrient demand is greatest (Mälkönen 1974).
Conclusions
We conclude that we could formulate regression equations where stand volume is used as an independent variable and provide an easy tool to get estimates of the nutrient content of stems and above-ground biomass in boreal Scots pine, Norway spruce, and birch stands. More data would be needed to increase the validity of the equations. Tree species, harvesting method, and the stage of stand development have a pronounced influence on nutrient removals. Nutrient removals per harvested stem m3 are smallest in pine stands both in SOH and WTH. Nutrient removals by WTH can be reduced by harvesting energy wood from mature forests instead of young thinning forests because the nutrient-rich crown accounts for a relatively higher proportion of biomass in young forests. If WTH is done at final cutting, nutrient export by harvesting is in many cases greater than the nutrient inputs through deposition and weathering during the rotation. In forests stands where nutrient leaching is high, the balances of P, K, and Ca can be negative even in SOH. In mineral soil sites, N removals, and in peatland sites, P and K removals by WTH during the rotation are relatively large compared with the respective soil nutrient pools.
References
Akselsson C, Westling O, Sverdrup H, Gundersen P (2007) Nutrient and carbon budgets in forest soils as decision support in sustainable forest management. For Ecol Manag 238:167–174
Akselsson C, Belyazid S, Hellsten S, Klarqvist M, Phil-Karlsson G, Karlsson P-E, Lundin L (2010) Assessing the risk of N leaching from forest soils across a steep N deposition gradient in Sweden. Environ Pollut 158:3588–3595
Albrektson A (1980) Relations between tree biomass fractions and conventional measurements. In: Persson T (ed) Structure and function of northern coniferous forests–an ecosystem study. Ecol Bull 32:315–327
Alriksson A, Eriksson HM (1998) Variations in mineral nutrient and C distribution in the soil and vegetation compartments of five temperate tree species in NE Sweden. For Ecol Manag 108:261–273
Asikainen A, Liiri H, Peltola S, Karjalainen T, Laitila J (2008) Forest Energy Potential in Europe (EU 27). Working Papers Finn For Res Inst, vol 69, 33pp
Augusto L, Ranger J, Ponette Q, Rapp M (2000) Relationships between forest tree species, stand production and stand nutrient amount. Ann For Sci 57:313–324
Augusto L, Meredieu C, Bert D, Trichet P, Porté A, Bosc A, Lagane F, Loustau D, Pellerin S, Danjon F, Ranger J, Gelpe J (2008) Improving models of forest nutrient export with equations that predict the nutrient concentration of tree compartments. Ann For Sci 65:808
Björheden R (2006) Drivers behind the development of forest energy in Sweden. Biomass Bioenergy 30:289–295
Björkroth G, Rosén K (1977) Biomassa och näringsmängder på fyra ståndorter. Projekt Helträdsutnyttjande. No. 49. Swedish University of Agricultural Sciences, Stockholm, 20 p
Brække FH, Håland B (1995) Above-ground biomass and mineral element distribution in a Scots pine stand of a virgin low-shrub pine bog. Meddelelser fra Communications of Skogforsk 47(7):1–17
Bringmark L (1977) A bioelement budget of an old Scots pine forest in central Sweden. Silva Fenn 11:201–209
Cajander AK (1949) Forest types and their significance. Acta For Fenn 56:1–71
Comerford NB, Leaf AL (1982a) An evaluation of techniques for sampling forest tree nutrient content. Part I. Sampling the crown for total nutrient content. Forest Sci 28:471–480
Comerford NB, Leaf AL (1982b) An evaluation of techniques for sampling forest tree nutrient content. Part II. Sampling for stem nutrient content. Forest Sci 28:481–487
DeLuca TH, Zackrisson O, Nilsson M-C, Sellstedt A (2002) Quantifying nitrogen fixation in feather moss carpets of boreal forests. Nature 419:917–920
Derome J, Lindgren M, Merilä P, Beuker E, Nöjd P (2007) Forest condition monitoring under the UN/UCE and EU programmes in Finland. In: Merilä P, Kilpua T, Derome J (eds) Forest condition monitoring in finland—national report 2002–2005. Working Papers of the Finnish Forest Research Institute 45:11–20
Egnell G, Valinger E (2003) Survival, growth and growth allocation of planted Scots pine trees after different levels of biomass removal in clear-felling. For Ecol Manag 177:65–74
Ericsson A, Nordén L-G, Näsholm T, Walheim M (1993) Mineral nutrient imbalances and arginine concentrations in needles of Picea abies (L.) Karst. from two areas with different levels of airborne deposition. Trees 8:67–74
Eriksson HM, Berden M, Rosén K, Nilsson IS (1996) Nutrient distribution in a Norway spruce stand after long-term application of ammonium nitrate and superphosphate. Water Air Soil Pollut 92:451–467
European Commission (1997) Energy for the future: renewable sources of energy. White paper for a community strategy and action plan. Com (97) 599 final
Finér L (1989) Biomass and nutrient cycle in fertilized and unfertilized pine, mixed birch and pine and spruce stands on a drained mire. Acta For Fenn 208:63
Finér L (1991) Effect of fertilization on dry mass accumulation and nutrient cycling in Scots pine on an ombotrophic bog. Acta For Fenn 233:42
Finér L, Mannerkoski H, Piirainen S, Starr M (2003) Carbon and nitrogen pools in an old-growth, Norway spruce mixed forest in eastern Finland and changes associated with clear-cutting. For Ecol Manag 174:51–63
Finér L, Kortelainen P, Mattsson T, Ahtiainen M, Kubin E, Sallantaus T (2004) Sulphate and base cation leaching from unmanaged forested catchments in Finland. For Ecol Manag 195:115–128
Finér L, Mannerkoski H, Piirainen S, Laurén A, Koivusalo H, Kokkonen T, Penttinen S (2005) Nutrient fluxes from managed boreal forest ecosystems to surface and ground waters. In: Jalkanen A, Nygren M 2005 (eds) Sustainable use of renewable natural resources—from principles to practices. University of Helsinki Department of Forest Ecology Publications 34:123–134
Finér L, Mattsson T, Joensuu S, Koivusalo H, Laurén A, Makkonen T, Nieminen M, Tattari T, Ahti E, Kortelainen P, Koskiaho J, Leinonen A, Nevalainen R, Piirainen S, Saarelainen J, Sarkkola S, Vuollekoski M (2010) Vesistökuormituksen laskenta metsäisiltä valuma-alueilta. Suomen ympäristö 10/2010, 33 p
Finney DJ (1941) On the distribution of a variate whose logarithm is normally distributed. J Roy Stat Soc Ser B7:155–161
Fisher RF, Binkley D (2000) Ecology and management of forest soils. Wiley, New York. 489 p
Hakkila P (1989) Utilization of residual forest biomass. Springer, Berlin, 568 p
Hakkila P (2002) Operations with reduced environmental impact. In: Richardson J, Björnheden R, Hakkila P, Lowe AT, Smith CT (eds) Bioenergy from sustainable forestry. Guiding principles and practice. Kluwer, Dordrecht, pp 244–261
Hakkila P (2006) Factors driving the development of forest energy in Finland. Biomass Bioenergy 30:281–288
Handson PD, Shelley BC (1993) A review of plant analysis in Australia. Aust J Exp Agric 33:1029–1038
Heikurainen L, Pakarinen P (1982) Peatland classification. In: Laine J (ed) Peatlands and their utilization in Finland. Finnish peatland Society. Helsinki, pp 14–23
Helmisaari H-S (1995) Nutrient cycling in Pinus sylvestris stands in eastern Finland. Plant Soil 168–169:327–336
Helmisaari H-S, Kukkola M, Luiro J, Saarsalmi A, Smolander A, Tamminen P (2009) Hakkuutähteen korjuu—muuttuuko typen saatavuus ? Metsätieteen aikakauskirja 1(2009):57–62
Holmén H (1964) Forest ecological studies on drained peat land in the provence of Uppland, Sweden. Parts I-III. Stud For Suec 16:236
Hynynen J, Ahtikoski A, Siitonen J, Sievänen R, Liski J (2005) Applying the MOTTI simulator to analyse the effects of alternative management schedules on timber and non-timber production. For Ecol Manag 207:5–18
Ilomäki S, Nikinmaa E, Mäkelä A (2003) Crown rise due to competition drives biomass allocation in silver birch. Can J For Res 33:2395–2404
Ingerslev M, Hallbäcken L (1999) Above ground biomass and nutrient distribution in a limed and fertilized Norway spruce (Picea abies) plantation part II. Accumulation of biomass and nutrients. For Ecol Manag 119:21–38
Jacks G (1990) Mineral weathering studies in Scandinavia. In: Mason BJ (ed) The surface waters acidification programme. Cambridge University Press, Cambridge, pp 215–222
Jacobson S, Kukkola M, Mälkönen E, Tveite B (2000) Impact of whole-tree harvesting and compensatory fertilization on growth of coniferous thinning stands. For Ecol Manag 129:41–51
Jokela EJ, Shannon CA, White EH (1981) Biomass and nutrient equations for mature Betula papyrifera Marsh. Can J For Res 11:298–304
Joki-Heiskala P, Johansson M, Holmberg M, Mattsson T, Forsius M, Kortelainen P, Hallin L (2003) Long-term base cation balances of forest mineral soils in Finland. Water Air Soil Pollut 150:255–273
Kantola A, Mäkelä A (2006) Development of biomass proportions in Norway spruce (Picea abies [L.] Karst.). Trees 20:111–121
Kärkkäinen L (2005) Evaluation of performance of tree-level biomass models for forestry modelling and analyses. Finn For Res Inst Res Pap 940:108 p + appendices
Kaunisto S, Paavilainen E (1988) Nutrient stores in old drainage areas and growth of stands. Commun Inst For Fenn 145:39
Kellomäki S, Väisänen H (1986) Kasvatustiheyden ja kasvupaikan viljavuuden vaikutus puiden oksikkuuteen taimikko- ja riukuvaiheen männikössä. Summary: Effect of stand density and site fertility on the branchiness of Scots pine at pole stage. Commun Inst For Fenn 139:38
Kubin E (1977) The effect of clear-cutting upon the nutrient status of a spruce forest in northern Finland (64° 28′ N). Acta For Fenn 155:1–40
Kubin E (1983) Nutrients in the soil, ground vegetation and tree layer in an old spruce forest in northern Finland. Ann Bot Fenn 20:361–390
Laiho R (1997) Plant biomass dynamics in drained pine mires in southern Finland, implications for carbon and nutrient balance. Finn For Res Inst Res Pap 631
Lehtonen I (1978) Ravinteiden kierto eräässä männikössä: IV fytomassan ja ravinteiden määrä. Nutrient cycle in a Scots pine stand: IV The amount of phytomass and nutrients (Summary in English). Silva Fenn 12:47–55
Lehtonen A, Mäkipää R, Heikkinen J, Sievänen R, Liski J (2004) Biomass expansion factors (BEFs) for Scots pine, Norway spruce and birch according to stand age for boreal forests. For Ecol Manag 188:211–244
Madgwick HAI, Satoo T (1975) On estimating the above-ground weights of tree stands. Ecology 56:1446–1450
Mäkelä A, Vanninen P (1998) Impacts of size and competition on tree form and distribution of aboveground biomass in Scots pine. Can J For Res 28:216–227
Maljanen M, Jokinen H, Saari A, Strömmer R, Martikainen PJ (2006) Methane and nitrous oxide fluxes, and carbon dioxide production in boreal forest soil fertilized with wood ash and nitrogen. Soil Use Manag 22:151–157
Mälkönen E (1972) Hakkuutähteiden talteenoton vaikutus männikön ravinnevaroihin. (Summary in English: Effect of harvesting logging residues on the nutrient status of Scots pine stands). Folia For 157:14
Mälkönen E (1974) Annual primary production and nutrient cycle in some Scots pine stands. Commun Inst For Fenn 84:87
Mälkönen E (1977) Annual primary production and nutrient cycle in a birch stand. Commun Inst For Fenn 91(5):1–35
Mälkönen E, Saarsalmi A (1982) Hieskoivikon biomassatuotos ja ravinteiden menetys kokopuunkorjuussa. English summary: biomass production and nutrient removal in whole tree harvesting of birch stands. Folia For 534:17
Nihlgård B (1972) Plant biomass, primary production and distribution of chemical elements in a beech and a planted spruce forest in south Sweden. Oikos 23:69–81
Nilsson U, Gemmel P (1993) Changes in growth and allocation of growth in young Pinus sylvestris and Picea abies due to competition. Scand J For Res 8:213–222
Nohrstedt HO (2001) Response of coniferous forest ecosystems on mineral soils to nutrient additions: a review of Swedish experiences. Scand J For Res 16:555–573
Nykvist N (1971) The effect of clearfelling on the distribution of biomass and nutrients. Ecol Bull 14:166–178
Nykvist N (1974) Växtnäringsförluster vid helträdsutnyttjande. En sammanställning av undersökningar i gran- och tallbestånd. Swedish University of Agricultural Sciences. Res Notes 76:74–90
Oleksyn J, Reich PB, Zytkowiak R, Karolewski P, Tjoelker MG (2003) Nutrient conservation increases with latitude of origin in European Pinus sylvestris populations. Oecologia 136:220–235
Olsson M, Rosén K, Melkerud P-A (1993) Regional modelling of base cation losses from Swedish forest soils due to whole-tree harvesting. Appl Geochem 2:189–194
Olsson BA, Staaf H, Lundkvist H, Bengtsson J, Rosén K (1996a) Carbon and nitrogen in coniferous forest soils after clear-felling and harvests of different intensity. For Ecol Manag 82:19–32
Olsson BA, Bengtsson J, Lundkvist H (1996b) Effects of different forest harvest intensities on the pools of exchangeable cations in coniferous forest soils. For Ecol Manag 84:134–147
Paarlahti K, Reinikainen A, Veijalainen H (1971) Nutritional diagnosis of Scots pine stands by needle and peat analysis. Commun Inst For Fenn 74:1–58
Paavilainen E (1980) Effect of fertilization on plant biomass and nutrient cycle on a drained dwarf shrub pine swamp. Commun Inst For Fenn 98(5):1–71
Palviainen M (2005) Logging residues and ground vegetation in nutrient dynamics of a clear-cut boreal forest. Dissertationes Forestales 12:38
Peltola A (ed) (2009) Finnish statistical yearbook of forestry. Finnish Forest Research Institute. Vammalan kirjapaino Oy, Sastamala, 452 p
Piirainen S, Finér L, Mannerkoski H, Starr M (2004) Effects of forest clear-cutting on the sulphur, phosphorus and base cations fluxes through podzolic soil horizons. Biogeochem 69:405–424
Ponette Q, Ranger J, Ottorini J-M, Ulrich E (2001) Above-ground biomass and nutrient content of five Douglas-fir stands in France. For Ecol Manag 142:109–127
Popović B, Burgtorf H (1964) Upptagningen av växtnäring efter gödsling av ett tallbestånd i Lappland. Department of Forest Ecology. Res Notes 4:15
Raulund-Rasmussen K, Stupak I, Clarke N, Callesen I, Helmisaari H-S, Karltun E, Varnagiryte-Kabasinskiene I (2008) Effects of very intensive forest biomass harvesting on short and long term site productivity. In: Röser D, Asikainen A, Raulund-Rasmussen K, Stupak I (eds) Sustainable use of forest biomass for energy. Managing forest ecosystems, vol 12. Springer, Dordrecht, pp 29–78
Reinikainen A, Veijalainen H, Nousiainen H (1998) Puiden ravinnepuutokset: Metsänkasvattajan ravinneopas. The Finnish Forest Research Institute, Research Papers, vol 688, 44pp
Repola J (2009) Biomass equations for Scots pine and Norway spruce in Finland. SilvaFenn 43:625–647
Rosén K (1982) Supply, loss and distribution of nutrients in three coniferous watersheds in central Sweden. Reports in Forest Ecology and Forest Soils. Department of Forest Soils. Swedish University of Agricultural Sciences. Report, vol 41, 70pp
Rosén K, Lindberg T (1980) Biological nitrogen fixation in coniferous watershed areas in central Sweden. Holarctic Ecol 3:137–140
Röser D, Asikainen A, Stupak I, Pasanen K (2008) Forest energy resources and potentials. In: Röser D, Asikainen A, Raulund-Rasmussen K, Stupak I (eds) Sustainable use of forest biomass for energy. A synthesis with focus on the Baltic and Nordic region. Managing Forest Ecosystems, vol 12. Springer, Dordrecht, pp 9–28
Ruoho-Airola T, Alaviippola B, Salminen K, Varjoranta R (2003) An investigation of base cation deposition in Finland. Bor Env Res 8:83–95
Saarsalmi A, Tamminen P, Kukkola M, Hautajärvi R (2010) Whole-tree harvesting at clear-felling: Impact on soil chemistry, needle nutrient concentrations, and growth of Scots pine. Scand J For Res 25:148–156
Salminen H, Lehtonen M, Hynynen J (2005) Reusing legacy FORTRAN in the MOTTI growth and yield simulator. Comput Electron Agric 49:103–113
Starr M, Westman CJ (1978) Easily extractable nutrients in the surface peat layer of virgin sedge-pine swamps. Silva Fenn 12:65–78
Starr M, Lindroos A-J, Tarvainen T, Tanskanen H (1998) Weathering rates in the Hietajärvi integrated monitoring catchment. Bor Env Res 3:275–285
Stupak I, Nordfjell T, Gundersen P (2008) Comparing biomass and nutrient removals of stems and fresh and predried whole trees in thinning in two Norway spruce experiments. Can J For Res 38:2660–2673
Sverdrup H, Rosen K (1998) Long-term base cation mass balances for Swedish forests and the concept of sustainability. For Ecol Manage 110:221–236
Tamm CO (1969) Site damage by thinning due to removal of organic matter and plant nutrients. Thinning and mechanization. IUFRO meeting, Stockholm, Sweden, pp 175–179
Tamminen P (1998) Maaperätekijät. In: Mälkönen E (ed) Ympäristömuutos ja metsien kunto. Metsäntutkimuslaitoksen tiedonantoja 691, 64–75
Thelin G, Rosengren U, Callesen I, Ingerslev M (2002) The nutrient status of Norway spruce in pure and mixed-species stands. For Ecol Manag 160:115–125
Ukonmaanaho L, Merilä P, Nöjd P, Nieminen TM (2008) Litterfall production and nutrient return to the forest floor in Scots pine and Norway spruce stands in Finland. Bor Env Res 13(suppl. B):67–91
Vanninen P, Ylitalo H, Sievänen R, Mäkelä A (1996) Effects of age and site quality on the distribution of biomass in Scots pine (Pinus sylvestris L.). Trees 10:231–238
Westman CJ, Laiho R (2003) Nutrient dynamics of drained peatland forests. Biogeochem 63:269–298
Acknowledgments
We thank the University of Helsinki, the Finnish Forest Research Institute, the Academy of Finland (project 640026) and Nordic Forestry CAR-ES, which is financed by the Nordic Ministers through SNS (Nordic Forest Research Co-operation Committee), for funding this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. Puettmann.
Rights and permissions
About this article
Cite this article
Palviainen, M., Finér, L. Estimation of nutrient removals in stem-only and whole-tree harvesting of Scots pine, Norway spruce, and birch stands with generalized nutrient equations. Eur J Forest Res 131, 945–964 (2012). https://doi.org/10.1007/s10342-011-0567-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-011-0567-4