Abstract
The codling moth (Cydia pomonella L.) is a significant pest of pome fruit throughout the world. Behavioral and ovicidal activities of five non-host plant extracts (Arctium lappa, Bifora radians, Humulus lupulus, Verbascum songaricum, Xanthium strumarium), synthetic sex pheromone, (E,E)-8,10-dodecadienol (codlemone), and the plant volatile lure, (2E,4Z)-2,4-decadienoate (pear ester) were evaluated against the codling moth, C. pomonella L. Codlemone elicited the greatest electroantennogram (EAG) response (6.2 ± 1.2 mV) of the compounds tested from male C. pomonella while pear ester elicited 1.7 ± 0.1 mV EAG response in female moths. Codlemone attracted 34.5% of male C. pomonella in olfactometer studies, and it was followed by the X. strumarium extract with 24.8%. There was a significant difference between the behavior of unmated and mated females. V. songaricum extract was the most active extract, attracting 25.4% of unmated females. However, mated C. pomonella females exhibited greatest attraction to pear ester. In a wind tunnel bioassay, combining X. strumarium with codlemone significantly increased the response of male upwind flight and source contact as compared with codlemone alone. All plant extracts, except for V. songaricum, significantly reduced the number of eggs laid. The plant extracts exhibited some toxic effects to eggs, and hatching rate of eggs was reduced as compared with the control. Our results indicate that some of the plant extracts tested are potential candidates for practical use after elucidation and characterization of active compound(s).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key message
-
New biologically active compounds are needed to improve the control of codling moth and discovery of non-host plant extracts holds potential.
-
The tested plant extracts had both behavioral and toxic effects on codling moth eggs, males, and females.
-
Some of the tested plant extracts could be further developed as management tools for codling moth.
Introduction
The codling moth (CM), Cydia pomonella L. (Lepidoptera: Tortricidae), is a cosmopolitan pest of deciduous fruits and causes heavy damage on apple (Malus domestica Borkh) and pear (Pyrus communis L.) in many parts of the world (Vickers and Rothschild 1991; Beers et al. 1993). The management of this pest heavily relies on the application of insecticides, ranging from organophosphates (OPs) to synthetic pyrethroids, neonicotinoids, and insect growth regulators (IGRs) (Croft and Reidl 1992; Dunley and Welter 2000; Reuveny and Cohen 2004; Mota-Sanchez et al. 2008). Due to increasing codling moth resistance to certain insecticide groups, e.g., organophosphorous, pyrethroids, oxadiazines, diacylhydrazine insecticides (Stara and Kocourek 2007; Mota-Sanchez et al. 2008) alongside with tight regulation of EPA and EU laws, new control methods to reduce the apple industry reliance on broad-spectrum insecticides have been explored (Varela et al. 1993; Knight et al. 1994; Witzgall et al. 2008). Several control strategies have been postulated and some of them, e.g., mating disruption, attract and kill technology, and biological control, have produced some promising results. Among these methods, mating disruption of CM appears to be the most promising one with a successful application under different climate conditions, despite reports of some failures with this method (Trimble 1995; Stelinski et al. 2007; Witzgall et al. 2010).
Host finding in the codling moth is largely guided by plant volatiles released from host plants, and these plant volatiles have been characterized extensively (Landolt and Guedot 2008). A multi-front attack strategy with using plant volatile compounds for improving CM management has been suggested (Light et al. 2001; Mitchell et al. 2008). This approach could increase the level of efficacy of pheromone mating disruption (Knight et al. 2005) and improve the efficacy of insecticides (Light and Knight 2011; Schmidt et al. 2008). Terpenoids may play an important role in host finding by the CM (Vallat and Dorn 2005). Masking of these terpenoids or reducing their concentration in the air could increase the success of the “push–pull” strategy that has been suggested in the CM management (Cook et al. 2007). Presence of particular non-host plants or presence of a diversity of plants could offer a new dimension in protecting plants from insect attack (Light et al. 1993; Bender et al. 1999). Our previous studies indicate that certain non-host extracts are toxic and behaviorally active, e.g., antioviposition, attractant against important lepidopteran pest species (e.g., oblique banded leafroller, Choristoneura rosaceana Harris, red-banded leafroller, Argyrotaenia velutinana Walker and grape berry moth, Paralobesia viteana Clemens) (Gökçe et al. 2005, 2006, 2010). Preliminary studies with plant extracts used in this study showed that these plants extracts had some biological activities, e.g., attractant, antioviposition against the CM. Further studies with these extracts against the CM may help us to explore full potential for using them in different control strategies, e.g., mating disruption, attract and kill and push–pull strategies of non-host plant extracts.
The objectives of this study were to determine: (1) the effects of the non-host plant extracts, pheromone, and pear ester on behavioral and antennal responses of mated and unmated CM; (2) the impact of non-host plant extracts on CM female oviposition rate; and (3) the ovicidal activity of non-host plant extracts.
Materials and methods
Insects
CM pupae were obtained in corrugated cardboard strips from the colony maintained at the USDA-APHIS Wapato, WA. Pupae were sorted by sex as described in Peterson (1965) into plastic (32.5 × 32.5 × 32.5 cm) Bugdorm cages (the BugDorm Store, http://bugdorm.megaview.com.tw) and held in Percival growth chambers (Percival Scientific, Perry, IA, USA) under environmental conditions of 24 °C, 60% relative humidity (RH), and 16:8 Light:Dark (L:D) photoperiod until emergence. Emerged moths were provided with 5% sucrose solution dispensed via dental wicking material sticking through the lids of 1.0 oz SOLO portion cups.
Tested materials
Plant extracts tested in behavioral studies are given in Table 1. These plants extracts were prepared in the laboratory as described previously in Gökçe et al. (2005). Plant materials were collected in Tokat, Turkey (43.4°N, 36.5°E), dried at room temperature and subsequently ground into a fine powder. Each plant sample (50 g) was soaked in 500 ml of methanol (Sigma) in a 1000 ml Erlenmeyer flask for 24 h, and then, the suspension was filtered through two layers of cheese cloth. Excess methanol was evaporated in a rotary evaporator, and the resulting residue was eluted with acetone to yield 20% (w/v) plant suspensions. The pear ester [ethyl(E,Z)-2,4-decadienoate] and codlemone [(E,E)-8,10-dodecadien-1-ol] were > 98% isomerically pure (Bedoukian Co, Danbury, CT, USA) and used as a chemical standard in behavioral tests.
Electroantennogram (EAG) recordings
The electroantennogram (EAG) system and test protocols were performed as described previously (Stelinski et al. 2003). A data acquisition interface board (Type IDAC-02) and a universal single-ended probe (Type PRS-1) from Syntech (Hilversum, The Netherlands) were used. The recording and different electrodes were comprised of silver wire in a 10 µl glass micropipette filled with a 0.5 M KCl solution. A computer equipped with an interface card and software (PC-EAG version 2.4) from Syntech was used to record data. EAG cartridges were produced by pipetting 0.25 mg of each plant extract (Table 1) in 25 µl of acetone or 5 mg codlemone and 5 mg pear ester in 25 µl of hexane onto 1.4 × 0.5 cm strips of Whatman No. 1 filter paper. To evaporate the solvent, filter papers were set in a fume hood for 15 min prior to testing. Treated strips were then inserted into disposable glass Pasteur pipettes. Using live insect preparations, 1 ml puffs of air were elicited through EAG cartridges to measure EAG response as maximum amplitude of depolarization.
Male and female codling moths were 2–4 days old when used for electroantennograms. Insects were mounted on a wax-filled, 3.5-cm diameter Petri dish with a clay strip (10 × 3 mm) covering their thorax and abdomen. A recording electrode was positioned above the apex of the antennae while the reference electrode was inserted near the base of the head in close proximity to the antenna. EAGs were recorded for 10 insects of each sex for all chemicals. Filter paper soaked with 20 µl of acetone solvent or hexane was delivered prior as well as after each stimulus presentation as reference control stimulations. For each replicate of moths, two puffs of each volatile treatment and control were applied (12 s apart) to the antenna to produce duplicate depolarization amplitudes. The experiment was performed in a randomized complete block design using chemical odor and CM sex as factors.
Olfactometer
The olfactometer system and test protocols as described by Gökçe et al. (2005) were used in the experiment. Males and females of CM used in this study were 3 days old adults. The sticky liners of pheromone traps (LPD Scenturion Guardpost, Suterra, Bend, OR) were cut to make 55-mm diameter disks for catching the moths. A 20-mm diameter Whatman Number one filter paper disk was placed centrally on top of each 55-mm sticky disk and then transferred into a sterile 90-mm disposable Petri dish prior to insertion in the olfactometer. Twenty-five milligrams of each plant extract was diluted in 250 µl acetone, and then, 25 µl of the plant extract suspension was applied to the central filter paper disk. In the control treatment, 25 µl of acetone was applied to the disk. In addition to these control disks, the CM pheromone and pear ester were also used as a standard. The treated disks were left to dry for 15 min in a fume hood prior to performing the assays. Using clean forceps, the disks (five plant extracts and one control) and rubber septa with pheromone and pear ester were transferred into an eight-arm olfactometer (Gökçe et al. 2005). The wheel olfactometer was connected to a vacuum pump set at 100 mmHg, which pumped clean air through a hydrocarbon filter into the olfactometer.
In the unmated CM experiment, male and female moths were tested separately. Ten-unmated codling moths were inserted into the central release point of the olfactometer and incubated at 24 °C under a 16:8 h L:D photoperiod. Counts of CM in each olfactometer arm were made after 24 h. The experiment was repeated on six different days, and for statistical analysis, the data were blocked by days. In the experiment with mated CM females, moths were allowed to mate for 24 h prior to the experiment. All preparation for extracts and other chemicals and experimental protocol was similar to those described above. After mating, 10 males and 10 females were released into the olfactometer and incubated at 24 °C under 16:8 h L:D photoperiod. When the experiment was completed, the females were examined for their mating status as described in Mantey and White (1975). Only mated females (n = 71) were recorded for statistical analysis.
Wind tunnel
Based on male CM behavioral data in the olfactometer, further bioassays were conducted in a Plexiglas flight tunnel (Stelinski et al. 2004) to evaluate male response to pheromone and pheromone + Xanthium strumarium extract. Male CM (2–3 days old) were collected 30 min prior to assay in aluminum wire mesh cylinder-shaped cages, 8 cm long × 8 cm diameter, with removable plastic Petri dish serving as lids on the open ends. Moths were caged in pairs of the same sex and held in the flight tunnel room maintained at 50–70% RH and 16–18 °C for 30 min to acclimate the conditions. Red rubber sleeve stoppers, (#S0511-Plasticoid) containing 5 mg codlemone and 20 mg plant volatile extract, were placed into the flight tunnel upwind of the cages. Stoppers were pinned 1 cm above a horizontal yellow index secured on the arm of a ring stand 25 cm above the wind tunnel floor. A release cage containing one moth was placed into the airflow at 2 m directly downwind of the stopper. Response of moths was recorded for 3 min until moths stopped movement. Moth behaviors were recorded as either: No Response, Wing Fanning—whereby moths flutter their wings rapidly, Upwind Orientation—flying toward the volatile source, contacting the source, or No Orientation—where the moths either did not respond or flew immediately to the back of the wind tunnel. For each replicate, 10 insects were used, and the whole experiment was repeated three different times for a total of 30 insects per treatment.
Oviposition bioassay
The protocol for testing antioviposition effects of plant extracts was conducted as previously described in Gökçe et al. (2005). One liter bioassay cups 140 mm in height and 110 mm in diameter were used. Four windows (30 × 30 mm), 90° apart around its circumference, were cut into each bioassay cup and covered with fine mesh. Wax paper strips were cut into 50 × 100 mm strips that were wiped with acetone prior to applying plant extracts. One hundred µl of acetone suspended plant extract (20% w/v) was applied to each side of the wax paper strips and spread with a sterile bent glass rod “hockey stick.” In the control treatment, 100 µl of acetone was applied to each side of the wax paper. The treated wax papers were left to dry for 15 m in a fume hood. Each cup contained one acetone-treated wax paper and one plant extract-treated wax paper, arranged 30 mm from the edge of cups and suspended by strings from the top of the cup, as a choice test. A 5% sucrose solution was provided within bioassay cups to act as food sources. Five female and five male CM adults (1–3 days old) were transferred into each bioassay cup. Freshly treated wax paper was replaced daily. The number of individual eggs per wax paper strip was counted for 7 days. A randomized complete block design was used in this study, with each block consisting of four treatment–control cups and one control–control cup, and the entire experiment was replicated six times.
Ovicidal activity of the plant extracts was also quantified in this experiment. The eggs, obtained in antioviposition study, were incubated at 24 °C with 16:8 h L:D photoperiod for 10 days. Hatch rate of eggs both in the treatment and in the control was recorded, and percentage of egg hatch was calculated for each treatment.
Statistical analysis
Analysis of variance (ANOVA) (α = 0.05) was conducted on EAG data, and differences in pairs of means between treatments were separated using Tukey’s multiple comparisons test (α = 0.05) (Minitab Release 16, McKenzie and Goldman 2011). In the olfactometer tests, the number of male or female insects arriving at each treatment arm was expressed as a percentage of the total number of insects tested in each replicate. The resulting preference values for the treatments resulted in 100%. The data were transformed and normalized using arcsine transformation (Zar 1999) and were then analyzed with ANOVA and followed by Tukey’s test to detect difference between treatments. Two-sample t tests (Minitab Release 16) were performed to determine whether moth response to plant extracts and pheromone varied significantly between the sexes.
For the oviposition choice test, the number of eggs counted on each treatment was presented as a percentage. Within replicates, the cumulative number of eggs laid on each treatment was divided by the total number of eggs laid. The resulting preference values for the treatments resulted in 100%. The data were transformed to normalized using arcsine transformation (Zar 1999) and were then subjected to paired t tests (α = 0.05) (Minitab Release 16, McKenzie and Goldman 2005).
The percentage of egg hatch was calculated by dividing the number of hatched eggs by the total number of egg on each treatment. Data were arcsine transformed and subjected to paired t tests (α = 0.05) for detecting difference between the treatment and the control (Minitab Release 16, McKenzie and Goldman 2005). The wind tunnel assay data were recorded as a percentage for each designated behavior, transformed into arcsine, and subjected to t tests (P < 0.05) for differentiating differences between treatments.
Results
Electroantennogram
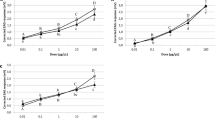
Male and female CM exhibited varying EAG responses to the tested chemicals and the control (Table 2). EAG responses of male CM to pheromones (Table 2) were nearly four times higher (F = 18.63, df = 7.79, P < 0.01) than those of females. However, females showed a significantly higher response (P < 0.01) to pheromone (Table 2) as compared with the control. The chemical standard, pear ester, caused high EAG responses from both sexes of CM as compared with the control; however, there was no significant difference between them. All plants extracts, except Arctium lappa and Humulus lupulus, elicited significantly higher EAG response from female CM than the control (F’s = 27.41, df = 7.79, P < 0.01) (Table 2). The greatest EAG responses from male CM were observed to X. strumarium extract (Table 2), while for female CM, the greatest EAG response was observed with Verbascum songaricum and X. strumarium among the tested plant extracts (Table 2).
Olfactometer study
Behavioral of male CM in the olfactometer differed significantly in response to the tested non-host extracts, pear ester, pheromone, and the control (F = 8.43, df = 7.40, P < 0.01). The greatest percentage of male moths (34.5%) was attracted to the pheromone (codlemone) treatment (Fig. 1). This response was similar to that recorded for the X. strumarium extract (24.8%), and there was no significant difference between these two treatments with respect to the total number of males captured. The Bifora radians extract and pear ester captured 8.4 and 7.5% of males, respectively, but these treatments were not significantly different from response to the control (P > 0.05) (Fig. 1). The fewest moths were captured in H. lupulus, V. songaricum, and A. lappa treatments with 2.7, 2.7, and 4.6% of males captured, respectively (Fig. 1).
The responses of female CM adults between treatments were also statistically different in the olfactometer assays (F = 9.53, df = 7.47, P < 0.01). V. songaricum and A. lappa treatments attracted most moths, capturing an average of 25.4 and 22.9% of the released females, respectively (Fig. 2). About 14.5% of the released moths were captured with the pear ester treatment, and this was followed by B. radians extract with 11% of CM females captured (Fig. 2). These four treatments were significantly different from the control treatment (P < 0.05). CM sex pheromone and X. strumarium captured only 7.2 and 5.6% of female CM, respectively, and these were statistically similar to the control captures.
In the mated CM experiment, although varying numbers of moth were captured among different treatments, there was no significant difference between them (F = 1.52, df = 7. 47, P = 0.19). Pear ester captured the most mated females with 18.4%, and it was followed by V. songaricum extract, which captured 14.9% of the females. Approximately 12.9% of the females were found captured by the A. lappa extract treatment. The other treatment captured varying percentage of females ranging from 7.1% for the sex pheromone to 12.6% for H. lupulus.
Wind tunnel assay
The plant extract, X. strumarium, was further investigated in the wind tunnel assay. There was significant difference between pheromone and pheromone + X. strumarium in all categories, except for non-oriented flight, and it appeared that the plant extract + pheromone caused greater response than pheromone alone (Fig. 3). The increase was clearer in upwind flight and touching on the source categories. Significantly, more moths performed upwind flight (94.9%) in response to pheromone + X. strumarium (t = − 4.89, df = 2, P < 0.05) than in response to pheromone alone (76.3%). Pheromone + X. strumarium also increased contact with the card containing the odor source (86.9%) as compared with pheromone alone (76.2%) (t = − 5.16, df = 2, P < 0.05).
Oviposition bioassay
All tested non-host plant extracts reduced the number of eggs laid by female CM (Fig. 4). The most pronounced effect was observed with H. lupulus extract, and only 10.6% of eggs was laid on the wax paper treated with this extract. This was significantly lower (t = − 9.95, df = 5, P < 0.01) than for the control (89.4%) (Fig. 4). Similar reduction was also observed on wax paper treated with X. strumarium, where 19.6% of deposited eggs was recorded which was significantly lower (t = − 6.20, df = 5, P < 0.01) than in the control (80.4%) (Fig. 3). B. radians (26.3%) and A. lappa (36.3%) treatments caused significant reduction in deposited eggs as compared with the control (P < 0.05). Although wax paper treated with V. songaricum extract contained 39.5% of total laid eggs, this was not statistically different from the control treatment (t = − 1.67, df = 5, P = 0.16) (Fig. 4).
Egg hatch was significantly reduced by the plant extracts (Fig. 5) (P < 0.05). Similar to the oviposition pattern, the lowest egg hatch rate was recorded with H. lupus (21.5%) and X. strumarium (21.9%) extracts. The greatest egg hatch rate was observed with V. songaricum extract (50%). The other two extracts, B. radians (t = − 9.84, df = 5, P < 0.05) and A. lappa (t = − 10.73, df = 5, P < 0.05), also significantly reduced egg hatch rate as compared with the control (Fig. 5).
Discussion
CM is a serious pest of pome fruits and a key pest of apples worldwide. Control strategies primarily rely on insecticide applications and mating disruption with pheromone (Knight 2008; Witzgall et al. 2008, 2010). These technologies generally provide good control of this pest and keep its population below injury thresholds (Witzgall et al. 2008). However, as reported by Trimble (1995) and Witzgall et al. (2008), the use of pheromone for mating disruption is only effective at low population densities of this pest and has failed to control CM at higher population densities. In recent years, plant volatiles have been admixed with sex pheromone (codlemone) in an effort to improve against mating disruption and monitoring of CM (Knight et al. 2005; Schmera and Guerin 2012). Neuroethological investigations have also elucidated that processing of sex pheromone signals and plant volatile cues are coded together in the CM central nervous system (Trona et al. 2013).
Pear esters, β-farnesene, and (E,E)-farnesol are main plant volatiles used to increase the attraction of both male and female CM. The pear ester attracted both male and female CM moths in combined numbers that were comparable to the attractiveness of conspecific sex pheromone. A series of structure–activity tests were conducted in orchard trials to determine CM attraction specificity to the pear ester kairomone (Doughlas and Knight 2005). Admixing pear ester (3 mg) with codlemone (3 mg) was reported to increase male CM captures in monitoring traps (Knight et al. 2005). However, Knight et al. (2012) reported that simultaneously releasing of codlemone and pear ester did not significantly increase capture male CM as compared with codlemone alone in both Washington and Michigan field tests. In the current study, pear ester caused moderate EAG responses from male moth antennae, which were lower than those recorded in response to codlemone and X. strumarium extract. A similar trend with behavioral response was observed in the olfactometer study.
CM pheromone released with X. strumarium extract resulted in greater capture of male CM than pheromone alone. X. strumarium is known to release sesquiterpene lactones (xanthanolides), which are responsible for most of the biological activities associated with Xanthium species (Kamboj and Saluja 2010). X. strumarium fruits contain various bioactive compounds, e.g., 7-hydroxymethyl-8,8-dimethyl-4,8-dihydrobenzol-thiazine-3,5-dione, chlorogenic acid, ferulic acid, and formononetin (Han et al. 2006). High terpenoid and acid contents of X. strumarium fruit extract may explain its possible combined effect with codlemone on CM behavior. Further elucidation and characterization of possible behaviorally active compounds, e.g., antioviposition and attractant from X. strumarium fruit extract, are planned to understand interactions between the extract and codlemone.
Green leaf volatiles play important role in host finding, especially for egg-laying females (Light et al. 1993; Pinero and Dorn 2007). Aldehydes, alcohols, and acetates are the main green leaf volatiles found among most plant species. Their proportion is believed to be key factor for host recognition and selection for herbivores. Generally, plant volatiles are more important in highly specialized insect species than polyphagous species. In the current study, female CM showed preference for V. songaricum in both the olfactometer and the antiovipoisition bioassays. Many saponins, iridoids, phenylethanoid glycosides, monoterpene glucosides, neolignan glucosides, flavonoids, steroids, and spermine alkaloids were isolated and characterized from V. songaricum extracts (Tatli and Akdemir 2004). These green leaf volatiles could play a role in CM female response to V. songaricum extract. This plant extract also elicited EAG responses in both A. velutinana and C. rosaceana and attracted significantly more female C. rosaceana than any other treatments (Gökçe et al. 2005).
Xanthium strumarium and H. lupulus extracts caused high levels of both antioviposition and ovicidal activity against female CM. These results are congruent with previous investigations (Gökçe et al. 2005, 2006), where similar activities of these plant extracts against other lepidopteran moths were reported. Both of these extracts are rich in secondary metabolites especially alkaloids, acids, and terpenoids (Zanoli and Zavatti 2008; Kamboj and Saluja 2010). Repellent and insecticidal effects of these compounds against many insect species were documented in previous studies (Ulubelen et al. 2001; Badgujar et al. 2011). Developing new tools for CM management using non-host volatiles to push female CM away from host trees and also reducing egg deposition and hatch rate could be useful additions to current management programs. X. strumarium and H. lupulus extracts should be further explored by refining these extracts or isolating and characterizing the active compounds. Bioassay-guided characterization with associated chemical identification is underway for elucidation and characterization of active compounds from these plant extracts.
In the current study, the effects of several non-host plant extracts on CM behavior and oviposition were examined. Certain extracts showed potential for modifying CM behavior and possible population reduction. Field studies with the promising extracts are underway, and these will help to better understand the role of non-host plant extracts for management of CM.
References
Badgujar KB, Kasar SS, Nemade NV (2011) Repellent and antifeedant activity of methanolic extract and alkaloidal fraction of Sphaeranthus indicus in stored grain pests Tribolium castaneum (Herbst). Res Artic Budding Res 2:23–26
Beers EH, Brunner JF, Willett MJ, Warner GM (eds) (1993) Orchard pest management. Good Fruit Grower, Yakima
Bender DA, Morrison WP, Frisbie RE (1999) Intercropping cabbage and Indian mustard for potential control of Lepidopterous and other insects. HortScience 34:275–279
Cook SM, Khan ZR, Pickett JA (2007) The use of push–pull strategies in integrated pest management. Annu Rev Entomol 52:375–400
Croft BA, Reidl HW (1992) Chemical control and resistance to insecticides of codling moth. In: van der Geest LPS, Evenhuis HH (eds) World crop pests. Tortricid pests: their biology, natural enemies, and control, vol 5. Elsevier, Amsterdam, pp 371–387
Doughlas ML, Knight A (2005) Specificity of codling moth (Lepidoptera: Tortricidae) for the host plant kairomone, ethyl (2E,4Z)-2,4-decadienoate: field bioassays with pome fruit volatiles, analogue, and isomeric compounds. J Agric Food Chem 53:4046–4053
Dunley JE, Welter SC (2000) Correlated insecticide cross-resistance in azinphosmethyle resistant codling moth (Lepidoptera: Tortricidae). J Econ Entomol 93:955–962
Gökçe A, Stelinski LL, Whalon ME (2005) Behavioral and electrophysiological responses of leafroller moths to selected plant extracts. Environ Entomol 34:1426–1432
Gökçe A, Stelinski LL, Isaacs R, Whalon ME (2006) Behavioural and electrophysiological responses of grape berry moth (Lep: Tortricidae) to selected plant extracts. J Appl Entomol 130:509–514
Gökçe A, Stelinski LL, Whalon ME, Gut LJ (2010) Toxicity and antifeedant activity of selected plant extracts against larval obliquebanded leafroller, Choristoneura rosaceana (Harris). Open Entomol J 4:18–24
Han T, Zhang LH, Zheng H, Qin L (2006) New thiazinediones and other components from Xanthium strumarium. Chem Nat Compd 42:567–570
Kamboj A, Saluja AK (2010) Phytopharmacological review of Xanthium strumarium L. (Cocklebur). Int J Green Pharm 4:129–139
Knight AL (2008) Codling moth areawide IPM. In: Koul O, Cupernus G, Elliott N (eds) Areawide pest management theory and implementation. CAB International, Oxfordshire, pp 159–190
Knight AL, Brunner JF, Alston D (1994) Survey of azinphosmethyl resistance in codling moth in Washington and Utah. J Econ Entomol 87:285–292
Knight AL, Hilton R, Light DM (2005) Monitoring codling moth (Lepidoptera:Tortricidae) in apple with blends of ethyl (E,Z)-2,4-decadienoate and codlemone. J Environ Entomol 34:598–603
Knight AL, Stelinski LL, Hebert V, Gut L, Light D, Brunner J (2012) Evaluation of novel semiochemical dispenser simultaneously releasing pear ester and sex pheromone for mating disruption of codling moth (Lepidoptera:Tortricidae). J Appl Entomol 136:79–86
Landolt PJ, Guedot C (2008) Field attraction of codling moths (Lepidoptera: Tortricidae) to apple and pear fruit, and quantitation of kairomones from attractive fruit. Ann Entomol Soc Am 101:675–681
Light DM, Knight AL (2011) Microencapsulated pear ester enhances insecticide efficacy in walnuts for codling moth (Lepidoptera: Tortricidae) and navel orangeworm (Lepidoptera: Pyralidae). J Econ Entomol 104:1309–1315
Light DM, Flath RA, Buttery RG, Zalom FG, Rice RE, Dickens JC, Jang EB (1993) Host-plant green-leaf volatiles synergize the synthetic sex pheromones of the corn earworm and codling moth (Lepidoptera). Chemoecology 4:145–152
Light DM, Knight AL, Henrick CA, Rajapaska D, Lingren B, Dickens JC, Reynolds KM, Buttery RG, Merrill G, Campbell BC (2001) A pear derived kairomone with pheromonal potency that attracts male and female codling moth, Cydia pomonella (L.). Naturwissenschaften 88:339–342
Mantey KD, White LD (1975) Removal of intact female reproductive organs from codling moths. Entomol Exp Appl 18:393–394
McKenzie JD, Goldman R (2005) The student guide to MINITAB release 14. Pearson Education, Boston
Mitchell VJ, Manning LA, Cole L, Suckling DM, El-Sayed AM (2008) Efficacy of the pear ester as a monitoring tool for codling moth, Cydia pomonella (Lepidoptera: Tortricidae) in New Zealand apple orchards. Pest Manag Sci 64:209–214
Mota-Sanchez D, Wise JC, Poppen RV, Gut LJ, Hollingworth RM (2008) Resistance of codling moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae), larvae in Michigan to insecticides with different modes of action and the impact on field residual activity. Pest Manag Sci 64:881–890
Peterson DM (1965) A quick method for sex determination of codling moth pupae. J Econ Entomol 58:576
Pinero JC, Dorn S (2007) Synergism between aromatic compounds and green leaf volatiles derived from the host plant underlies female attraction in the oriental fruit moth. Entomol Exp Appl 125:185–194
Reuveny H, Cohen E (2004) Resistance of the codling moth, Cydia pomonella (L.) (Lepedoptera:Tortricidae) to pesticides in Israel. J Appl Entomol 128:645–651
Schmera D, Guerin PM (2012) Plant volatile compounds shorten reaction time and enhance attraction of the codling moth (Cydia pomonella) to codlemone. Pest Manag Sci 68:454–461
Schmidt S, Tomasi C, Pasqualini E, Ioriatti C (2008) The biological efficacy of pear ester on the activity of granulosis virus for codling moth. J Pest Sci 81:29–34
Stara J, Kocourek F (2007) Insecticidal resistance and cross-resistance in populations of Cydia pomonella (Lepidoptera: Tortricidae) in Central Europe. J Econ Entomol 100:1587–1595
Stelinski LL, Miller JR, Gut LJ (2003) Presence of long-lasting peripheral adaptation in oblique-banded leafroller, Choristoneura rosaceana and absence of such adaptation in redbandedleafroller, Argyrotaenia velutinana. J Chem Ecol 29:405–423
Stelinski LL, Gut LJ, Vogel KJ, Miller JR (2004) Behaviors of native vs. pheromone-exposed leafroller moths in plumes from high-dosage pheromone dispensers in a sustained-flight wind tunnel: implications for mating disruption of these species. J Insect Behav 17:533–554
Stelinski LL, Gut LJ, Haas M, McGhee P, Epstein D (2007) Evaluation of aerosol devices for simultaneous disruption of sex pheromone communication in Cydia pomonella and Grapholita molesta (Lepidoptera: Tortricidae). J Pest Sci 80:225–233
Tatli II, Akdemir ZS (2004) Chemical constituents of Verbascum L. species. FABAD J Pharm Sci 29:93–107
Trimble RM (1995) Mating disruption for controlling the codling moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae), in organic apple production in southwestern Ontario. Can Entomol 127:493–505
Trona F, Anfora G, Balkenius A, Bengtsson M, Tasin M, Knight A, Janz N, Witzgall P, Ignell R (2013) Neural coding merges sex and habitat chemosensory signals in an insect herbivore. Proc R Soc B 280:20130267. https://doi.org/10.1098/rspb.2013.0267
Ulubelen A, Meriçli AH, Meriçli F, Kilinçer N, Ferizli AG, Emekci M, Pelletier SW (2001) Insect repellent activity of diterpenoid alkaloids. Phytother Res 15:170–171
Vallat A, Dorn S (2005) Changes in volatile emissions from apple trees and associated response of adult female codling moths over the fruit-growing season. J Agric Food Chem 53:4083–4090
Varela LG, Welter SC, Jones VP, Brunner JF, Riedl H (1993) Monitoring and characterization of insecticide resistance in codling moth (Lepidoptera: Tortricidae) in four western states. J Econ Entomol 86:1–10
Vickers RA, Rothschild GHL (1991) Use of sex pheromones for control of codling moth. In: van der Geest LPS, Evenhuis HH (eds) World crop pests. Tortricid pests: their biology, natural enemies, and control, vol 5. Elsevier, Amsterdam, pp 339–354
Witzgall P, Stelinski L, Gut L, Thomson D (2008) Codling moth management and chemical ecology. Annu Rev Entomol 53:503–522
Witzgall P, Kirsch P, Cork A (2010) Sex pheromones and their impact on pest management. J Chem Ecol 36:80–100
Zanoli P, Zavatti M (2008) Pharmocognastic and pharmocological profile of Humulus hupulus L. J Ethnopamocol 116:383–396
Zar JH (1999) Biostatiscal analyses. Prentice-Hall, Englewood Cliffs
Acknowledgements
We would like to thank Bedrettin Selvi for taxonomically identifying the plant species, Juan Huang, Willy Bryan, and others in Department of Entomology for their help during the study. Some of the plant extracts used in the study were obtained from BOPAM project supported by Turkish Republic Prime Minister State Planning Organization (27-DPT-01-07-01).
Author information
Authors and Affiliations
Contributions
AG, LLS, and MEW designed the research. AG, LLS, and MEW conducted the experiments. AG and LLS analyzed the data. AG, LLS, and MEW wrote the manuscript. All authors read and approved the current manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
This manuscript does not contain any studies involving human participants and/or animals.
Additional information
Communicated by M. B. Isman.
Rights and permissions
About this article
Cite this article
Gökçe, A., Stelinski, L.L. & Whalon, M.E. The effects of non-host plant extracts on electroantennogram responses, behavior and egg hatching of codling moth, Cydia pomonella. J Pest Sci 91, 681–690 (2018). https://doi.org/10.1007/s10340-018-0953-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-018-0953-5