Abstract
The Brazilian Merganser is one of the rarest birds of South America with three known remnant populations in Brazil and only 250 individuals existing in the wild. Thus, evaluating the genetic variability within the remaining population is essential to its conservation. This study assessed genetic diversity of remnant populations by mtDNA (Dloop and cytb) and nDNA (MUSK 13 intron, five microsatellites) markers, giving support to management and conservation efforts. Seventy-one samples were collected during eight years from Chapada dos Veadeiros National Park (PNCV), Serra da Canastra National Park (PNSC), and Alto do Paranaíba region (APR). Cytochrome b, Dloop, and MUSK intron showed only one haplotype, indicating a loss of genetic variability for the Brazilian Merganser. The microsatellites confirmed low genetic variability (1–4 alleles). Despite the low variability observed, it was possible to infer minor differentiation between remnant populations, with PNCV locale being the most differentiated subgroup, due to its being isolated by greater distance from the others. Low genetic diversity observed in Brazilian Merganser, is expected to be found in Critically Endangered species, due to small population size, being result of high inbreeding rates, and gene drift effects. Thus, our genetic data have confirmed the extremely critical status of the Brazilian Merganser, making it necessary to create an immediate recovery program for their populations, and develop a management plan to integrate and preserve all remnant populations to avoid the species extinction.
Zusammenfassung
Nachweis eines gravierenden populationsgenetischen Engpasses beim Brasilianischen Dunkelsäger ( Mergus octosetaceus )
Der Brasilianische Dunkelsäger ist mit nur noch drei bekannten Populationen und 250 wildlebenden Individuen einer der seltensten Vögel Südamerikas. Für seinen Erhalt ist es daher unbedingt notwendig, die genetische Variabilität in den verbliebenen Populationen zu bestimmen. Zur Unterstützung der Bemühungen um den Schutz und Erhalt der Art erfassten wir in dieser Untersuchung die genetische Vielfalt der Populationen anhand der mtDNS- (Dloop und cytb) und nDNS- (MUSK 13 intron, fünf Mikrosatelliten) –Markern. Über acht Jahre hinweg wurden 71 Proben im Chapada dos Veadeiros-Nationalpark (PNCV), dem Serra da Canastra-Nationalpark (PNSC) und der Umgebung des Alto do Paranaiba erhoben. Bei Cytochrome b, Dloop und MUSK introns gab es nur einen Haplotyp, was auf einen Verlust an genetischer Bandbreite beim Brasilianischen Dunkelsäger hindeutete. Mit 1 – 4 Allelen bestätigten die Mikrosatelliten eine geringe genetische Variabilität. Dennoch war es möglich, auf eine schwache Differenzierung zwischen den Populationen zu schließen, wobei sich die Population im PNCV, die aufgrund ihrer größten Entfernung zu den anderen am isoliertesten ist, als die am stärksten differenzierte Untergruppe erwies. Diese beim Brasilianischen Dunkelsäger festgestellte geringe genetische Diversität wird als Ergebnis einer hohen Inzuchtrate und genetischer Drift-Effekte wegen der geringen Populationsgrößen vermutlich generell bei extrem bedrohten Arten zu finden sein. Unsere zur Genetik erfassten Daten haben den extrem kritischen Status des Brasilianischen Dunkelsägers bestätigt. Sie machen es notwendig, ein sofortiges Schutzprogramm für ihre Populationen aufzustellen und einen Organisations-Plan zur Einbeziehung und zum Erhalt aller übriggebliebenen Populationen auszuarbeiten, um sie vor dem Aussterben zu bewahren.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endangered species normally have evidence limited population size, leading to suffering from strong effects of inbreeding, and genetic drift (Frankham et al. 2002). Thus, it is expected that these populations lose genetic diversity which affects short- and long-term viability of the wild population. Small population size can be due to habitat fragmentation, hunting pressures, environmental pollution, and disease. Effects of population reduction are proportional to intensity of reduction and the time that population remains small (Luikart et al. 1998; Funk et al. 2010). Genetic bottlenecks can increase the probability of mating among related individuals, consequently increasing frequencies of otherwise rare alleles and causing inbreeding depression, that can decrease individual adaptive potential (fitness) (Funk et al. 2010). Thus, bottlenecks can reduce genetic diversity, leading to concerns about population viability over the long term.
Modern genetic investigation methods allow researchers to evaluate population bottlenecks, detecting changes in Ne (effective number of alleles), and loss of diversity (Funk et al. 2010). Several markers have been used to estimate demographic events in natural populations; these include microsatellites, nuclear introns, and mitochondrial regions. Nuclear DNA (nDNA) and mitochondrial DNA (mtDNA) have distinct inheritance, ploidy, and mutation rates, mtDNA being derived via maternal inheritance, while nDNA is of biparental origin; and effective population size of nDNA is four times higher than mtDNA (Zink and Barrowclough 2008). Thus, it is expected that demographic events that mark the nuclear genome occur more slowly than those of mitochondrial (Solovyeva et al. 2011). However, nuclear loci tend to retain ancestral polymorphism, when they are not subject to selection or drastic population reductions (Warn 2009). On the other hand, microsatellites that also are nuclear markers are characterized by high mutational rates and thus are used in various population studies to recover evidence of recent genetic events and kinship analysis (Grativol et al. 2001; Bristol 2013). For these reasons, inferences based on multiloci approaches demonstrate robust results for recovery of evolutionary history of populations/species (Zink and Barrowclough 2008).
The Brazilian Merganser (Mergus octosetaceus Vieillot, 1817) is considered critically endangered on the Red List of Threatened Species of IUCN (International Union for Conservation of Nature, Birdlife 2016) and the National Official Species of Endangered Fauna (IBAMA 2014), due to having a world population estimated as around 250 individuals and being subjected to continued habitat loss. Historically, Brazilian Mergansers had been recorded in Argentina, Paraguay, and Brazil. However, currently this species occurs only in Brazil, as four remaining relict populations (Silveira and Bartmann 2001; Hughes et al. 2006; Lins et al., 2011): existing in Chapada dos Veadeiros at Goiás State, (Yamashita and Valle 1990; Bianchi et al. 2005); in Jalapão State Park at Tocantins State (Braz et al. 2003; Barbosa and Almeida, 2010), and in the Alto do Paranaíba region and the Serra da Canastra National Park at Minas Gerais State (Bartmann 1988; Lamas 2006).
Only one prior study of Brazilian Mergansers characterized genetic diversity and used exclusively mitochondrial genetic markers (Vilaça et al. 2012). These authors found two distinct lineages with high polymorphism between them at Serra da Canastra population, suggesting that current Brazilian Merganser lineages could be the outcome of an ancient geographic isolation structure which was later erased by second contact. However, there are few cases of high genetic diversity being observed in small endangered populations (Kuro-o et al. 2010). One alternative hypothesis to explain observed Brazilian Merganser genetics is that past bottleneck events could have erased genetic diversity in natural populations, leaving only two common ancestral haplotypes remaining.
In this context, the present study sought to discern demographic history of Brazilian Mergansers covering a great part of the whole species actual distribution, three out of four remnants populations, searching for estimated genetic diversity, gene flow, population structure, and testing the bottleneck hypothesis on the relict populations, through a multilocus study approach.
Methods
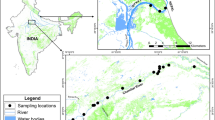
For this study, we used a total of 71 samples from blood, muscles, eggshell, feathers, and skulls of dead animals, that were collected in the period from 2006 to 2014 in three isolated remaining populations of Brazilian Merganser: National Park Chapada dos Veadeiros (PNCV) (n = 08); Alto do Paranaíba region (APR) (n = 08); and National Park Serra da Canastra (PNSC) (n = 55) (Fig. 1). All samples were submitted to DNA genomic extraction by digestion with Proteinase K (20 mg/ml) and posterior purification Phenol:Clorophormio:isoamilic alcohol (25:24:1), and ethanol precipitation protocol (Sambrook et al. 2001). Genomic DNA was quantified using the NanoDrop 2000 (Spectrophotometer Thermo Scientific Uniscience) and diluted to 25 ng/μl.
Control region from mitochondrial DNA was amplified by primers DUCK L81 (5ʹ-TATTTGGYTATGYAYRTCGTGCAT-3ʹ) and DUCK H768 (5ʹ-TATACGCMAACCGTCTCATYGAG-3ʹ) (Muñoz-Fuentes et al. 2005). PCR reactions were performed in final volumes of 10 ul, containing approximately 25 ng of genomic DNA, 2.5 μM MgCl2; 2.5 μM dNTPs; 5 M of each primer; 4 mg/ml BSA; 1 × buffer and 0.5 unit of Taq DNA polymerase tube. The reactions were conducted in an initial denaturation step at 94 °C for 5 min, followed by 35 cycles of denaturation at 95 °C for 30 s, hybridization of primers at 55 °C for 30 s, and chain extension at 72 °C for 45 s, and one final extension step at 72 °C for 7 min. Cytochrome b (Cytb) was amplified by primers L1481 (F: 5ʹ-CCATCCAACATCTCAGCATGATGAAA-3ʹ) and H 160,565 (5ʹ-GTCTTCAGTTTTTGGTTTACAAGAC-3ʹ). (Kocher et al. 1989; Lougheed et al. 2000). PCR reactions were performed in final volumes of 20 μl, containing approximately 25 ng of genomic DNA, 1.5 μM MgCl2; 2.5 μM dNTPs; 5 M of each primer; 4 mg/ml BSA; 1 × buffer, and 0.5 unit of Taq DNA polymerase tube. The reactions were conducted with an initial denaturation step at 94 °C for 5 min, followed by 35 cycles of denaturation at 95 °C for 30 s, hybridization of primers at 55 °C for 30 s, and chain extension at 72 °C for 45 s, and one final extension step at 72 °C for 7 min.
Nuclear intron from skeletal Muscle Gene Receptor tyrosine kinase (MUSK) was amplified by primers MUSK13F (5ʹ-CTTCCATGCACTACAATGGGAAA-3ʹ) and MUSK13R (5ʹ-CTCTGAACATTGTGGATCCTCAA-3ʹ) (Clark and Witt 2006). PCR reactions were performed in final volume of 21 μL, and 50 ng of genomic DNA, 2.5 μM MgCl2; 2.5 uM dNTPs; 0.5 pmol of each primer; 1 × buffer and 0.5 unit of Taq DNA polymerase tube. The thermal program used has an initial denaturation step at 94 °C for 5 min, followed by 35 cycles of denaturation at 95 °C for 30 s, hybridization of primers at 55 °C for 30 s, and chain extension at 72 °C for 45 s and over a final extension step at 72 °C for 7 min. PCR products were cleaned by precipitation using 20% polyethyleneglicol with 2.5 M NaCl (Paithankar and Prasad 1991). The sequencing reactions were performed using standard protocol by BIGDYE v.3.1, performed in automatic sequencer ABI 3130.
Five microsatellite (MOCC3, MOCD4, MOCH3, MOCH5, APH08) were amplified following conditions described for Maia et al. (2017). For genotyping, 1 μl of each amplified product was added to a solution composed 0.25 μl molecular weight marker GeneScan ROX-500 and 7.75 Hi-Di Formamide, performed in an automatic sequencer ABI 3130.
The sequence alignments in SeqScape v5 and haplotype diversity (h) and nucleotide diversity (π) were estimated in DNAsp v.5.10.01 (Librado and Rozas 2009). Microsatellites were checked in GeneMapper software, null allele, and dropout was verified by MICROCHECKER 2.2.3 (Van-Oosterhout et al. 2004). The program version GenAlEx 6501 (Peakall and Smouse, 2006) was used to calculate the allele frequency, number of alleles (Na), effective number of alleles (Ne), expected heterozygosity (He), observed heterozygosity (Ho), the alleles fixation index (F), and the FST pairwise population. In addition to variation in the composition of microsatellites, individuals were evaluated by PCoA (Principal Coordinates Analysis). In addition, microsatellites data were used in the software STRUCTURE program. 2.3.4 (Pritchard et al. 2000) and Structure Harvester (Earl and Vonholdt 2012) to estimate distinct groups inside the populations (delta K) considering ten independent runs were performed with K assumption ranging between 1 and 10, parameters 100,000 interactions of MCMC in a period of burn-in of 10,000 interactions. BOTTLENECK V.1.2 (Cornuet and Luikart 1996) was used to check if there are bottleneck effects of influence on the pattern of genetic variability of populations, under the hypothesis of mutation-drift equilibrium. The assumed mutation models were the model of alleles Infinite (IAM) and Mutation by Step Model (SMM). The distribution of allele frequency (-Shift Mode) was generated to verify the signs of such a bottleneck. The statistical test used to assess the significance of differences was the Wilcoxon sign rank test, to have greater statistical power when used for less than 20 loci, and small number of samples.
Results
Control region DNA fragments were obtained from 58 individuals (579 bp), Cytochrome b from 32 individuals (846 bp), and MUSK (560pb) fragments from 56 samples covering all three regions. MUSK, Cytb, and Control regions showed only one haplotype for all samples that included the three remaining populations of Brazilian Merganser, thereby revealing no genetic variations between them.
The microsatellite loci did not show evidence of null alleles or of drop out to MOCC3, MOCD4, MOCH5, Aph08, MOCH3 to PNCV and Serra do Salitre. However, PNSC samples yielded results which detected an excess of homozygosity for MOCD4, MOCH3 and MOCH5, and Aph08 that may represent the presence of null alleles. Low genetic variability also was observed in microsatellite loci at all populations (Table 1), with low allelic richness (1–4 alleles) and heterozygosity (0.00–0.32). Despite the low genetic diversity observed, it was possible to estimate population genetic differentiation and identify PNCV samples as the most differentiated population (Table 2). However, the observed genetic differentiation among populations in Brazilian Mergansers probably is due to their isolation by distance (Mantel test r = 0.99, p < 0.001). However, STRUCTURE analysis found three distinct clusters (groups), but these clusters were not associated with geographic localization of the samples. Thus, these analyses corroborate the low genetic structure to recovery by F-statistic (Fig. 2). In addition, BOTTLENECK indicated significant evidence of a possible bottleneck for the Serra da Canastra population for MOCD4, MOCH3, and APH08 under SMM.
Discussion
Control region of mitochondrial DNA is considered to be the regions with the highest mutational rate, thus it was expected to find higher genetic diversity there than in nuclear intron (MUSK), but both markers showed only one haplotype. Low genetic diversity such as that observed in Brazilian Mergansers is expected to be found when a population has passed through an intense bottleneck or due to selective sweep on the specific loci (Frankham et al. 2002, Dantas et al. 2012). In addition, microsatellites loci also indicated low genetic diversity, with several loci monomorphic, corroborating the scenario of intense bottleneck on Brazilian Merganser. Our results do not corroborate the prior Brazilian Merganser study, which had reported finding two distinct mitochondrial lineages (cytochrome b and Dloop region) with high genetic differentiation (cytb-20 polymorphic sites and Dloop-24 polymorphic sites) (Villaça et al. 2012). All markers used in deriving our results (Dloop, Cytb and MUSK) revealed only one haplotype and seem to be more realistic for one species with restricted distribution and extreme habitat loss, whose world population is estimated at only 250 individuals (Birdlife 2016). One study on Common Mergansers (Mergus merganser) a species that is widely distributed across the northern hemisphere in both Europe and North America, showed high genetic variability in control region, showed 25 mutational points between haplo-groups (Canadian and Swiss) (Hefti-Gautschi 2009). On the other hand, a study of the Scaly-sided Merganser, M. squamatus, one of the most endangered of known Palearctic species of ducks revealed low nucleotide diversity and haplotype between individuals sampled (Solovyeva et al. 2011). We believe that genetic diversity observed by Villaça et al. (2012) could be the result of nuclear copying or heteroplasmy, that has been reported in other bird species (Gandolfi et al. 2017; Kvist et al 2003; Zhang and Hewitt 1996), since we used the same locations and even re-sequenced some Villaça et al. (2012) samples.
Low genetic diversity has been reported in other threatened species that have gone through population bottlenecks, as in the case of Leontopithecus rosalia, the golden lion tamarin (Grativol et al 2001), Terpsiphone corvina, the Seychelles Paradise Flycatcher (Bristol et al 2013), and Petroica traversi, the black robin (Arden & Lambert 1997). Despite of the weak signal, we recovered on Bottleneck tests for Brazilian Merganser, we suggest stronger historical bottleneck probably occurred in this species since observed loss of genetic diversity in loci from both mitochondrial and nuclear, probably resulted from a demographic event. In addition, the bottleneck test is known to have limitations in detecting slower rates of population declines and work most efficiently to detect catastrophic population crashes (Cornuet and Luikart 1996; Goossens et al. 2006). The effects of population bottlenecks in natural populations are directly related to the intensity (proportion individuals of the population survival) and the event duration time (Kuo and Janzen 2004; Frankham et al. 2008). However, this critical factor can be minimized by increasing the number of markers sampled, it being suggested by Hogan et al. (2013) that using higher than 50 markers, for this genomic approach is fundamental to elucidate evolutionary history of endangered species. One other great problem of loss of genetic diversity is continuing decline of populations, despite protection of its remaining population areas, as observed in the North American spotted owl, Strix occidentalis caurina (Funk et al. 2010). In future, monitoring both population numbers and of genetic diversity of population stocks will be fundamental to conservation efforts of these endangered species. However, it is possible that the northern Brazilian Merganser species is already caught in an extinction vortex.
Despite the low genetic diversity, we observed it was possible to infer differentiation among remnant populations through microsatellites loci. Our outcome indicated that the Alto do Paranaíba region and the Serra da Canastra National Park, distant by about 80 km, showed low genetic differentiation. National Park of Chapada of Veadeiros distant 665 km of PNCV and 550 km to APR evidenced the greatest differentiation. Thus, we recommended that each population receive intense conservation attention and that this population as a whole be managed together as one unit. In addition, it is important to note that one of the remaining populations was not sampled in this study. Thus, the sampling effort in the Tocantins region is extremely important for the consolidation of observed patterns.
We concluded from our study and this genetic analysis context, that the Brazilian Merganser should be considered extremely endangered due to habitat degradation, low population size, and low genetic diversity. Thus, it is extremely important to conserve all remnant populations and execute an integrated management plan, using genetic tools for design of management strategies for breeding and reintroductions into the wild. However, it is essential to continue trying to develop new genetic markers to access evolutionary and demographic history to help retain or increase genetic diversity to help manage and avoid extinction of this species.
References
Arden SL, Lambert DM (1997) Is the black Robin in genetic peril? Mol Ecol 6:21–28
Barbosa MO, Almeida ML (2010) Novas observações e dados reprodutivos do pato-mergulhão Mergus octosetaceus na região do Jalapão, Tocantins, Brasil. Cotinga 32:40–45
Bartmann W (1988) New observations on the Brazilian Merganser. Wildfowl 39:7–14
Bianchi CA, Brant S, Brandão RA, Brito BF (2005) New records of Brazilian Merganser Mergus octosetaceus in the rio das Pedras, Chapada dos Veadeiros, Brazil. Cotinga 24:72–74
BirdLife International (2016) Mergus octosetaceus. The IUCN red list of threatened species 2016: e.T22680482A92863947. Accessed 07 June 2017.
Braz V, Abreu TLS, Lopes LE, Leite LO, França FGR, Vasconcellos MM, Balbino SF (2003) Brazilian Merganser Mergus octosetaceus discovered in Jalapão State Park, Tocantins, Brazil. Cotinga 20:68–71
Bristol RM, Tucker R, Dawson DA, Horsbugh G, Jones RP, Frantz AC, Krupa A, Shah NJ, Burke T, Groombridfe JJ (2013) Comparison of historical bottleneck effects and genetic consequences of re-introduction in a critically endangered island passerine. Mol Ecol 22:4644–4662
Clark WS, Witt CC (2006) First known specimen of a hybrid Buteo: Swainson’s Hawk (Buteo swainsoni) Rough-legged Hawk (B. Lagopus) from Louisiana. Wilson J Ornithol 118:42–52
Dantas GPM, Meyer D, Godinho R, Ferrand N, Morgante JS (2012) Genetic variability in mitochondrial and nuclear genes of Larus dominicanus (Charadriiformes, Laridae) from the Brazilian coast. Genet Mol Biol 35:874–885
Earl D, Vondholdt B (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Cons Genet Res 4:359–361
Frankham R, Ballou JD, Bricoe DA (2008) Introduction to conservation genetic. Cambridge University Press, Cambridge
Frankham R (2002) Population viability analysis. Nature 419:18–19
Funk WC, Forsman ED, Johnson M, Mullins TD, Haig SM (2010) Evidence for recent population bottlenecks in northern spotted owls (Strix occidentalis caurina) Conserv Genet 11:1013–1021
Gandolfi A, Crestanello B, Fagotti A, Simoncelli F, Chiesa S, Girardi M, Giovagnoli E, Marongoni C, Di Roza I, Lucentini L (2017) New evidences of mitochondrial DNA heteroplasmy by putative paternal leakage between the rock partridge (Alectoris graeca) and the Chukar Partridge (Alectoris chukar). PLoS ONE 12(1):e0170507. https://doi.org/10.1371/journal.pone.0170507
Goossens BT, Chikhi L, Ancrenaz M, Lackman-Ancrenaz I, Andau P, Bruford MW (2006) Genetic signature of anthropogenic population collapse in Orangutans. PLoS Biol 4:e25
Grativol AD, Ballou JD, Fleischer RC (2001) Microsatellite variation within and among recently fragmented populations of the golden lion tamarin (Leontopithecus rosalia). Conserv Genet 2:1–9
Hefti-Gautschi B, Pfunder M, Jenni L, Keller V, Ellegren H (2009) Identification of conservation utits in the European Mergus merganser based on nuclear and mitochondrial DNA markers. Conserv Genet 10:87–99
Hogan SM, Gaggiotti OE, Bertorelle G (2012) The number of markers and samples needed for detecting bottlenecks under realistic scenarios, with and without recovery: a simulation-based study. Mol Ecol 22:3444–3450
Hughes B, Dugger B, Cunha H.J, Lamas I, Goerck J, Lins L, Silveira LF, Andrade R, Bruno SF, Rigueira S, Barros YM (2006) Action plan for the conservation of the Brazilian Merganser Mergus octosetaceus. Brasília: Ibama. (Threatened Species Series, 3).
IBAMA (2014) Ministério do Meio Ambiente. Portaria n.444, de 17 de dezembro de 2014. Lista Nacional Oficial de Espécies da Fauna Ameaçadas de Extinção Diário Oficial da União, Brasília, 18 de dez. P 122, 2014
Kocher TD, Thomas W, Meyer A, Edwards SV, Paabo S, VillaBlanca FX, Wilson AC (1989) Dynamics of mitochondrial DNA evolution in animals: amplification and sequencing with conserved primers. Proc Natl A Sci 86:6196–6200
Kou CH, Janzen FJ (2004) Genetic effects of a persistent bottleneck on a natural population of ornate box turtles (Terrapene ornata). Conserv Genet 5:425–437
Kuro-o M, Yonekawa H, Saito S, Eda M, Higuchi H, Koike H, Hasegawa H (2010) Unexpectedly high genetic diversity of mtDNA control region through severe bottleneck in vulnerable albatross Phoebastria albatrus. Conserv Genet 11:127–137
Kvist L, Martens J, Nazarenko AA, Orell M (2003) Paternal Leakage of Mitochondrial DNA in the Great Tit (Parus major). Mol Biol Evol 20:243–247
Lamas IR (2006) Census of Brazilian Merganser Mergus octosetaceus in the region of Serra da Canastra National Park, Brazil, with discussion of its threats and conservation. Bird Conserv Int 16:145–154
Librado P, Rozas J (2009) DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452
Lins VL, Andrade DR, Ribeiro F, Rigueira ES (2011) Distribuição e biologia reprodutiva do pato-mergulhão (Mergus octosetaceus) na região da serra da Canastra. Minas Gerais, MG BIOTA, Belo Horizonte
Lougheed SC, Freeland JR, Handford P, Boag PT (2000) A molecular phylogeny of Warbling-Finches (Poospiza): paraphyly in a Neotropical Emberizid genus. Mol Phylogenet Evol 17:367–378
Luikart G, Allendorf FW, Cournuet J-M, Shewing WB (1998) Distortion of allele frequency distributions provides a test for recent population bottlenecks. J Hered 89:238–247
Maak S, Neumann K, Von Lengerker G, Gattermann R (2000) First seven microsatellites developed for the Peking duck (Anas platyrhynchos). Anim Genet 31:233
Maia TA, Vilaça ST, Silva LR, Santos FR, Dantas GPM (2017) DNA sampling from eggshells and microsatellite genotyping in rare tropical birds: case study on Brazilian Merganser. Genet Mol Biol 40:808–812
Muñoz-Fuente V, Gyllenstrand N, Negro JJ, Green AJ, Sorenson M (2006) Population structure and loss of genetic diversity in the endangered white-headed duck, Oxyura leucocephal. Conserv Genet 6:999–1015
Paithankar KR, Prasad KSN (1991) Precipitation of DNA by polyethylene glycol and ethanol. Nucleic Acids Res 19:1346
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Pritchard JK, Stephens M, Donnely P (2000) Inference of population structure using multilocus genotype data. Genetics 15:945–959
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. CSHL press, New York
Silveira LF, Bartmann W (2001) Natural history and conservation of the Brazilian merganser Mergus octosetaceus at Serra da Canastra National Park, Minas Gerais, Brazil. Bird Conserv Int 11:287–300
Solovyeva D, Pearce J (2011) Comparative mitochondrial genetics of North American and Eurasian merganser with an emphasis on the endangered scaly-sided merganser (Mergus squamatus). Conserv Genet 12:839–844
Van Oosterhout C, Hutchinson WF, Wills D, Shipley P (2004) Micro-checker: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538
Vilaça ST, Redondo RAF, Lins LV, Santos FR (2012) Remaining genetic diversity in Brazilian Merganser (Mergus octosetaceus). Conserv Genet 13:293–298
Yamashita C, Valle MP (1990) Ocorrência de duas aves raras no Brasil Central: Mergus octosetaceus e Tigrisoma fasciatum fasciatum. Ararajuba 1:107–109
Zhang D-X, Hewitt GM (1996) Nuclear integrations: challenges for mitochondrial DNA markers. Tree 11:247–251
Zink RM, Barrowclough GF (2008) Mitochondrial DNA under siege in avian phylogeography. Mol Ecol 17:2107–2121
Acknowledgements
This work was supported by Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG APQ00504-12), Conselho Nacional de Desenvolvimento Cientifico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Petrobras and Fundação de Boticário de Proteção à Natureza. We also thank the Instituto Terra Brasilis for collecting samples. We thank Dr. John M. Pearce (Alaska Science Center, USGS) for kindly sending us the primers for the heterologous loci. The study complies with all current laws of the country. We appreciate the improvements in English usage made by Phil Whitford through the Association of Field Ornithologists' program of editorial assistance.
Author information
Authors and Affiliations
Contributions
TAM, GPM analyzed data and designed the manuscripts. TAM, DPC, LRS developed laboratory procedures, LV, FR, FS given samples to study. All authors contributed to manuscript text.
Corresponding author
Additional information
Communicated by M. Wink.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maia, T.A., Campos, D.P., Silva, L.R. et al. Evidence of strong population bottleneck in genetics of endangered Brazilian Merganser (Mergus octosetaceus). J Ornithol 161, 521–527 (2020). https://doi.org/10.1007/s10336-020-01747-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-020-01747-y