Abstract
Aging is often associated with reduced behavioral performance such as decreased locomotion or food consumption, related to a deterioration in physiological functions. In orb-web spiders, webs are used to capture prey and aging can affect web-building behavior and web structure. Here, we investigated the effect of aging on prey capture in the orb-web spider Zygiella x-notata. The ability of adult females to capture flies was examined at different ages. The rate of prey capture did not change with age, but older spiders took more time to subdue and capture the prey. Alterations which appeared in web structure with age (increase in the number of anomalies affecting radii and capture spiral) affected prey capture behavior. Furthermore, the analysis of individual performance (carried out on 17 spiders at two different ages) showed that older females spent more time handling the prey and finding it in the web. Our results suggest that, in the laboratory, age does not affect prey capture rates but it influences prey capture behavior by affecting web structure or/and spider motor functions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aging is a progressive natural process in which deterioration of physiological functions is often associated with deficits in behavioral performance, which ultimately lead to death (Arking 1998). Many studies on vertebrate models, both in the laboratory and natural populations, have led to a better understanding of aging mechanisms and its consequences for the organism (Austad and Fischer 1991; Holmes and Austad 1995; Perret and Aujard 2006; Ricklefs 2010). In invertebrates, dipterans such as Drosophila melanogaster have been used extensively to study age-related behavioral changes (Grotewiel et al. 2005; Simon et al. 2006; Lliadi and Boulianne 2010; Jones and Grotewiel 2011). Most of the aging studies in flies focused on the decline in behavioral functions linked with age, including memory, olfaction, and biological rhythms (for review, see Grotewiel et al. 2005). Furthermore, several studies have demonstrated a decrease in locomotion with increasing age in insects (Le Bourg and Minois 1999; Ridgel et al. 2003). This loss in mobility could influence an animal’s ability to acquire resources. Food intake is one of the vital functions that decrease with age. In mammals, aging is also associated with declines in food consumption (quantity and quality ingested) (Blanton et al. 1998; McCue 1995). Increasing age has also been shown to be associated with a decrease in foraging efficiency in the honeybee Apis mellifera (Tofilsky 2000) and other invertebrates (Grotewiel et al. 2005; Moya-Larano 2002). However, none of these studies examined the link between locomotor abilities and foraging efficiency in aging animals. Orb-web spiders can be used for this purpose because there is a direct link between locomotor activity and prey capture efficiency (Foelix 2011). In fact, spiders first use their legs (and their whole body) to build a web that will intercept prey and second to capture prey according to a specific behavioral sequence of capture (go to the prey, catch it, immobilize it, and, for some spiders, transport it) (Foelix 2011). Web construction and prey capture are thus two complementary aspects of orb-spider foraging behavior.
Orb-web spiders are sit-and-wait predators that, unlike Lycosidae or Salticidae spiders, do not actively search for prey but instead invest time and energy in the construction of an orb-web, a geometrical structure which serves as a trap for prey capture (Heiling and Herberstein 2000; Scharf et al. 2011). Once the web is built, the spiders wait in the center or hide in a retreat until a prey strikes the structure. Prey capture efficiency depends on the orb-web capacity to (1) intercept prey, (2) inform the spider about the location of the prey, (3) retain prey long enough for the spider to subdue it before it escapes or tumbles away, and (4) on the spider’s capture behavior after prey interception (Coslovsky and Zschokke 2009; Sensenig et al. 2011). These capture abilities depend on silk properties and on the amount of silk invested for web construction (capture thread length, capture area) as well as on the arrangement of the radii and capture threads (Vollrath 1992; Foelix 2011; Harmer et al. 2011). The arrangement of radii and capture threads is particularly important for successful capture because, when the spider hides in a retreat at the edge of the web, silky threads serve to intercept and retain the prey by absorbing the energy of the struggling prey without breaking. Vibrations are also transmitted by radii and a signal thread connecting the center of the web to the retreat (Zschokke 2000). Recently, we demonstrated that aging affects orb-web structure. Webs built by old spiders are smaller, geometrically less regular, and have more anomalies than webs built by younger spiders (Anotaux et al. 2012); we suggested that these web modifications could be due to a decline in spider mobility during web construction and/or in neurological function.
In this study, we used the orb-web spider Zygiella x-notata (Araneae, Araneidae, Clerk) to test the effects of age on foraging efficiency. In this species, spiders build an orb-web and then wait for a prey in a retreat. When a prey strikes the web, the spider captures it by following a characteristic capture behavioral sequence: after approaching the prey, it immobilizes it by biting and wrapping it with silk, then it transports the prey to the retreat to consume it. By comparing prey capture sequences, their duration and the frequency of their different components with age, we expect that (1) the rate of prey capture decreases with aging, (2) the duration of the behavioral acts increases, and (3) the access to prey and prey manipulation to subdue it is more difficult.
Materials and methods
Zygiella x-notata is a widespread medium-sized spider (carapace width for adult females: 1.5 mm) in northern Europe, which builds its web preferentially in the vicinity of human buildings. It constructs an orb-web, which is generally characterized by the presence of a free sector in the upper part (Fig. 1), and feeds primarily on flying prey (generally Diptera). In eastern France, its development cycle is annual: the juveniles leave the eggs sacs at the beginning of spring, reproduction starts in summer with mating, females lay eggs in September–October, and juveniles hatch 3 weeks later and stay in cocoons until spring. While males die after reproduction, the majority of females disappear when winter arrives, but some can survive to the next spring (Jones 1983; Thévenard et al. 2004; Bel-Venner and Venner 2006). Once adult, the lifespan of females is approximately from 5 to 7 months (Juberthie 1954; Thévenard et al. 2004).
The spiders used in this study were captured as subadults (in August and September) and reared in the laboratory in plastic boxes (10 × 7 × 2.5 cm). They were sprayed with water and fed once a week with a fly (Lucilia caesar). They had their last meal 4 days before the tests. All the spiders used for the tests molted and became adults in the laboratory, so their exact adult age was known. They were all virgin females.
Since the body mass of spiders may influence web construction characteristics and varies with age (Venner et al. 2003), spiders were weighed before being placed in experimental conditions (balance: Sartorius BASIC BA110S, precision 0.1 mg).
Procedure
To collect data on prey capture efficiency, spiders were placed individually into wooden frames (50 × 50 × 10 cm) closed by two windowpanes in which they could build their web. Frames were incubated under controlled conditions (temperature 20°–22°, hygrometry 55 %, and luminous cycle of 12 h, light from 0800 to 2000 hours) for 72 h. The spiders were then put back into their respective boxes once they had made a web or after a maximum of 72 h even if they had not constructed a web. The presence of a web was checked every day. As soon as a spider completed a web, the frame was opened and web parameters were directly measured using electronic callipers. Photos (Lumix FZ18 camera) were taken by placing webs in front of a black panel using artificial light.
After web measurements, we carried out a prey capture trial. For this, a living prey was placed in the middle of the lower section of the web (Fig. 1) and then we observed and quantified spider capture behaviors. The prey used was a fly (Lucilia caesar) of smaller size than the spider but that could be considered as a large prey for this spider species (prey size approximately 2/3 of the spider size and with a weight of 25 ± 5 mg). The observations of spider behavior began once the prey became entangled in the web.
Parameters
Web parameters
From direct measurements, we estimated the spider’s investment in the web by calculating the total length of the capture spiral (capture thread length, CTL) (following Venner et al. 2000, 2001 method).
Anomalies
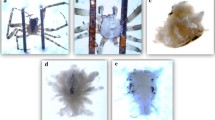
Anomalies in web construction were identified on photos and counted (as defined by Pasquet et al. 2013; Fig. 2). Anomalies can affect the radii or capture spiral, which is the silky area defined by the presence of sticky threads. For radii, we counted: the number of supernumerary, deviated, and “Y” radii (Pasquet et al. 2013; Fig. 2a). For the capture spiral, we counted the number of stops and returns, holes, silk threads of the capture spiral stuck together between two radii, and the number of nonparallel and discontinuous silk threads in the capture spiral (Pasquet et al. 2013; Fig. 2b).
a The different type of radius anomalies defined for Zygiella x-notata orb-webs (from Pasquet et al. 2013). A ‘super-numerary radius’ takes its origin from a spiral thread unit and not from the center of the web; a ‘deviated radius’ presents a deviation >5° compared to a rectilinear trajectory; a ‘Y’ radius is a radius separated abnormally into two radii. b The different types of capture spiral anomalies defined for Zygiella x-notata orb-webs (Pasquet et al. 2013). A ‘return’ is when two spiral units end in one point (triangular ending) or two points (rectangular ending) of a radius, interrupting the spiral; a ‘hole’ is defined by the absence of at least one spiral unit between two adjacent spiral units; ‘two or more spiral units stuck together’ and ‘discontinuity of the spiral’ define anomalies of interruptions in the capture spiral; ‘non-parallel spiral units’ defines a spiral thread stuck to another one on a radius, forming a triangle
Prey capture parameters
Before placing the prey in the web, all spiders were in the same position: they were all in their retreat with a foreleg (L1) in contact with a signal thread (Fig. 1). We took into account the following parameters:
-
Contact latency Its first movement characterizes the first reaction of the spider after the prey is introduced into the web; contact latency is the time between this first movement and the first contact with the prey. In general, the spider reaches the prey following the sequence: retreat, signal thread, center of the web and prey (see Fig. 1).
-
Handling time Handling is a spider behavior (bites and wrappings) used to subdue the prey. Handling time begins after the first contact between the spider and the prey and stops when the spider leaves the capture site with or without the prey.
-
Number of bites A bite is characterized by the introduction of the spider’s fangs into the prey. A spider can bite the prey several times during a capture before it finds a suitable site (i.e. articulation of the prey or specific places with a thinner cuticle). A bite stops when the spider’s fangs leave the prey. The number of bites was noted. There was no way of knowing whether a spider bite was associated with an injection of venom.
-
Number of wrappings Wrapping is a special behavior during which the spider uses silk to immobilize the prey. During this behavior, the silk is extruded from the spinnerets and projected onto the prey; it is visible and easy for the observer to count the number of wrapping sequences. This behavior may be reproduced several times during a capture. For each prey capture, we took into account the number of wrappings.
-
Prey transport time After subduing the prey, the spider then transports it to its retreat before ingestion. To transport the prey back to the retreat, the spider hangs it on its spinnerets. Several events can interrupt the transport on the way back: the prey can become entangled in the sticky spiral and the spider can lose it, or the spider can go the wrong way and not find the best way back to its retreat. The spider might also return to its retreat without the prey and stay motionless before coming back and going on with its previous activity. Each interruption during its return to the retreat was counted, and the total duration of all interruptions was included in the time of transport, which ended when the spider reached its retreat with the prey.
-
Activity time This is the total time during which the spider was actively capturing the prey. This was calculated by adding the contact latency, the handling time, and the transport time.
Statistical analyses
Variation in predatory performance with age could be observed at two different levels: the inter-individual level between spiders of different ages or the intra-individual level by comparing the capture performance of spiders at two different ages.
For the inter-individual analysis, a total of 78 spiders (aged from 17 to 261 days) weighing between 12 and 73 mg were used. Multiple linear regressions, using the ‘lm’ function of the ‘nlme’ package (Pinheiro et al. 2005) for R (v.2.15.0), were performed where (1) age and body mass were the independent variables and web parameter was the dependant variable, and (2) age, body mass, CTL, number of anomalies affecting radii, and number of anomalies of the capture spiral were the independent variables and each prey capture parameter was the dependant variable. In the tables showing the results of multiple linear regression analysis, the multiple regression coefficient “b” referred to a non-standardized coefficient, and “β” to a standardized coefficient.
In the second analysis, we compared the performance of 17 spiders tested at two different ages. The spiders used in this analysis were at least 50 days older between test 1 and test 2 (age of the spiders for test 1: mean = 109 ± 19 days; and age for test 2: mean = 197 ± 33 days). The age of the laboratory-reared spiders in test 1 was similar to that reached by adult females in natural conditions (Bel-Venner and Venner 2006), whereas the age of the spiders in test 2 was much higher than the average age in natural conditions. To take into account repeated measurements for each spider, linear mixed-effects regression models, using the ‘lmer’ function of the ‘lme4’ package (Bates et al. 2013) for R, were carried out where (1) age and body mass were the independent variables, and web parameters were the dependant variables, and (2) age, body mass, CTL, number of anomalies affecting radii, and number of anomalies of the capture spiral were the independent variables, and each prey capture parameter was the dependant variable. Random individual effects accounted for repeated measures. The final restricted maximum-likelihood model, including only significant effects, was achieved by deletion of the non-significant interactions and additive effects from the primary model using the backward stepwise method. F tests of the significance of effects were computed with models derived from the final model. P values for each effect were obtained by likelihood ratio tests of the full model with the effect in question against the model without the effect in question.
A Shapiro–Wilk test was applied to determine whether the sample data were likely to derive from a normally distributed population. Most variables were not normally distributed, thus most prey capture parameters were logarithmically transformed. The normality of model residuals was verified by calculating a Q–Q plot and a Q–Q line. Significance was considered at p < 0.05.
Results
Variation in the predatory performance with age at Inter-individual level
Relationship between age and web properties
All 78 webs constructed by the spiders retained the prey deposited in the lower region of the web. Thus, the rate of prey capture by web retention was 100 %. The two-predictor model was able to account for 20 % of the variance in CTL (F 2,75 = 9.51, p < 0.001, R 2 = 0.20). Body mass had a significant positive coefficient, indicating that spiders with higher body mass are expected to build webs with a longer CTL (Table 1). Spider age had a significant negative coefficient, indicating that older spiders built smaller webs (Fig. 3). Multiple linear regression analysis, with capture spiral anomalies (F 3,69 = 1.19, p = 0.32, R 2 = 0.05) or radius anomalies (F 3,69 = 0.35, p = 0.79, R 2 = 0.02), found that none of the independent variables contributed to the multiple regression models.
Relationship between age and prey capture parameters
Since all spiders successfully caught the prey, the rate of capture due to capture behavior was 100 %. Multiple linear regression analysis was conducted to examine the relationship between prey capture behavior—contact latency, handling time, number of bites, number of wrappings, transport time, and activity time—and five potential predictors—age and spider body mass, CTL, number of capture spiral anomalies, and number of radius anomalies. The best model for predicting the contact latency only included the independent variables: body mass, age, number of radii anomalies, and the interaction between body mass and age (F 4,68 = 2.07, p = 0.09, R 2 = 0.11). Only the number of radius anomalies had a significant positive coefficient, indicating that the spider needed more time to detect the prey when there was a higher number of radii anomalies (Table 2; Fig. 4).
To predict the variance in handling time, only the number of radius anomalies was removed, and the four predictor model was able to account for 30 % of the variance in handling time (F 4,65 = 7.06, p < 0.001, R 2 = 0.30). Body mass and CTL had significant negative coefficient, indicating that spiders with higher body mass or who built webs with a higher CTL were expected to handle prey for shorter periods (Table 3; Fig. 5).
The two predictors used in the best model accounted for 10 % of the variance of the transport time (F 2,74 = 3.90, p = 0.02, R 2 = 0.10). Body mass had a significant negative regression coefficient, indicating that larger spiders spent less time transporting their prey (Table 4).
In contrast, age had a significant positive coefficient, indicating that older spiders transport their prey more slowly (Fig. 6). When multiple linear regression analysis was carried out with bite number and the number of wrappings, none of the independent variables contributed to the multiple regression models.
Only three of the independent variables were used in the best model and these were able to explain 31 % of the variance of the activity time (F 3,74 = 11.06, p < 0.001, R 2 = 0.31). Once again, body mass and CTL had significant negative regression coefficient, indicating that spiders with higher body mass or who made webs with a higher CTL were expected to be active for shorter time periods (Table 5). Age had a significant positive coefficient, suggesting that older spiders were active for longer periods of time (Fig. 7).
Variation in the predatory performance with age at the intra-individual level
Relationship between age and web properties
Linear mixed-effect analysis was used to study the effects of age and body mass on web parameters (CTL, number of capture spiral anomalies and number of radius anomalies) for spiders at two different ages. None of the predictors, however, was able to explain the variance in the dependent variables.
Relationship between age and prey capture properties
Linear mixed-effects regression models showed that there was a significant association between age and handling time (χ 2 = 7.24, p = 0.007), with an increase in handling time of 1.43 ± 1.95 s for each day increase in age. Transport time varied significantly with the number of radius anomalies (χ 2 = 16.40, p = 0.006), increasing with the increase in radius anomalies (log, 0.05 ± 0.04 s for each radius anomalies). Age tended to affect transport time (χ 2 = 3.52, p = 0.06), increasing it by log: 0.07 ± 0.01 s each day. Activity time varied significantly with age (χ 2 = 11.80, p < 0.001) and CTL (χ 2 = 4.03, p = 0.04), increasing for each day increase in age (log, 0.020 ± 0.005 s) and for each increase in CTL of one cm (log, 0.007 ± 0.001 s). However, none of the predictors were able to explain the variance in contact latency.
Discussion
In the present study, we examined the effect of age on the efficiency and behavioral changes in spider foraging. Contrary to our predictions, the rate of prey capture by spiders did not differ with age. The prey (flies) was directly deposited in the web and thus it was perhaps easier for the spiders to capture it than in nature. Our results showed that with age there are some changes in time management during spider capture behavior, and that age-induced changes in web properties also appeared to have an indirect effect on these behaviors.
The consequences of aging on behavioral performance are variable depending on the activity (foraging, reproduction, locomotion; Grotewiel et al. 2005). These are often linked to a decrease in locomotor activity in vertebrates (i.e. in rats; Altun et al. 2007) and invertebrates (i.e. in nematodes; Murakami and Murakami 2005), or in Drosophila (Le Bourg and Minois 1999). In particular, in Drosophila melanogaster, locomotor behavior, such as negative geotaxis, exploratory activity, fast phototaxis, and flight ability, decline with age (Grotewiel et al. 2005). In spiders, it has also been shown that age can influence locomotor behavior, i.e., Lycosa tarantula, (Araneae, Lycosidae), an iteroparous species that reproduces in two successive years, is more productive in the first year than in the second (Moya-Larano 2002). Furthermore, in this latter study, the author showed that, in the second year, females were less active with a lower locomotor activity than the first-year females. For the orb-web spider Zygiella x-notata, it was recently shown that aging affects web geometry, and thus web-building behavior (Anotaux et al. 2012; Toscani et al. 2012). In these papers, the authors also suggested that these age-induced changes in the webs could have a locomotor origin with an associated degradation of the mechanical properties of the spiders’ legs.
In our study, activity time during prey capture increased with age; comparison of prey capture parameters for the same spider at least 50 days later showed that this increase in the activity time was due to an increase in handling time. It is possible that the locomotor behavior used to immobilize the prey becomes more difficult with age because old spiders tire more rapidly during the struggle with prey than younger spiders.
It is known that body mass can lead to variations in web-building behavior (Venner et al. 2003; Kunter et al. 2010) and foraging effort (Venner et al. 2003). When spider body mass increased, the amount of silk used per web decreased, while their foraging effort increased (Venner et al. 2003). As the feeding conditions were uniform for all spiders used in our laboratory experiment, body mass should not have had an effect and cannot explain differences in capture performance. However, in nature, spiders rarely feed to satiation and body mass may play an important role in determining differences in performance in young and old adults.
We also showed that changes in web construction affect foraging effort: the activity time increased and spiders took longer to handle the prey when less silk was used to make a web. Anotaux et al. (2012) suggested that spiders invest less silk in their web with aging because aging affects the glands that produce spiral silk and leads to a reduction in silk reserves. In our study, it appeared that, to compensate for the decrease in silk investment, which occurs with age and can lower the probability of capturing prey by reducing the capture area, spiders took longer to reach the prey to ensure that it did not escape.
Our results showed that the contact latency between the spider and the prey increased with the number of anomalies affecting radii; spiders took more time to detect the prey because radii with anomalies do not correctly transmit prey vibrations, or because spiders are less skilled at moving on radii affected by anomalies. This could also explain why, in the intra-individual approach, transport time was found to increase with the number of anomalies.
We found a significant relationship between mass/age and some web properties at the inter-individual level but not in the second approach based on intra-individual relationships. This difference is probably due to a methodological problem. For the inter-individual comparison, 78 spiders aged from 17 to 261 days were used whereas, in the intra-individual comparison, we used only 17 spiders and the range of ages was reduced (range, 109–197 days) (see “Materials and methods”). Thus in the second case, we reduced the variability in spider age and mass and this could explain the absence of a relationship between these parameters and web properties found in the inter-individual comparison.
Initially, we expected that prey capture efficiency would decrease with age. However, the observed changes in handling behavior and age-induced changes in web geometry did not alter prey capture rates. Nevertheless, the time to complete capture behavior activities did increase with age and access to prey appeared to be more difficult. These findings therefore suggest that, although in the laboratory aging did not have a dramatic impact on spider capture performance, in the wild, these age-induced changes in behavior could affect spider survival due to longer exposure to predators.
References
Altun M, Bergman E, Edstrom E, Johnson H, Ulfhake B (2007) Behavioral impairments of the aging rat. Physiol Behav 92:911–923
Anotaux M, Marchal J, Châline N, Desquilbet L, Leborgne R, Gilbert C, Pasquet A (2012) Ageing alters spider orb-web construction. Anim Behav 84:1113–1121
Arking R (1998) The biology of aging: observations and principles. Sinauer, Sunderland
Austad SN, Fischer KE (1991) Mammalian aging, metabolism, and ecology: evidence from the bats and marsupials. J Gerontol 46:B47–B53
Bates DM, Maechler M, Bolker B (2013) lme4: linear mixed-effects models using S4 classes. R package version 0.999999-2
Bel-Venner MC, Venner S (2006) Mate-guarding strategies and male competitive ability: results from a field study in an orb-weaving spider. Anim Behav 71:1315–1322
Blanton CA, Horwitz BA, Murtagh-Mark C, Gietzen DW, Griffey SM, McDonald RB (1998) Meal patterns associated with the age-related decline in food intake in the Fischer 344 rat. Am Physiol Soc 44(5):R1494–R1502
Coslovsky M, Zschokke S (2009) Asymmetry in orb-webs: an adaptation to web building costs? J Insect Behav 22:29–38
Foelix RF (2011) Biology of spiders, 3rd edn. Oxford University Press, Oxford
Grotewiel MS, Martin I, Bhandari P, Cook-Wiens E (2005) Functional senescence in Drosophila melanogaster. Ageing Res Rev 4:372–397
Harmer AMT, Blackledge TA, Madin JS, Herberstein ME (2011) High-performance spider webs: integrating biomechanics, ecology and behaviour. J R Soc Interface 8:457–471
Heiling AM, Herberstein ME (2000) Interpretations of orb-web variability: a review of past and current ideas. Ekol Bratisl 19:97–106
Holmes DJ, Austad SN (1995) The evolution of avian senescence patterns: implications for understanding primary aging processes. Am Zool 35:307–317
Jones D (1983) Guide des Araignées et des Opilions d’Europe. Delachaux et Niestlé, Paris
Jones M, Grotewiel MS (2011) Drosophila as a model for age-related impairment in locomotor and others behaviors. Exp Gerontol 46(5):320–325
Juberthie C (1954) Sur les cycles biologiques des Araignées. Bull Soc Hist Nat Toulouse 89:299–318
Kunter M, Gregoric M, Li D (2010) Mass predicts web asymmetry in Nephila spiders. Naturwissenschaften 97:1097–1105
Le Bourg E, Minois N (1999) A mild stress, hypergravity exposure, postpones behavioral aging in Drosophila melanogaster. Gerontology 34(2):157–172
Lliadi KG, Boulianne GL (2010) Age-related behavioral changes in Drosophila. Ann N Y Acad Sci 1197:9–18
McCue JD (1995) The naturalness of dying. JAMA 273:1039–1043
Moya-Larano J (2002) Senescence and food limitation in a slowly ageing spider. Funct Ecol 16:734–741
Murakami S, Murakami H (2005) The effects of aging and oxidative stress on learning behavior in C. elegans. Neurobiol Aging 26:899–905
Pasquet A, Marchal J, Anotaux M, Leborgne R (2013) Imperfections in a perfect architecture: the orb web of spiders. Eur J Entomol 110(3):413–500
Perret M, Aujard F (2006) Vieillissement et rythmes biologiques chez les primates. Méd Sci 22:279–283
Pinheiro JC, Bates DM, Debroy S, Sarkar D (2005) Nlme: linear and non linear mixed-effects models. R package version 3.1-83, R foundation for statistical computing, Vienna
Ricklefs RE (2010) Insights from comparative analyses of aging in birds and mammals. Aging Cell 9(2):273–284
Ridgel AL, Ritzmann RE, Scheafer PL (2003) Effects of aging on behaviour and leg kinematics during locomotion in two species of cockroach. J Exp Biol 206:4453–4465
Scharf I, Lubin Y, Ovadia O (2011) Foraging decisions and behavioural flexibility in trap-building predators: a review. Biol Rev Camb Philos Soc 86(3):626–639
Sensenig AT, Agnarsson I, Blackledge TA (2011) Adult spiders use tougher silk: ontogenetic changes in web architecture and silk biomechanics in the orb-weaver spider. J Zool 285:28–38
Simon AF, Liang DT, Krantz DE (2006) Differential decline in behavioral performance of Drosophila melanogaster with age. Mech Ageing Dev 127:647–651
Thévenard L, Leborgne R, Pasquet A (2004) Web-building management in an orb-weaving spider, Zygiella x-notata: influence of prey and conspecifics. C R Biol 327:84–92
Tofilsky A (2000) Senescence and learning in honeybee (Apis mellifera) workers. Acta Neurobiol Exp 60:35–39
Toscani C, Leborgne R, Pasquet A (2012) Analysis of web building anomalies in the orb weaving spider Zygiella x-notata (Araneae, Araneidae). Arachnol Mitt 43:79–83
Venner S, Pasquet A, Leborgne R (2000) Web-building behaviour in the orb weaving spider Zygiella x-notata: influence of experience. Anim Behav 59:603–611
Venner S, Thevenard L, Pasquet A, Leborgne R (2001) Estimation of the web’s capture thread length in orb-weaving spiders: determining the most efficient formula. Ann Entomol Soc Am 94(3):490–496
Venner S, Bel-Venner MC, Pasquet A, Leborgne R (2003) Body-mass-dependant cost of web-building behavior in an orb weaving spider, Zygiella x-notata. Naturwissenschaften 90:269–272
Vollrath F (1992) Analysis and interpretation of orb spider exploration and web-building behavior. Adv Stud Behav 21:147–199
Zschokke S (2000) Form and function of the orb-web. Eur Arachnol 2002:99–106
Acknowledgments
The University of Nancy supported this work with a grant to M. Anotaux. The study is supported by a grant of the CNRS program “Longévité et Vieillissement”. We thank the reviewers for their invaluable comments. We also thank J. Marchal and L. Bahans who helped collect the spiders in 2008 and 2009 and assisted with spider breeding in the laboratory in 2009. Finally, we thank Dr. Leigh Gebbie, LGK Australia, for corrections.
Author information
Authors and Affiliations
Corresponding authors
Additional information
M. Anotaux and C. Toscani equally contributed to this study.
About this article
Cite this article
Anotaux, M., Toscani, C., Leborgne, R. et al. Aging and foraging efficiency in an orb-web spider. J Ethol 32, 155–163 (2014). https://doi.org/10.1007/s10164-014-0404-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10164-014-0404-6