Abstract
Background
Concerns about the adverse effects of excessive oxygen have grown over the years. This study investigated the relationship between high oxygen saturation and short-term prognosis of patients with spontaneous intracerebral hemorrhage (sICH) after liberal use of oxygen.
Methods
This retrospective cohort study collected data from the Medical Information Mart for Intensive Care III (MIMIC-III) database (ICU cohort) and a tertiary stroke center (general ward cohort). The data on pulse oximetry-derived oxygen saturation (SpO2) during the first 24 h in ICU and general wards were respectively extracted.
Results
Overall, 1117 and 372 patients were included in the ICU and general ward cohort, respectively. Among the patients from the ICU cohort, a spoon-shaped association was observed between minimum SpO2 and the risk of in-hospital mortality (non-linear P<0.0001). In comparison with minimum SpO2 of 93–97%, the minimum SpO2>97% was associated with a significantly higher risk of in-hospital mortality after adjustment for confounders. Sensitivity analysis conducted using propensity score matching did not change this significance. The same spoon-shaped association between minimum SpO2 and the risk of in-hospital mortality was also detected for the general ward cohort. In comparison with the group with 95–97% SpO2, the group with SpO2>97% showed a stronger association with, but non-significant risk for, in-hospital mortality after adjustment for confounders. The time-weighted average SpO2>97% was associated significantly with in-hospital mortality in both cohorts.
Conclusion
Higher SpO2 (especially a minimum SpO2>97%) was unrewarding after liberal use of oxygen among patients with sICH and might even be potentially detrimental.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
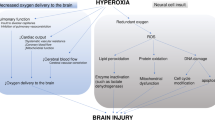
Since the advent of oxygen therapy, liberal oxygen has been routinely administrated for the management of emergent diseases [1, 2]. Concerns about the adverse effects of excessive oxygen, however, have grown over the years. Hyperoxic conditions may contribute to oxidative stress through the production of reactive oxygen species (ROS), which is responsible for the oxidation of lipids, proteins, and nucleic acids and consequently leading to cellular dysfunction or death [3, 4]. The mortality rate was documented to be higher among critically ill patients under a hyperoxic state than in those under a normoxic state, and an optimal value of oxygen saturation or partial pressure of oxygen is still inconclusive [5,6,7,8].
In patients with spontaneous intracerebral hemorrhage (sICH), activated phagocytes, mitochondria, endoplasmic reticulum, and red blood cell (RBC) lysates all cause excess release of ROS, which further exacerbates the inflammatory response, apoptosis, autophagy, and blood-brain barrier (BBB) disruption [9, 10]. Excessive oxygen-mediated generation of ROS can further aggravate oxidative stress and contribute to the progression of secondary brain injury. Hence, it is imperative to carefully determine the endpoint of oxygen therapy in ICH.
While existing studies have reported the potential drawbacks of hyperoxia in stroke, the endpoint of liberal use of oxygen in patients with sICH has not been clearly defined [11, 12]. Previous studies tried to use partial oxygen pressure (PaO2) rather than pulse oximetry-derived oxygen saturation (SpO2) to define oxygenation [13,14,15]. A U-shaped relationship between PaO2 and mortality has been discovered in patients with acute brain injury [15], whereas PaO2 is an invasive assay and cannot be continuously tested, which results in the limited resolution in detecting hyperoxemia or hypoxemia and would potentially restrict the analysis on the correlation of outcomes with time. Herein, we chose SpO2 as an indicator to evaluate oxygenation, given its noninvasiveness, continuity, and proven ability to detect hyperoxia in a neurointensive care unit (detection of PaO2 > 100 mmHg: sensitivity = 0.715 [95% confidence interval, CI = 0.712–0.718]; specificity = 0.700 [95% CI = 0.697–0.703]) [16]. Recent research also probed the association between hyperoxemia (measured by SpO2) and in-hospital mortality in patients with cerebral injury [17]. Albeit no significant nonlinear relationship was detected in the ICH subgroup when SpO2 ranged from 93 to 100%, the adjusted ratio for in-hospital mortality still increased as SpO2 increased. However, the threshold of SpO2 for this subgroup was still left undiscussed.
Based on the preceding background, the aim of our research was to investigate the relationship between high SpO2 and short-term mortality among patients with sICH after liberal oxygen therapy.
Methods
Data source
Two datasets (the ICU dataset and the general ward dataset) were used in our study. The ICU dataset was derived from the publicly available Medical Information Mart for Intensive Care (MIMIC) III database version 1.4, as per the ethical standards of the institutional review board of the Massachusetts Institute of Technology (no. 53032805) [18]. The database gathers information from 2002 to 2011. Hourly physiologic readings from bedside monitors, validated by ICU nurses, were recorded.
The general ward dataset obtained from a local tertiary academic stroke center was approved by the local institutional review boards and covered the time period between February 2018 and February 2023. Hourly physiologic readings from bedside monitors were validated and manually entered into electronic health records by nurses. Data were systematically collected through electronic health records during June 2023. The requirement for consent was waived. This research was conducted in accordance with the Helsinki Declaration.
Study cohorts
This was an exploratory, retrospective cohort study conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
-
(1)
ICU cohort
The inclusion criteria were as follows: (1) diagnosis of sICH based on the International Classification of Diseases (ICD)-9 code; (2) additional oxygen, including invasive ventilation, non-invasive ventilation, high-flow nasal cannula, and other supplementary oxygen therapy such as regular nasal cannula or oxygen mask in the first 24 h in ICU; and (3) first SpO2 at admission > 90% or first PaO2 at admission > 80 mmHg without supplementary oxygen inhalation.
Patients were excluded if they met any of the following exclusion criteria: (1) repeated ICU stays; (2) individuals under 16 years old; (3) patients with medical history that influences respiration and circulation (chronic obstructive pulmonary disease, pulmonary embolism, pulmonary fibrosis, pulmonary hypertension, acute or chronic heart failure, acute myocardial infarction, and dilated cardiomyopathy); (4) failure of circulation at admission, defined as a mean arterial pressure (MAP) < 70 mm Hg, or vasopressor requirement to maintain systolic blood pressure (SBP) > 90 mm Hg or MAP > 70 mm Hg, and duration > 1 h in the first 24 h during ICU stay; and (5) dysfunction of microcirculation, defined as lactic acid > 2.0 mmol/dL at admission.
-
(2)
General ward cohort
Given that regular cannula and oxygen masks dominated the oxygen support in general wards of the local stroke center, we included individuals who received oxygen therapy through a regular nasal cannula or oxygen mask only. Patients receiving invasive ventilation, non-invasive ventilation, or high-flow nasal cannula were excluded from the general ward cohort.
The inclusion criteria were as follows: (1) diagnosis of sICH based on the ICD-9 code; (2) hospitalization in general wards, (3) additional oxygen through a regular nasal cannula or oxygen mask in the first 24 h during a hospital stay, and (4) first SpO2 at admission > 90% or first PaO2 at admission > 80 mmHg without supplementary oxygen inhalation.
Patients were excluded if they met any of these criteria: (1) repeated hospital stays; (2) individuals under 16 years old; (3) patients with a medical history that influences respiration and circulation (chronic obstructive pulmonary disease, pulmonary embolism, pulmonary fibrosis, pulmonary hypertension, acute or chronic heart failure, acute myocardial infarction, and dilated cardiomyopathy); (4) failure of circulation at admission, defined as MAP < 70 mm Hg, or vasopressor requirement to keep SBP > 90 mm Hg or MAP > 70 mm Hg, and duration > 1 h in the first 24 h during the in-hospital stay; (5) dysfunction of microcirculation, defined as lactic acid > 2.0 mmol/dL at admission; and (6) brain herniation at admission.
Data extraction
SpO2 was chosen as our exposure. SpO2 during the first 24 h in ICU or general ward stay was extracted. The minimum and median values of SpO2 were calculated, and we focused on the analysis of minimum SpO2 (The reasons were detailed in Supplementary data 1). Other admission information (age, gender, comorbidities, hemoglobin value, sequential oxygen failure assessment (SOFA) score, acute physiology score III (APSIII), Glasgow coma score (GCS), first arterial blood gas results, vital signs), duration time of oxygen therapy, main practice of oxygen supplement in the first 24 h (invasive positive pressure ventilation [IPPV], non-invasive positive pressure ventilation [NIPPV], high-flow nasal cannula [HFNC], regular nasal cannula, and oxygen mask), length of ICU and hospital stay, any positive culture of pathogen from pulmonary specimen, and surgical procedures were also collected. For patients who received multiple oxygen support therapies in the first 24 h, only the one with the longest duration was recorded. For the general ward cohort, data on the location and volume of hematoma and intraventricular hemorrhage (IVH) were also extracted.
Outcome measurements
The primary outcome was in-hospital mortality, while the secondary outcome was all-cause death and survival time.
Statistical analysis
Variables with > 10% missing data were excluded before imputation. Missing data were otherwise imputed by an iterative imputation method (missForest) based on a random forest [19]. Continuous variables were expressed as mean with standard deviation (SD) when normally distributed or median with interquartile range (IQR) if the distribution was skewed. Categorical variables were presented as event numbers with percentages.
-
(1)
Analysis on the ICU cohort
To investigate the non-linear relationship between SpO2 and in-hospital mortality, a restricted cubic splines (RCS) regression model was employed. A spoon-shaped association was noted through the RCS regression analysis, with five knots observed at the 5th, 27.5th, 50th, 72.5th, and 95th percentiles of minimum and median SpO2. In light of the nonlinear curves, we divided patients into four groups based on minimum SpO2 values as follows: intermediate group (93–97%, the flattest part of the bottom of the curve), high group (>97%), slightly lower group (88–93%), and low group (<88%).
The independent effect of minimum SpO2 on mortality was investigated by adjusting other confounders (including the severity of sICH, the physiological status of patients, and treatment after admission). Baseline characters with P<0.1 in the univariate analysis or factors considered to be associated with mortality by physicians were primarily analyzed. Lasso regression analysis was then performed to eliminate variables with collinearity. Other remaining variables such as GCS, age, gender, APSIII at admission, therapies for ICH (surgery or conservative treatment), and main practices in oxygen support (IPPV, NIPPV or HFNC, nasal cannula or oxygen mask) were included in the multivariate logistic regression model to measure the independent effect of SpO2 on in-hospital mortality. To further focus on the association between hyperoxia and mortality, the time-weighted average SpO2 > 97% (TWA-SpO2>97%) was calculated. The detailed algorithm of TWA-SpO2 is illustrated in Supplementary data 2.. RCS regression and multivariate logistic regression were undertaken to evaluate the associations between hyperoxia exposure (TWA-SpO2>97%) and in-hospital death.
Subgroup analyses were conducted to explore if the spoon-shaped association between minimum SpO2 and in-hospital mortality interacts with other factors such as degree of consciousness (evaluated by GCS of 3–8, 9–12, 13–15), age of patients (classified by approximal quantiles: <60, 60–70, 70–80, >80 years), the main practice of oxygen support (high support: IPPV; moderate support: NIPPV or HFNC; low support: regular nasal cannula or oxygen mask), any positive culture of pathogen from pulmonary specimens, and ICH therapy (surgery or conservative treatment).
To assess the robustness of our results and minimize bias, a sensitivity analysis using propensity score matching (PSM) was performed. The analysis focused on the comparison of high and intermediate SpO2 groups. The results of PSM were incorporated into the final multivariable logistic model. Finally, restricted mean survival time (RMST) analyses were also performed to compare life expectancy differences between the selected SpO2 groups (high and intermediate SpO2 groups) after PSM [20].
-
(2)
Analysis on the general ward cohort
The ICU cohort was the major cohort, given its larger sample size than the general ward cohort. The latter was used for the supplementary analysis of the primary outcome, as its relatively small sample size prevented a comparable subgroup analysis. Univariate and multivariate RCS were undertaken to test the nonlinear relationship between minimum and median SpO2 and in-hospital mortality. Factors such as location of hematoma, volume of hematoma, IVH, GCS, age, gender, and ICH therapy (surgery or conservative treatment) were included in the multivariate logistic regression model.
Depending on the non-linear RCS curves, minimum SpO2 was divided into several groups as follows: intermediate group (95–97%, the flattest part of the bottom of the curve), higher group (>97%), and slightly lower group (<95%). Univariate and multivariate logistic regression models were generated to measure the independent effect of SpO2 on in-hospital mortality. To further focus on the association between hyperoxia and mortality, TWA-SpO2>97% was also calculated. RCS regression and multivariate logistic regression were undertaken to evaluate the associations between hyperoxia exposure (TWA-SpO2>97%) and in-hospital death.
The significance level was set to P<0.05 using the two-sided test. Statistical analyses were performed by R version 4.2.0.
Results
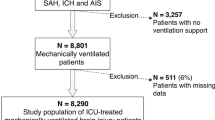
In total, 1367 patients with sICH from the MIMIC III database and 400 individuals from the local stroke center were screened. Following the application of the exclusion criteria, 1117 and 372 patients for the ICU cohort and general ward cohort, respectively, were included in the final analysis (Fig. 1).
Variables with >10% missing data (PaO2, partial pressure of carbon dioxide [PaCO2], PH (potential of hydrogen), base excess, SOFA score, and APS III for the general ward cohort) were excluded from the analysis. For the remaining variables, the missing data were imputed by random forest. In the ICU cohort, the median age of patients was 69 years and the majority of them chose conservative treatment (84.9%) instead of surgical treatment (15.1%). Their median GCS was 8. In the general ward cohort, the median age of patients was 66 years; their median GCS was 12, and the median hematoma volume was 20 mL. The median SpO2 value at admission was 94% and 98% for the ICU and general ward cohort, respectively. The characteristics of the two cohorts are detailed in Tables 1 and 2.
Analysis of the ICU cohort
Primary outcome: SpO2 and in-hospital mortality in the ICU cohort
Among 1117 patients, 406 in-hospital deaths were identified. Variables, including age, hemoglobin, GCS, SOFA score, APSIII, surgical procedure, first blood gas results, SpO2, and main practice in oxygen support, were all significantly associated with in-hospital mortality. Variables with collinearity were eliminated, while other remaining variables such as GCS, age, gender, APSIII at admission, main practices in oxygen support (IPPV, NIPPV or HFNC, nasal cannula or oxygen mask), and ICH therapy (surgery or conservative treatment) were adjusted in further multivariate models.
The roughly nonlinear relationship of minimum SpO2 with in-hospital mortality is descriptively illustrated using the locally weighted scatterplot smoothing (LOWESS) algorithm in Fig. 2A. Multivariate restricted cubic splines regression revealed the spoon-shaped association between the minimum (nonlinear P<0.0001), median SpO2 (nonlinear p=0.0002), and the risk of in-hospital mortality. These results highlight the potential harm of the hyperoxia state (Fig. 2B, C). A minimum SpO2 value of 96% and a median SpO2 of 97% were associated with the lowest odds for in-hospital mortality. We transformed the continuous minimum SpO2 values into four levels as follows: intermediate group (93–97%, the flattest part of the bottom of the curve), high group (>97%), slightly lower group (88–93%), and low group (<88%).
Relationship between SpO2 and in-hospital mortality in the ICU cohort. A. The roughly nonlinear relationship between minimum SpO2 and mortality is descriptively illustrated by the locally weighted scatterplot smoothing (LOWESS) algorithm. B. Spoon-shaped association of minimum SpO2 with in-hospital mortality in the multivariate restricted cubic spline regression model (adjusted to GCS, age, gender, APSIII at admission, practices in oxygen support, and therapy for ICH [surgery or conservative treatment]). The nonlinear p was < 0.0001. The minimum SpO2 value of 96% is associated with the lowest odds for in-hospital mortality. The vertical dashed lines indicate SpO2 values of 88%, 93%, and 97%. The shadowed area represents the 95% confidence intervals for the spline model. OR indicates odds ratio. C. Spoon-shaped association of median SpO2 with in-hospital mortality in the multivariate restricted cubic spline regression model (adjusted to GCS, age, gender, APSIII at admission, practices in oxygen support, and therapy for ICH [surgery or conservative treatment]). The nonlinear p was = 0.0002. The median SpO2 value of 97% is associated with the lowest odds for in-hospital mortality
We measured the relative ORs for in-hospital mortality by performing univariate and multivariate logistic regression analyses. The unadjusted and adjusted ORs for in-hospital mortality were significantly higher for the low SpO2 group (<88%, adjusted OR, 4.41; 95% CI, 2.66–7.45) and high SpO2 group (>97%, OR, 1.54; 95% CI, 1.03–2.29) than for the intermediate group (93–97%) (Fig. 3A, B, Table 3). Further, we focused on the exposure to SpO2>97%, the risk for in-hospital death rises as the TWA-SpO2 increases (Fig. 3C). Each percent increment in TWA-SpO2 was associated with increased odds of death by about 41% (adjusted OR=1.41, 95% CI=1.19–1.68, p=0.005).
Comparison of different SpO2 levels according to in-hospital mortality for the ICU cohort. A. Univariate logistic regression: In comparison with the intermediate SpO2 group (93–97%), the low SpO2 group (<88%, OR, 6.82; 95% CI, 4.56–10.39) and the high SpO2 group (>97%, OR, 2.67; 95% CI, 1.91–3.75) showed significantly higher ORs for in-hospital mortality. B. Multivariate logistic regression conducted after adjusting for GCS, age, gender, APSIII at admission, practices in oxygen support, and ICH therapy (surgery or conservative treatment). Multivariate logistic regression on the four groups of minimum SpO2 also demonstrates the significant higher ORs in the low SpO2 and high SpO2 groups than in the intermediate SpO2 group. C. Multivariate RCS regression conducted after adjusting for GCS, age, gender, APSIII at admission, practices in oxygen support, and ICH therapy (surgery or conservative treatment): when exposed to SpO2>97%, the risk for in-hospital death rises as the TWA-SpO2 increases.
We performed subgroup analyses to investigate if the spoon-shaped association between SpO2 and in-hospital mortality was influenced by other factors such as degree of consciousness (evaluated by GCS of 3–8, 9–12, 13–15), age of patients (classified by approximal quantiles: <60, 60–70, 70–80, >80 years), practice of oxygen support (high support: IPPV; moderate support: NIPPV or HFNC; low support: regular nasal cannula or oxygen mask), any positive culture of pathogen from pulmonary specimen (positive or negative), and ICH therapy (surgery or conservative treatment). The death risk was significantly higher in both low and high SpO2 groups than in the intermediate group considering the subgroups of GCS 3–8, negative pathogen culture from the pulmonary specimen, conservational therapy for sICH, and low oxygen support (Table 4, Fig. 4).
Subgroup analyses to estimate the associations of minimum SpO2 with in-hospital mortality in the ICU cohort. In comparison with the intermediated group, both low and high SpO2 groups had significantly higher death risks considering the subgroups of GCS 3–8, negative pathogen culture from the pulmonary specimen, conservational therapy for sICH, and low oxygen support
Sensitivity analysis and secondary outcome in the ICU cohort
We performed a sensitivity analysis using PSM to assess the robustness of our results and minimize bias. We focused on a comparison of high (>97%) and intermediate (93–97%) SpO2 groups and balanced the between-group baseline characters (Table 5). We added the result of the propensity score to the final multivariable logistic model and still found that the high SpO2 group had a significantly higher risk of in-hospital mortality than the intermediate SpO2 group (OR 1.64, 95% CI 1.05–2.59).
To further clarify the link between high SpO2 and survival time, we conducted PSM and then compared the RMST in the matched cohort at different follow-up times (30, 60, and 90 days). The difference in RMST between the groups, calculated as life expectancy difference (LED), was found to be significantly preponderant in the intermediate SpO2 group at 30- and 90-day follow-up (Table 6).
Analysis in the general ward cohort
Eighty-six in-hospital deaths were identified among 372 patients from the general ward cohort. Confounders, including GCS, age, hemoglobin, therapy for ICH (surgery or conservative treatment), location of hematoma, IVH, and volume of hematoma, were considered as covariates and adjusted in further multivariate models.
The roughly nonlinear relationship between minimum SpO2 and in-hospital mortality in the general ward cohort is descriptively illustrated by LOWESS in Fig. 5A. The multivariate RCS regression analysis results confirmed the spoon-shaped association between minimum SpO2 (nonlinear, P=0.0056), median SpO2 (nonlinear, P=0.0018), and the risk of in-hospital mortality in the general ward cohort (Fig. 5B, C). We found that a minimum SpO2 of 96% and a median SpO2 of 97% were associated with the lowest odds for in-hospital mortality in the multivariate model (Fig. 5B, C). Based on the nonlinear curve of minimum SpO2 values, we divided patients into three groups as follows: intermediate group (95–97%, the flattest part of the bottom of the curve), higher group (>97%), and slightly lower group (<95%).
Relationship between SpO2 and in-hospital mortality in the general ward cohort. A. The roughly nonlinear relationship between minimum SpO2 and mortality is descriptively illustrated by the locally weighted scatterplot smoothing (LOWESS) algorithm. B. Spoon-shaped association between minimum SpO2 and in-hospital mortality in the multivariate restricted cubic spline regression model (adjusted for GCS, age, hemoglobin, therapy for ICH [surgery or conservative treatment], location of hematoma, IVH, and volume of hematoma), nonlinear P = 0.0056. The minimum SpO2 of 96% is associated with the lowest odds for in-hospital mortality. The vertical dashed lines indicate SpO2 values of 95% and 97%. The shadowed area represents the 95% confidence intervals for the spline model. OR indicates the odds ratio. C. Spoon-shaped association between median SpO2 and in-hospital mortality in the multivariate restricted cubic spline regression model (adjusted for GCS, age, hemoglobin, therapy for ICH [surgery or conservative treatment], location of hematoma, IVH, and volume of hematoma), nonlinear P = 0.0018. The median SpO2 of 97% is associated with the lowest odds for in-hospital mortality
We also measured the relative OR for in-hospital mortality by performing univariate and multivariate logistic regression analyses. The unadjusted OR for in-hospital mortality was significantly higher for the slightly low SpO2 group (<95%, OR=4.15, P<0.001) and high SpO2 group (>97%, OR=2.33, P=0.015) than for the intermediate group (95–97%). In the multivariate model, the odds of in-hospital death were still higher in the high SpO2 group, but no statistical significance was observed (Table 7). Whereas in the analysis of TWA-SpO2, the risk for in-hospital death rises as the TWA-SpO2 increases (Supplementary data 3). When exposed to SpO2 > 97%, each percent increment in TWA-SpO2 was associated with increased odds of death by about 11% (adjusted OR=1.11, 95% CI=1.03–1.44, p = 0.035).
Discussion
In this retrospective cohort study, we investigated the relationship between high SpO2 levels and in-hospital mortality and survival outcomes. To this end, we found SpO2 to be associated with in-hospital mortality, and their relationship exhibited a spoon-shaped pattern. An SpO2 value >97% showed no benefits to patients with sICH and was thought to be related to a higher risk of in-hospital mortality.
The deleterious effects of supplemental oxygen are prominently linked to its physiological and biochemical action [4, 22]. Physiologically, oxygen induces hemodynamic changes such as vasoconstriction of the systemic circulation and vasodilation of the pulmonary vasculature. Inhaling a high concentration of oxygen can lead to a replacement of nitrogen in the alveoli. The alveoli collapse as oxygen rapidly diffuses into the blood, resulting in resorptive atelectasis [23]. Biochemically, oxygen inhalation could promote the formation of ROS, including hydroxyl radical and peroxynitrite, which are extremely unstable with unpaired electrons. These molecules frequently react with lipids, proteins, and nucleic acids and contribute to uncontrolled oxidative stress, followed by cellular necrosis or apoptosis [24]. These previous findings serve as a potential theoretical basis for the spoon-shaped pattern of the interaction of SpO2 with mortality observed herein.
While previous preclinical studies have underlined the potential toxicity of excessive oxygen, there has been no consensus on the relationship between high SpO2 and mortality. A large meta-analysis of acutely ill adults reported that supplemental oxygen might become unfavorable above an SpO2 range of 94–96% [7]. The British Thoracic Society issued guidelines on oxygen use and recommended a targeted SpO2 of 94 to 98% in acutely ill patients without type 2 respiratory failure [25]. The same targeted SpO2 range was reiterated by another retrospective study [5]. While the subsequent guidelines recommended an SpO2 < 96% for patients receiving oxygen therapy, a target SpO2 range of 90–94% was deemed reasonable for most patients and 88–92% was advisable for those at risk of hypercapnic respiratory failure [6]. In our analysis, the OR for in-hospital mortality was the lowest when the minimum SpO2 ranged from 93 to 97% in patients with sICH after liberal use of oxygen. Noticeably, the optimal interval in our study was the minimum of SpO2, while a median SpO2 of 97% was associated with the lowest mortality. In terms of the optimal minimum and median SpO2 in our sICH cohorts, the relatively safe SpO2 interval was actually higher than most studies on the general ICU population, which echoes a previous study on acute brain injury [15]. This could be explained by the extreme vulnerability of the brain to hypoxia compared with other organs. What is more, autophagy could be initiated under oxidative stress in the brain, and according to previous studies, activated autophagy can provide antioxidant defense for the brain after stroke, which confers certain tolerance or remediation for stroked brain [26, 27].
While the various target SpO2 values seemed dazzling, the nonlinear dose-dependent relationship between SpO2 and mortality observed herein is consistent with that reported in a previous research [5]. One might infer that the shape and the position of the spoon-shaped curve could be contingent upon situations, leading to the heterogeneity of the targeted SpO2 range. Such presumption could be verified in our subgroup analysis. In the separate analysis of the surgery subgroup, the spoon-shaped association became much flatter and was hardly recognized as compared with that in the conservative treatment subgroup (Fig. 4A). The line graph inferred that patients with sICH undergoing surgery may exhibit better tolerance to high SpO2 than those receiving conservative therapy, consistent with the recent findings on the ROS-mediated aggravation of cerebral edema through the destruction of endothelial cells of the blood-brain barrier (BBB) and downregulation of AQP4 proteins on the astrocytes of BBB [10, 21]. High SpO2 is associated with the potential exacerbation of perihematomal edema (PHE). Surgery can alleviate the occupancy effect of hematoma and PHE and increase the tolerance to hyperoxia. The same alteration in the spoon-shaped relation between minimum SpO2 and mortality was observed in different GCS groups (Fig. 4B). In comparison with individuals with GCS 3–8, those with GCS 9–12 and 13–15 showed no positive correlation between high SpO2 and mortality. Thus, patients with higher GCS scores might suffer from less occupancy effects of hematoma and can be tolerant to PHE exacerbated by excessive ROS.
In the general ward cohort, SpO2 and mortality exhibited a spoon-liked relationship in the RCS regression, whereas the significance of higher death risk in the high SpO2 interval (minimum SpO2 >97% vs minimum SpO2 of 95–97%) was lost in the logistic regression. This observation was different from that in the ICU cohort. The relationship between the exposure to SpO2>97% and in-hospital mortality remains significant in the general ward cohort (adjusted OR for TWA-SpO2=1.11, p=0.035), while the risk seems lower than that in the ICU cohort (adjusted OR for TWA-SpO2=1.41, p=0.005). These discrepancies might be explained by different consciousness statuses in the two cohorts. According to the subgroup analysis of the ICU cohort, the positive correlation between high SpO2 and mortality lost its significance in the subgroups of GCS 9–12 and 13–15 as compared with that in individuals with GCS 3–8. As the median GCS score was 12 in the general ward cohort, the risk of hyperoxia might be weakened for these patients. Still and all, a cumulatively high dose of oxygen exposure might also be dangerous for those patients according to our time-weighted analysis.
The present study has a few limitations. First, we only enrolled individuals with first SpO2 >90% or first PaO2 at admission >80 mmHg without any oxygen support. Our findings may not be generalizable among patients with severe respiratory failure at admission. Second, the time points for our two cohorts differ a lot, which brings about potential time bias. Third, we focused on the analysis of minimum SpO2, which is a single value without consideration of the time that holds at the minimum SpO2. Fourth, due to the origin of the ICU patients (MIMIC-III database), our result might be influenced by many factors such as hematoma volume, location of hematoma, hematoma expansion in the first 24 h, hydrocephalus, intake of anticoagulants, and time between injury and surgery. Fifth, as this was a retrospective cohort study, it could hardly infer causality. Further large-sample prospective research is warranted to verify our viewpoint.
Despite these limitations, our study elucidated the nonlinear spoon-shaped relationship between SpO2 and short-term mortality and further explored the interval related to the lowest risk for death among patients with sICH. These results lay a foundation for future prospective studies and clinical work.
Conclusion
After liberal oxygen use in patients with sICH, higher SpO2 (especially a range of minimum SpO2 >97%) is associated with worse outcomes in the most severe patients according to the level of consciousness (GCS 3–8 points). The potential harm of cumulatively high doses of oxygen exposure should not be neglected in mild or moderate patients in the general ward either.
Data Availability
The source datasets used in this study are available from the following URL: https://figshare.com/s/242dff4a2bbc2f7cc575 or https://doi.org/10.6084/m9.figshare.23614446
Abbreviations
- SpO2 :
-
Pulse oximetry-derived oxygen saturation
- ICH:
-
Intracerebral hemorrhage
- MIMIC-III:
-
Medical Information Mart for Intensive Care III database
- OR:
-
Odds ratio
- CI:
-
Confidence interval
- RCS:
-
Restricted cubic splines
- PSM:
-
Propensity score matching
- FiO2:
-
Fraction of inspires oxygen
- ROS:
-
Reactive oxygen species
- BBB:
-
Blood-brain barrier
- ICU:
-
Intensive care unit
- STROBE:
-
Strengthening the Reporting of Observational Studies in Epidemiology
- SOFA:
-
Sequential Organ Failure Assessment
- APS:
-
Acute Physiology Score
- GCS:
-
Glasgow Coma Scale
- IPPV:
-
Invasive positive pressure ventilation
- NIPPV:
-
Non-invasive positive pressure ventilation
- HFNC:
-
High-flow nasal cannula
- SD:
-
Standard deviation
- IQR:
-
Interquartile range
- RMST:
-
Restricted mean survival time
- LED:
-
Life expectancy difference
- LOWESS:
-
Locally weighted scatterplot smoothing
- PaO2:
-
Partial pressure of oxygen in arterial blood
- HR:
-
Hazard ratio
References
Helmerhorst HJ et al (2014) Self-reported attitudes versus actual practice of oxygen therapy by ICU physicians and nurses. Ann Intensive Care 4:23. https://doi.org/10.1186/s13613-014-0023-y
Albin RJ et al (1992) Pattern of non-ICU inpatient supplemental oxygen utilization in a university hospital. Chest 102(6):1672–1675. https://doi.org/10.1378/chest.102.6.1672
Alva R et al (2022) Oxygen toxicity: cellular mechanisms in normobaric hyperoxia. Cell Biol Toxicol:1–33. https://doi.org/10.1007/s10565-022-09773-7
Nakane M (2020) Biological effects of the oxygen molecule in critically ill patients. J Intensive Care 8(1):95. https://doi.org/10.1186/s40560-020-00505-9
van den Boom W et al (2020) The search for optimal oxygen saturation targets in critically ill patients: observational data from large ICU databases. Chest 157(3):566–573. https://doi.org/10.1016/j.chest.2019.09.015
Siemieniuk RAC et al (2018) Oxygen therapy for acutely ill medical patients: a clinical practice guideline. BMJ 363:k4169. https://doi.org/10.1136/bmj.k4169
Chu DK et al (2018) Mortality and morbidity in acutely ill adults treated with liberal versus conservative oxygen therapy (IOTA): a systematic review and meta-analysis. Lancet 391(10131):1693–1705. https://doi.org/10.1016/s0140-6736(18)30479-3
Girardis M et al (2016) Effect of conservative vs conventional oxygen therapy on mortality among patients in an intensive care unit: the oxygen-ICU randomized clinical trial. JAMA 316(15):1583–1589. https://doi.org/10.1001/jama.2016.11993
Yao Z, Bai Q, Wang G (2021) Mechanisms of oxidative stress and therapeutic targets following intracerebral hemorrhage. Oxid Med Cell Longev 2021:8815441. https://doi.org/10.1155/2021/8815441
Jeon H et al (2021) Upregulation of AQP4 improves blood-brain barrier integrity and perihematomal edema following intracerebral hemorrhage. Neurotherapeutics 18(4):2692–2706. https://doi.org/10.1007/s13311-021-01126-2
Roffe C et al (2017) Effect of routine low-dose oxygen supplementation on death and disability in adults with acute stroke: the stroke oxygen study randomized clinical trial. JAMA 318(12):1125–1135. https://doi.org/10.1001/jama.2017.11463
Rincon F et al (2014) Association between hyperoxia and mortality after stroke: a multicenter cohort study. Crit Care Med 42(2):387–396. https://doi.org/10.1097/CCM.0b013e3182a27732
Fallenius M et al (2016) Association between high arterial oxygen tension and long-term survival after spontaneous intracerebral hemorrhage. Crit Care Med 44(1):180–187. https://doi.org/10.1097/ccm.0000000000001281
Fukuda S et al (2019) Hyperoxemia during the hyperacute phase of aneurysmal subarachnoid hemorrhage is associated with delayed cerebral ischemia and poor outcome: a retrospective observational study. J Neurosurg:1–8. https://doi.org/10.3171/2019.9.Jns19781
Robba C et al (2023) Individualized thresholds of hypoxemia and hyperoxemia and their effect on outcome in acute brain injured patients: a secondary analysis of the ENIO study. Neurocrit Care. https://doi.org/10.1007/s12028-023-01761-x
de Courson H et al (2022) The ability of Oxygen Reserve Index® to detect hyperoxia in critically ill patients. Ann Intensive Care 12(1):40. https://doi.org/10.1186/s13613-022-01012-w
Yin H et al (2022) In-hospital mortality and SpO2 incritical care patients with cerebral injury: data from the MIMIC-IV database. BMC Anesthesiol 22(1):386. https://doi.org/10.1186/s12871-022-01933-w
Johnson AE et al (2016) MIMIC-III, a freely accessible critical care database. Sci Data 3:160035. https://doi.org/10.1038/sdata.2016.35
Stekhoven DJ, Bühlmann P (2012) MissForest--non-parametric missing value imputation for mixed-type data. Bioinformatics 28(1):112–118. https://doi.org/10.1093/bioinformatics/btr597
Dehbi HM, Royston P, Hackshaw A (2017) Life expectancy difference and life expectancy ratio: two measures of treatment effects in randomised trials with non-proportional hazards. Bmj 357:j2250. https://doi.org/10.1136/bmj.j2250
Chan PH et al (1984) Brain injury, edema, and vascular permeability changes induced by oxygen-derived free radicals. Neurology 34(3):315–320. https://doi.org/10.1212/wnl.34.3.315
Thomson L, Paton J (2014) Oxygen toxicity. Paediatr Respir Rev 15(2):120–123. https://doi.org/10.1016/j.prrv.2014.03.003
Aboab J et al (2006) Effect of inspired oxygen fraction on alveolar derecruitment in acute respiratory distress syndrome. Intensive Care Med 32(12):1979–1986. https://doi.org/10.1007/s00134-006-0382-4
Pacher P, Beckman JS, Liaudet L (2007) Nitric oxide and peroxynitrite in health and disease. Physiol Rev 87(1):315–424. https://doi.org/10.1152/physrev.00029.2006
O'Driscoll BR et al (2017) BTS guideline for oxygen use in adults in healthcare and emergency settings. Thorax 72(Suppl 1):ii1–ii90. https://doi.org/10.1136/thoraxjnl-2016-209729
Liu M et al (2022) TIGAR alleviates oxidative stress in brain with extended ischemia via a pentose phosphate pathway-independent manner. Redox Biol 53:102323. https://doi.org/10.1016/j.redox.2022.102323
Liu J et al (2023) ATM-CHK2-TRIM32 axis regulates ATG7 ubiquitination to initiate autophagy under oxidative stress. Cell Rep 42(11):113402. https://doi.org/10.1016/j.celrep.2023.113402
Funding
This study was funded by the Senior Medical Talents Program of Chongqing for Young and Middle-aged.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The conception and design of the study were proposed by Yutong Zhao and Zongyi Xie; data collection and analysis were performed by Yutong Zhao, Ye Yuan, Hai Zhou, Yuguang Tang, and Shuwei Zhang. The first draft of the manuscript was written by Yutong Zhao. The revised manuscript was written by Yutong Zhao and Ye Yuan. The reviewed draft of the manuscript was revised by Zongyi Xie and Ye Yuan. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Yutong Zhao and Ye Yuan contributed equally to this work and should be considered co-first authors.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the ethical standards of the institutional review board of the Massachusetts Institute of Technology (no. 53032805), the local institutional review boards (The National Drug Clinical Trial Institution, clinical study approval number: 2023-6), and the requirement for consent was waived.
Competing interests
All authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 152 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Yt., Yuan, Y., Tang, Yg. et al. The association between high-oxygen saturation and prognosis for intracerebral hemorrhage. Neurosurg Rev 47, 45 (2024). https://doi.org/10.1007/s10143-024-02283-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10143-024-02283-6