Abstract
Preserving cortical frontal bridging veins draining into the superior sagittal sinus is a factor of good neurological outcome in anterior interhemispheric transcallosal approaches, classically performed to reach intraventricular tumors. Challenging the idea that veins are utterly variable, we propose a statistical analysis of 100 selective cerebral angiographies to determine where to place the craniotomy in order to expose the most probable vein-free area. The mean distance to the first pre-coronal vein was 6.66 cm (± 1.73, 1.80 to 13.00) and to the first post-coronal vein 0.94 cm (± 0.92, 0 to 3.00) (p < 0.001). The probability of absence of bridging veins was 92.0% at 4 cm anterior to the coronal suture versus 37.5% at 1 cm and 12.5% at 2 cm posteriorly. The length of the surgical corridor (distance between the first pre-coronal and post-coronal vein) was 7.60 cm (± 1.72, 3.00 to 14.10). Overall, the ideal centering point of the craniotomy was 2.86 cm (± 1.08, − 0.65 to 6.50) ahead of the coronal suture. The mean number of veins within 6 cm behind the coronal suture was 8.47 (± 2.11, from 3 to 15) versus 0.530 (± 0.82, from 0 to 3) ahead of the coronal suture (p < 0.001). These findings support a purely pre-coronal 5 cm craniotomy for interhemispheric approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The anterior interhemispheric transcallosal approach is a very useful technique to resect intraventricular tumors, located in the frontal horn of the lateral ventricle or the anterior part of the third ventricle. The principle of this approach is to reach the ventricles without creating a corticectomy nor interrupting frontal white matter tracts, by following the natural interhemispheric space and using a minimal opening of the anterior part of the body of the corpus callosum. This aims at avoiding neurological deficits, in particular frontal symptoms and epilepsy, and at giving anatomic access to the ventricles, even when they are not dilated. The first obstacles met in this approach are cortical frontal bridging veins that drain into the superior sagittal sinus.
Although sacrificing frontopolar cortical veins is theoretically possible and—fortunately—does not always have relevant clinical consequences [7], it should be avoided: surgical interruption of bridging veins during the approach has been shown to be a significant predictor of major neurological complications in patients operated through the transcallosal approach, both in adults and children [2, 5], and a significant factor of venous infarction, even though brain retraction can also cause it [16].
In this context, performing an adapted craniotomy is the first step towards a safe surgical resection. Most of the recent neurosurgical technique textbooks use the coronal suture as a bony landmark to locate the craniotomy but give heterogeneous advice: it is taught for instance to make the craniotomy halfway anterior and halfway posterior to the suture [6, 11]; or 4 cm anterior, 2 cm posterior to the suture [4]; 2/3 anterior, 1/3 posterior [14]; or 3–6 cm anterior, 1 cm posterior [1, 10].
Probably because of the idea that venous anatomy is highly variable, anatomical studies based on cadavers or imaging have been limited to small samples of patients, privileging meticulous description rather than statistically reproducible patterns [3]. They mostly describe the location and dimension of veins in relation to the brain parenchyma. Using CT scan and arteriography, we chose to describe these veins in relation to the coronal suture, in order to get a schematic picture of the veins repartition in this area and to establish the most reproducible safe entry zone, if it exists.
Materials and methods
Data acquisition
One hundred selective cerebral angiographies free from any venous abnormalities and performed in patients who did not undergo cranial neurosurgery were extracted from the local anonymous database. Analyses of the angiographies with systematic concomitant CT scan were then performed to provide a description of the venous anatomy (SA & PB): number of veins in the zone extending 6 cm behind and 6 cm in front of the coronal suture; distance between the coronal suture and the first vein anteriorly; distance between the coronal suture and the first vein posteriorly. The coronal suture was visualized on CT scan and distances were measured by drawing a curved line on the skull. These analyses were performed separately for both sides of the sagittal sinus.
All patients gave their approval for the anonymous use of their selective cerebral angiography for research, according to the French law (n° 2012-300).
Data analysis and statistics
The distance between the first vein ahead and behind the coronal was analyzed through a survival analysis using Kaplan-Meier curves, the event being defined by the presence of a vein and the time replaced by the geometric distance from the coronal suture measured on the cerebral angiography. The comparison of the survival distributions of the two samples (pre- and post-coronal) was performed by using a logrank test. This comparison was done for both sides and the overall data separately.
The length of the surgical corridor for each patient was defined as the distance between the first pre-coronal and the first post-coronal vein. The ideal centering of the craniotomy was the middle of the surgical corridor. The left and right length of the surgical corridor was compared by using Welch’s t test, as well as the left and right relative position of the ideal centering of the craniotomy in relation to the coronal suture.
All statistical analyses were performed with Matlab© R2017a v9.2.0.556344 (Copyright © 1984-2017, The MathWorks©, Natick, Massachusetts, USA) and Medistica. pvalue.io, a Graphic User Interface to the R statistical analysis software for scientific medical publications 2019. Figures and artworks were performed with Glimpse Image Editor © 0.1.2.
Results
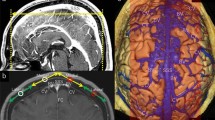
Among the 100 selective cerebral angiographies analyzed, the mean distance to the first pre-coronal vein was significantly higher than the one to the first post-coronal vein (p < 0.001). The mean distance to the first pre-coronal vein was 6.67 cm (± 1.77, from 2.00 to 13.00) on the left-hand side and 6.66 cm (± 1.73, from 1.80 to 13.00) on the right-hand side when the distance to the first post-coronal vein was 0.95 cm (± 1.08, from 0 to 5.80) on the left-hand side and 0.94 cm (±0.92, from 0 to 3.00) on the right-hand side. The difference between left and right was not significant. It is worth noticing that while the probability of the absence of bridging veins is 37.5% at 1 cm and 12.5% at 2 cm posteriorly to the coronal suture, this probability is 92.0% at 4 cm anteriorly to the coronal suture (Fig. 1).
On the top corner left: representation of the distribution of the probability of vein free approach (blue curve) and of the distribution of the position of the ideal centering of the craniotomy (red curve). The ideal centering of the craniotomy is marked by a black dot; on the bottom left: example of one of the 100 selective cerebral angiographies used for the study, with MRI fusion available in this particular case, showing the relation to the corpus callosum and deep anatomical structures; on the right, Kaplan-Meier curves representing the absence of bridging veins according to the distance from the coronal suture
The mean length of the surgical corridor (defined by the distance between the first pre-coronal and the first post-coronal vein) was not significantly different (p = 0.95) between the left-hand side 7.62 cm (± 2.17, from 2.40 to 14.70) and the right-hand side 7.60 cm (± 1.72, from 3.00 to 14.10). Unsurprisingly, the ideal centering point of the craniotomy (defined as the center of the vein-free surgical corridor) in relation to the coronal suture was not significantly different (p = 0.99) between the left-hand side 2.86 cm (± 0.99, from 2.34 to 3.51) and the right-hand side 2.86 cm (± 1.08, from − 0. 65 to 6.50).
The mean number of veins (left and right) within 6 cm behind the coronal suture was 8.47 (± 2.11, from 3 to 15) and was significantly higher than the mean number of veins within 6 cm ahead of the coronal suture, 0.530 (± 0.82, from 0 to 3) (p < 0.001).
Discussion
Frontal cortical bridging veins drain not only the convexity of the frontal lobe but also the medial part in 47% of cases, and the basal surface in 21% [13]. Their interruption can lead to severe venous infarction or hemorrhage, with permanent or temporary neurological consequences in up to 44.5% of patients, and it is a significant predictor of increased incidence of major neurological complications in patients operated through this approach, both in adults and children [2, 5]. Consequently, their surgical sacrifice should be avoided whenever possible, making it crucial to describe and use a vein-free surgical corridor.
Anatomical cadaveric descriptions of frontal bridging veins have calculated the average distance between the fontal pole, defined as the most anterior point of the frontal lobe, and the bridging veins draining into the sagittal sinus. They include the frontopolar vein, located 2.5 cm (from 1.2 to 5.0) to 3.11 cm of the frontal pole [9, 13], the anterior frontal one (6.8 cm, from 3.2 to 9.6), the middle one (8.3 cm, from 4.0 to 11.8), and the posterior one (10.5 cm, from 9.0 to 12.5). The surgical corridor devoid of tributaries is located either between the anterior and the middle frontal vein or between the middle and the posterior one [9], which is congruent with our findings. However, the frontal pole cannot be used as a surgical landmark to predict the position of the veins.
Bony landmarks are the most reliable references neurosurgeons have per-operatively in this context, and, in the case of intraventricular lesions, the physiological anatomy of cortical veins is unchanged, by the opposition for instance to meningiomas that modify the local superficial vasculature. We found that the coronal suture can be used as a very reliable landmark to position the craniotomy for an interhemispheric approach. While an extension of the craniotomy posterior to the coronal figure is frequently recommended in textbooks, the analysis of the anatomy of bridging veins reveals that a 1-cm extension is unusable in 62.5% of patients and a 2-cm extension in 87.5% of patients, making it controversial to perform such post-coronal extension (Fig. 2). Conversely, the probability of the presence of a bridging vein within the 4 cm anterior to the coronal suture is low (8.6%) and remains acceptable within 6 cm (22.5%). These findings encourage to propose to perform a purely pre-coronal craniotomy of 5 cm for the interhemispheric approach. In case a bridging vein is exposed by the craniotomy, it is also more acceptable to sacrifice a bridging vein that drains into the anterior third of the superior sagittal sinus, which is thought to have less function than the middle or posterior third of the sinus.
Examples of craniotomies for interhemispheric approach. The crosshatched zones correspond to the region having a high probability to be inaccessible due to bridging veins. a Craniotomy anterior to the coronal suture, centered 2.5 cm ahead. The proposition made by the authors. b Craniotomy two-thirds anterior to the coronal suture [7, 8]. c Craniotomy 1 cm posterior to the coronal suture and 6 cm ahead [9, 10]. d Craniotomy with the coronal suture at midway [5, 6]. To access the intracranial area exposed by the part of the craniotomy posterior to the coronal suture, the neurosurgeon will have to sacrifice bridging veins in 9.5% in case a, in 62.5% of patients in case c, and in 87.5% of patients in cases b and d
This study does not replace a thorough preliminary analysis of venous anatomy on preoperative imaging, MRI, or CT scan with contrast. It aims at giving reliable landmarks that can be used in the majority of cases when no vascular particularity is observed. If a major vein is visible on the anterior frontal lobe and will presumably hinder the interhemispheric approach, a trans-frontal approach should be considered. If an interhemispheric approach is still performed, dissecting the bridging vein from the cortex is usually possible since the bridging segment, which includes the subarachnoid and subdural segments, is rarely less than 1.6 cm [13]. This will help minimizing the risk of thrombosis since retracting the veins leads to minimal damage compared to coagulation [12]. Some authors also propose to cut the vein from the sinus and anastomose it to a more posterior venous drainage, giving a large interhemispheric access [8, 15]. This approach is nevertheless technically demanding and its safety remains to be proven.
References
Apuzzo MLJ (1993) Brain surgery: complication avoidance and management. Churchill Livingstone
Aryan HE, Ozgur BM, Jandial R, Levy ML (2006) Complications of interhemispheric transcallosal approach in children: review of 15 years experience. Clin Neurol Neurosurg 108:790–793. https://doi.org/10.1016/j.clineuro.2005.10.009
Brockmann C, Kunze SC, Schmiedek P, Groden C, Scharf J (2012) Variations of the superior sagittal sinus and bridging veins in human dissections and computed tomography venography. Clin Imaging 36:85–89. https://doi.org/10.1016/j.clinimag.2011.05.003
Greenberg MS Handbook of neurosurgery
Hassaneen W, Suki D, Salaskar AL, Levine NB, DeMonte F, Lang FF, McCutcheon IE, Dorai Z, Feiz-Erfan I, Wildrick DM, Sawaya R (2010) Immediate morbidity and mortality associated with transcallosal resection of tumors of the third ventricle. J Clin Neurosci 17:830–836. https://doi.org/10.1016/j.jocn.2009.12.007
Hernesniemi J, Romani R, Dashti R, Albayrak BS, Savolainen S, Ramsey C, Karatas A, Lehto H, Navratil O, Niemelä M (2008) Microsurgical treatment of third ventricular colloid cysts by interhemispheric far lateral transcallosal approach-experience of 134 patients. Surg Neurol 69:447–453. https://doi.org/10.1016/j.surneu.2007.11.005
McNatt SA, Sosa IJ, Krieger MD, McComb JG (2011) Incidence of venous infarction after sacrificing middle-third superior sagittal sinus cortical bridging veins in a pediatric population: clinical article. J Neurosurg Pediatr 7:224–228. https://doi.org/10.3171/2010.11.PEDS09261
Ohara K, Inoue T, Ono H, Kiyofuji S, Tamura A, Saito I (2017) Technique for rerouting a bridging vein that hinders the anterior interhemispheric approach: a technical note. Acta Neurochir (Wien) 159:1913–1918. https://doi.org/10.1007/s00701-017-3285-y
Oka K, Rhoton AL, Barry M, Rodriguez R (1985) Microsurgical anatomy of the superficial veins of the cerebrum. Neurosurgery 17:711–748. https://doi.org/10.1227/00006123-198511000-00003
Quiñones-Hinojosa A (2012) Schmidek and sweet operative neurosurgical techniques: indications, methods, and results: Sixth Edition. Elsevier Inc.
Rhoton AL, Yamamoto I, Peace DA (1981) Microsurgery of the third ventricle: Part II. Operative approaches. Neurosurgery 8:357–373. https://doi.org/10.1227/00006123-198103000-00007
Sakaki T, Kakizaki T, Takeshima T, Miyamoto K, Tsujimoto S (1995) Importance of prevention of intravenous thrombosis and preservation of the venous collateral flow in bridging vein injury during surgery: an experimental study. Surg Neurol 44:158–162. https://doi.org/10.1016/0090-3019(95)00160-3
Sampei T, Yasui N, Okudera T, Fukasawa H (1996) Anatomic study of anterior frontal cortical bridging veins with special reference to the frontopolar vein. Neurosurgery 38:971–975. https://doi.org/10.1097/00006123-199605000-00024
Sindou M (2009) Practical handbook of neurosurgery: from leading neurosurgeons. Springer-Verlag
Sindou MP, Auque J, Jouanneau E (2005) Neurosurgery and the intracranial venous system. Acta Neurochir Suppl 94:167–175. https://doi.org/10.1007/3-211-27911-3_27
Tsutsumi K, Shiokawa Y, Sakai T, Aoki N, Kubota M, Saito I (1991) Venous infarction following the interhemispheric approach in patients with acute subarachnoid hemorrhage. J Neurosurg 74:715–719. https://doi.org/10.3171/jns.1991.74.5.0715
Author information
Authors and Affiliations
Contributions
S.A. performed the analysis, drafted the manuscript, and approved the final version of the manuscript.
C.A. performed the analysis, drafted the manuscript, and approved the final version of the manuscript.
D.C. critically reviewed the manuscript and approved the final version of the manuscript.
C.L.G. critically reviewed the manuscript and approved the final version of the manuscript.
P.B. designed the study, performed the analysis, drafted the manuscript, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Local committee approval.
Informed consent
Use of anonymized data, with patients’ consents
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aldea, S., Apra, C., Chauvet, D. et al. Interhemispheric transcallosal approach: going further based on the vascular anatomy. Neurosurg Rev 44, 2831–2835 (2021). https://doi.org/10.1007/s10143-021-01480-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-021-01480-x