Abstract
Fusiform intracranial aneurysms (FIA) are associated with significant morbidity and mortality. We carried out a systematic review and meta-analysis of individual participant data with propensity score adjustment to compare the functional and angiographic outcomes between surgical and endovascular approaches to FIA. We conducted a systematic review for articles on the treatment of FIA with individual patient-level detailing. Data from patients treated for FIA in our institution from 2010 to 2018 were also collected. The primary studied outcome was morbidity, and secondary outcomes were angiographic results and retreatment. Propensity score–adjusted mixed-effects logistic regression models evaluated treatment options, stratified by anatomical location. Compiling original and published data, there were 312 cases, of which 79 (25.3%) had open surgery, and 233 (74.5%) were treated with endovascular procedures. There were no differences between treatment groups, for neither cavernous ICA (OR 1.04, 95% CI 0.05–23.6) nor supraclinoid aneurysms (OR 7.82, 95% CI 0.65–94.4). Both size (OR 1.11, 95% CI 1.03–1.19) and initial mRS (OR 2.0, 95% CI 1.2–3.3) were risk factors for morbidity, independent of location. Neither age nor rupture status influenced the odds of posterior morbidity. Unfavorable angiographic outcomes were more common in the endovascular group for supraclinoid and vertebrobasilar aneurysms (χ2, P < 0.01). There were no differences between morbidity of surgical and endovascular treatments for FIA, regardless of aneurysm location. Size and initial mRS were correlated with functional outcomes, whereas age and rupture status were not. Microsurgery seems to yield better long-term angiographic results compared to endovascular procedures.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fusiform intracranial aneurysms (FIA) are circumferential arterial dilatations 1.5 times the normal diameter of an arterial segment with any degree of tortuosity, without a definable neck. [9, 36] Significant morbidity and mortality are attributable to those spindle-shaped aneurysms, which have exhibited annual growth rates of 12% and a 3% annual rupture risk. [12, 13, 36, 37] In a landmark study on the topic, Mizutani and colleagues reported a mortality rate of 35.3%. [30]
The origin and development of FIA remain subject to debate. There are probably many different mechanisms of formation, as histopathological analyses have proven. [6, 15, 21, 30, 35] However, as stressed by Horie and colleagues, [15] the term fusiform refers mainly to a specific morphology, since pathological examination is not possible preoperatively. Multiple treatment strategies for FIA exist, which are often classified as deconstructive or reconstructive, both presenting microsurgical and endovascular choices.
The literature on this topic is composed mostly of case reports and small series [40], and few studies to date directly compare different treatment options. We performed a systematic review and meta-analysis of individual participant data, including original data from our institution, to compare the functional and angiographic outcomes between surgical and endovascular approaches to FIA.
Methods
Literature search
We conducted a systematic review based on the PRISMA-IPD guidelines [22, 31], searching the Medline database for articles published on the treatment of FIA. Last date of search was March 3, 2020. The PICOS components of the research question we sought to answer were (P) patients with FIA, (I) neurosurgery, (C) endovascular treatments, (O) long-term functional and angiographic outcomes, and (S) any study design presenting individual patient data. The search strategy combined the terms “fusiform,” “intracranial,” “cerebral,” “vertebrobasilar,” and “aneurysm,” in both “AND” and “OR” combinations, in the “all fields” modality. The term “vertebrobasilar” was included due to the higher prevalence of fusiform morphology in this specific location. No time or language restrictions were applied, and additional articles were identified by hand searching the articles’ references.
Study selection
Abstracts were screened and selected for full article consideration according to the following inclusion criteria: studies of intracranial aneurysms in humans detailed at patient-level describing clinical outcomes in terms of performance scales (e.g., Modified Rankin Scale, Glasgow Outcome Scale). To optimize the number of patients included, we sought not only for studies exclusively about FIAs but also papers on intracranial aneurysms in general that included at least five patients with FIAs.
Studies were excluded if not fulfilling all the inclusion criteria, written in languages other than English, patients who had both endovascular and surgical treatments, single case reports or technical notes, or if the number of subjects with fusiform aneurysms < 5. All pediatric patients (< 18 years old) were excluded from the analysis, regardless of studies being focused exclusively on this population or not. Two independent authors retrieved the data, and disagreements were solved with a senior author’s opinion.
Data extraction
Abstracted data included age (in years), gender, location of the aneurysm, size (defined as the largest diameter), rupture status, treatment modality (surgical or endovascular), and conflict of interest in the study. For cases in which age was reported, for example, as “early 40s,” “mid 40s,” or “late 40s,” these were considered 40, 45, and 50, respectively.
The primary studied outcome was morbidity, defined as mRS worsening compared to pretreatment status. For unknown baseline assessment, mRS worsening was defined as final mRS > 1. Functional status pre and postoperatively was converted to modified Rankin Scale (mRS) whenever possible.
Secondary outcomes were long-term angiographic results and retreatment. Long-term angiographic outcomes (> 3 months) were coded as Favorable (complete occlusion, aneurysm obliteration, bypass patent, stable wrapping) or Unfavorable (recanalization, residual aneurysm, incomplete occlusion, bypass occluded).
Original data
Data from patients treated for FIA in our institution from 2010 to 2018, both surgically or endovascularly, were also collected according to the same specifications above. Medical records were retrospectively analyzed, including admissions and outpatient visits. The final mRS refers to the latest available consultation, which was > 6 months for all cases except the deceased and one lost to follow-up.
Decision on treatment options for FIA at our institution is multidisciplinary. A board of vascular neurologists, vascular neurosurgeons, and expert endovascular practitioners discusses each case. The proposed alternatives are then explained to the patient, until a final decision is reached.
Data analysis
Since treatment nuances vary widely among anatomical sites, analyses were stratified according to three location groups—cavernous ICA (cICA), supraclinoid (ACA, MCA, and supraclinoid ICA), and vertebrobasilar (vertebral, basilar, and posterior cerebral arteries).
Mixed-effects logistic regressions with propensity score (PS) adjustment were modeled to evaluate predictors of morbidity. The PS regressed treatment category (surgical or endovascular) on age, size, rupture status, and initial mRS. Details of PS calculation are reported in the Electronic Supplementary Material. Studies were treated as clusters on the univariate and multivariable analyses [1], and we used a one-step meta-analytical approach. [7, 14, 17, 44]
Sensitivity analyses were conducted to model the primary outcome without propensity score adjustment. Secondary outcomes were studied with chi-squared tests. Risk of bias was assessed across studies by identifying papers that did not meet our eligibility criteria, excluding series with less than five participants, and excluding studies that did not report the primary outcome.
All tests were 2-sided, and final P values under 0.05 were considered statistically significant. Normality assumptions were checked with Shapiro-Wilk’s test. Results are presented as count (valid percentage) or mean (standard deviation/interquartile range). Analyses were conducted with R (R Foundation for Statistical Computing. Vienna, Austria, 2018) and the STATA software (StataCorp. 2013. Stata Statistical Software: Release 13. College Station, TX: StataCorp LP). This study was approved by the local Ethics Committee (ID: 12089), and patient consent was waived because of the retrospective design.
Results
Systematic review
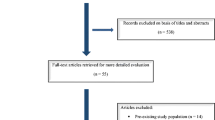
We identified 1219 records in the database, plus three additional relevant studies by hand search. After duplicates were removed, 891 were screened, 159 full articles were assessed for eligibility, and 29 were included in the final synthesis, providing 296 individual patient data. [3, 5, 8, 10, 11, 16, 18,19,20, 24,25,26,27,28,29, 32,33,34, 38, 41,42,43, 45, 46, 48,49,50,51,52] The selection process is detailed in a flow diagram (Fig. 1).
Original data
Medical records of patients followed at our institution’s cerebrovascular surgery clinic were reviewed, identifying 16 patients who underwent interventions for FIA (Table 1). Ten of them were submitted to endovascular procedures: 1 balloon occlusion, 6 embolizations, and 3 flow diverting stents. Six were treated with microsurgery: 2 clippings, 3 clip-wrappings, and 1 saccular bleb clipped.
Two patients died in the same admission: one from complications of SAH from the fusiform lesion (patient 5), one from SAH by another aneurysm later in the course of her hospital stay (patient 6). The only unfavorable angiographic outcome was a 28-year-old male who had a flow diverting stent placed in V4, whose 12-month angiographic follow-up demonstrated incomplete aneurysm obliteration.
Patient characteristics and treatment options
Compiling original and published data, there were 312 cases, of which 79 (25.3%) were treated with microsurgery and 233 (74.5%) were treated with endovascular procedures. Patient characteristics stratified by aneurysm location for 283 of them are summarized in Table 2, because anatomical site was not available for the other 29.
Table 3 lists the treatment options for FIA within each group. Open interventions tackled cICA aneurysms in 3 cases (2 wrappings and 1 clip wrapping) and by endovascular procedures in 27 cases (16 stents, 9 stents + coil/balloon, and 2 balloon occlusions). Supraclinoid lesions were approached surgically in 58 patients. The two commonest strategies were bypassing (56.9%, either alone or combined with clipping, trapping, or excision) and clipping/clip reconstruction (15.6%). For the endovascular group, supraclinoid aneurysms were stented in 29 cases, in one case also deploying coils. Vertebrobasilar FIA were operated in 17 cases (6 parent artery occlusions, 5 bypasses, 4 wrappings, and 2 clip-wrappings) and endovascularly treated in 149 instances (70 stents, 46 parent artery occlusions, and 33 stents + coil/balloon).
Clinical outcomes
For each of the location strata, we modeled propensity score–adjusted regressions to study the relationship between treatment category (microsurgery or endovascular) and morbidity, and there were no significant associations (Table 4).
Among cICA aneurysms, post-procedure morbidity inflicted one surgically treated patient (33%) and seven endovascularly treated ones (26%). No differences were observed between groups (OR 1.04, 95% CI 0.05–23.6). Among the patients treated for supraclinoid FIA, 19 (33%) of the microsurgical group and 2 (6.9%) of the endovascular group worsened their clinical status after the intervention. The PS-adjusted analysis failed to demonstrate an association (OR 7.82, 95% CI 0.65–94.4). Patients with vertebrobasilar lesions had an mRS worsening in 4 (23.5%) surgical cases and 43 (28.9%) endovascular ones. There was no significant association between treatment category and morbidity (OR 0.45, 95% CI 0.05–3.9).
Risk factors for morbidity were evaluated in a separate, multivariable model that demonstrated a significant influence of size (each additional mm, OR 1.11, 95% CI 1.03–1.19) and initial mRS (each additional score, OR 2.0, 95% CI 1.2–3.3). Interaction terms were evaluated, all demonstrating that these associations were independent of location groups (all terms, P > 0.1). Neither age nor rupture status seemed to alter the odds of posterior morbidity (Table 5 and Fig. 2).
Risk factors for postoperative morbidity. Forest plot showing the results of the mixed-effects logistic regression, evaluating risk factors for morbidity after treatment of fusiform aneurysms, both surgically and endovascularly. The odds ratios (OR) are shown, together with the 95% confidence intervals and P values for the coefficients
Angiographic outcomes, retreatment, and conflict of interests
Unfavorable long-term angiographic outcomes occurred in 42 cases from the endovascular group and in one case who underwent microsurgery, although this outcome was often unreported (Table 6). Within the group of cICA aneurysms, 7 (30.4%) patients submitted to endovascular procedures had unfavorable angiographic findings, but there were no long-term imaging assessments for any of the surgical cases, and applying the statistical test was considered inappropriate. Within the group of supraclinoid aneurysms, 6 (23.1%) endovascular cases had this unfavorable outcome, compared to none of those submitted to microsurgery (χ2 = 33.5, P < 0.01). In the vertebrobasilar territory, endovascular cases yielded worse angiographic results: 29 unfavorable outcomes (26.4%) compared to 1 (16.6%) in the surgical group (χ2 = 140.1, P < 0.01).
Retreatment rates were not significantly different between surgery and endovascular procedures in any of the location strata (all P > 0.05, Table 7). Data from 101 patients (32.4%) originated from studies reporting conflicting interests, all of them belonging to the endovascular group (43.3% of those). Conflicting interest was not associated with the primary outcome (OR 1.55, 95% CI 0.86–2.8).
Sensitivity analyses
Sensitivity analyses were performed to evaluate the impact of the PS-adjustment on the results regarding the primary endpoint, morbidity (see Figure 3 of the Electronic Supplementary Material). When removing the PS, surgical treatment is associated with higher rates of morbidity compared to endovascular interventions (P < 0.04, see Figure 4, Electronic Supplementary Material). This difference is likely due to the unbalanced mean initial mRS between groups (surgery 1.6, endovascular 0.6), which was corrected via PS-adjustment. Comparisons within the other groups remained unassociated with the primary outcome.
Discussion
Post-procedure morbidity was not significantly different between surgical and endovascular treatments for fusiform intracranial aneurysms. Larger aneurysms and higher initial mRS were correlated with morbidity, but age and rupture status were not. Angiographic results tend to be more favorable in surgically treated patients, except for cICA aneurysms, in which no analyses were possible. Retreatment rates did not differ between treatment categories, and conflicting interests did not seem to influence the primary outcome.
Conservative management and interventions
Many authors have proposed conservative management for asymptomatic lesions. [23, 39, 40] This is indeed a reasonable option because, as we have found in our study, treatment for FIAs still harbors significant risks and often leads to functional impairment. Other studies with designs focused on comparing watchful waiting and invasive management are needed to determine when and which interventions are beneficial.
However, when clinical presentation prompts intervention, the strategy will depend on patient status and characteristics of the lesion. [4, 40, 41] Arguments in favor of endovascular treatments include the invasiveness of their surgical counterparts, while open surgeries are usually seen as definitive solutions with frequently satisfying angiographic results.
Treatment options differ among anatomical locations
The procedural risk and technical considerations for approaching lesions vary widely among anatomical locations. For instance, a large M1 aneurysm cannot be compared to a small V4 one. In order to provide an adequate comparison, we chose to analyze three territories separately: cICA, supraclinoid, and vertebrobasilar.
Cavernous ICA aneurysms present very specific particularities due to their anatomical location, rarely presenting with SAH. Reports of cICA FIAs treated microsurgically were very scarce, and intervention options included stenting and parent artery occlusion.
On the other hand, supraclinoid are more easily accessed using standard pterional and orbitozygomatic craniotomies, and this group concentrated most of the microsurgically treated lesions. Reconstructive techniques in these cases, such as wrapping or clip reconstruction, are focused on reestablishing the normal vessel lumen. [47] Endovascular techniques in this territory have to avoid occlusion of crucial perforators and preserve distal irrigation. [27]
Vertebrobasilar FIAs presented multiple treatment strategies in the included studies. While some vertebral aneurysms can be treated with parent artery occlusion without neurological compromise, exclusion of other arteries in this territory (such as the PICA) would lead to devastating outcomes due to cerebellar and brainstem infarcts. In these latter cases, stents and anastomoses are needed to preserve distal blood flow. [24, 47]
Microsurgical and endovascular outcomes
We collected results from patients with fusiform lesions treated at our institution in the last 8 years and performed a systematic review to study a large number of cases from various centers. Our meta-analysis included individual patient data from over 300 patients, including our original sample and 29 published series. Selection bias is a critical concern in case reports and series, and studies with less than five patients were excluded in an attempt to minimize this problem. Furthermore, the lack of randomization also entails several biases, and we attempted to address this concern through propensity score adjustment. [14]
Surgery and endovascular did not result in significantly different morbidity for any of the anatomical sites. For the supraclinoid aneurysms, the unadjusted model demonstrated significance, but the adjusted regression did not. We believe the large difference between baseline functional assessment was a confounder in the unadjusted analysis. However, the large confidence intervals for cICA and supraclinoid anterior circulation aneurysms should be interpreted carefully, signaling that a larger sample could be necessary for more robust conclusions.
Size and initial mRS seem to be more important risk factors for morbidity than age and rupture status. Although patient independence and functionality is always the priority, angiographic outcomes are another essential part of the treatment evaluation. In our analysis, microsurgery seems to have yielded better angiographic results after the treatment of supraclinoid and vertebrobasilar aneurysms, but these were exploratory analyses that should be validated in other studies.
This sort of information is valuable when facing difficult clinical decisions where both surgical and endovascular treatments are feasible. For patients with small aneurysms and good initial functional assessment, surgical intervention could provide definitive angiographic resolution with minimal functional impairment, regardless of rupture status. Especially in younger patients undergoing elective procedures, a definitive solution is extremely desirable, and microsurgery still yields adequate long-term solutions.
Our results also suggest that FIA are still associated with poor functional outcomes, much worse compared to saccular aneurysms. In a meta-analysis including 2404 clipped MCA aneurysms [2], unfavorable neurological outcomes were reported for 2.1% of the patients, while in our study, the rates range from 23.5 to 33.3%, depending on the location. The same meta-analysis also reported less than 5% of unfavorable neurological outcomes for coiling, whereas morbidity rates for endovascular procedures in the present study range from 6.9 to 28.9%. [2] There is an urgent necessity to devise more efficient operative techniques, clinical management practices, or neurointerventional devices to improve the quality of life of patients with FIA.
Study limitations
This study presents several limitations. The design brings a significant amount of selection and publication biases that were minimized with study eligibility criteria and statistical methods, but most certainly not abolished. Particularly for angiographic outcomes, reporting was very heterogeneous, which is why we chose to conduct only chi-squared analyses, not more sophisticated regression models. These results should be interpreted as exploratory and need clarifying in further studies.
Moreover, both endovascular and surgical techniques evolve rapidly with time, and even though the earliest included study is from 2002, some of the outcomes could be different using current techniques. Conclusions must be evaluated carefully, and randomized clinical trials are ideally necessary to compare the different techniques.
Conclusion
There were no differences between morbidity of surgical and endovascular treatments, regardless of aneurysm location. Size and initial mRS were correlated with functional outcomes, whereas age and rupture status were not. Microsurgery seems to yield better long-term angiographic results compared to endovascular procedures.
References
Abo-Zaid G, Guo B, Deeks JJ, Debray TPA, Steyerberg EW, Moons KGM, Riley RD (2013) Individual participant data meta-analyses should not ignore clustering. J Clin Epidemiol 66:865–873.e4. https://doi.org/10.1016/j.jclinepi.2012.12.017
Alreshidi M, Cote DJ, Dasenbrock HH, Acosta M, Can A, Doucette J, Simjian T, Hulou MM, Wheeler LA, Huang K, Zaidi HA, Du R, Aziz-sultan MA, Mekary RA, Smith TR (2018) Coiling versus microsurgical clipping in the 0:1–11. doi: https://doi.org/10.1093/neuros/nyx623/4847331
Byoun HS, Yi HJ, Choi KS, Chun HJ, Ko Y, Bak KH (2016) Comparison of endovascular treatments of ruptured dissecting aneurysms of the intracranial internal carotid artery and vertebral artery with a review of the literature. J Korean Neurosurg Soc 59:449–457. https://doi.org/10.3340/jkns.2016.59.5.449
Chiaradio JC, Guzman L, Padilla L, Chiaradio MP (2002) Intravascular graft stent treatment of a ruptured fusiform dissecting aneurysm of the intracranial vertebral artery: technical case report. Neurosurgery 50:213–217. https://doi.org/10.1097/00006123-200201000-00034
Dabus G, Lin E, Linfante I (2014) Endovascular treatment of fusiform intracranial vertebral artery aneurysms using reconstructive techniques. J Neurointerv Surg 6:589–594. https://doi.org/10.1136/neurintsurg-2013-010897
Day AL, Gaposchkin CG, Yu CJ, Rivet DJ, Dacey RG (2003) Spontaneous fusiform middle cerebral artery aneurysms: characteristics and a proposed mechanism of formation. J Neurosurg 99:228–240. https://doi.org/10.3171/jns.2003.99.2.0228
Debray TPA, Moons KGM, van Valkenhoef G, Efthimiou O, Hummel N, Groenwold RHH, Reitsma JB (2015) Get real in individual participant data (IPD) meta-analysis: a review of the methodology. Res Synth Methods 6:293–309. https://doi.org/10.1002/jrsm.1160
Deshmukh VR, Kakarla UK, Figueiredo EG, Zabramski JM, Spetzler RF (2006) Long-term clinical and angiographic follow-up of unclippable wrapped intracranial aneurysms. Neurosurgery 58:434–441. https://doi.org/10.1227/01.NEU.0000199158.02619.99
Drake CG, Peerless SJ (1997) Giant fusiform intracranial aneurysms: review of 120 patients treated surgically from 1965 to 1992. J Neurosurg 87:141–162. https://doi.org/10.3171/jns.1997.87.2.0141
Ertl L, Holtmannspötter M, Patzig M, Brückmann H, Fesl G (2014) Use of flow-diverting devices in fusiform vertebrobasilar giant aneurysms: a report on periprocedural course and long-term follow-up. Am J Neuroradiol 35:1346–1352. https://doi.org/10.3174/ajnr.A3859
Findlay JM, Hao C, Emery D (2002) Non-atherosclerotic fusiform cerebral aneurysms. Can J Neurol Sci 29:41–48. https://doi.org/10.1017/S0317167100001700
Flemming KD, Wiebers DO, Brown RD, Link MJ, Huston J, McClelland RL, Christianson TJH (2005) The natural history of radiographically defined vertebrobasilar nonsaccular intracranial aneurysms. Cerebrovasc Dis 20:270–279. https://doi.org/10.1159/000087710
Flemming KD, Wiebers DO, Brown RD, Link MJ, Nakatomi H, Huston J, McClelland R, Christianson TJH (2004) Prospective risk of hemorrhage in patients with vertebrobasilar nonsaccular intracranial aneurysm. J Neurosurg 101:82–87
Haukoos JS, Lewis RJ (2015) The propensity score. JAMA 314:1637–1638. https://doi.org/10.1001/jama.2015.13480
Horie N, Takahashi N, Furuichi S, Mori K, Onizuka M, Tsutsumi K, Shibata S (2003) Giant fusiform aneurysms in the middle cerebral artery presenting with hemorrhages of different origins: report of three cases and review of the literature. J Neurosurg 99:391–396. https://doi.org/10.3171/jns.2003.99.2.0391
Hu P, Zhang HQ, Li XY, Tong XZ (2019) Double-barrel superficial temporal artery to proximal middle cerebral artery bypass to treat complex intracranial aneurysms: a reliable high blood flow bypass. World Neurosurg 125:e884–e890. https://doi.org/10.1016/j.wneu.2019.01.203
Jolani S, Debray TPA, Koffijberg H, van Buuren S, Moons KGM (2015) Imputation of systematically missing predictors in an individual participant data meta-analysis: a generalized approach using MICE. Stat Med 34:1841–1863. https://doi.org/10.1002/sim.6451
Joo SP, Kim TS, Choi JW, Lee JK, Kim YS, Moon KS, Kim JH, Kim SH (2007) Characteristics and management of ruptured distal middle cerebral artery aneurysms. Acta Neurochir 149:661–667. https://doi.org/10.1007/s00701-007-1061-5
Kalani MYS, Zabramski JM, Nakaji P, Spetzler RF (2013) Bypass and flow reduction for complex basilar and vertebrobasilar junction aneurysms. Neurosurgery 72:763–775. https://doi.org/10.1227/NEU.0b013e3182870703
Kivipelto L, Niemelä M, Meling T, Lehecka M, Lehto H, Hernesniemi J (2014) Bypass surgery for complex middle cerebral artery aneurysms: impact of the exact location in the MCA tree. J Neurosurg 120:398–408. https://doi.org/10.3171/2013.10.JNS13738
Krings T, Mandell DM, Kiehl T-R, Geibprasert S, Tymianski M, Alvarez H, TerBrugge KG, Hans F-J (2011) Intracranial aneurysms: from vessel wall pathology to therapeutic approach. Nat Rev Neurol 7:547–559. https://doi.org/10.1038/nrneurol.2011.136
L.A. S, M. C, M. R, R.D. R, M. S, G. S (2015) Preferred reporting items for a systematic review and meta-analysis of individual participant data: the PRISMA-IPD statement. JAMA - J. Am. Med. Assoc.
Lanzino G, Kaptain G, Kallmes DF, Dix JE, Kassell NF (1997) Intracranial dissecting aneurysm causing subarachnoid hemorrhage: the role of computerized tomographic angiography and magnetic resonance angiography. Surg Neurol 48:477–481. https://doi.org/10.1016/S0090-3019(97)00178-X
Laukka D, Rautio R, Rahi M, Rinne J (2019) Acute treatment of ruptured fusiform posterior circulation posterior cerebral, superior cerebellar, and posterior inferior cerebellar artery aneurysms with fred flow diverter: report of 5 cases. Oper Neurosurg 16:549–556. https://doi.org/10.1093/ons/opy194
Leibowitz R, Do HM, Marcellus ML, Chang SD, Steinberg GK, Marks MP (2003) Parent vessel occlusion for vertebrobasilar fusiform and dissecting aneurysms. Am J Neuroradiol 24:902–907
Lin N, Brouillard AM, Keigher KM, Lopes DK, Binning MJ, Liebman KM, Veznedaroglu E, Magarik JA, Mocco J, Duckworth EA, Arthur AS, Ringer AJ, Snyder KV, Levy EI, Siddiqui AH (2015) Utilization of pipeline embolization device for treatment of ruptured intracranial aneurysms: US multicenter experience. J Neurointerv Surg 7:808–815. https://doi.org/10.1136/neurintsurg-2014-011320
Lin N, Lanzino G, Lopes DK, Arthur AS, Ogilvy CS, Ecker RD, Dumont TM, Turner RD, Gooch MR, Boulos AS, Kan P, Snyder KV, Levy EI, Siddiqui AH (2016) Treatment of distal anterior circulation aneurysms with the pipeline embolization device: a US multicenter experience. Neurosurgery 79:14–22. https://doi.org/10.1227/NEU.0000000000001117
Lubicz B, Collignon L, Raphaeli G, Pruvo JP, Bruneau M, De Witte O, Leclerc X (2010) Flow-diverter stent for the endovascular treatment of intracranial aneurysms: a prospective study in 29 patients with 34 aneurysms. Stroke 41:2247–2253. https://doi.org/10.1161/STROKEAHA.110.589911
Meckel S, McAuliffe W, Fiorella D, Taschner CA, Phatouros C, Phillips TJ, Vasak P, Schumacher M, Klisch J (2013) Endovascular treatment of complex aneurysms at the vertebrobasilar junction with flow-diverting stents: initial experience. Neurosurgery 73:386–394. https://doi.org/10.1227/01.neu.0000431472.71913.07
Mizutani T, Miki Y, Kojima H, Suzuki H (1999) Proposed classification of nonatherosclerotic cerebral fusiform and dissecting aneurysms. Neurosurgery. 45:253–259. https://doi.org/10.1097/00006123-199908000-00010
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097
Monteith SJ, Tsimpas A, Dumont AS, Tjoumakaris S, Fernando Gonzalez L, Rosenwasser RH, Jabbour P (2014) Endovascular treatment of fusiform cerebral aneurysms with the Pipeline Embolization Device. J Neurosurg 120:945–954. https://doi.org/10.3171/2013.12.JNS13945
Munich SA, Tan LA, Keigher KM, Chen M, Moftakhar R, Lopes DK (2014) The Pipeline Embolization Device for the treatment of posterior circulation fusiform aneurysms: lessons learned at a single institution. J Neurosurg 121:1077–1084. https://doi.org/10.3171/2014.7.JNS132595
Murchison AG, Young V, Djurdjevic T, Cellerini M, Corkill R, Küker W (2018) Stent placement in patients with acute subarachnoid haemorrhage: when is it justified? Neuroradiology 60:735–744. https://doi.org/10.1007/s00234-018-2020-6
Nakatomi H, Segawa H, Kurata A, Shiokawa Y, Nagata K, Kamiyama H, Ueki K, Kirino T (2000) Clinicopathological study of intracranial fusiform and dolichoectatic aneurysms: insight on the mechanism of growth. Stroke. 31:896–900. https://doi.org/10.1161/01.STR.31.4.896
Nasr DM, Brinjikji W, Rouchaud A, Kadirvel R, Flemming KD, Kallmes DF (2016) Imaging characteristics of growing and ruptured vertebrobasilar non-saccular and dolichoectatic aneurysms. Stroke. 47:106–112. https://doi.org/10.1161/STROKEAHA.115.011671
Nasr DM, Flemming KD, Lanzino G, Cloft HJ, Kallmes DF, Murad MH, Brinjikji W (2018) Natural history of vertebrobasilar dolichoectatic and fusiform aneurysms: a systematic review and meta-analysis. Cerebrovasc Dis 45:68–77. https://doi.org/10.1159/000486866
Natarajan SK, Lin N, Sonig A, Rai AT, Carpenter JS, Levy EI, Siddiqui AH (2016) The safety of pipeline flow diversion in fusiform vertebrobasilar aneurysms: a consecutive case series with longer-term follow-up from a single US center. J Neurosurg 125:111–119. https://doi.org/10.3171/2015.6.JNS1565
Niikawa S, Yamada J, Sumi Y, Yamakawa H (2002) Dissecting aneurysm of the middle cerebral artery manifesting as subarachnoid hemorrhage and hemorrhagic infarctions - case report. Neurol Med Chir (Tokyo) 42:62–66. https://doi.org/10.2176/nmc.42.62
Park S-H, Yim M-B, Lee C-Y, Kim E, Son E-I (2008) Intracranial fusiform aneurysms: It’s pathogenesis, clinical characteristics and managements. J Korean Neurosurg Soc 44:116–123. https://doi.org/10.3340/jkns.2008.44.3.116
Park W, Kwon DH, Ahn JS, Lee SH, Park JC, Kwun BD (2015) Treatment strategies for dissecting aneurysms of the posterior cerebral artery. Acta Neurochir 157:1633–1643. https://doi.org/10.1007/s00701-015-2526-1
Pumar JM, Arias-Rivas S, Rodríguez-Yáñez M, Blanco M, Ageitos M, Vazquez-Herrero F, Castiñeira-Mourenza JA, Masso A (2013) Using Leo Plus stent as flow diverter and endoluminal remodeling in endovascular treatment of intracranial fusiform aneurysms. J Neurointerv Surg 5:iii22–iii27. https://doi.org/10.1136/neurintsurg-2013-010661
Qu S, Lv X, Wu Z (2009) Clinical outcomes of basilar artery aneurysms. Neuroradiol J 22:228–238. https://doi.org/10.1177/197140090902200215
Resche-Rigon M, White IR, Bartlett JW, Peters SAE, Thompson SG (2013) Multiple imputation for handling systematically missing confounders in meta-analysis of individual participant data. Stat Med 32:4890–4905. https://doi.org/10.1002/sim.5894
Rho MH, Park HJ, Chung EC, Choi YJ, Lee SY, Won YS, Kim BM (2013) Various techniques of stent-assisted coil embolization of wide-necked or fusiform artherosclerotic and dissecting unruptured vertebrobasilar artery aneurysms for reducing recanalization: mid-term results. Acta Neurochir 155:2009–2017. https://doi.org/10.1007/s00701-013-1866-y
Roh HG, Kim SS, Han H, Kang HS, Moon WJ, Byun HS (2008) Endovascular treatment of posterior cerebral artery aneurysms using detachable coils. Neuroradiology 50:237–242. https://doi.org/10.1007/s00234-007-0321-2
Safavi-Abbasi S, Kalani MYS, Frock B, Sun H, Yagmurlu K, Moron F, Snyder LA, Hlubek RJ, Zabramski JM, Nakaji P, Spetzler RF (2017) Techniques and outcomes of microsurgical management of ruptured and unruptured fusiform cerebral aneurysms. J Neurosurg 127:1353–1360. https://doi.org/10.3171/2016.9.JNS161165
Seo BR, Kim TS, Joo SP, Lee JM, Jang JW, Lee JK, Kim JH, Kim SH (2009) Surgical strategies using cerebral revascularization in complex middle cerebral artery aneurysms. Clin Neurol Neurosurg 111:670–675. https://doi.org/10.1016/j.clineuro.2009.06.002
Stroke T, Gates MF, Carolina N (2012) S 1258. 116:1258–1266
Suh SH, Kim BM, Chung TS, Kim DI, Kim DJ, Hong CK, Kim CH, Ahn JY, Kim SS (2010) Reconstructive endovascular treatment of intracranial fusiform aneurysms: a 1-stage procedure with stent and balloon. Am J Neuroradiol 31:155–160. https://doi.org/10.3174/ajnr.A1784
Wang Q, Leng B, Song D, Chen G (2010) Fusiform aneurysms of the vertebrobasilar arterial trunk: choice of endovascular methods and therapeutic efficacy. Acta Neurochir 152:1467–1476. https://doi.org/10.1007/s00701-010-0691-9
Xu F, Xu B, Huang L, Xiong J, Gu Y, Lawton MT (2018) Surgical treatment of large or giant fusiform middle cerebral artery aneurysms: a case series. World Neurosurg 115:e252–e262. https://doi.org/10.1016/j.wneu.2018.04.031
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
the authors declare that they have no conflict of interest.
Ethics approval
This study was approved by the local Ethics Committee (ID: 12089), and patient consent was waived because of the retrospective design.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 3616 kb)
Rights and permissions
About this article
Cite this article
Telles, J.P.M., Solla, D.J.F., Yamaki, V.N. et al. Comparison of surgical and endovascular treatments for fusiform intracranial aneurysms: systematic review and individual patient data meta-analysis. Neurosurg Rev 44, 2405–2414 (2021). https://doi.org/10.1007/s10143-020-01440-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-020-01440-x