Abstract
Epidermoid cysts constitute less than 1 % of intracranial tumors with the majority of them involving cerebellopontine angle (CPA). Although several mechanisms for cranial nerve dysfunction due to these tumors have been proposed, no direct evaluation for hyper- or hypoactive dysfunction has been done. In this case series, pathophysiology of cranial nerve dysfunction in CPA epidermoid cysts was evaluated with special attention to a new mechanism of capsule strangulation caused by stratified tumor capsule. Twenty-two cases with epidermoid cysts of CPA micro-neurosurgically treated in our departments since 2005 were reviewed. Clinical status of the patients before the surgery and post-operative functional outcome were recorded. Available data from the English literature were summarized for comparison. Mass reduction of cyst contents in most cases was usually associated with prompt and marked improvement of the symptoms suggesting neuroapraxia caused by compression of the tumor content and/or mild ischemia. Among them, two cases showed strangulation of the affected nerves by the tumor capsule whose preoperative dysfunction did not improve after surgery in spite of meticulous microsurgical removal of the lesion. Involved facial and abducent nerves in these two cases showed distortion of nerve axis and nerve atrophy distal to the strangulation site. We report the first direct evidence of etiology of cranial nerve dysfunction caused by cerebellopontine angle epidermoid tumors. Young age and rapidly progressive neurological deficit might be the characteristics for strangulation of the affected nerves by the cyst capsule. Even though the number of cases might be limited, immediate decompression and release of the strangulating band might be urged in such patients to prevent irreversible deficits.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intracranial epidermoid cyst is a rare embryonal tumor of ectodermal origin accounting for about 1 % of all intracranial tumors [8, 12–14, 25, 29, 42]. It arises in early gestation as a result of ectopic inclusion of the skin ectoderm [3, 24]. Cerebellopontine angle (CPA) is the most common intracranial location for these pathologies [45] constituting 40 % of all cases [23]. However, these lesions make up only 7 % of the tumors in this region [26].
Patients with CPA epidermoids may present with symptoms of cerebellar and/or brainstem dysfunction, hydrocephalus, chemical meningitis, as well as cranial nerves (CNs) dysfunction. Headache is the most common symptom [9, 12], and involvement of the vestibule-cochlear nerve [9, 21, 35, 43, 45], trigeminal nerve [6, 15, 17, 23], or cerebellar dysfunction [46] has been reported as the most common sign in different case series. Usually, accumulation of the cyst contents causes direct compression and/or raised intracranial pressure. Acoustic nerve’s tumors often stretch the facial nerve but do not induce the palsy until advanced stages. On the contrary, epidermoid cysts usually show facial palsy at earlier stages [46]. The “strangling” mechanism, meaning that the tumor tissue encases the nerve entirely, was first described by Antoli-Candela and Stewart in 1974 and considered as a probable mechanism by other authors [9, 35] and is postulated to cause facial palsy at earlier stages compared with acoustic tumor. The “strangulation” is supposed to reduce blood supply of the nerve bundles resulting in nerve dysfunction [4], and this term was originally coined to describe the cholesteatoma in the temporal bone not the CPA epidermoid cysts [4]. The evidence of this mechanism has not been described at all, and details of this phenomenon are still missing.
In this study, we reviewed recent cases operated in our department with emphasis on the mechanisms underlying CN deficits in them, which could be mostly explained by neuroapraxia, among which we noticed a new strangulation mechanism not described in the literature previously and named it “capsular strangulation.”
Patients and methods
Since 2005, 22 patients with CPA epidermoid cyst have been operated by the two senior surgeons (18 cases by MH and 4 by YH) in our institutes. Clinical and surgical records, radiological studies, and intraoperative photographs and videos were reviewed retrospectively. All patients were pre- and post-operatively evaluated with CT-scan and MRI. Retrosigmoid approach was adopted in the lateral position except for one recurrent case for which an anterior petrosal approach was used to access the tumor in the Meckel’s cave and to avoid dissection of the adhesive retrosigmoid route after multiple operations in another center. After the tumor capsule was incised, flaky white contents were evacuated and non-adhesive cyst wall was removed as much as possible. The same strategy was applied to the tumors with supratentorial or contralateral CPA extensions where a unilateral suboccipital route would be used for decompression of the affected nerves (Fig. 1). Extreme care was exercised to avoid any irritant debris seeding into the subarachnoid space. We do not advocate routine administration of corticosteroids before or during the operation. In cases with hyperactive dysfunction of the CNs, displaced arterial loop into the root entry/exit zone was carefully sought. The completeness of cyst removal and relationship between the affected nerves and the cyst wall and contents were assessed by intraoperative observations by the neurosurgeon and post-operative images. Follow-up clinical and radiological evaluations were performed at half to 1 year intervals. For statistical analysis, SPSS software version 14.0 (2005, SPSS Inc.) was utilized. Mann–Whitney U (MWU), chi-square (χ 2), and Fisher’s exact tests were used when applicable and statistical significance was defined as p value less than 0.05. Data are presented as mean ± standard error of mean.
A 47-year-old male, with left trigeminal neuralgia, left hemifacial spasm, and double vision for several months (case 10). Preoperative MRI (a diffusion weighted; b T2-weigted) showed epidermoid cyst in cerebellopontine angle cistern in both sides. Left lateral suboccipital craniotomy was performed, and complete resection of the epidermoid cyst at left side(c before removal; f after removal) and tumor remnant at right side are evident. Post-operative MRI shows tumor remnant at right side (d diffusion weighted; e T2-weigted). Patient showed no tumor recurrence or new symptoms at follow-ups for 4.5 years after the operation
Results
Details of cases with functional outcome are summarized in Table 1. In brief, 22 cases (ten males) with the average age of 41.05 ± 2.82 years (17–64) at the time of operation were treated using micro-neurosurgical techniques.
No gender differences were observed in any of the symptoms or complication rates (χ 2, p > 0.5). All cases were symptomatic, and trigeminal nerve symptoms were the most common at presentation (n = 15:68.2 % of cases) where trigeminal neuralgia (TN) was the dominant form and observed in 13 patients. The second most common signs and symptoms were related to the facial nerve (n = 11; 50.0 % of cases) where facial nerve palsy and hemifacial spasm (HFS) were seen equally in 6 cases. As it can be expected, patients with TN come earlier to the hospital (15.58 ± 5.93 months) compared with the patients without pain (36.63 ± 15.63 months) though this was not statistically significant (MWU, p > 0.05). Symptoms at presentation had no correlation with complications (χ 2, p > 0.5). Pre- and post-operative cranial nerve deficits are summarized in Table 2.
Four patients were recurrent cases operated at least once previously with mean age at the present surgery of 50.75 ± 5.26 years. Although HFS was more common in recurrent cases (Fisher’s exact test, p = 0.04), other symptoms showed no significant differences. Patients with hyperactive symptoms (i.e., TN or HFS) tend to be older than those with other symptoms (MWU, p = 0.02). However, age was not statistically different in the presence of other symptoms, complications, or recurrent cases (MWU, p > 0.05). Also, hyperactive symptoms were not different in complicated cases or according to the gender (χ 2 or Fisher’s exact test, p > 0.5). The interval between the onset of symptoms and the operation ranged between 1 month to 10 years (24.00 ± 7.33 months), and those with trigeminal neuralgia had a shorter interval to diagnosis (15.58 ± 5.93 vs. 36.63 ± 15.63 months) though, this was not statistically significant (MWU, p = 0.27). Interestingly, this interval was much longer in those with hearing loss compared with those with intact hearing (MWU, p = 0.01). The duration of symptoms was not significantly affected by presence or absence of other CN deficits.
Location and extension of the tumors are demonstrated in Table 3. In most cases, cyst capsule was stratified and projected between the nerves in a finger-like fashion. Cranial nerves were compressed, deviated, and/or embedded in the stratified cyst wall, but no apparent strangulating effect could be observed except for two cases (case 2 and case 17: details are described below). After careful inspection of the root entry zone, no direct arterial compression was observed in any case except for one patient with trigeminal neuralgia. The affected nerves were compressed/embedded and distorted by the tumor content and the capsule. Based on neurosurgeon’s intraoperative observations and post-operative MRI studies, the rate of total/near total removal of the cyst was 81.8 % (18 of 22 cases). In case of contralateral or supratentorial tumor extension, the tumor would be resected through the ipsilateral corridor as much as possible and the contralateral remnant would be followed for any symptomatic or radiological growth. All the patients were followed up until now where the range of follow-up period was between 2 months to 9 years. No tumor growths requiring surgical intervention were observed in any of the patients. In our series, there were no patients experiencing post-operative wound dehiscence, pseudomeningocele, CSF leakage, or meningitis. New post-operative neurological deficits are demonstrated in Table 1.
Case illustrations
Case 2
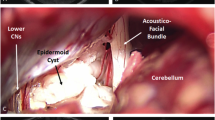
A 17-year-old female noticed slight asymmetry of countenance about 1 year ago which progressed to House–Brackmann (HB) grade IV brining her to our outpatient clinic. No other neurological signs such as hearing loss or double vision were seen. She was diagnosed with CPA epidermoid after a brain MRI. Suboccipital craniotomy revealed that the facial nerve was embedded in the cyst content and severely strangulated by the firm, fibrous, and tenaciously adherent cyst wall at the portion between the normal-looking facial nerve at the brainstem and red, atrophied distal side of the nerve at the internal acoustic meatus (Fig. 2a–c). The stratified, firm cyst wall was incised in a meticulous fashion to release and decompress the nerve. The atrophied distal facial nerve was extremely thin compared with the proximal portion with normal appearance (Fig. 2b). One year after the operation, only slight improvement in the facial nerve function could be observed (HB grade III).
Two patients with cranial nerve strangulation by the tumor’s capsule. Upper lane: a 17-year-old girl with right facial palsy. a Preoperative photograph shows facial palsy House-Brackmann (HB) grade IV. b and c Intraoperative photo shows severe strangulation between two arrows. Nerve diameter of the distal side becomes extremely thin, and the surface color changed red, indicating atrophy of the facial nerve due to Wallerian degeneration with blood congestion. Lower lane: a 21-year-old man with left abducent nerve palsy. a Preoperative photograph shows complete abducent nerve palsy. b Intraoperative photo shows capsule strangulation with thick capsule (between the arrows). c Blood congestion after removal of the capsule
Case 17
A 21-year-old man was referred to our clinic from another hospital with the complaint of double vision and image-based diagnosis of posterior fossa epidermoid cyst. His symptoms were fluctuating in the beginning but turned constant later in the course of the disease as his deficit was fixed in the last 4 months before being referred to us. The operation revealed that the cyst content around the abducent nerve was relatively firm, and after removal of the cyst content, the abducent nerve strangulated by the firm, fibrous, and tenaciously adherent cyst wall came into sight (Fig. 2d–f). Although nerve atrophy was not apparent, the axis of the nerve fibers was distorted and the surface of the nerve looked congested. Post-operatively, no remarkable improvement of the abducent nerve function happened, and 15 months later, he underwent a neuro-ophthalmological surgery to have his globe dislocation corrected.
Discussion
Epidermoid cyst is considered a rare, congenital, slow-growing, extra-axial tumorous malformation. This lesion is usually located in cerebellopontine angle or suprasellar region [1], although intradiploic epidermoids with rapid growth rate have been described [28]. Intracranial epidermoids arise from displaced epithelial tissues between the 3rd and 5th weeks of gestation during closure of the neural tube. In contrast with most neoplastic lesions, epidermoid is believed to grow at a linear rate similar to that of normal epidermis [3]. For this reason, it requires more than usual time to make patients symptomatic.
Incidence of cranial nerves dysfunction
Rate of each CN dysfunction has shown wide variation in each institute. According to a report which summarized 14 published papers including overall 263 cases, the incidence of CNs dysfunction was as follows: CN-VIII 73 % > CN-V 56 % > CN-VII 24 % whereas double vision was observed in 44 (16.7 %) patients [17]. In our series, the most common sign or symptom at presentation was trigeminal nerve dysfunction (68.2 %) followed by facial nerve disorders (50.0 %). Although facial palsy is not a common feature in epidermoids, it usually happens at early stages of the disease and is mild in nature [46]. Table 4 shows summary of incidence of CN-VI and VII palsies according to the previous case series of CPA epidermoid from the English literature [2, 9, 10, 11, 18, 21, 23, 29, 30, 32, 33, 35-37, 40, 43, 45, 46] along with our present series.
Pathoetiology
Symptoms and signs are caused by various mechanisms. Compression caused by the cyst wall and content results in stretching, bending, and kinking of the nerves and the brain stem. This will interfere with blood supply and axonal flow of the nerves leading to their demyelination. In larger tumors, raised intracranial pressure causes various neurological deficits [42]. In cases with CN hyperactive dysfunction, the etiology might be more complicated. Different pathological tumor types show different characteristics for hemifacial spasm [19]. Also, there are variations in epidermoids with trigeminal neuralgia: nerve encasement by the tumor with/without displacement, compression and displacement of the nerve, pressing the arterial vessels to the nerve by moving the vessel or nerve by the tumor, or compression to the artery which is located in the opposite site of the tumor [17, 39]. The cyst capsule and its content may irritate the nerves as they induce chemical meningitis [44]. Although strangulation of the CNs by the cyst content has been described before, an additional mechanism was observed among our cases: cyst wall may choke the nerve bundles resulting in nerve degeneration as clearly showed in two cases illustrated above. Whether this capsular strangulating mechanism happens congenitally or develops gradually cannot be determined. Multiple finger-like projections of the cyst wall penetrating into the space between the CNs and the vessels may overlap and adhere to each other. The gradual accumulation of cyst content might strain the cyst walls and accelerate the choking mechanism or might reduce both blood and axonal flow to the nerve bundles. Both illustrated cases (i.e., case 2 and case 17) were young, experienced relatively rapid deterioration of function in the affected nerves, and showed severe Wallerian degeneration by capsule-straggling mechanism. This mechanism might reflect one of the reasons for which the amount of tumor resection does not correlate with the functional outcome [29]. It is well known that the closer the facial nerve injury to the brainstem, the more severe the retrograde axonal degeneration of the facial nucleus in the pons [22]. The poorer functional prognosis of these cases might be due to retrograde degeneration of the neurons in each nucleus, although the model of retrograde degeneration of the abducens nerve has not yet been reported. Further cases with similar mechanism are required to better clarify its characteristics.
Diagnosis
CT scans demonstrate a hypo- or slightly hyper-dense area compared with CSF, and MRI studies show iso- to slightly hyper-intense lesion relative to CSF on both T1- and T2-weighted images. Although it has been described that FLAIR modality is useful to display a higher contrast ratio and contrast-to-noise ratio of the tumor to CSF than the conventional MRI [16], in some cases, lesion delineation is not sufficient [20]. Metrizamide CT-scan is useful, but subarachnoid puncture and intrathecal injection is invasive and dangerous in some cases. Tsuruda and colleagues described the usefulness of diffusion-weighted imaging in differentiating bright epidermoids from dark CSF and arachnoid cysts [41]. This MRI modality is very sensitive and specific for diagnosis of CPA epidermoids.
Approach
The goal to cure CPA epidermoids is ideally total removal of the cyst content. Subtotal excision requires multiple operations for symptomatic tumor recurrences [2]. However, their close proximity to the CNs and brain stem and their tendency to adhere to them make their complete removal a real technical challenge in some cases. In these cases, the primary surgical objective is to decompress the affected nerves and brain stem by evacuating the cyst contents and removing the non-adherent capsule, leaving some parts of the capsule adherent to the surrounding critical structures behind [7, 34]. Suboccipital retrosigmoid approach is most suitable to remove as much tumor and capsule as possible to delay symptomatic recurrence as long as possible. However, it has been described that there is no correlation between the degree of tumor removal and neurological outcome [5]. In our cases, micro-neurosurgical treatment was applied via retrosigmoid approach except one recurrent case for whom an anterior petrosal approach was used to get better visualization of the Meckel’s cave and pre-pontine cistern [38] and to avoid exploration of the scarred, fibrotic structures in the retrosigmoid route.
Surgical technique
With recent advances in neuroendoscopic techniques, reports of endoscopic assisted [31] and purely endoscopic [27] removal of CPA epidermoids have emerged. Use of angled views by endoscope at the conclusion of the surgery may assure the surgeon of total removal of the cyst contents. Closer observation by endoscope might give us additional information for further understanding of the etiology of nerve dysfunction in CPA epidermoids.
Outcome and histopathological importance
We should note that in our series, one of the recurrent cases showed histologically inflammatory reaction in addition to typical histology of the epidermoids (case 19). Altschuler et al. reported three cases with combined histopathological features of both epidermoid and cholesterol granuloma in which subtotal removal resulted in multiple operations [2]. We had two other recurrent cases with inflammatory reactions in their histopathology, but as their lesions were not in the CPA region but in the temporal fossa, they were not included in this series. These cases might support the hypothesis that congenital epidermoid cysts in the petroclival bone may incite a local inflammatory reaction. We should be aware that an epidermoid cyst with cholesterol granuloma reaction is a distinct clinical entity with poor control and higher chance of recurrence.
In conclusion, we report the first direct evidence of etiology of cranial nerve dysfunction caused by cerebellopontine angle epidermoid tumors other than neuroapraxia. Young aged, rapidly progressive neurological deficit might indicate a risk of capsule strangulating mechanism of affected nerves by cyst capsule. Even though the number of cases might be extremely limited, immediate decompression and release of strangulating band might be urged in such special cases, for getting better functional prognosis.
References
Abramson RC, Morawetz RB, Schlitt M (1989) Multiple complications from an intracranial epidermoid cyst: case report and literature review. Neurosurgery 24:574–578
Altschuler EM, Jungreis CA, Sekhar LN, Jannetta PJ, Sheptak PE (1990) Operative treatment of intracranial epidermoid cysts and cholesterol granulomas: report of 21 cases. Neurosurgery 26:606–614
Alvord EC Jr (1977) Growth rates of epidermoid tumors. Ann Neurol 2:367–370
Antoli-Candela F Jr, Stewart TJ (1974) The pathophysiology of otologic facial paralysis. Otolaryngol Clin N Am 7:309–330
Apfelbaum MI (1987) Epidermoid cysts and cholesterol granulomas centered on the posterior fossa: twenty years of diagnosis and management. Neurosurgery 21:805
Bauman CH, Bucy PC (1956) Paratrigeminal epidermoid tumors. J Neurosurg 13:455–468
Berger MS, Wilson CB (1985) Epidermoid cysts of the posterior fossa. J Neurosurg 62:214–219
Committee of Brain Tumor Registry of Japan (2014) Report of brain tumor resistor of Japan (2001–2004). Neurol Med Chir 54(Suppl):9–102
De Souza CE, de Souza R, da Costa S et al (1989) Cerebellopontine angle epidermoid cysts: a report on 30 cases. J Neurol Neurosurg Psychiatry 52:986–990
Gagliardi FM, Vagnozzi R, Caruso R, Delfini R (1980) Epidermoids of the cerebellopontine angle (CPA): usefulness of CT scan. Acta Neurochir (Wien) 54:271–281
Gopalakrishnan CV, Ansari KA, Nair S, Menon G (2014) Long term outcome in surgically treated posterior fossa epidermoids. Clin Neurol Neurosurg 117:93–99
Grant FC, Austin GM (1950) Epidermoids; clinical evaluation and surgical results. J Neurosurg 7(3):190–198
Guidetti B, Gagliardi FM (1977) Epidermoid and dermoid cysts: clinical evaluation and late surgical results. J Neurosurg 47:12–18
Hamel E, Frowein RA, Karimi-Nejad A (1980) Intracranial intradural epidermoids and dermoids. Neurosurg Rev 3:215–219
Hori T, Numata H, Hokama Y, Muraoka K, Tamaki M, Saito Y (1983) Trigeminal pain caused by a parapontine epidermoid cyst. Surg Neurol 19:517–519
Karantanas AH (2001) MR imaging of intracranial epidermoid tumors: specific diagnosis with Turbo-FLAIR pulse sequence. Comput Med Imaging Graph 25:249–255
Kobata H, Kondo A, Iwasaki K, Nishioka T (1998) Combined hyperactive dysfunction syndrome of the cranial nerves: trigeminal neuralgia, hemifacial spasm and glossopharyngeal neuralgia—11-year experience and review. Neurosurgery 43:1351–1362
Kobata H, Kondo A, Iwasaki K (2002) Cerebellopontine angle epidermoids presenting with cranial nerve hyperactive dysfunction: pathogenesis and long-term surgical results in 30 patients. Neurosurgery 50:276–285
Lee SH, Rhee BA, Choi SK, Koh JS, Lim YJ (2010) Cerebellopontine angle tumors causing hemifacial spasm: types, incidence, and mechanism in nine reported cases and literature review. Acta Neurochir (Wien) 152(11):1901–1908
Liu P, Saida Y, Yoshioka H, Itai Y (2003) MR imaging of epidermoids at the cerebellopontine angle. Magn Reson Med Sci 2:109–115
Mallucci CL, Ward V, Carney AS, O’Donoghue GM, Robertson I (1999) Clinical features and outcomes in patients with non-acoustic cerebellopontine angle tumours. J Neurol Neurosurg Psychiatry 66(6):768–771
Mattsson P, Meijer B, Svensson M (1999) Extensive neuronal cell death following intracranial transection of the facial nerve in the adult rat. Brain Res Bull 49:333–341
Mohanty A, Venkatrama SK, Rao BR, Chandramouli BA, Jayakumar PN, Das BS (1997) Experience with cerebellopontine angle epidermoids. Neurosurgery 40(1):24–29
Netsky MG (1988) Epidermoid tumors. Review of literature. Surg Neurol 29:477–483
Obrador S, Lopez-Zafra JJ (1969) Clinical features of the epidermoids of the basal cisterns of the brain. J Neurol Neurosurg Psychiatry 32:450–454
Olivecrona H (1932) On suprasellar cholesteatomas. Brain 55:122–134
Peng Y, Yu L, Li Y, Fan J, Qiu M, Qi S (2014) Pure endoscopic removal of epidermoid tumors of the cerebellopontine angle. Childs Nerv Syst 30(7):1261–1267
Rengachary A, Kishore PRS, Watanabe I (1978) Intradiploic epidermoid cyst of the occipital bone with torcular obstruction. J Neurosurg 48(3):475–478
Rubin G, Scienza R, Pasqualin A, Rosta L, Da Pian R (1989) Craniocerebral epidermoids and dermoids. A review of 44 cases. Acta Neurochir (Wien) 97:1–16
Sabin HI, Bordi LT, Symon L (1987) Epidermoid cysts and cholesterol granulomas centered on the posterior fossa: twenty years of diagnosis and management. Neurosurgery 21:798–805
Safain MG, Dent WC, Heilman CB (2014) An endoscopic assisted retrosigmoid approach to the cerebello-pontine angle for resection of an epidermoid cyst. Neurosurg Focus 36(1 Suppl):1. doi:10.3171/2014
Safavi-Abbasi S, Di Rocco F, Bambakidis N et al (2008) Has management of epidermoid tumors of the cerebellopontine angle improved? A surgical synopsis of the past and present. Skull Base 18(2):85–98
Salazar J, Vaquero J, Saucedo G, Bravo G (1987) Posterior fossa epidermoid cysts. Acta Neurochir 85:34–39
Samii M, Draf W (eds) (1989) Surgery of the skull base. An interdisciplinary approach. Springer-Verlag, Berlin, pp 340–342
Samii M, Tatagiba M, Piquer J, Carvalho GA (1996) Surgical treatment of epidermoid cysts of the cerebellopontine angle. J Neurosurg 84(1):14–19
Schiefer TK, Link MJ (2008) Epidermoids of the cerebellopontine angle: a 20-year experience. Surg Neurol 70(6):584–590
Schroeder HW, Oertel J, Gaab MR (2004) Endoscope-assisted microsurgical resection of epidermoid tumors of the cerebellopontine angle. J Neurosurg 101(2):227–232
Shimamoto Y, Kawase T, Sasaki H, Shiobara R, Yamada F (1999) Anterior transpetrosal approach to the prepontine epidermoids. Skull Base Surg 9(2):75–80
Shulev Y, Trashin A, Gordienko K (2011) Secondary trigeminal neuralgia in cerebellopontine angle tumors. Skull Base 21(5):287–294
Talacchi A, Sala F, Alessandrini F, Turazzi S, Bricolo A (1998) Assessment and surgical management of posterior fossa epidermoid tumors: report of 28 cases. Neurosurgery 42(2):242–251
Tsuruda JS, Chew WM, Moseley ME, Norman D (1990) Diffusion-weighted MR imaging of the brain: value of differentiating between extraaxial cysts and epidermoid tumors. AJR Am J Roentgenol 155:1059–1065
Ulrich J (1964) Intracranial epidermoids. A study on their distribution and spread. J Neurosurg 21:1051–1058
Vinchon M, Pertuzon B, Lejeune JP, Assaker R, Pruvo JP, Christiaens JL (1995) Intradural epidermoid cysts of the cerebellopontine angle: diagnosis and surgery. Neurosurgery 36(1):52–56
Wakabayashi T, Tamaki N, Satoh H, Matsumoto S (1983) Epidermoid tumor presenting as painful tic convulsif. Surg Neurol 19(3):244–246
Yamakawa K, Shitara N, Genka S, Manaka S, Takakura K (1989) Clinical course and surgical prognosis of 33 cases of intracranial epidermoid tumors. Neurosurgery 24:568–573
Yasargil MG, Abernathey CD, Sarioglu AÇ (1989) Microsurgical treatment of intracranial dermoid and epidermoid tumors. Neurosurgery 24:561–567
Acknowledgments
The authors confirm that no governmental, institutional, or industrial fund or grant was given for the material of this study or to support any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Kazunari Yoshida, Tokyo, Japan
The symptoms of the brain tumor usually occur by compression or destruction of the neural tissue by the tumor mass. Epidermoid cysts in the cerebellopontine angle (CPA) are known to present variable dysfunction of the cranial nerves, including hemifacial spasm, trigeminal neuralgia, and variable cranial nerve palsy. The distinct finding, described in this manuscript, is that the mechanism of the cranial nerve dysfunction in some cases is the nerve degeneration of the distal to the strangulation site by the tumor capsule. Nobody has recognized this type of the mechanism of the cranial nerve dysfunction so far. Dr. Hasegawa and his colleagues have clearly demonstrated this mechanism of the cranial nerve dysfunction in cases of the CPA epidermoid cysts. We should notice this mechanism of the cranial nerve dysfunction and decide the appropriate timing of the management of these tumors.
Henry W. S. Schroeder, Greifswald, Germany
The authors report their experience with the microsurgical treatment of 22 CPA epidermoids. The discussion of the mechanisms of the cranial nerve deficits is interesting. In two of their patients, the authors found a strangulation of the facial and abducent nerve respectively. Although the neurological deficits improved in most of the patients, in the two patients with strangulation of the nerve, there was no satisfying improvement. The authors conclude that in the case of rapidly developing cranial nerve palsy, early surgical intervention is required to increase the chance of recovery of the cranial nerve function. This is an interesting finding that has not frequently been reported in the literature. In our series, the cranial nerve dysfunction was caused by nerve compression and distortion, but we never observed such a strangulation as described by the authors. Early nerve decompression seems to be very reasonable in case of a rapid neurological deficit.
I agree that gross total removal of the epidermoid including the cyst capsule should be the aim of the surgery for epidermoids. I always try a very aggressive resection of the cyst capsule. However, in most patients, I had to leave a small portion of the capsule behind because it was very adherent to the nerves, vessels, or brain stem. Mostly, in virgin cases, that was only a very tiny part (a few square millimeters), sometimes even at only one location in the surgical field. This is referred to as near total resection.
The authors stated that in case of contralateral or supratentorial tumor extension, the tumor would be resected through the ipsilateral corridor as much as possible and the contralateral remnant would be followed for any symptomatic or radiological growth. In epidermoid resections, I almost always use endoscope assistance to increase the rate of total removal of the cyst contents. With 30° or 45° endoscopes, one may look around the tentorium or the brain stem to the contralateral side and remove the residual tumor under endoscopic visualization in a safe way. Additionally, I found the endoscope very helpful to remove extensions of the epidermoid located in Meckel’s cave.
Thomas Kretschmer, Oldenburg, Germany
The paper’s emphasis is on a considerable case series of CP-angle epidermoids with evaluation of functional outcome and correlation to intraoperative findings. This is compared to the pertinent literature. As such, the reader finds condensed information on clinical findings, functional deficits and expectable outcomes from microsurgery. An additional mechanism for cranial nerve deficit other than neurapraxia is identified and introduced: namely “strangulation” of CN. The proposed mechanism is detailed by two cases. As the functional results in these cases were that CNs were unfavorable and distinct from other cases, the authors drew the following conclusion: A rapidly progressive CN deficit in a young patient with CP-angle epidermoid is suggestive of a strangulation mechanism. In such cases early surgery is recommended.
This is an interesting observation in a large case series, which was nicely worked-up for outcome and compared to existing series. The paper adds to our knowledge about cp-angle epidermoid, congratulation.
Rights and permissions
About this article
Cite this article
Hasegawa, M., Nouri, M., Nagahisa, S. et al. Cerebellopontine angle epidermoid cysts: clinical presentations and surgical outcome. Neurosurg Rev 39, 259–267 (2016). https://doi.org/10.1007/s10143-015-0684-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-015-0684-5