Abstract
Glioblastoma multiforme is the most common and most malignant primary brain tumour. Prognosis after diagnosis remains poor despite recent advances in adjuvant therapy. Treatment of choice is gross surgical resection and combined radio-chemotherapy with temozolomide as chemotherapeutic agent. Experimental continuous low-dose chemotherapy with temozolomide in combination with a cyclooxygenase-2 inhibitor has shown encouraging effects on progression-free survival and overall survival in patients, but leads to a high proportion of distant recurrences. Here, we describe extreme far-distant metastases along the neural axis of glioblastoma multiforme in four patients receiving metronomic antiangiogenic chemotherapy and review the literature to discuss possible mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gliobastoma multiforme is the most common primary brain tumour in adults [10]. Therapy nowadays consists of surgical removal of the tumour and concomitant radio-chemotherapy with temozolomide. The radio-chemotherapy is followed by six cycles of conventional high-dose chemotherapy according to the so-called “Stupp scheme” [35]. Despite of prolongation of progression-free and overall survival with this therapy, there is still a lot of room for improvement, since the median survival is still 14.6 months.
Recent findings of our group showed a good effect on progression-free and overall survival in patients with glioblastoma multiforme when low-dose temozolomide is continuously administered in combination with a cyclooxygenase-2 (COX-2) inhibitor [38], but this therapy regime leads to an increase in distant recurrences, i.e., recurrences >3 cm away from the original tumour location [37].
Metastases of glioblastoma multiforme are rarely reported [33, 39]. Brain stem, spinal axis and visceral organs (liver, lung, pleura), local lymph nodes, skin and bone or bone marrow occur in the literature as a target for distant metastases [21, 29, 41].
We describe far-distant seeding of glioblastoma multiforme along the neural axis after surgical tumour removal, radiotherapy and during continuous low-dose chemotherapy with temozolomide and celecoxib in four patients.
Clinical cases
Case 1
A 64-year-old female patient presented with hemianopsia and gait disturbance. Magnetic resonance imaging (MRI) scan showed a temporo-dorsal gadolinium-enhancing tumour on the right side. Tumour was resected and the histopathological diagnosis was glioblastoma multiforme. Further treatment was radiation and continuous low-dose chemotherapy with temozolomide and celecoxib. Six months later, follow-up MRI scan showed local tumour control during chemotherapy. However, a gadolinium-enhancing mass in the brain stem next to the fourth ventricle had developed (Fig. 1, upper row). Local radiotherapy was performed, and chemotherapy was continued. In the next 3 months, the patient suffered from progredient swallow and gait disturbances. Two months later, the patient died.
Upper row: T1-weighted axial MRI scan after gadolinium showing baseline tumour volume in the dorsal temporal lobe on the right side before treatment (a), after radiotherapy and antiangiogenic chemotherapy (b) with regressive contrast enhancement. Persistent local tumour control and far-distant metastasis in the brain stem (c) in T1-weighted coronal MRI scan after gadolinium. Lower row: T1-weighted axial MRI scan showing tumour volume on the left temporo-mesial side (d), after radiotherapy and antiangiogenic chemotherapy (e) with regressive contrast enhancement and extremely far-distant metastasis under persistant local tumour control in T1-weighted coronal MRI scan after gadolinium in the cervical spine (f, arrow)
Case 2
A 43-year-old female patient presented with a single seizure. MRI scan showed a right temporal gadolinium-enhancing lesion with central necrosis. After tumour resection, the histopathological diagnosis was glioblastoma multiforme. Further therapy included radiation and continuous low-dose chemotherapy with temozolomide and celecoxib. Four months later, the follow-up MRI showed local recurrent tumour. A second operation and tumour resection was performed. After second radiation therapy and further metronomic chemotherapy with temzolomide and celecoxib, the patient presented with neck pain without radicular symptoms and progressive fatigue. MRI showed frontolateral gadolinium-enhancing mass on the right side and a metastasis in the cervical spine (Fig. 1, lower row). The patient refused further therapy and died 4 weeks later.
Case 3
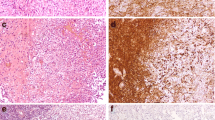
A 42-year-old male patient presented with headache and cognitive deficits. MRI scan showed a gadolinium-enhancing mass in the left temporal lobe with central necrosis. After craniotomy and tumour removal, histopathological workup diagnosed glioblastoma multiforme. Combined radio-chemotherapy, according to the Stupp scheme, was initiated. Four months later, the patient's follow-up MRI scan showed an in-loco recurrent tumour and continuous low-dose chemotherapy when temozolomide and celecoxib was initiated. Two months later, there was a regression in the temporal recurrent tumour, but the patient suffered from lower back pain. He was then administered to our department a few weeks later with radicular pain in both legs and walking impairment for 2 weeks. MRI of the spinal axis detected multiple intraspinal lesions with compression of the myelon in the thoracic spine. The patient had septic temperature, and lumbar puncture showed malignant glioma cells (Fig. 2). There was no evidence for meningitis. Two days later, the patient died in septic status, most probably due to fulminant pneumonia. The relatives rejected autopsy of the patient.
Upper row: T1-weighted axial MRI scan after gadolinium showing the resection cavity on the left temporal side after radiotherapy and antiangiogenic chemotherapy (G), initial increase (G) in contrast enhancement followed by regressive contrast enhancement in the further course (H). Lower row: T1-weighted MRI sagittal scan with fat suppression showing spinal seeding with epidural enhancement and intraspinal tumour mass (J, arrow) with a tumour cell in CSF (K)
Case 4
A 68-year-old male patient presented with aphasia; MRI scan detected left-sided gadolinium-enhancing mass lesion. After surgical gross total tumour removal, histopathological diagnosis was glioblastoma multiforme. Adjuvant radiotherapy and continuous low-dose chemotherapy with temozolomide and celecoxib was administered. The further course was uneventful, aphasia resolved postoperatively, and the MRI follow-up examinations showed good local tumour control. MRI follow up was evident for local recurrent tumour in the left temporal lobe, 9 months after surgery and diagnosis routine; furthermore, there was a new gadolinium-enhancing lesion at the pontomedullar junction on the right side (Fig. 3) suspicious for distant metastasis of glioblastoma multiforme. In the further course, the status of the patient deteriorated rapidly due to pulmonal infection, and no further therapy was started. The patient died 4 weeks later, 10 months after diagnosis of glioblastoma multiforme.
Discussion
We describe far-distant metastases of glioblastoma multiforme along the neural axis in patients treated with adjuvant metronomic antiangiogenic low-dose temozolomide and celebrex following surgical treatment and radiotherapy. This issue is illustrated by a series of four patients harbouring a far-distant relapse of their disease. The discussion further contains possible mechanisms leading to this phenomenon.
In glioblastoma multiforme, local relapse is common. In more than 90%, tumour recurrence occurs within a 2-cm margin around the resection cavity [8]. Systemic metastases of intracranial glioblastoma multiforme are rare. According to Ley, this is due to the absence of lymphatic tissue connecting the brain with the rest of the body, dense dural coating of the sinuses preventing malignant invasion, collapse of the thin-walled blood vessel due to the mass effect preventing hematogeneous spread, immune response against the malignant cells in cerebrospinal fluid (CSF) or foreign territories and the rapid progress of the disease leading to death before developing metastases [20]. Martin et al. reported that the absence of CD15 in human glioma cells partially explains the failure of brain gliomas to metastasize by precluding the adhesion of circulating neoplastic glioma cells to target organ endothelium [23].
There are factors that might promote metastases in glioblastomas, like immunosuppression by chemotherapy, radiotherapy or steroid therapy, surgical procedures and close neighbourhood to the ventricular system. There are even reports about extracranial metastases in regional lymph nodes despite of the absent lymphatic system in the brain [29]. Rare locations of systemic glioblastoma multiforme metastases are visceral organs, bone and parotid gland [9, 29, 36]. Metastases via venous drainage from the infiltrated dura in the middle fossa to pleura or lung [5] or via dural vessels in the vertebral bones occur in the literature as well [9, 25].
Spinal leptomeningeal metastases of glioblastoma are reported in the literature with an incidence range from 6% to 21% [13, 32, 43]. This data is mostly from autopsy series and published about a decade ago; therefore, the incidence of symptomatic seeding along the neural axis is difficult to estimate, since therapy has changed. In a recent series, Saito et al. reported about symptomatic spinal leptomeningeal seeding in five patients with glioblastoma multiforme and in one patient with anaplastic astrocytoma out of 68 patients with malignant astrocytoma (35 hemispheric glioblastoma, 33 hemispheric anaplastic astrocytoma) within a range from 7 to 35 months after initial diagnosis following radiotherapy and chemotherapy with ACNU. Their patients, except one, died within 4 to 8 months after diagnosis of the metastases regardless of further therapy [31].
Taken together, spontaneous systemic metastases of glioblastoma or seeding of malignant glioma cells along the spinal axis is rare; this manuscript is focused on therapy-induced factors leading to increased invasiveness and spreading of the tumour cells. In general, two main factors can be pointed out: (1) metastases related to operative procedures and (2) metastases following adjuvant antiangiogenic treatment.
Metastases related to operative procedures
After vetriculoperitoneal shunt implantation, abdominal and pulmonal metastases are known [17]. Triggered by stereotactic biopsies, craniotomies as well as spontaneous metastases in the dura mater, cranial vault, muscle and skin occur in the literature [1, 6, 30, 34]. In a recent publication, Mujic et al. described a patient with glioblastoma metastases in small bowel, pancreas and pleura. After initial surgical and radiotherapeutical treatment, their patient developed second mass lesion next to the original lesion. After biopsy and surgery of the second intracerebral lesion-metastatic seeding in small bowel, pancreas and pleura occurred 2 years after initial diagnosis of glioblastoma multiforme [27]. They discuss that initial treatment of the first tumour alters the natural barriers around the lesion, giving rise to metastases.
According to the literature, risk factors for leptomeningeal spreading of malignant glioma cells are multiple resections, ventricular entry during surgery and tumour adjacent to the ventricular system [12, 14, 41]. Second surgery or biopsy might increase the risk for hematogeneous spread of malignant cells [12, 14, 26, 41]. In patients with space-occupying mass lesions, surgical cytoreductive therapy remains the only treatment option to relieve symptoms. Furthermore, in recurrent glioblastoma multiforme, reoperation results in a median survival of 3 to 5 months without significant increase in morbidity or mortality and leads to an improvement in response to chemotherapy and/or radiotherapy, according to a review of Barbagello et al [4]. Although there might be a risk for tumour cell spread and secondary metastases up to now, there are no alternative treatment strategies for primary or recurrent space-occupying glioblastoma multiforme.
Metastases following adjuvant antiangiogenic treatment
For chronic administration of temozolomide alone, antiangiogenic effects have been shown as well in vitro and in vivo by decreasing expression levels of proangiogenic factors like HIF-1 alpha, ID-1, ID-2 and c-myc [24]. Adding COX-2 inhibition to metronomic temozolomide therapy, this treatment provides good effects on progression-free survival and even lower side effects in patients with highly vascularised glioblastomas multiforme compared to the “Stupp scheme” standard therapy [31]. However, antiangiogenic therapy seems to increase the rate of distant metastases despite local tumour control. This phenomenon has been well described under experimental conditions especially with perivascular infiltration of tumour cells “coopting” the preexisting vasculature to guarantee nutrition under hypoxic conditions [15, 18]. Taken together, these studies suggest that antiangiogenic therapy may be changing the natural history of tumours by inducing heightened tumour cell invasiveness and, in solid tumour models, enhancing distant metastasis [11, 28]. We could recently describe a rate of 62.5% of distant metastases (>3 cm of the original localisation) in a clinical series of 32 patients harbouring glioblastoma multiforme under continuous low-dose chemotherapy with temozolomide and a COX-2 inhibitor [37]. The mechanism of this increased invasiveness is not yet fully understood. Possible explanations are overexpression of EGFR under hypoxic conditions [19], activation of met-protooncogene [7], TGF-β triggered invasion [42] or overexpression of tyrosine kinase Axl [40] promoting invasion and escape of tumour cells.
In contrast to our recently published series, the patients in this case series developed a far-distant seeding along the neural axis with possible spread along the CSF pathway. There is evidence that GFAP expression in metastatic malignant glioma is less than in the original tumour [2]. Asano et al. described CSF dissemination, dural and extracranial metastases like skin metastases in 9 of 11 patients harbouring glioblastoma multiforme. The lesions occurred within an average time of 14.6 months after initial diagnosis of glioblastoma multiforme. They found a decreased expression of N-cadherin, suggesting that this leads to the spread by a decrease of cell adhesion [3]. This is in congruence with findings in the literature showing that a loss of cadherin expression in epithelial tumours is associated with a more invasive phenotype and metastasis [16, 22]. Even under conditions of systemic metronomic chemotherapy with local tumour control, metastases cannot be prevented, but the precise mechanism leading to these distant metastases is unclear up to now.
In two of our four cases, the initial tumour was controlled by chemotherapy, and distant metastasis occurred without second surgical approach. In three of our tumours, there was no intraoperative opening of the ventricular system, but gadolinium-enhancing tumour adjacent to the ventricle wall was seen. In one patient, the temporal horn of the left side ventricle was opened during surgery. In this patient, the metastasis occurred most distant from the original tumour location in the thoracic spine, and CSF was suspicious for malignant glioma cell.
In our selected cases, the localisation of the metastases was extremely far distant to the original tumour site along the neural axis representing another thread in the merging issue of increased invasiveness and seeding of malignant gliomas under antiangiogenic therapy. Further investigations are required to better understand the biological changes and possible seeding mechanisms to find an effective preventive therapy against metastases similar to other extracranial cancers. Due to the increasing survival period, metastases in glioblastoma multiforme patients will be one of the future challenges for further treatment options.
Conclusion
In glioblastoma multiforme patients, metastases have to be considered especially after tumour recurrence. In patients under antiangiogenic chemotherapy, distant metastases should be kept in mind despite of good local tumour control. Surgical treatment of recurrent glioblastoma multiforme in selected patients should always be considered, whereas in primary or recurrent malignant mass lesions adjacent to or in the ventricular system or in tumour approaches with opening of the basal cisterns, malignant cell spread along the neural axis has to be kept in mind. Follow-up MRI scans of the spine, especially when antiangiogenic chemotherapy is administered, should be performed early, when patients suffer from spinal symptoms.
The prevention and further treatment of glioblastoma multiforme metastases in these patients will be a future challenge.
References
Aichholzer M, Mazal PR, Haberler C, Dietrich W, Bertalanffy A, Roessler K et al (2001) Epidural metastasis of a glioblastoma after stereotactic biopsy: case report. Minim Invasive Neurosurg 44:175–177
Arita N, Taneda M, Hayakawa T (1994) Leptomeningeal dissemination of malignant gliomas. Incidence, diagnosis and outcome. Acta Neurochir 126:84–92
Asano K, Kubo O, Tajika Y, Takakura K, Suzuki S (2000) Expression of adherin and CSF dissemination in malignant astrocytic tumors. Neurosurg Rev 23:39–44
Barbagallo GM, Jenkinson MD, Brodbelt AR (2008) 'Recurrent' glioblastoma multiforme, when should we reoperate? Br J Neurosurg 22:452–455
Beauchesne P, Soler C, Mosnier JF (2000) Diffuse vertebral body metastasis from a glioblastoma multiforme: a technetium-99 m sestamibi single-photon emission computerized tomography study. J Neurosurg 93:887–890
Bouillot-Eimer S, Loiseau H, Vital A (2005) Subcutaneous tumoural seeding from a glioblastoma following stereotactic biopsy: case report and review of the literature. Clin Neuropathol 24:247–251
Brockmann MA, Ulbricht U, Grüner K, Fillbrandt R, Westphal M, Lamszus K (2003) Glioblastoma and cerebral microvascular endothelial cell migration in response to tumor-associated growth factors. Neurosurgery 52:1391
Burger PC, Dubois PJ, Schold SC Jr, Smith KR Jr, Odom GL, Crafts DC et al (1983) Computerized tomographic and pathologic studies of the untreated, quiescent, and recurrent glioblastoma multiforme. J Neurosurg 58:159–169
Cervio A, Piedimonte F, Salaberry J, Alcorta SC, Salvat J, Diez B et al (2001) Bone metastasis from secondary glioblastoma multiforme: a case report. J Neurooncol 52:141–148
De Angelis LM (2001) Brain tumours. N Engl J Med 344:114–123
Ebos JM, Lee CR, Cruz-Munoz W, Bjarnason GA, Christensen JG, Kerbel RS (2009) Accelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesis. Cancer Cell 15:232–239
Erlich SS, Davis RL (1978) Spinal subarachnoid metastasis from primary intracranial glioblastoma multiforme. Cancer 42:2854–2864
Fakhrai N, Czech T, Diekmann K, Fazeny-Dörner B, Birner P, Hainfellner JA et al (2004) Glioblastoma with spinal seeding. Strahlenther Onkol 180:455–457
Grabb PA, Albright AL, Pang D (1992) Dissemination of supratentorial malignant gliomas via the cerebrospinal fluid in children. Neurosurgery 30:64–71
Holash J, Wiegand SJ, Yancopoulos GD (1999) New model of tumor angiogenesis: dynamic balance between vessel regression and growth mediated by angiopoietins and VEGF. Oncogene 18:5356–5362
Juliano RL (2002) Signal transduction by cell adhesion receptors and cytoskeleton: functions of integrins, cadherins, selectins, and immunoglobulin-superfamily members. Annu Rev Pharmacol Toxicol 42:283–323
Kumar R, Jain R, Tandon V (1999) Thalamic glioblastoma with cerebrospinal fluid dissemination in the peritoneal cavity. Pediatr Neurosurg 31:242–245
Kunkel P, Ulbricht U, Bohlen P, Brockmann MA, Fillbrandt R, Stavrou D et al (2001) Inhibition of glioma angiogenesis and growth in vivo by systemic treatment with a monoclonal antibody against vascular endothelial growth factor receptor-2. Cancer Res 61:6624–6628
Lamszus K, Brockmann MA, Eckerich C, Bohlen P, May C, Mangold U et al (2005) Inhibition of glioblastoma angiogenesis and invasion by combined treatments directed against vascular endothelial growth factor receptor-2, epidermal growth factor receptor, and vascular endothelial-cadherin. Clin Cancer Res 11:4934–4940
Ley A, Campillo D, Oliveras C (1961) Extracranial metastases of glioblastoma multiforme. J Neurosurg 18:313–330
Longee DC, Friedman HS, Phillips PC, Oakes WJ, Heffez D (1991) Osteoblastic metastases from astrocytomas: a report of two cases. Med Pediatr Oncol 19:318–324
Mareel M, Leroy A (2003) Clinical, cellular, and molecular aspects of cancer invasion. Physiol Rev 83:337–376
Martin K, Akinwunmi J, Roopmai HK et al (1995) Nonexpression of CD15 by neoplastic glia: a barrier to metastasis? Anticancer Res 15:1159–1166
Mathieu V, De Nève N, Le Mercier M, Dewelle J, Gaussin JF, Dehoux M, Kiss R, Lefranc F (2008) Combining bevacizumab with temozolomide increases the antitumor efficacy of temozolomide in a human glioblastoma orthotopic xenograft model. Neoplasia 10:1383–1392
Mihara F, Rothman IM, MI NY, Kristt D (1994) Vertebral body metastasis of glioblastoma multiforme with epidural mass formation. Contrast enhanced MRI study. Clin Imaging 18:386–389
Morley TP (1959) The recovery of tumour cells from venous blood draining cerebral gliomas. Can J Surg 2:363–365
Mujic A, Hunn A, Taylor AB, Lowenthal RM (2006) Extracranial metastases of a glioblastoma multiforme to the pleura, small bowel and pancreas. J Clin Neurosci 13:677–681
Paez-Ribes M, Allen E, Hudock J et al (2009) Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15:220–231
Pasquier B, Pasquier D, Ngolet A, Panh MH, Couderc P (1980) Extraneural metastases of astrocytomas and glioblastomas: clinicopathological study and review of literature. Cancer 45:112–125
Pierallini A, Caramia F, Piatella MC, Pantano P, Santero A, Di Stefano D et al (1999) Metastasis along the stereotactic biopsy trajectory in glioblastoma multiforme. Acta Neurochir 141:1011–1012
Saito R, Kumabe T, Jokura H, Shirane R, Yoshimoto T (2003) Symptomatic spinal dissemination of malignant astrocytoma. J Neurooncol 61:227–235
Salazar OM, Rubin P (1976) The spread of glioblastoma multiforme as a determining factor in the radiation treated volume. Int J Radiat Oncol Biol Phys 2:627–63725
Smith DR, Hardman JM, Earle KM (1969) Metastasizing neuroectodermal tumours of the central nervous system. J Neurosurg 31:50–58
Steinmetz MP, Barnett GH, Kim BS, Chidel MA, Suh JH (2001) Metastatic seeding of the stereotactic biopsy tract in glioblastoma multiforme: case report and review of the literature. J Neurooncol 55:167–171
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for gliobastoma. N Engl J Med 35:978–996
Taha M, Ahamd A, Wharton S, Jellinek D (2005) Extra-cranial metastasis of glioblastoma multiforme presenting as acute parotitis. Br J Neurosurg 19:248–251
Tuettenberg J, Grobholz R, Seiz M, Brockmann MA, Lohr F, Wenz F et al (2009) Pattern of tumour recurrence in patients with glioblastoma multiforme undergoing anti-angiogenic therapy with metronomic temozolomide and COX-II-inhibition. J Cancer Res Clin Oncol 135(9):1239–1244
Tuettenberg J, Grobholz R, Korn T, Wenz F, Erber R, Vajkoczy P (2005) Continuous low dose chemotherapy plus inhibition of cyclooxygenase-2 as an antiangiogenic therapy in glioblastoma multiforme. J Cancer Res Clin Oncol 131:31–40
Utsuki S, Tanaka S, Oka H, Iwamoto K, Sagiuchi T, Fujii K (2005) Glioblastoma multiforme metastasis to the axis. Case report. J Neurosurg 102:540–542
Vajkoczy P, Knyazev P, Kunkel A, Capelle HH, Behrndt S, von Tengg-Kobligk H et al (2006) Dominant-negative inhibition of the Axl receptor tyrosine kinase suppresses brain tumor cell growth and invasion and prolongs survival. Proc Natl Acad Sci USA 103:5799–5804
Vertosick FT Jr, Selker RG (1990) Brain stem and spinal metastases of supratentorial glioblastoma multiforme: a clinical series. Neurosurgery 27:516–521
Wick W, Naumann U, Weller M (2006) Transforming growth factor-beta: a molecular target for the future therapy of glioblastoma. Curr Pharm Des 12:341–349
Yung WA, Horten BC, Shapiro WR (1980) Meningeal gliomatosis: a review of 12 cases. Ann Neurol 8:605–608
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Karl Frei, Zurich, Switzerland
Despite optimal treatment, the median survival for patients is only 12 to 15 months, and therefore, more effective and targeted therapies are still needed for this devastating disease.
Seiz et al. describes in a series of four patients far-distant relapses of GBM along the neural axis after surgical tumor removal, radiotherapy and during continous low-dose chemotherapy with temozolomide and the antiangiogenic COX-2 inhibitor celecoxib. This is in contrast to their recent findings showing a good effect on progression-free and overall survival in GBM patients when using this therapy regime, but leading to an increased rate (20/32 patients) of metastasis (>3 cm of the original localisation), but not far distant along the neural axis. The manuscript is focused on therapy-induced factors leading to increased invasiveness and spreading of the tumor cells. The two main factors are pointed out: (1) metastases related to operative procedures (in one patient, the temporal horn of the left ventricle was opened during surgery) and (2) metastases following adjuvant antiangiogenic treatment (in two patients, the initial tumor was controlled by chemotherapy and distant metastsasis occurred without second surgical approach). The phenomenon that angiogenic therapy increases the rate of distant metastasis has been described under experimental conditions especially with perivascular infiltration of tumor cells “coopting” the preexisting vasculature to guarantee nutrition under hypoxia. However, the underlying mechanisms of this increased invasiveness and escape of tumor cells are still not known.
Extra-cranial metastases from GBM are quite rare, with an incidence of <2% reported in the published literature. In general, they are asymptomatic and found only at autopsy. Factors contributing to the limited incidence of metastatic disease include the dense impassable dura, the unique extracellular matrix of the brain, the tough basement membrane that surrounds intracerebral blood vessels, the lack of true lymphatics and limited patient survival. However, by the development of novel therapies for this aggressive tumor, we may witness an increase in the incidence of neural and extraneural metastasis of these challenging tumors. As described in this study, in GBM patients, metastases have to be considered after tumor recurrence and under antiangiogenic therapy; distant metastases should be kept in mind despite of good local tumor control.
Benoit JM Pirotte, Brussels, Belgium
Marcel Seiz and coworkers from Heidelberg addressed the unusual problem of extreme far-distant metastases along the CSF pathway of glioblastoma multiforme in four patients receiving metronomic antiangiogenic chemotherapy with temozolomide and celecoxib and discussed the possible mechanisms. Indeed, seeding of glioblastoma is rarely observed by everyone of us. Usually, this represents an additional feature of darker prognosis in an individual. However, when seeding occurs, it always reminds the clinician that some mechanism of tumor progression might remain unknown or different from one tumor to another. It illustrates as well the heterogeneous phenotypic patterns of high-grade gliomas. Additionally, it also suggests that increasing patients' survival might allow other mechanisms of tumor invasiveness to take place. Herein, the authors provide an analysis of the microscopic tumor behavior. They suggest to take into account not only the mitotic activity but also the motility and invasiveness of cancer cells and emphasize that a potential effect of some treatments might be to select some other metabolic pathways and to orientate cell activity in unpredicted directions. Last but not least, the present paper has the merit to emphasize that whereas in primary or recurrent malignant tumors adjacent to or in the ventricular system or in approaches with opening basal cisterns, malignant cell seeding along the neural axis has to be kept in mind. The first consequence of that statement is that patients who died from glioblastomas are not candidates to organ donation anymore.
Rights and permissions
About this article
Cite this article
Seiz, M., Nölte, I., Pechlivanis, I. et al. Far-distant metastases along the CSF pathway of glioblastoma multiforme during continuous low-dose chemotherapy with temozolomide and celecoxib. Neurosurg Rev 33, 375–381 (2010). https://doi.org/10.1007/s10143-010-0253-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-010-0253-x