Abstract
The epidemiology of non-tuberculous mycobacteria (NTM) in Spain is largely unknown because systematic reporting is not compulsory. The aim of our study was to describe the frequency and diversity of NTM species in our region and their distribution according to the source sample, gender, and age of the patients. We performed a multicenter study of all NTM isolated in 24 public hospitals in Madrid from 2013 to 2017. A total of 6.923 mycobacteria were isolated: 4535 (65.5%) NTM, and 2.388 (34.5%) Mycobacterium tuberculosis complex (MTB). Overall, 61 different NTM species were identified. The most frequently isolated species were Mycobacterium avium complex (47.7%), M. lentiflavum (12.2%), M. gordonae (9.2%), M. fortuitum (8.9%), and M. abscessus (3.9%). Whereas MTB cases were stable during the study period, the number of NTM isolates increased considerably from 930 isolates in 2013 to 1012 in 2017; a sharp increase occurred in the last year. The rise in NTM isolates was mostly due to M. lentiflavum, M. kansasii, and M. abscessus mainly isolated from respiratory specimens in patients older than 60. The increase in isolation rate of NTM in our region is consistent with the increasing rates reported worldwide in the last decades. The rise in NTM isolates was mainly attributed to M. lentiflavum but it also should be noted the increasing of species with high pathogenic potential such as M. kansasii and M. abscessus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Non-tuberculous mycobacteria (NTM) include all Mycobacterium species other than Mycobacterium tuberculosis complex and Mycobacterium leprae. These environmental bacteria are considered opportunistic pathogens, and several species are frequently associated with human disease. Although there are significant differences in geographic distribution of NTM, the most commonly isolated species are Mycobacterium avium complex (MAC), M. gordonae, M. kansasii, M. marinum, M. xenopi, M. fortuitum, M. chelonae, and M. abscessus [1, 2].
In recent years, a considerable increase in NTM isolation rates has been observed worldwide [3,4,5]. In fact, NTM account for almost half of the total number of isolated mycobacteria [6]. The reasons for this increase are not entirely clear; it may be due to development of more sensitive molecular assays for NTM identification, but also to a true increase of NTM disease cases, or even to environmental changes in the habitats where these organisms are usually found [7].
Reporting NTM infection to public health authorities is not mandatory in most countries. Therefore, precise epidemiological data are lacking. Some European countries have published studies on the epidemiology of NTM showing an increase in NTM isolation [3, 5, 8, 9]. However, in Spain, studies about MNT human infections are scarce [10,11,12], and therefore the epidemiology of MNT infections remains unknown.
The aim of this study was to determine the frequency and diversity of NTM species in our region and their distribution according to the source sample, gender, and age of the patients.
Methods
We retrospectively reviewed all laboratory reports from 15 microbiology laboratories that perform the laboratory diagnosis of mycobacterial diseases from 24 public hospitals in Madrid (mainly an urban area with a mean population of 6.640.705 inhabitants). All patients with at least 1 isolate of NTM obtained from clinical specimens during a 5-year study period from 1st of January 2013 to 31st of December 2017 were included in the study. MTB cases from the same hospitals and period were also registered for comparison purposes.
In the case of multiple consecutive positive cultures from the same patient, only the first isolate was included. Patients’ information, including age, gender, and sample, was obtained from laboratory records.
All strains were isolated using liquid culture systems (BACTEC™ MGIT™ 960, BD, USA, and/or Bact/ALERT ® MP, Biomérieux, France), and/or solid culture media (Löwenstein-Jensen). Species level identification was performed using matrix-assisted laser desorption ionization–time of flight (MALDI-TOF) and/or molecular probes (Genotype CM/AS [Hain Lifescience, Germany). In some cases, the strain was submitted to the National Microbiology Center for a detailed identification at species level.
Research ethics approval was not necessary for retrospective studies in our Institutions when clinical charts are not reviewed, and informed consent was not required as the data were analyzed anonymously.
Results
During the study period, we identified 6.923 mycobacterial isolates from the same number of patients, 4535 (65.5%) were NTM and 2.388 (34.5%) were MTB. NTM isolates were obtained 2.061 for men and 2.474 for women (45.4 vs. 54.5%, p < 0.001) with a mean age of 61.5 years (64.4 for men and 62.3 for women).
The distribution over time of the NTM and MTB isolates is showed in Fig. 1. Whereas MTB cases were stable during the study period, the number of NTM isolates increased considerably from 930 isolates in 2013 to 1012 in 2017; a sharp increase occurred in the last year.
Table 1 shows the isolation rates of the different NTM species identified in the study. A total of 61 different NTM species were isolated. The most frequently isolated species were MAC (47.7%), M. lentiflavum (12.2%), M. gordonae (9.2%), M. fortuitum (8.9%), and M. abscessus (3.9%).
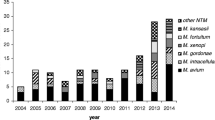
Figure 2 shows the distribution over time of the eight most prevalent species excluding MAC isolates that represented almost 50% of all MNT and which isolation rate was stable during the study period. Most species showed stable isolation rates; however, we observed that some species increased considerably during the study period. Particularly, the isolation rate of M. lentiflavum, the second most prevalent species, increased from 73 isolates in 2014 to 161 in 2017. Although M. kansasii only represented 1.5% of total MNT isolates, it should be noted that the number of isolates increased from 5 in 2013 to 22 in 2017. Regarding rapidly growing mycobacteria (RGM), M. abscessus also increased more than 2.5 times between 2013 and 2017.
NTM isolates were obtained mainly from respiratory samples (89.8%); sputum was the most common respiratory sample, followed by bronchial aspirate (BAS), bronchoalveolar lavage (BAL), and pleural effusion. Most extrapulmonary strains were isolated from urine, followed by lymph node biopsies, skin biopsies, and blood (Table 2).
When the data were analyzed by sex and age range, we observed that NTM infections were more common in women than in men (54.5 vs. 45.4%, p < 0.001). Regarding to age distribution, NTM isolates were largely obtained from patients older than 60 years (n = 3151, 69.5%). The most commonly isolated species from patients aged 1–10 years old were MAC and M. lentiflavum.
Discussion
NTM diseases detection and communication are not compulsory in Spain; therefore, the overall epidemiological situation of NTM in our setting is largely unknown. To the best of our knowledge, this is the largest study of NTM epidemiology conducted in Madrid (one of the most populated regions in Spain) during several years when the same microbiological protocols were used. Our data show that whereas the number of MTB cases was stable during the study period, an increase in isolation rate of NTM in our setting was detected, especially in the last year of the study.
More isolates of NTM were obtained from woman than from men, a data different from those of other series [3, 6, 13–14], but similar to other reports [4]. According to other previous studies, the highest number of isolates (approximately 70%) was obtained from respiratory samples of patients older than 60 years [14].
A total of 61 different NTM species were identified, and only 2% of the isolates could not be identified as valid species. MAC, M. lentiflavum, M. gordonae, M. fortuitum, and M. abscessus were the most frequently isolated species. The distribution was similar to that reported in previous European studies [3, 9, 10, 13], but differently to those reports, we found a high rate of M. lentiflavum detection. In fact, it was the second prevalent species, comprising 12% of all the NTM in our setting. In Europe, Portugal has also recorded high rates of M. lentiflavum isolates (6%) [8]. We have found that M. lentiflavum isolates were obtained mainly from respiratory specimens where these mycobacteria are commonly considered a colonizer or contaminant. However, it should be noted that 11% of the isolates of this species were from lymph node biopsies, mostly in patients aged 1–10 years old. This observation is in agreement with recent studies [11, 15, 16] that describe M. lentiflavum as an important etiologic agent of cervical lymphadenitis in children.
Remarkably, M. kansasii, which is usually considered a pulmonary pathogen, increased considerably from 5 isolates in 2013 to 24 in 2017. Despite this increase, it only represents 1.5% of all MNT isolates, a difference with other geographical areas where this species is frequently isolated [8].
According to previous reports, we observed that M. fortuitum and M. abscessus were the most frequently isolated RGM [3, 9, 13]. Whereas the isolation rates of M. fortuitum remained stable during the study, the number of M. abscessus isolates gradually increased from 20 isolates in 2013 to 50 in 2017. This fact must be analyzed in the context of considering M. abscessus as an emerging pathogen [17], especially among some specific populations like cystic fibrosis patients, as many reports in the literature has shown in the recent years [18, 19], even with the detection of hyperpathogenic clones that have been described worldwide [20, 21]. Because this is a retrospective microbiological study, we are not able to analyze neither the strains nor the clinical data, but the increase in the number of isolates of this pathogenic species could be a matter of concern for the next years.
It must be noted that Madrid is mainly an urban region with a continental climate. This must be taken into account for comparison purposes of MNT isolation rates with other regions of a rural environment that have wetter climates. Since NTM are environmental organisms, these data are of importance, because it has been speculated that changes in climate or in the water system disinfection could affect the number or frequency of the different isolated species [2, 7].
In conclusion, our data show an increase in NTM isolation rate in our region that is according to the general increase in NTM infections reported worldwide in the last decades. Unexpectedly, M. lentiflavum, which is considered a nonpathogenic environmental contaminant, was the second most commonly isolated species. The increase in NTM isolation frequency was mainly attributed to M. lentiflavum, but it also should be noted the increasing of M. kansasii and M. abscessus, common pathogens that could be of great importance in the next future.
Further studies involving the clinical significance of increased isolation rate of NTM from clinical samples to better understand changes in the epidemiology of NTM and its impact on clinical practice are needed.
References
Brown-Elliott BA, Nash KA, Wallace RJ Jr (2012) Antimicrobial susceptibility testing, drug resistance mechanisms, and therapy of infections with nontuberculous mycobacteria. Clin Microbiol Rev 25(3):545–582
Falkinham JO 3rd (1996) Epidemiology of infection by nontuberculous mycobacteria. Clin Microbiol Rev 9(2):177–215
Moore JE, Kruijshaar ME, Ormerod LP, Drobniewski F, Abubakar I (2010) Increasing reports of non-tuberculous mycobacteria in England, Wales and Northern Ireland, 1995-2006. BMC Public Health 10:612
Ringshausen FC, Apel RM, Bange FC, de Roux A, Pletz MW, Rademacher J, Suhling H, Wagner D, Welte T (2013) Burden and trends of hospitalisations associated with pulmonary non-tuberculous mycobacterial infections in Germany, 2005-2011. BMC Infect Dis 13:231
van der Werf MJ, Kodmon C, Katalinic-Jankovic V, Kummik T, Soini H, Richter E, Papaventsis D, Tortoli E, Perrin M, van Soolingen D, Zolnir-Dovc M, Ostergaard Thomsen V (2014) Inventory study of non-tuberculous mycobacteria in the European Union. BMC Infect Dis 14:62
Martinez Gonzalez S, Cano Cortes A, Sota Yoldi LA, Garcia Garcia JM, Alba Alvarez LM, Palacios Gutierrez JJ (2017) Non-Tuberculous mycobacteria. An emerging threat? Arch Bronconeumol 53(10):554–560
Falkinham JO 3rd (2016) Current epidemiologic trends of the nontuberculous mycobacteria (NTM). Curr Environ Health Rep 3(2):161–167
Hoefsloot W, van Ingen J, Andrejak C, Angeby K, Bauriaud R, Bemer P, Beylis N, Boeree MJ, Cacho J, Chihota V, Chimara E, Churchyard G, Cias R, Daza R, Daley CL, Dekhuijzen PN, Domingo D, Drobniewski F, Esteban J, Fauville-Dufaux M, Folkvardsen DB, Gibbons N, Gomez-Mampaso E, Gonzalez R, Hoffmann H, Hsueh PR, Indra A, Jagielski T, Jamieson F, Jankovic M, Jong E, Keane J, Koh WJ, Lange B, Leao S, Macedo R, Mannsaker T, Marras TK, Maugein J, Milburn HJ, Mlinko T, Morcillo N, Morimoto K, Papaventsis D, Palenque E, Paez-Pena M, Piersimoni C, Polanova M, Rastogi N, Richter E, Ruiz-Serrano MJ, Silva A, da Silva MP, Simsek H, van Soolingen D, Szabo N, Thomson R, Tortola Fernandez T, Tortoli E, Totten SE, Tyrrell G, Vasankari T, Villar M, Walkiewicz R, Winthrop KL, Wagner D (2013) The geographic diversity of nontuberculous mycobacteria isolated from pulmonary samples: an NTM-NET collaborative study. Eur Respir J 42(6):1604–1613
Rindi L, Garzelli C (2016) Increase in non-tuberculous mycobacteria isolated from humans in Tuscany, Italy, from 2004 to 2014. BMC Infect Dis 16:44
Martin-Casabona N, Bahrmand AR, Bennedsen J, Thomsen VO, Curcio M, Fauville-Dufaux M, Feldman K, Havelkova M, Katila ML, Koksalan K, Pereira MF, Rodrigues F, Pfyffer GE, Portaels F, Urgell JR, Rusch-Gerdes S, Tortoli E, Vincent V, Watt B (2004) Non-tuberculous mycobacteria: patterns of isolation. A multi-country retrospective survey. Int J Tuberc Lung Dis 8(10):1186–1193
Miqueleiz-Zapatero A, Santa Olalla-Peralta C, Guerrero-Torres MD, Cardenoso-Domingo L, Hernandez-Milan B, Domingo-Garcia D (2018) Mycobacterium lentiflavum as the main cause of lymphadenitis in pediatric population. Enferm Infecc Microbiol Clin 36(10):640–643
Santin M, Barrabeig I, Malchair P, Gonzalez-Luquero L, Benitez MA, Sabria J, Palau-Benavent M, Canete C, Lloret-Queralto JA, Grijota-Camino MD, Dorca J, Alcaide F (2018) Pulmonary infections with nontuberculous mycobacteria, Catalonia, Spain, 1994-2014. Emerg Infect Dis 24(6):1091–1094
Jankovic M, Samarzija M, Sabol I, Jakopovic M, Katalinic Jankovic V, Zmak L, Ticac B, Marusic A, Obrovac M, van Ingen J (2013) Geographical distribution and clinical relevance of non-tuberculous mycobacteria in Croatia. Int J Tuberc Lung Dis 17(6):836–841
Russell CD, Claxton P, Doig C, Seagar AL, Rayner A, Laurenson IF (2014) Non-tuberculous mycobacteria: a retrospective review of Scottish isolates from 2000 to 2010. Thorax 69(6):593–595
Jimenez-Montero B, Baquero-Artigao F, Saavedra-Lozano J, Tagarro-Garcia A, Blazquez-Gamero D, Cilleruelo-Ortega MJ, Ramos-Amador JT, Gale-Anso I, Marin N, Gomez-Garcia R, Santiago-Garcia B, Garrido J, Lopez G (2014) Comparison of Mycobacterium lentiflavum and Mycobacterium avium-intracellulare complex lymphadenitis. Pediatr Infect Dis J 33(1):28–34
Piersimoni C, Goteri G, Nista D, Mariottini A, Mazzarelli G, Bornigia S (2004) Mycobacterium lentiflavum as an emerging causative agent of cervical lymphadenitis. J Clin Microbiol 42(8):3894–3897
Bryant JM, Grogono DM, Rodriguez-Rincon D, Everall I, Brown KP, Moreno P, Verma D, Hill E, Drijkoningen J, Gilligan P, Esther CR, Noone PG, Giddings O, Bell SC, Thomson R, Wainwright CE, Coulter C, Pandey S, Wood ME, Stockwell RE, Ramsay KA, Sherrard LJ, Kidd TJ, Jabbour N, Johnson GR, Knibbs LD, Morawska L, Sly PD, Jones A, Bilton D, Laurenson I, Ruddy M, Bourke S, Bowler IC, Chapman SJ, Clayton A, Cullen M, Daniels T, Dempsey O, Denton M, Desai M, Drew RJ, Edenborough F, Evans J, Folb J, Humphrey H, Isalska B, Jensen-Fangel S, Jonsson B, Jones AM, Katzenstein TL, Lillebaek T, MacGregor G, Mayell S, Millar M, Modha D, Nash EF, O'Brien C, O'Brien D, Ohri C, Pao CS, Peckham D, Perrin F, Perry A, Pressler T, Prtak L, Qvist T, Robb A, Rodgers H, Schaffer K, Shafi N, van Ingen J, Walshaw M, Watson D, West N, Whitehouse J, Haworth CS, Harris SR, Ordway D, Parkhill J, Floto RA (2016) Emergence and spread of a human-transmissible multidrug-resistant nontuberculous mycobacterium. Science 354(6313):751–757
Gilljam M, Schersten H, Silverborn M, Jonsson B, Ericsson Hollsing A (2010) Lung transplantation in patients with cystic fibrosis and Mycobacterium abscessus infection. J Cyst Fibros 9(4):272–276
Leung JM, Olivier KN (2013) Nontuberculous mycobacteria: the changing epidemiology and treatment challenges in cystic fibrosis. Curr Opin Pulm Med 19(6):662–669
Bryant JM, Grogono DM, Greaves D, Foweraker J, Roddick I, Inns T, Reacher M, Haworth CS, Curran MD, Harris SR, Peacock SJ, Parkhill J, Floto RA (2013) Whole-genome sequencing to identify transmission of Mycobacterium abscessus between patients with cystic fibrosis: a retrospective cohort study. Lancet 381(9877):1551–1560
Harris KA, Kenna DT, Blauwendraat C, Hartley JC, Turton JF, Aurora P, Dixon GL (2012) Molecular fingerprinting of Mycobacterium abscessus strains in a cohort of pediatric cystic fibrosis patients. J Clin Microbiol 50(5):1758–1761
Acknowledgments
Parts of this manuscript were communicated at the 29th ECCMID (Amsterdam, 2019) and the 23rd Congress of SEIMC (Madrid, 2019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
López-Roa, P., Aznar, E., Cacho, J. et al. Epidemiology of Non-Tuberculous Mycobacteria isolated from clinical specimens in Madrid, Spain, from 2013 to 2017. Eur J Clin Microbiol Infect Dis 39, 1089–1094 (2020). https://doi.org/10.1007/s10096-020-03826-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-020-03826-7