Abstract
Vancomycin-resistant Enterococcus (VRE) is considered to be a major nosocomial pathogen that results in serious morbidity and mortality worldwide. Limited information is available concerning the prevalence of VRE infections in Iran. We carried out a systematic search by using different electronic databases including: Medline (via PubMed), Embase, Web of Science, and the Iranian Database. Meta-analysis was performed using comprehensive meta-analysis software. The meta-analyses revealed that the prevalence of VRE infections was 9.4 % (95 % confidence interval [95 % CI] 7.3–12) among culture-positive cases for Enterococcus species. The prevalence of VRE in Iran is compared with the results of developed countries. The prevalence of VRE in Germany, the United Kingdom (UK), and Italy was 11.2 %, 8.5–12.5 %, and 9 % respectively. Additionally, the frequency of vancomycin resistance among E. faecalis isolates was higher than for E. faecium. The results of this study indicate that a comprehensive infection control strategy based on hand hygiene, educating the hospital staff members, providing clinical guidance and principles for the appropriate use of antibiotics, sanitizing the hospitals, contact precautions, and active surveillance systems on the basis of international criteria is urgently needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Enterococcus is a common commensal bacterium originating from the human gastrointestinal tract and may cause serious infections in humans under certain conditions [1]. Enterococci constitute the second most common cause of nosocomial urinary tract infections [2]. This species has been recognized as the third leading cause of healthcare-associated bloodstream infections in the USA, generating an enormous cost burden for the healthcare system [3]. Enterococci have gained resistance to almost the entire antimicrobial spectrum, including vancomycin, undoubtedly the most potential antimicrobial agent as a treatment for enterococcal infections [4]. In 1986, vancomycin-resistant Enterococcus (VRE) was initially was reported from clinical isolates in the UK and France. According to the data, a year later VRE was documented in US hospitals and emerged as one of the crucial hospital-acquired infectious agents [4]. In 2004, VRE strains were initially reported in Iran; and presently pertain to be a highly relevant nosocomial pathogen [5, 6]. It is quite significant to mention that VRE has been identified as a global public health dilemma, as the therapeutic options for the treatment are utterly limited [3]. Infections due to VRE are significantly associated with an increased rate of morbidity, mortality, prolonged hospitalization, and elevated cost burden [7]. Based on the afore-mentioned data, the prevalence rates of VRE in clinical samples has been reported in several scientific works in Iran [5, 8–12]. However, most of these studies have shown local information, and a comprehensive analysis from different parts of Iran has not yet been performed. The aim of this surveillance review was to assess the prevalence of VRE in Iran by using a systematic review and meta-analysis based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISM) statement [13].
Materials and methods
Search strategies
We performed a systematic search by using different electronic databases including: Medline (via PubMed), Embase, Web of Science, and the Iranian Database from January 2000 to January 2016.
“Enterococcus,” “vancomycin,” vancomycin-resistant enterococci, and related terms were used as scientific keywords in this survey. Two independent reviewers screened the titles and abstracts for its relevance. The original articles were obtained and assessed in detail for inclusion. The scientific studies had to meet all the following criteria for inclusion: a standard method had to be used to detect VRE, reported data on the number of VRE, and enrolled patients who were hospitalized and presented with symptoms of VRE. According to the Clinical and Laboratory Standards Institute (CLSI) guidelines [14], broth microdilution, agar dilution, and E-test are all methods of accurately detecting vancomycin resistance in enterococci.
We excluded studies that did not report the number of VRE. In addition, we disqualified the reports that particularly discuss specific groups of samples. For instance, articles that only focused on stool or urine samples were eliminated. We excluded reports that did not use standardized methods. We neglected reports that did not have abstracts in English. Furthermore, to minimize the potential bias caused by an inadequate sample size, articles with less than 100 subjects were omitted.
Data extraction and definitions
The following details were extracted from the manuscripts: the first author’s name, the publication year, the date of the study, the study setting, the number of cases involved in the studies, the study method, the source of isolates, the sample size, and the prevalence of VRE infections. Two independent reviewers extracted all data from the articles included and the results were reviewed by the third reviewer. Inconsistencies between the reviewers were resolved by a general consensus.
Quality assessment of studies
Two reviewers independently assessed the study quality by using a checklist, which was provided by the Joanna Briggs Institute [15].
Statistical analysis
Meta-analysis was performed using comprehensive meta-analysis (Biostat V2.2) software. We reported the amount of residual heterogeneity by using the t 2 statistic and the Q statistic to test the heterogeneity between the inquiries. To assess any possible publication bias, Begg’s rank correlation and Egger’s weighted regression methods were used in combination with a funnel plot (p < 0.05 was considered indicative of statistically significant publication bias).
Results
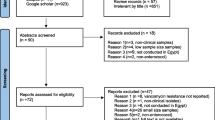
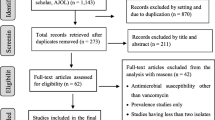
We identified 296 articles, 20 of which fulfilled the criteria mentioned (Table 1). Figure 1 presents the reasons why some records were excluded because of the assessment of title, abstract, and full article texts.
Based on the 20 selected articles, the pooled prevalence of VRE was estimated at 9.4 % (95 % CI 7.3–12.0) among culture-positive cases of VRE in Iran. However, the results were found to be periodically heterogeneous (I2 = 85.0; p = 0.00 test for heterogeneity).
The prevalence of E. faecalis and E. faecium was 77.5 % (95 % CI 60.0–84.0) and 22 % (95 % CI 15.6–30.0) respectively. Details of the meta-analysis for the sub-groups can be found in Table 2. A forest plot for the meta-analysis of VRE isolates is depicted in Fig. 2. As shown in Table 2 and Fig. 3, no evidence of publication bias was observed (p = 0.1 for Begg’s rank correlation analysis; p = 0.2 for Egger’s weighted regression analysis).
Discussion
To our knowledge, this study is the first comprehensive systematic review on the prevalence of VRE in Iran. Based on the meta-analysis results, the overall estimate of VRE prevalence in Iran was 9.4 %. The prevalence of VRE in Iran is compared with results from developed countries [16–20]. The prevalence of VRE in Germany, the UK, and Italy was 11.2 %, 8.5–12.5 % and 9 % respectively [16–20]. Several factors may explain the prevalence of VRE in Iran. First, infection control programs are not very effective in Iran. For instance, hand hygiene is the most important infection control measure for reducing transmission of VRE in hospitals [21, 22]. In most Iranian university hospitals, compliance with hand hygiene among healthcare workers is quite poor. Heavy workload, insufficient numbers of healthcare staff, limited infrastructures (lack of sinks, hygiene products that are difficult to access), and behavioral aspects are the major reasons for noncompliance [23–25]. Second, wards with a high bed occupancy rate are the leading cause of poor attention to infection control protocols in Iranian university hospitals. Third, the lack of isolation rooms could be considered another concern in Iranian hospitals, as patients infected with VRE are admitted to the multi-bed rooms together with uninfected patients. Fourth, in developing countries such as Iran, the inappropriate use of antibiotics and empiric therapies by physicians is prevalent. As a consequence, there is an increasing rate of infections with VRE in different parts of Iran [24]. Fifth, sanitizing the hospital environment is generally ineffective [24]. Thus, VRE can directly transfer through contact with contaminated surfaces [3]. Finally, the disk diffusion agar method (Kirby–Bauer method) is currently extensively used by medical microbiology laboratories in Iran. Although not very accurate, it is better to use the minimum inhibitory concentration method of detection.
Stratified analyses were conducted in the present study with regard to the geographic areas. The majority of studies included were performed in Iran’s capital (Tehran), where a relatively high prevalence of VRE has been reported. These findings suggest that the many tertiary hospitals in Tehran might play a referral role in almost all regions and most patients from different parts of country are referred to these centers for diagnosis, treatment, and further follow-up. According to our analysis, the rate of vancomycin resistance among E. faecalis isolates was higher than for E. faecium. This finding could be explained by the fact that E. faecalis was the dominant species isolated from infections [5, 9, 10].
In the present review article, we were faced with several constraints. First, the studies could not fully indicate the prevalence of VRE infections in Iran as the magnitude of VRE infections was not identified in different regions of the country. Second, we only considered published articles in the current meta-analysis, exactly like any other meta-analysis; thus the potential for publication bias had to be considered as well. Third, heterogeneity was observed among the studies included.
In conclusion, the present systematic review summarized the prevalence of VRE in Iran. According to the results, the hospital’s infection control program in Iran has been dramatically ineffective. Therefore, a comprehensive infection control strategy based on hand hygiene, educating the hospital staff members, sanitizing the hospitals, taking precautions with contact, and an active surveillance system according to international criteria should be enforced in the community.
References
Prematunge C, MacDougall C, Johnstone J, Adomako K, Lam F, Robertson J et al (2016) VRE and VSE bacteremia outcomes in the era of effective VRE therapy: a systematic review and meta-analysis. Infect Control Hosp Epidemiol 37(1):26–35
Cetinkaya Y, Falk P, Mayhall CG (2000) Vancomycin-resistant enterococci. Clin Microbiol Rev 13(4):686–707
Balli EP, Venetis CA, Miyakis S (2014) Systematic review and meta-analysis of linezolid versus daptomycin for treatment of vancomycin-resistant enterococcal bacteremia. Antimicrob Agents Chemother 58(2):734–739
Corso AC, Gagetti PS, Rodriguez MM, Melano RG, Ceriana PG, Faccone DF et al (2007) Molecular epidemiology of vancomycin-resistant Enterococcus faecium in Argentina. Int J Infect Dis 11(1):69–75
Emaneini M, Aligholi M, Aminshahi M (2008) Characterization of glycopeptides, aminoglycosides and macrolide resistance among Enterococcus faecalis and Enterococcus faecium isolates from hospitals in Tehran. Pol J Microbiol 7(2):173–178
Feizabadi MM, Asadi S, Zohari M, Gharavi S, Etemadi G (2004) Genetic characterization of high-level gentamicin-resistant strains of Enterococcus faecalis in Iran. Can J Microbiol 50(10):869–872
Cosgrove SE (2006) The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis 42 [Suppl 2]:S82–S89
Dadfarma N, Imani Fooladi AA, Oskoui M, Mahmoodzadeh Hosseini H (2013) High level of gentamicin resistance (HLGR) among enterococcus strains isolated from clinical specimens. J Infect Public Health 6(3):202–208
Sharifi Y, Hasani A, Ghotaslou R, Varshochi M, Hasani A, Soroush MH et al (2012) Vancomycin-resistant enterococci among clinical isolates from north-west Iran: identification of therapeutic surrogates. J Med Microbiol 61(Pt 4):600–602
Shokoohizadeh L, Mobarez AM, Zali MR, Ranjbar R, Alebouyeh M, Sakinc T et al (2013) High frequency distribution of heterogeneous vancomycin resistant Enterococcus faecium (VREfm) in Iranian hospitals. Diagn Pathol 8:163
Shokri D, Zaghian S, Khodabakhsh F, Fazeli H, Mobasherizadeh S, Ataei B (2014) Antimicrobial activity of a UV-stable bacteriocin-like inhibitory substance (BLIS) produced by Enterococcus faecium strain DSH20 against vancomycin-resistant Enterococcus (VRE) strains. J Microbiol Immunol Infect 47(5):371–376
Talebi M, Sadeghi J, Pourshafie MR (2014) Molecular characterization of vancomycin-resistant Enterococcus faecium isolated from intensive care units. Curr Microbiol 68(5):615–620
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264–269
Performance Standards for Antimicrobial Susceptibility Testing (2014) Twenty-fourth informational supplement, CLSI, M100-S24
Joanna Briggs Institute (2014) Joanna Briggs Institute Reviewers’ Manual: 2014 edition. The Joanna Briggs Institute Australia
Werner G, Coque TM, Hammerum AM, Hope R, Hryniewicz W, Johnson A et al (2008) Emergence and spread of vancomycin resistance among enterococci in Europe. Euro Surveill 13(47):1–11
The European Antimicrobial Resistance Surveillance System. EARSS results [database on the internet]. [Cited 20.10.2008]. Available from: http://www.rivm.nl/earss/result/
Brown DF, Hope R, Livermore DM, Brick G, Broughton K, George RC et al (2008) Non-susceptibility trends among enterococci and non-pneumococcal streptococci from bacteraemias in the UK and Ireland, 2001–06. J Antimicrob Chemother 62 [Suppl 2]:ii75–85
British Society for Antimicrobial Chemotherapy (BSAC). Resistance Surveillance Project. Available from: http://www.bsac.org.uk/resistance_surveillance.cfm
Fontana R, Ligozzi M, Mazzariol A, Veneri G, Cornaglia G (1998) Resistance of enterococci to ampicillin and glycopeptide antibiotics in Italy. The Italian Surveillance Group for Antimicrobial Resistance. Clin Infect Dis 27 [Suppl 1]:S84–S86
Larson EL, Early E, Cloonan P, Sugrue S, Parides M (2000) An organizational climate intervention associated with increased handwashing and decreased nosocomial infections. Behav Med 26(1):14–22
Zhang S, Sun X, Chang W, Dai Y, Ma X (2015) Systematic review and meta-analysis of the epidemiology of vancomycin-intermediate and heterogeneous vancomycin-intermediate Staphylococcus aureus isolates. PLoS One 10(8):e0136082
Alp E, Leblebicioglu H, Doganay M, Voss A (2011) Infection control practice in countries with limited resources. Ann Clin Microbiol Antimicrob 10:36
Mamishi S, Pourakbari B, Teymuri M, Babamahmoodi A, Mahmoudi S (2014) Management of hospital infection control in Iran: a need for implementation of multidisciplinary approach. Osong Public Health Res Perspect 5(4):179–186
Pittet D, Allegranzi B, Storr J, Bagheri Nejad S, Dziekan G, Leotsakos A et al (2008) Infection control as a major World Health Organization priority for developing countries. J Hosp Infect 68(4):285–292
Akhi MT, Farzaneh F, Oskouei M (2009) Study of enterococcal susceptibility patterns isolated from clinical specimens in Tabriz, Iran. Pak J Med Sci 25(2):211–216
Aleyasin A, Mobarez AM, Sadeghizadeh M, Hosseini Doust R, Khoramabadi N (2007) Resistance to vancomycin in Enterococcus faecium and faecalis clinical isolates. Pak J Med Sci 23(3):390–393
Talebi M, Pourshafie MR, Oskouii M, Eshraghi SS (2008) Molecular analysis of vanHAX element in vancomycin resistant enterococci isolated from hospitalized patients in Tehran. Iran Biomed J 12(4):223–228
Pourshafie MR, Talebi M, Saifi M, Katouli M, Eshraghi S, Kuhn I et al (2008) Clonal heterogeneity of clinical isolates of vancomycin-resistant Enterococcus faecium with unique vanS. Trop Med Int Health 13(5):722–727
Japoni A, Farshad S, Ziyaeyan M, Ziaian S (2009) Detection of Van-positive and negative vancomycin resistant enterococci and their antibacterial susceptibility patterns to the newly introduced antibiotics. Pak J Biol Sci 12(11):844–851
Yasliani S, Mohabati Mobarez A, Hosseini Doust R, Satari M, Teymornejad O (2009) Linezolid vancomycin resistant Enterococcus isolated from clinical samples in Tehran hospitals. Indian J Med Sci 63(7):297–302
Ghasemi A, Moniri R, Mosavi GH (2009) Investigation of multi-drug resistant strains of Enterococcus faecalis isolated from clinical samples in Shahid Beheshti Hospital and Kashan during 2008. Iran J Med Microbiol 3:21–26, Persian
Hosseinizadeh A, Abtahi H, ShojaPour M, Akbari M, Nazari R, Sofian M (2012) Prevalence and antimicrobial susceptibility pattern of vancomycin resistant enterococci isolated from clinical sample of educational hospitals in Arak. Arak Medical Univ J 15(64):11–16, Persian
Nikouie M, Meidani M, Khorvash F, Karimi M, Parsaei P (2014) Evaluation of the frequency of phenotype and genotype of Van A and Van B genes in vancomycin resistant enterococcus isolated from clinical sample of Alzahra Hospitals in Isfahan. J Shahrekord Univ Med Sci 16(3):61–69, Persian
Mirzaei B, Babaei R, Asiabar AP, Bameri Z (2015) Detection of both vanA & vanB genes in vanA phenotypes of Enterococci by Taq Man RT-PCR. Braz J Microbiol 46(1):161–165
Balaei Gajan E, Shirmohammadi A, Aghazadeh M, Alizadeh M, Sighari Deljavan A, Ahmadpour F (2013) Antibiotic resistance in Enterococcus faecalis isolated from hospitalized patients. J Dent Res Dent Clin Dent Prospects 7(2):102–104
Naserpour Farivar T, Najafipour R, Johari P, Aslanimehr M, Peymani A, Jahani Hashemi H et al (2014) Development and evaluation of a Quadruplex Taq Man real-time PCR assay for simultaneous detection of clinical isolates of Enterococcus faecalis, Enterococcus faecium and their vanA and vanB genotypes. Iran J Microbiol 6(5):335–340
Abbasi S, Zamanzad B (2015) Genotypic and phenotypic characteristics of vancomycin-resistant Enterococcus isolated from clinical specimens of patients in Shahrekord. J Mazandaran Univ Med Sci 259(122):98–106, Persian
Acknowledgements
This research has been supported by Tehran University of Medical Sciences and Health Services grant no. 25177/93-02-30.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None.
Conflicts of interest
None.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Rights and permissions
About this article
Cite this article
Emaneini, M., Hosseinkhani, F., Jabalameli, F. et al. Prevalence of vancomycin-resistant Enterococcus in Iran: a systematic review and meta-analysis. Eur J Clin Microbiol Infect Dis 35, 1387–1392 (2016). https://doi.org/10.1007/s10096-016-2702-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-016-2702-0