Abstract
Necrotizing fasciitis is a soft-tissue infection with a high risk of fatality. Infection with Vibrio vulnificus can lead to development of necrotizing fasciitis and primary septicemia, and occurs mostly in immunocompromised host-associated diseases such as hepatic disease, diabetes mellitus, chronic renal insufficiency, and adrenal insufficiency. Early recognition and treatment of the infection, which are unclear, are vital to patient welfare. We studied the disease epidemiology and reviewed the prognosis and clinical features of patients treated using our developed protocol. Clinical manifestations and outcomes were retrospectively analyzed for 67 patients with V. vulnificus-mediated necrotizing fasciitis and sepsis. All patients who had contacted seawater or raw seafood with positive culture for vibrio were included. Patients were divided into two groups based on the timing of first fasciotomy and injury; within 24 h (group A) and beyond 24 h (group B). Twenty-three of the 67 patients (40%) had hepatic disease, 17 (25.4%) had chronic renal insufficiency, and 12 (17.9%) exhibited adrenal insufficiency. The most common site of infection was the upper extremity (74.7%). Group B presented with more clinical symptoms including fever (p = 0.02), hemorrhagic bullae (p < 0.0001), and shock (p = 0.007). Group A patients exhibited enhanced survival compared to group B (in hospital mortality: 4.9% vs. 23%; p = 0.005). We conclude that early and appropriate diagnosis for V. vulnificus infection should be made, especially in patients presenting with atypical clinical findings. Early fasciotomy within 24 h remains the highest priority and decreases the mortality rate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Infection caused by the Vibrio vulnificus, initially described in 1979 [1], remains one of the most invasive and rapidly fatal human infections yet documented. V. vulnificus is a naturally occurring, Gram-negative, halophilic bacterium that is a free-living inhabitant of estuarine and marine environments throughout the world. Infections caused by V. vulnificus exhibit a seasonable pattern, with a peak in summer [2, 3].
Vibrio species have been found in warm coastal waters ranging in temperature from 9–21°C in geographically diverse regions that include the Gulf of Mexico, South America, Asia (Thailand, Taiwan, Hong Kong), and Australia. The bacterium is frequently found in oysters, crustaceans, and shellfish. Infection of wounds by the microbe occurs by direct invasion or contact with contaminated seawater [3, 4]. The estimated prevalence of V. vulnificus infection for residents of Taiwan ranges from 0.354–1.237 per million people, with the reported number of cases increasing in recent years [5].
Patients are often middle-aged to elderly, and are compromised with other co-morbidities, particularly chronic liver disease, diabetes mellitus, adrenal insufficiency, diseases that compromise the immune system, hemochromatosis, thalassemia, or chronic renal insufficiency [6, 7, 8]. The infection presents hours after introduction of the organism and is manifest as swelling, pain, tenderness, and blistering at the site of infection. Predominant skin lesions include hemorrhagic bulla, purpura, petechia, or cellulitis that can progress to necrotic ulcers. Rapidly spreading gangrenous tissue death (necrotizing fasciitis) necessitates prompt treatment, which can include limb amputation, to halt the infection and minimize morbidity and mortality. Muscle and skin necrosis, further deterioration in the patient’s general condition, multiorgan dysfunction, and disseminated intravascular coagulation will tend to develop. V. vulnificans septicemia is a prominent aspect of the infection, which can prelude multi-organ failure within 24 h. Finally, a hallmark of V. vulnificans growth is the production of virulent and cytotoxic factors, which can quickly lead to fatal sepsis. Delay in recognition and treatment may result in a poor outcome [9].
The relatively high susceptibility of people in Taiwan to V. vulnificus infections has stimulated interest in the development of an optimal therapeutic regimen or algorithm. The purpose of this retrospective study was to evaluate the risk factors for mortality in a large group of Taiwanese patients with V. vulnificus-mediated necrotizing fasciitis in a single institute, and to examine the current incidence and mortality of this disease in this institute.
Patients and methods
We retrospectively reviewed the medical records of all patients treated for V. vulnificus-mediated necrotizing fasciitis with sepsis at National Cheng Kung University Hospital, Tainan, Taiwan between February 1997 and October 2005. Necrotizing fasciitis was defined by the presence of necrosis of subcutaneous tissue and fascia, with variable involvement of the skin and muscle. The presence of necrosis was confirmed by permanent histopathologic tissue examination in all patients.
In a study approved by the institutional review board of National Cheng Kung University Hospital, we developed a treatment protocol summarized in Fig. 1. Patients with a high suspicion for necrotizing fasciitis arriving at our emergency room underwent a thorough physical examination by checking the presence of open wound and lesion morphology. Additionally, clinical signs including edema, patchiness, erythema, and tenderness of skin lesions; and the presence of hemorrhagic bullae or compartment syndrome were evaluated. The medical history obtained included information on other underlying diseases such as chronic liver disease, diabetes mellitus, adrenal insufficiency, immunocompromised diseases, hemochromatosis, thalassemia, and chronic renal insufficiency. All patients were queried regarding their contact with seawater and raw seafood including details about any fish or oyster harvesting they engaged in. Finally, wounds exudates and blister fluid obtained by fine needle aspiration were used to prepare smears and for culture of Gram-negative bacteria. The microscopic detection of comma-shaped, Gram-negative rods in smears was considered positive. Patients with these presentations were suspected of having V. vulnificus-mediated necrotizing fasciitis, and were included and treated, based on our treatment protocol.
When a vibrio infection was suspected upon examination in the emergency room, an antibiotic regimen was initiated for all patients. The treatment involved two combinations of antibiotics; ceftriaxone (2 g every 6 h intravenously) and doxycycline (100 mg every 12 h orallly, with a 200 mg loading dose) or ceftazidime (2 g every 6 h intravenously) and doxycycline (100 mg every 12 h orally, with a 200 mg loading dose). For the cases of chronic renal insufficiency, the antibiotic dosages were adjusted according to the creatinine clearance rate.
Next, the plastic surgeon was consulted and an emergency fasciotomy was performed. Microbiological cultures of the tissue samples obtained at the first operative debridement were used to confirm the diagnosis. Following the fasciotomy, all patients were admitted to the intensive care unit for supportive care, which routinely includes intravenous administration of antibiotics, maintenance of adequate electrolyte balance and nutritional support fluid, and combined enteral-parenteral nutrition. Intravenous nutritional support was subsequently reduced as enteral intake increased.
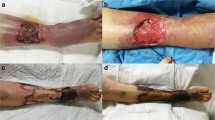
Wound surfaces were kept moist by 8-h application of the wet-to-wet technique. Aggressive debridement was planned over the next 2–4 days depending on the wound status. As patients’ general condition got stabilized, they were transferred to a regular ward and further wound reconstruction with a skin graft or flap was arranged by the attending plastic surgeon (Fig. 2).
Medical records were reviewed for age, gender, type of co-morbidities, anatomical site of infection, physical findings and laboratory values at the time of admission, interval between injury and admission, initial antimicrobial treatment, number of operative debridements, number of amputations, duration of hospitalization, and in-hospital mortality rate. Information concerning the interval between seawater contact and admission, interval between diagnosis of necrotizing fasciitis and operative intervention, blood pressure on arrival in the emergency room, length of hospitalization, and survival were also retrieved for each patient.
Statistical analyses were performed with use of SPSS Version 12.0 statistical software (SPSS, Chicago, Illinois). Student t and chi-square tests were used to assess significant relationships among the various factors. A p value of <0.05 was considered to be significant.
Results
The patients were collected from an overall group of 89 individuals diagnosed with necrotizing fasciitis treated at National Cheng Kung University Hospital during the designated time period. Twenty-two patients were excluded from the study; 12 patients refused surgical intervention or were lost to follow up, six patients had no evidence of trauma history, and four patients were diagnosed with cellulitis in the absence of typical tissue involvement. The remaining 67 patients with necrotizing fasciitis were treated using the above-detailed protocol. The study group included 52 men and 15 women (male-female ratio 77.6:22.4). The average age was 54 years (range 42–76 years); all patients had a history of handling seafood and prolonged occupational exposure to warm seawater (mean contact time of 17 h, with a range of 5–83 h) during oyster harvesting or fishing.
Patient characteristics and clinical information
The most common site of involvement were the upper extremities, with right upper extremity involvement in 33 patients (49.3%), and left upper extremity involvement in 17 patients (25.4%) (Table 1). Lesions involving the lower limbs were less prevalent, with left lower limb involvement in eight patients (11.9%) and right lower limb involvement in nine patients (13.4%). Underlying medical conditions included hepatic disease (chronic hepatitis B or C, liver cirrhosis or hepatoma; n = 23, 40%) chronic renal insufficiency (n = 17, 25.4%), adrenal insufficiency (n = 12, 17.9%), and diabetes mellitus (n = 5, 7.5%). Eight patients (11.9%) had septic shock upon emergency room arrival and 38 patients (56.7%) showed presence of bacteremia based on a positive blood culture for vibrios.
Antimicrobial treatment
All patients received combination therapy with third-generation cephalosporins (ceftriaxone or ceftazidime) and doxycycline. A proportion of patients received other antibiotic treatment regimens at arrival such as combination therapy with a first-generation cephalosporin (10.4%), or an aminoglycoside, ciprofloxacin, amikacin, penicillin, or amoxicillin/clavulanate (9%), and were shifted to the combination therapy with third-generation cephalosporins and doxycycline following confirmation of V. vulnificus infection.
Group comparison based on timing of the first fasciotomy
The patients were divided into two groups based on the timing of the first fasciotomy. Group A (n = 41) consisted of patients treated within 24 h after the injury. Group B (n = 26) consisted of patients treated beyond 24 h after the injury. The demographic characteristics of pre-existing medical conditions and clinical findings upon admission are summarized in Table 2. Seven (17%) group A and 16 group B (61.5%) patients had hepatic disease. The difference was significant (p < 0.0001). Diabetes mellitus was evident in three group A (7%) and two group B (7.7%) patients. Twelve (29%) patients in group A had chronic renal insufficiency compared to five (19%) patients in group B. Finally, five (12%) group A and seven (27%) group B patients had adrenal insufficiency. The difference was significant (p = 0.002).
The main presenting symptoms included fever, hemorrhagic bullae, compartment syndrome, and shock (Table 3). All patients had chills and general malaise at the time of presentation to the emergency room. Seven patients (17%) in group A and eight (31%) patients in group B presented with a body temperature exceeding 38.5°C (101.3°F); the difference was significant (p = 0.02). A single group A patient (2.4%) was hypotensive, displaying a systolic blood pressure of 90 mm Hg, compared to the seven group B patients. The difference was significant (p = 0.007).
Initial laboratory studies (Table 4) revealed median white blood cell counts of 8,300/mm2 (range 2,100–14,300) in group A and 9,130/mm2 (range 1,000–27,000) in group B. The median platelet count was 125,000/mm2 (range 76,400–245,000) in group A and 113,800/mm2 (range 76,400–245,000) in group B. The median serum C-reactive protein was higher in group A (3.2 mg/L; range 0.1–5) although the difference did not reach statistical significance. The median value of renal parameter creatinine in group A was 1.42 mg/dl (range 0.5–7) and 0.97 mg/dl (range 0.1–8.4) in group B.
All 67 patients initially underwent an emergency fasciotomy. The numbers of debridements and amputations performed are summarized in Table 5. The average number of debridements was 3.6 (group A) and 2.9 (group B), with an overall average of 3.4. The fatal and nonfatal subgroups were compared within each group. In group A, the difference was minimal. In contrast, in group B the number of surgical debridements was significantly higher. Fasciotomy was followed by amputation because of progressive deterioration of sepsis in 1/41 patients (2.4%) in group A and 2/26 patients in group B (7.7%). All three patients had an above-the-knee amputation.
Eight patients died within three to eleven days after admission and 59 patients survived, resulting in a mortality rate of 12%. Bivariate analysis revealed that a delay of more than 24 h from injury to surgery was significantly associated with increased mortality (p = 0.005).
The mean hospital stay for the group A and B patients was 31 days (range 7–54) and 29 days (range 3–49), respectively. Comparisons in group B demonstrated higher differences between the fatal (7 days) and nonfatal (56 days) subgroups.
Discussion
Necrotizing fasciitis develops as the infectious organisms proliferate within subcutaneous tissue along superficial and deep fascial planes. Infections with etiologies characteristic of V. vulnificus infection can be traced back to the fifth century BC [10], when Hippocrates mentioned a resident on an island in the Aegean Sea, who developed a “violent pain in his foot” with shivering, nausea, and fever followed by confusion. One day later, the whole foot became erythematous and swollen with small black blisters, with death ensuing. Interestingly, after the first reported case of a V. vulnificus infection in Taiwan in 1985 [11], an increased prevalence has been apparent [6, 12, 13]. One reason could be the warm coastal waters; the water temperature, which typically exceeds 20°C (60°F), is ideal for the growth of V. vulnificus. The prolongation of warmer weather that is apparently linked to global warming may be a contributing factor [14, 15].
The immunocompromised condition of patients can be crucial for the fulminant development of infections [3], and is one among the many reasons for V. vulnificus infections. Underlying chronic illnesses (such as cirrhosis, alcoholic liver disease, gouty arthritis, chronic renal failure, and diabetes mellitus) or the chronic use of steroids are associated with primary vibrio wound infections [6, 16]. In addition to these diverse risk factors, the location of our institution could be a significant factor contributing to the higher incidence of admission. The reasons for this geographical involvement are as follows: firstly, Taiwan is an endemic area of liver diseases such as hepatitis B and liver cirrhosis that affect liver function and decrease host resistance to pathogens. Secondly, the city of Tainan, which is located in the coastal southern area of the country, is an area of concentrated aquaculture and ostreiculture facilities, increasing the likelihood of contact with seawater or raw seafood. Thirdly, traditional Chinese herb ingestion prevails among the local population, which may imply the illegal use of steroid-containing herbal medications and analgesics; these substances greatly influence immune competence [17, 18, 19].
Our data is consistent with previous reports [2, 3, 6] chronicling a higher risk of V. vulnificus infection in patients with liver disease and adrenal insufficiency. Clinical symptoms develop tardily and show a later onset compared to the normal population. This is due to altered neutrophil and macrophage functions, immunosuppression, and the presence of increased levels of cytokines, tumor necrosis factor (TNF), and interleukins (IL) 1 and 6 [20]. Furthermore, V. vulnificus virulent factors that include surface protein and toxins [21] may stimulate an overproduction of TNF, IL-1, and IL-6, setting in action a destructive process and playing a pivotal role in the subsequent inflammatory reaction [22, 23]. It remains to be determinated whether there is an interaction between the increased levels of cytokines TNF, IL-1, and IL-6 of V. vulnificus and either liver disease or adrenal insufficiency. The potential role in the modulation of the host cytokine response for this reaction and the possible systemic activation should be further elucidated.
V. vulnificus produces extracellular enzymes such as mucinase, lipase, chondroitinase, hyaluronidase, deoxyribonuclease, sulfatase, and hemolysis [21, 24]. These toxins cause fever, destruction of perforating vessels of skin flaps, and thrombosis of vessels. The toxic effects are clinically manifest as a spreading subcutaneous tissue infection above the deep fascia, resulting in gangrene of subcutaneous fat and dermis [4]. As the infection extends along the fascial planes, bullae formation, ischemia, and tissue necrosis may occur. Therefore, patients with liver disease and adrenal insufficiency, associated with immunocompromised situation, are at increased risk of vibrio skin infections and septic shock, resulting in higher mortality.
The difficulty of making an early diagnosis is due to the absence of a well-established management protocol [25, 26]. Symptoms typically begin anywhere from several hours to four days later (median 12 h), consisting of swelling, erythema, and often intense pain. These symptoms are commonly the warning signs of the onset of necrotizing fasciitis. The presence of edema or bullae formation with serous or serosanguinous fluid is another important diagnostic clue. However, bullae formation, fever, and hypotension are slated evidence of infection. Septicemia with severe toxicity and mortality often develops when appropriate therapy is not given. Therefore, treatment of infection must be initiated promptly before definite identification of the organism is made [13], as a delay in diagnosis of vibrio-mediated necrotizing fasciitis is associated with increased mortality. Gram stains of wound and blister fluids, or discharge from open lesions provides information for possible infecting organisms and is a useful method for immediate differential diagnosis. Vibrio infection is associated with the presence of many polymorphonuclear leukocytes and Gram-negative bacilli [3]. Direct comparison of the present patient population, who were treated at different times during the study period, has revealed a mortality rate of 31.2 and 11.9% prior to and following, respectively, the implementation of our protocol. Patients with surgical debridement and fasciotomy within 24 h had the lowest mortality rate (4.9%).
Necrotizing fasciitis is a surgical emergency. Early recognition and prompt aggressive debridement of all necrotic tissue is critical for survival and improves the rate of survival [27, 28]. The cause of death in most cases is multiple-organ failure, acute respiratory distress syndrome, or overwhelming sepsis [9, 12]. With appropriate early surgical intervention, mortality varies from 8.7–50% [24, 26], depending on a number of variables including mode of infection, treatment modalities, and host factors. Without surgical intervention, necrotizing fasciitis is usually fatal because antibiotics alone are ineffective against the large soft tissue bacterial inocula resulting from the invasive nature of these infections. Surgical debridement is therefore essential to reduce bacterial load. In this period, the mortality of the nonsurgical patients was 50% (6/12), which is consistent with previous results of our institute [26].
Surgical debridement of all necrotic tissue is of paramount importance as antibiotic delivery to the involved area is ineffective due to the thrombosis of the supplying blood vessels. Debridement must be aggressive, all necrotic tissue with overlying skin should be excised deeply and beyond the necrotic areas, all necrotic fascia and fat should be removed until healthy viable tissue is evident. A second examination should be done within 24 h to assess the progression of the condition and the need for further debridement. This process should be repeated as frequently as necessary until the infection is controlled [5].
In addition to aggressive surgical debridement, efficient and early, presurgical antimicrobial treatment is also essential for management of V. vulnificus infection. Antibiotic use should be initiated as soon as the diagnosis is considered possible. Numerous studies documented the role of antimicrobial treatment. The combination of antimicrobial therapy with cefotaxime and minocycline demonstrates a better outcome than monotherapy with cefotaxime or minocycline [16, 29, 30]. The efficacy of the combinations is better than first or second-generation cephalosporin [16, 30]. The combination therapy remains effective for V. vulnificus infection both in vitro and in vivo [16, 30, 31]. More recently, the newer fluoroquinolones have been demonstrated to be as effective as the combination of cefotaxime plus minocycline in vitro and in vivo [32]. However, their clinical use remains unclear.
Nutritional interventions also play a substantial role in surgically critically ill patients [33, 34]. The hypermetabolic status in patients with sepsis and open surgical wounds increases their metabolic needs and necessitates aggressive enteral or parenteral nutrition. In the immediate postoperative period, all patients require nutritional support via intravenous or enteral routes, either using partial or total parenteral nutrition and oral or tube enteral feeding, respectively, with meticulous monitoring of body weight, intake, liver function, and electrolyte balance [34]. With an appropriate immunonutrition regimen, the outcomes in the critically ill population with sepsis can be improved.
Prevention is one of the most important determinants of outcome of the disease process. We recommended following treatment modality for necrotizing fasciitis caused by vibrio V. vulnificus, summarized in Fig. 2. Early recognition of the disease process using standardized protocol begins with Gram stain and culture examination of the wound. Initiation of broad-spectrum antibiotic coverage with third-generation cephalosporin and doxycycline should also be prompt. An emergency fasciotomy and comprehensive debridement of all non-viable tissues follows and is supported by aggressive resuscitation, intensive medical care, and nutritional support. Debridement of the involved site should be repeated every 24–48 h until the infection is controlled. Once the infection is controlled and the patient’s condition has been stabilized, reconstruction of the surgical site with skin graft or flaps can be undertaken as is judged appropriate.
Conclusion
Early and appropriate diagnosis for a V. vulnificus infection should be made especially in a patient presenting with atypical clinical findings. Performance of a fasciotomy within 24 h remains the highest priority and decreases the mortality rate. A global treatment using the standardized protocol would ensure efficient and appropriate diagnosis and therapy.
References
Blake PA, Merson MH, Weaver RE et al (1979) Disease caused by a marine Vibrio: clinical characteristics and epidermiology. N Engl J Med Jan 4;300(1):1–5
Neil MA, Carpenter CCJ (2000) Vibrio vulnificus infection. In: Mandell GL, Bennet JE, Dolin R (eds) Principles and practice of infectious diseases, 5th edn. Livingston, Edinburgh, Scotland, pp 2274
Seal DV (2001) Necrotizing fasciitis. Curr Opin Infect Dis 14:127–132
Oliver JD (2005) Wound infection caused by Vibrio vulnificus and other marine bacteria. Epidermiol Infect 133:383–391
Hsueh PR, Lin CY, Tang HJ et al (2004) Vibrio vulnificus in Taiwan. Emerg Infect Dis 8:1363–1368
Hor LI, Chang TT, Wang ST (1999) Survival of Vibrio vulnificus in whole blood from patients with chronic liver diseases: association with phagocytosis by neutrophils and serum ferritin levels. J Infec Dis 179:275–278
Bullen JJ, Spalding PB, Ward CG et al (1991) Hemochromatosis, iron and septicemia caused by Vibrio vulnificus. Arch Intern Med 151:1606–1609
Katz BZ (1988) Vibrio vulnificus meningitis in a boy with thalassemia after eating raw oysters. Pediatrics 82:784–786
Kihiczak GG, Schwartz RA, Kapila R (2006) Necrotizing fasciitis: a deadly infection. J Eur Acad Dermatol Venereol 20:365–369
Baethge BA, West BC (1988) Vibrio vulnificus: did Hippocrates describe a fatal case? Rev Infect Dis 10:614–615
Yuan CY, Yuan CC, Wei DC et al (1987) Septicemia and gangrenous change of the leg caused by marine vibrio, Vibrio vulnificus: report of a case. J Formos Med Assoc 86:448–451
Wang SM, Liu CC, Chiou YY et al (2000) Vivrio vulnificus infection complicated by acute respiratory distress syndrome in a child with nephritic syndrome. Pediatr Pulmonol 29:400–403
Tsai YH, Hsu WW, Huang KC et al (2004) Systemic vibrio infection presenting as necrotizing fasciitis and sepsis: a series of thirteen cases. J Bone Joint Surg. Am 86:2497–2502
Jaenisch T, Patz J (2002) Assessment of associations between climate and infectious diseases. Global Change Human Health 3(1):67–72
Paz S, Bisharat N, Paz E et al (2007) Climate change and the emergence of Vibrio vulnificus disease in Israel. Environ Res 103:390–396
Chuang YC, Ko WC, Wang ST et al (1998) Minocycline and cefotaxime in the treatment of experimental murine Vibrio vulnificus infection. Antimicrob Agents Chemother 42(6):1319–1322
Liu YC, Shyu TG, Suen TT (1989) Investigation of the adulteration of synthetic chemical drugs in marketed Chinese herbal rheumatic and analgesic preparations. Annual Report of the National Laboratories of Foods and Drugs, vol 7. Department of Health, Taipei, Taiwan, pp 197–199
Huang WF, Wen KC, Hsiao ML (1997) Adulteration by synthetic therapeutic substances of traditional Chinese medicine in Taiwan. J Clin Pharmacol 37:344–350
Ernst E (2002) Adulteration of Chinese herbal medicines with synthetic drugs: a systemic review. J Inter Med 252:107–113
Espat NJ, Auffenberg T, Abouhamze A et al (1996) A role for Tumor Necrosis Factor Alpha in the increased mortality associated with Vibrio vulnificus infection in the presence of hepatic dysfunction. Ann Surg 223(4):428–433
Howard RJ, Lieb S (1988) Soft-tissue infections caused by halophilic marine vibrios. Arch Surg 123:245–249
Powell JL, Wright AC, Wasserman SS et al (1997) Release of Tumor Necrosis Factor Alpha in response to Vibrio vulnificus capsular polysaccharide in in vivo and in vitro models. Infec Immun 65(9):3713–3718
Shin SH, Shin DH, Ryu PY et al (2002) Proinflammatory cytokine profile in Vibrio vulnificus septicemic patients’ sera. FEMS Immun Med Microb 33(2):133–138
Klontz KC, Lieb S, Schreiber M et al (1988) Syndromes of Vibrio vulnificus infections: clinical and epidemiological features in Florida case, 1981–1987. Ann Intern Med 109:318–323
Chuang YC, Yuan CY, Liu CY et al (1992) Vibrio vulnificus infection in Taiwan: report of 28 cases and review of clinical manifestations and treatment. Clin Infect Dis Aug 15:271–276
Yau BC, Lee JW, Chen CL et al (1995) Surgical treatment in vibrio infection. J Plast Reconst Surg ROC 4 1:21–27
Wong CH, Chang HC, Pasupathy S et al (2003) Necrotizing fasciitis: clinical presentation, microbiology, and determinants of mortality. J Bone Joint Surg Am Aug 85(8):1454–1460
Halow KD, Harner RC, Fontenelle LJ (1996) Primary skin infection secondary to Vibrio vulnificus: the role of operative intervention. J Am Coll Surg 183:329–334
Chuang YC, Liu JW, Ko WC et al (1997) In vitro synergism between cefotaxime and minocycline against Vibrio vulnificus. Antimicrob Agents Chemother 41(10):2214–2217
Chiang SR, Chuang YC (2003) Vibrio vulnificus infection: clinical manifestation, pathogenesis, and antimicrobial therapy. J Microbiol Immunol Infect 36:81–88
Liu JW, Lee IK, Tang HJ et al (2006) Prognostic factors and antibiotics in Vibrio vulnificus septicemia. Arch Intern Med 166:2117–2123
Tang HJ, Chang MC, Ko WC et al (2002) In Vitro and In Vivo activities of newer fluoroquinolones against Vibrio vulnificus. Antim Agen Chem Nov:3580–3584
Elliott DC, Kufera JA, Myers RAM (1996) Necrotizing soft tissue infections: risk factors for mortality and strategies for management. Ann Surg 224(5):672–683
Childers BJ, Potyondy LD, Nachreiner R et al (2002) Necrotizing fasciitis: a fourteen-year retrospective study of 163 consecutive patients. Am Surg Feb 68(2):109–116
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuo, YL., Shieh, SJ., Chiu, HY. et al. Necrotizing fasciitis caused by Vibrio vulnificus: epidemiology, clinical findings, treatment and prevention. Eur J Clin Microbiol Infect Dis 26, 785–792 (2007). https://doi.org/10.1007/s10096-007-0358-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-007-0358-5