Abstract
Background
Three-Hz postural leg tremor has recently been identified as highly prevalent in patients with the cerebellar type of multiple system atrophy, but its impact on posture maintenance remains poorly understood.
Patients and methods
Thirty-seven patients with spinocerebellar ataxia and 58 others with cerebellar type of multiple system atrophy were given Synapsys posturography examinations. Fifty-three healthy controls were also tested. Low, medium, and high-frequency sway were recorded to compute energy values. Frequency shift and postural strategy predominance were evaluated from the postural sway distributions, mainly from the proportions of higher frequency values among the total values. The trajectories of postural sway components were evaluated with the generalized additive mixed model. Distributions of the components and their relationships with falls and tremors were assessed through repeated measures correlation analysis.
Results
As the test difficulty increased, the standard controls showed slight increases in the energy values at every frequency. Distributions of the higher frequency (>0.5 Hz) values increased escalatingly with test difficulty, illustrating frequency shifts and hip strategy predominance. Medium and high-frequency values were strongly and positively inter-correlated in normal stances, but this was not observed among the spinocerebellar ataxia or multiple system atrophy patients. Unlike normal stances, the proportion of medium frequency values was negatively related to the total value among the spinocerebellar ataxia and multiple system atrophy patients, implying a failure of frequency shift in response to perturbation. Medium frequency proportions were also inversely correlated with tremors among the multiple system atrophy patients.
Conclusions
The observed synchronized changes in medium and high-frequency postural sway indicate that they constitute a complete hip strategy for posture control. The strategy was rigid in those with spinocerebellar ataxia but completely disrupted in those with multiple system atrophy. Three Hertz postural leg tremors destabilize the ankle joints and interfere with postural adjustment among those with multiple system atrophy.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Postural instability is a central clinical feature in degenerative ataxia, and increased sway during a quiet stance is associated with gait and balance impairments in patients with cerebellar ataxia [1]. However, there is limited research on distinct postural sway patterns for different types of ataxia [2, 3]. In a previous study using posturography, we examined center of pressure (COP) sway in patients with spinocerebellar ataxia (SCA), multiple system atrophy-cerebellar type (MSA-C), and a group of healthy individuals [4]. Both ataxia groups exhibited significantly increased postural sway during the four-step sensory organization test (SOT). Still, SCA patients had better postural performance than MSA-C, as indicated by a significantly lower occurrence of falls [4]. However, the overall body sway in the SCA and MSA-C groups was similar across all frequency bands, and the medium frequency sway in the SCA group was even higher. As a result, the discrepancy in their postural performance was attributed to the high-frequency sway component, which was induced by a 3-Hz postural leg tremor [5] and was more pronounced in the MSA-C group [4]. This is plausible since the nature of the 3-Hz sway significantly differs from the stabilizing high-frequency physiological tremor (approximately 8 Hz) induced by isometric contractions of the soleus muscle during quiet standing [6].
COP sway and its derivatives are commonly used as dependent variables in the analysis of postural control. Over 50 sway measures have developed from COP data to describe and characterize postural instability [7, 8]. However, establishing a unified relationship between total sway power and postural function has been challenging due to the variability across study subjects, influenced by factors such as physical indices [6, 9], demographic features [10, 11], and lifestyle choices [12,13,14]. Direct comparisons among sway components have been commonly employed in postural control analysis. Increases in low, medium, and high-frequency sway have been attributed to deficits in visual-vestibular [15, 16], cerebellar [3, 17], or proprioceptive [18, 19] control of static posture. Neurophysiological studies have also investigated peripheral mechanisms of different sway components. Low-frequency sway originated from frequent, ballistic bias impulses from the soleus and gastrocnemius muscles [20]. Medium-frequency sway of ~2 Hz was mainly oscillations around the hips [3]. High-frequency COP sway was short-latency, somatosensory-triggered destabilizing bursts of the gastrocnemius muscle [13, 18, 19, 21]. However, concluding solely from one component of the power spectrum can be a potential source of error, as the amplitude and velocity of postural sway may increase in a nonspecific fashion.
After analysis of human body kinematics, muscle activations, and ground reaction forces during acute postural responses, two distinct postural strategies have been identified [22,23,24,25,26]. It is widely accepted that a normal stance is maintained predominantly by ankle strategy, assisted by the hip strategy in response to external or internal perturbation, accompanied by postural sway shifting toward higher frequency bands [24, 27,28,29,30,31,32]. However, sway increase at higher frequencies has been observed in vestibular diseases [26], musculoskeletal and joint disorders, and even in adults of older age, indicating a wide range of conditions associated with spectral change. Deciphering these sway patterns as normal, compensatory, or pathological responses to postural perturbation remains complex. The process termed frequency shift has yet to be well defined.
The SOT (Sensory Organization Test) has been incorporated into computerized posturography examinations for several decades [17, 24, 27, 31,32,33]. Neurologically intact adults typically exhibit minimal body sway and can maintain balance for approximately 20 to 30 s under all test conditions. By sequentially depriving vision, deep sensory inputs, and both, the SOT test introduces increasing difficulty levels and presents a potentially effective protocol for investigating frequency shift and postural control strategies [24, 34,35,36]. However, the whole picture of SOT-related postural sway patterns has yet to be fully revealed. In a previous study, we observed an escalating increase in sway at every frequency for normal adults and individuals with ataxia [4]. We hypothesized that the ankle strategy prevails during low-amplitude, low-velocity, and low-frequency motions. In contrast, the hip strategy dominates during larger and faster sway perturbations [37]. The proportion of higher-frequency sway would also increase with total sway power in the presence of frequency shift. Exploring the frequency composition of postural sway can aid in identifying indicators of postural strategies and potentially unveil distinct patterns between individuals with normal postural control and those with ataxia.
To test these hypotheses, we examined the postural sway profiles of two ataxia groups and a group of healthy controls. We examined the trajectories of these sway components and their distributions throughout the test, and we also assessed the correlations between the sway components and their distributions with total postural sway power, fall occurrence, and tremor. The study’s objectives were to identify indicators of efficient posture adjustment, understand the dynamic relationship between postural sway components and posture maintenance, and evaluate the impact of 3Hz leg tremor on postural instability.
Patients and methods
Demographics of the study subjects and administrations of the posturography have been reported before [4]. MSA-C diagnoses were based on the Movement Disorder Society criteria for the diagnosis of MSA [38]. The SCA was diagnosed based on typical clinical features and positive genetic findings. Exclusion criteria were the following: (1) acute cerebellar dysfunction; (2) a history of head trauma; (3) a history of prescribed medication known to induce cerebellar atrophy such as phenytoin; or (4) any active disease of the central nervous system that affects the cerebellum. None of the study’s subjects had a history of alcohol misuse or dependency. Under four conditions, the static balance was assessed using an SYNAPSYS force plate (Marseille, France). SOT1 involved standing on a firm platform with the eyes open, while SOT2 involved a firm platform with the eyes closed. SOT3 utilized a foam platform with the eyes open, and SOT4 utilized a foam platform with the eyes closed. Each condition consisted of 20-s trials of quiet standing. Sensors underneath the platform captured postural sway, and the amplitude was measured and transformed into an energy value using a Fourier analyzer. The low, medium, and high-frequency components of postural sway were represented by low (LFV), medium (MFV), and high-frequency values (HFV) as per the posturography system. The proportions of these components, commonly referred to as low-frequency proportion (LFP), medium-frequency proportion (MFP), and high-frequency proportion (HFP), can be calculated by dividing their respective values by the total sway power. Three-Hz postural tremor was confirmed through visual inspection of rhythmic leg shaking and an isolated ~3-Hz peak wave in the anterior-posterior direction stabilogram [4]. Trials were marked as fall occurrences if the subject required assistance from safety harness to prevent falling, if the subject took one or more steps, or if the subject touched or held onto the research assistant. Frequent fallers were those who experienced two or more falls during the total four stance tasks. The trajectories of postural sway components and their distributions during the four-step SOT test were analyzed using a generalized additive mixed model (GAMM) with version 2.17.8 of the Empower Stats software suite and version 4.1.0 of the R-project software. To examine for potential influences of falls and tremors on postural sway, the GAMM analysis was performed both before and after adjusting for age, sex, fall occurrences and postural tremors. The relationships among postural sway components and the associations between total postural sway, fall occurrences, and tremor were quantified using repeated measures correlation analysis [39]. Since human static posture is a well-organized system controlled by various muscle synergies and coordinated movements of multiple joints, there may be relationships and interactions between postural sway at different frequencies. So interactions among different postural sway components were also evaluated through repeated measures correlation analysis.
Results
General postural sway patterns of three groups
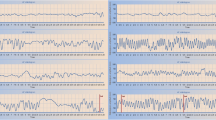
A total of 53 healthy controls, 37 patients with SCA (35 with SCA3 and 2 with SCA2) and 58 patients with MSA-C (48 clinical established, 10 clinical probable) were included in this study. Generally, compared with the SCA patients, those with MSA-C had an older age of onset (56.8±8.2 vs33.5±10.8, p<0.001), shorter disease duration (defined by the onset of motor symptoms, Q1-Q3 range of 1–2 years with a median one year, versus a Q1-Q3 range of 4–8 years with a median of 5 years, p<0.001), higher ICARS scores (21–34 with a mean of 28, versus 18–29 with a mean of 23) and higher prevalence of cross sign (69% versus 8.1%, p<0.001). The severity of cerebellar vermis atrophy was comparable in the two ataxia groups, although patients with moderate or severe vermis atrophy accounted for a higher percentage in MSA-C. During the posturography study, falls and tremors were also significantly higher in the MSA-C group [4]. Table 1 and Fig. 1 present the results of the GAMM analysis, which examined the changes in LFV (Fig. 1a), MFV (Fig. 1b), and HFV (Fig. 1c) among the three groups across the four stance tasks. The postural performance of the SOT 1 task was used as a baseline reference, and the subjects were observed for changes in sway magnitude over the subsequent three conditions. The increments of sway (Table 1) and sway distributions (Table 2) in ataxia patients were compared with those in healthy controls at each condition (Fig. 1). Additionally, the postural performance of MSA-C was compared with SCA (supplementary information Table 1-2). Initially, all the sway components were comparable among the three groups. During normal stances, only slight and insignificant increases in body sway were observed throughout the testing period (Table 1, Fig. 1). However, in the ataxia groups, the postural sway components at all frequencies increased significantly with the increasing difficulty of the test (Table 1, Fig. 1).
Changes in the low (a), medium (b), and high (c) frequency band energy with test difficulty in the three groups. The normal controls showed very slight increases in the energy values at every frequency. Compared with normal adults, postural sway at every frequency was significantly elevated with test difficulties in SCA and MSA patients. The LFV of SCA and MSA was comparable at every stance (a). In the SCA group, MFV increased significantly, especially in SOT 2 and SOT 4 tests (b). Similar to LFV, MFV was also significantly elevated with test difficulties in SCA and MSA patients. Increments of MFV of SCA were higher than MSA in the SOT4 task. Increments of HFV were much more prominent in MSA-C subjects on foam tests (SOT 3 and SOT 4) (c). SOT: sensory organization test, MSA-C: multiple system atrophy-cerebellar types, SCA: spinocerebellar ataxia, HC: healthy controls
In our previous study, direct comparison of postural sway did not reveal significant differences between the ataxia groups. In this study, LFV of SCA and MSA-C was comparable before and after adjustment (Table 1, Supplementary information Table 1, Fig. 1a). However, the SCA group exhibited higher increments in MFV, particularly in the SOT4 test, compared to the MSA-C group (Table 1, Supplementary information Table 1, Fig. 1b). This difference persisted even after adjustment (Supplementary information Table 1, Fig. 1b). Regarding HFV, the MSA-C subjects initially showed larger increments in the SOT3 test, but the differences compared to SCA became negligible after adjustment (Supplementary information Table 1, Fig. 1c).
Frequency shift and hip strategy predominance in normal stance
Table 2 presents the results of the GAMM analysis associating LFP, MFP (see also Fig. 2a), and HFP changes in the three groups with the four stance tasks. In a normal stance, a characteristic finding was the continuous upward tendency of MFPs (Fig. 2a). Conversely, HFP demonstrated minimal changes with varying test difficulties in healthy controls and SCA patients. The dynamic changes of LFP generally followed an inverted pattern compared to MFP (Table 2, Figures not shown).
Changes of the medium frequency proportion with test difficulty in ataxia groups before and after stratification by fall occurrence. a MFP gradually increased in normal stance. In SCA, there was a transient increase in the firm platform. However, the MFP of SCA and MSA dropped in foam tests. Noted that MFP was already elevated in ataxia patients and was, on average higher than that of the healthy controls in the initial stance task (SOT1). b After stratification of fall occurrence, MSA patients with worse postural stability (more fall occurrence) had higher MFP at the initial stance task. Also, they had a more severe drop with the increase of test difficulties. Frequency shift was suspected in a minority of MSA-C patients who did not experience any falls, in whom a slight upward trend in MFPs was present. c In the SCA group, there was an increase of MFPs in SOT 2 tests in patients with better postural stability (fall≤1). SCA patients with worse postural stability (fall≥2) seem to present with stable MFP levels during the tests, indicating no frequency shift. SOT: sensory organization test, MSA-C: multiple system atrophy-cerebellar type, SCA: spinocerebellar ataxia, HC: healthy controls. FALL_TOTAL indicates the total times of fall occurrence of the subjects during four-step SOT tests, ranging from 0 to 3 times
Initially, the MFPs of normal subjects were lower than those of ataxia patients. However, as the test difficulty increased, the MFPs gradually increased and eventually became more remarkable than the two ataxia groups in foam tests (Fig. 2a). Consistent with the GAMM findings, repeated measures correlation analysis revealed that total postural sway power (total energy value) during normal stance was positively related to both MFP (r=0.33, p≤0.05) and HFP (r=0.25, p≤0.05), and negatively associated with LFP (r=−0.32, p≤0.05). A strong and positive correlation between MFV and HFV was also observed (r=0.78, p≤0.05, Table 3), suggesting an intrinsic coupling of higher frequency sway components. The postural adjustment patterns depicted the frequency shift and hip strategy predominance characteristic of a normal stance. Notably, there was a negative correlation between LFP and the higher frequency components (including MFP and HFP) in both normal adults and ataxia patients (Table 3). This supports the fundamental concept that ankle and hip strategies complement or compete with each other and indirectly validate the predetermined cut-off values of the SYNAPSYS system for the three frequency bands (below 0.5 Hz for LF, 0.5–2 Hz for MF, over 2 Hz for HF).
Increase of MFPs was constrained or diminished in ataxia
Upon initial analysis, the MFPs of the two ataxia groups at baseline (SOT1) were comparable, and their trajectories during the four stance tasks were also similar. In the SCA group, MFPs increased in SOT2 but then decreased continuously during the foam tests (SOT3 and SOT4) (Fig. 2a, Table 2). In MSA-C, the increase in SOT2 was almost invisible, while the decrease in the foam tests was more pronounced (Fig. 2a, Table 2). These findings suggest that the increase in MFPs was limited in ataxia patients and thus can be interpreted as a surrogate for implementing the hip strategy. The transient frequency shift and the predominant use of the hip strategy on a firm platform suggest a strong reliance on proprioceptive input for effective postural adjustments in ataxia.
Upon further stratification, it remained evident that MFP levels had a momentary surge among non-fallers during the SOT2 test. This can be observed in Fig. 2b for the MSA-C group and Fig. 2c for the SCA group. The increase in MFP was diminished in frequent fallers, although their trajectory differed significantly between the two ataxia groups. In the SCA group, frequent fallers exhibited almost fixed MFP levels throughout the serial stance tasks (Fig. 2c). Frequent fallers with MSA-C showed a cascading trajectory of declining MFPs, characterized by higher initial MFPs and lower final MFPs (Fig. 2b).
Fall occurrence was accompanied by a null or reversed frequency shift
Falls are widely recognized as a reliable indicator of postural instability, and the relationship between fall occurrence and postural sway power is well established in research. This study observed a strong association between fall occurrence and postural sway power across all frequency bands in both groups with ataxia (Table 3). Specifically, fall occurrence showed the closest correlation with low-frequency postural sways in both SCA (r=0.783) and MSA (r=0.690) groups (Table 5). Given that the hip strategy is known to be faster and more efficient in postural adjustment, the predominance of the hip strategy may be lacking in patients with a higher occurrence of falls.
Indeed, in MSA-C patients, there was a positive correlation between total sway and LFP and a negative correlation between total sway and MFP, suggesting a reversed frequency shift. These correlations became even stronger in the subgroup of frequent fallers (Table 4). Fall occurrence was directly associated with MFPs in MSA-C (r=−0.352, P<0.05, Table 5). Also, the GAMM analysis revealed a declining trend in MFPs across the tests for the majority of MSA-C patients, as depicted in Fig. 2b. This disproportionate dependence on the ankle strategy can be seen as invalid and inefficient in the context of postural adjustment. These findings established a relationship between decreased MFPs, the predominance of ankle strategy, and postural instability in MSA.
Somewhat unexpectedly, in the SCA group, frequent fallers consistently demonstrated stagnant levels of MFPs across the serial stance tasks. This suggested the absence of frequency shift and the presence of a rigid postural strategy (Fig. 2c). Accordingly, no correlations were found between total sway power and any sway proportions before or after the stratification of falls (Table 4). In SCA there was no correlation between fall occurrence and sway proportions either. (Table 5).
Consistent with these findings, no coupling was observed between higher frequency sways (MFV and HFV) in any ataxia group. Instead, in SCA there was a significant coupling between low-frequency and medium-frequency sway (r=0.711, p<0.05). Conversely, in MSA-C, the strongest coupling was observed between low-frequency and high-frequency sway (r=0.591, p<0.05), as indicated in Table 3.
Tremor, frequency shift, and postural instability
In the SCA group, the correlation between tremor and postural sway contents was neglectable (Table 4). With the presence of tremor, no frequency shift was observed, as there was no correlation between total sway and any postural sway contents in tremor sub-groups (Table 4). Among those without tremors, negative correlations were found between total sway with MFP (r=−0.19, p>0.05) and HFP (r=−0.32, p≤0.05) (Table 4). These correlations provide supporting evidence for an inherent deficit in physiological high-frequency sway in SCA.
Postural performance of MSA-C patients exhibited a correlation with tremors, similar to its associations with falls. Generally, tremor may serve as another indicator of postural instability in MSA-C, as it demonstrated a significant correlation with sway power across all frequency ranges. The robust association between tremor and high-frequency sway power, as indicated by the correlation coefficients of r=0.532 (p<0.05) for HFV and r=0.545 (p<0.05) for HFP, underscores the overriding influence of the abnormal 3-Hz tremor over the expected physiological sway (Table 5). Despite the substantial increase in HFV, the negative correlation coefficient between total sway and MFPs exhibited a more significant growth, while the positive correlations between total sway and HFPs unexpectedly attenuated with the increasing occurrence of tremors (Table 4). These findings provided strong evidence that the 3-Hz tremor observed differed significantly from the physiological tremor (occurring at approximately 8 Hz) typically associated with the soleus muscles, which play a crucial role in stabilizing static posture.
Not surprisingly, coupling between higher frequency sway was disrupted, and a clear and distinct frequency shift to the lower band was observed in patients with tremors. Correlation analysis of MSA-C provided convincing evidence of ankle strategy predominance (Table 3). Similar to the postural performance of subjects with falls, MSA-C subjects with a higher occurrence of tremors also demonstrated higher initial MFPs levels, followed by a more significant decline in subsequent tests (Fig. 3).
Medium-frequency sway proportion changes in MSA-C patients after stratification by tremor occurrence. The influence of tremors on postural performance was similar to those of the fall occurrence in MSA-C. Note: SOT: sensory organization test, TREMOR: total tremor occurrence during four-step SOT tests, ranging from 0 to 3 times. Patients with 2 or 3 tremor occurrences were pooled together for GAMM analysis
Discussion
The term “frequency shift” has been frequently referenced in prior research. However, the concepts of frequency shift have yet to be well defined. Besides that, the connections among postural sway distribution, frequency shift, and postural instability could have been more linear and straightforward. The introduction of falls (indicative of postural dysfunction) and a 3-Hz tremor have added complexity to this intricate puzzle. One significant contribution of this study is that we have elucidated the concept of frequency shift and established its correlation with postural stability, a linkage established primarily through observations from a normal stance. Generally, frequency shift refers to the alteration in the composition of postural sway content. This shift involves increasing the contribution of higher-frequency sway components and a simultaneous decrease in lower-frequency sway components. This pattern signifies that the body is rapidly and effectively adapting its posture. Notably, this phenomenon aligns closely with the concept of hip strategy predominance. This strategy entails the body’s prioritized use of hip movements to counterbalance and stabilize itself swiftly.
On the other hand, the absence of the frequency shift, characterized by an unchanged proportion of sway components (like SCA) or a shift towards the lower frequency band (with an increased ratio of lower-frequency components, like MSA-C), both indicate an unsuccessful frequency shift. To describe these phenomena, we’ve termed them the “null shift” and the “inverse shift,” respectively. With fall occurrence as a landmark for postural instability, and by referring to the characteristic postural adjustment patterns of normal stance, we could unravel the complex cause-effect relationships between sway components, frequency shift, and postural stability. We identified MFP as an effective and easily measurable marker to trace frequency shifts and to evaluate postural strategy during serial stance tasks. We discovered a subtle and elusive deficit in high-frequency physiological sway in both SCA and MSA-C patients. This deficit was likely attributable to the cerebellar vermis atrophy, which led to a reduced loop gain and increased dependence on a firm surface for proprioceptive input. Consequently, there was a limited (SCA) or diminished (MSA-C) increase in MFPs among patients with ataxia. Additionally, the lack of high-frequency sway (SCA), together with the presence of a 3-Hz tremor (MSA-C), disrupts the coordination of higher-frequency sway and resulted in a null (SCA) or reversed (MSA-C) frequency shift.
MFPs, frequency shift, and hip strategy
MFP increase is easy to obtain and evaluate from static posturography data, plus it seldom gets influenced by pathological tremor (which usually has a frequency of above 2 Hz). So, we choose it as a surrogate measure for assessing frequency shift and postural stability. This study revealed a distinctly escalating pattern of MFPs in a normal stance. In contrast, individuals with compromised postural control displayed a constrained or diminished MFP elevation during demanding balancing tasks. These findings suggest that the elevation of MFPs can be a surrogate measure for assessing frequency shift and postural stability. Even though, in this study we could not establish a logistic regression model to determine the contribution of MFP to posture stability. This is mainly because the relationship between MFPs and postural control was not linear or straightforward. Relying solely on MFPs from a single stance task to predict postural stability can be misleading due to the variability of MFPs across different stance tasks and disease entities.
For example, we observed divergent interpretations regarding the significance of elevated MFPs in healthy adults, SCA and MSA-C patients. We noted a progressive increase in MFPs during demanding stance tasks in healthy adults. In individuals with impaired postural control (SCA and a minority of MSA-C with fair balance), the capacity for MFP elevation is limited. Still, higher MFPs indicated better postural performance, irrespective of the faller sub-groups or stance tasks considered. However, these patterns contradicted the situation of most MSA-C patients, as the highest MFPs were observed in the easiest stance tasks within the subgroup of individuals with the highest frequency of falls. In the natural stance, where the ankle strategy predominates, proximal muscles such as the erector spinae, external oblique, and gluteus medius activate to limit excessive motion in the hip joints, ensuring stability [37]. However, in cases of severe cerebellar vermis atrophy, as observed in the MSA-C group, these automated postural reflexes may also be impaired, leading to undesired hip oscillations. Consequently, an elevation in MFP occurred, which differed significantly from the intentional trunk sway observed during attempts to restore balance and should be interpreted as a consequence of unstable hips or a misleading frequency shift rather than as an indication of the implementation of a hip strategy.
Isolated MFP elevation and incomplete hip strategy
The exact energy value of high-frequency physiological sway is relatively low and hard to capture from the posturography system. This consideration was a significant factor in our decision not to use it as a replacement marker for frequency shifts. However, the significance of high-frequency sway in postural control is not neglectable. High-frequency sway serves at least two crucial roles in balance control. Firstly, it establishes a robust foundation to facilitate the translation of overt sway energy from the hips to the ground, which is subsequently attenuated by ground shear force. This collaborative process harmonizes with hip oscillation to restore balance. Secondly, aberrant high-frequency sway, such as a 3-Hz postural leg tremor, significantly undermines this process, even inciting a reverse shaking sequence originating from the ankles, traveling through the hips, and culminating in head movement. This disruptive sequence entirely dismantles the classical postural adjustment patterns typically guided by the hip strategy.
Previously, body oscillations of medium frequency have been explained by an increased gain of posturally stabilizing (long loop) reflexes that involve cerebellar control [40]. It is well known that cerebellar ataxia patients cannot scale the size of their postural responses to perturbations, as they tend to over-respond [41,42,43]. In SCA patients, the disrupted cerebellar circuitry, including the loss of Purkinje cells and dysfunction in the cerebellar output pathways, leads to uncoordinated and exaggerated muscle activity. This results in a sustained and uncontrolled shaking of the trunk and legs, even in tasks that would typically use ankle strategy to maintain balance in healthy individuals. By reducing the amplitudes of long-latency stretch reflexes, cerebellar transcranial direct current stimulation (tDCS) has demonstrated efficacy in improving ataxia symptoms [44].
In this study, SCA patients exhibited a distinctive rigid postural strategy, evidenced by the lack of correlation between MFPs and total sway across all sub-groups. Moreover, there was no observed association between LFPs or MFPs and the occurrence of falls or tremors. The rigidity of MFPs was particularly pronounced in individuals with poorer postural control, as their MFPs remained unchanged during the four-step stance tasks. This limited adaptability in postural adjustments resonated with the characteristic “locking knees” commonly observed in SCA. While the rigid knee posture was previously considered a compensatory mechanism to minimize interactions between body segments and reduce excessive postural responses in SCA, it may also contribute to impaired postural control [45].
By investigating automatic postural responses, Torres-Oviedo G et al. identified several muscle synergies that could satisfactorily simulate ankle and hip strategies [37]. Tight coupling of lower leg muscle activities has also been revealed, providing evidence of muscle cooperation during stance [46]. As is well known, the hip strategy consists mainly of the activation of the trunk and thigh muscles, radiating in a proximal-to-distal fashion to other muscle groups. However, as delayed feedback of a closed loop control, these oscillations are a source of instability themselves. Since medium-frequency postural sway consists mainly of oscillations around the hips, high-frequency physiological sway that contain stabilizing efforts of the knee and ankle joints is extremely important to facilitate the transmission of the excessive oscillations downward, thus making them counteracted by the shear force from the ground.
However, increments of physiological high-frequency sway were negligible in SCA. In SCA patients without 3-Hz tremors, total sway was positively correlated with LFP and negatively correlated with HFP, indicating high-frequency physiological sway deficit and failure of frequency shift. High-frequency physiological sway during static stance is predominantly mediated by a short-latency reflex, which receives proprioceptive information from velocity-sensitive muscle spindle Ia afferents and regulates muscle stiffness. Cerebellectomy reduced the gain of the proprioceptive feedback loops and induced a drop in fusimotor tone [47]. With high-frequency sway, steady fixations of static posture are readily obtained otherwise a conscious effort is continually needed. The findings of significant coupling of low and medium frequency sway in SCA depicted the adaptive picture involving enhanced visual and vestibular contributions.
3-Hz tremor, decreased MFPs, and reversed frequency shift
Limited research has been performed on the association between 3 Hz tremor and postural instability. In a study carried out by Diener HC et al, changes in 3-Hz tremor power were synchronous with alterations in total anterior-posterior body sway in individuals with alcohol use disorder [48]. In another study conducted by Sullivan et al. [49], a correlation was found between 3-5Hz leg tremor and overall postural sway. Furthermore, the researchers also observed that vermian atrophy exhibited a correlation with the ratio of higher frequency sway power (>2Hz) to lower frequency sway power (<2Hz). This study identified significant correlations between 3-Hz tremor and sway power across all frequency ranges in MS-C patients, highlighting its potential as an indicator of postural instability. The strong correlation between 3-Hz tremor and high-frequency sway (HFV and HFP) underscores the inherent deviation from the anticipated patterns of physiological sway.
Ankle stability during stance relies on steady plantar flexor force and co-activation of antagonist muscles [50]. The soleus muscles are key stabilizers [6]. Fluctuations during quiet standing correlate with 8–10 Hz EMG power in the medial gastrocnemius [6]. Pathological changes in the ankle muscles and ankle joints pose a risk to static posture [50]. Surface EMG recordings of ataxia patients revealed coherent 3-Hz rhythmic activities in bilateral tibialis anterior (TA) muscles and alternating firing patterns in the medial gastrocnemius (MG) muscles [4]. The pronounced effect of 3-Hz tremor on ankle joints significantly hinders the execution of the ankle strategy and disrupts the transmission of torques and forces from the hips to the ground. In essence, both ankle and hip strategies are impaired in MSA-C.
In this study, the ankle strategy predominated in MSA-C patients, accompanied by a shift towards lower frequencies. The muscles crossing the ankle joint, particularly the TA muscle, provide crucial sensory information for maintaining upright standing [51,52,53]. Rhythmic 3-Hz contraction of the muscles complicates proprioceptive input and distorts perceived sway boundaries when utilizing the ankle strategy. Furthermore, the alternating oscillations between the TA and MG muscles dissociated ankle joint angles from the center of mass position changes. As a result, the negative correlation between total sway power and MFPs exhibited a stepwise increase in the presence of higher levels of tremor.
In pioneering research by Maruitz et al. [54], kinematics of segmental body movement was investigated in patients with cerebellar anterior lobe atrophy, revealing a 3-Hz rhythmic anti-phase movement pattern between the head, trunk, and lower limbs. At a time when the concept of postural strategy was not yet developed, they astutely observed the remarkable similarity between the intersegmental postural response pattern and the characteristic posture exhibited by individuals with a narrowed base of stance, which was typical of "hip strategy". Interestingly, they also found that the 3-Hz tremor was induced by sudden perturbation of the body or vision deprivation, both of which also triggered hip strategy. Our study observed a significant decrease in MFPs among MSA patients. This decrease was further amplified in the presence of increased tremor activity. This process could also be taken as some kind of "frequency shift".
On the other hand, according to Mauritzs et al. [54], the 3-Hz tremor represented delayed and enhanced long-loop reflex, and the tremor frequency increased with disease severity. In line with their findings, we also observed lower frequency rhythmic tremor (2.4–2.8 Hz) in patients with degenerative ataxia of intermediate severity, falling between the severity of MSA and SCA [4]. This suggests a gradation of tremor frequency corresponding to the varying levels of disease severity. More interestingly, the characteristic hip oscillations of mild to moderated SCA (1.2–2.4 Hz) have also been associated with the exact central mechanisms [3]. Taken together, the observed postural oscillations, including excess hip oscillations in mild-moderate SCA patients, lower frequency rhythmic tremor (2.4–2.8Hz) in severe SCA patients, and the typical 3-Hz tremor in MSA-C patients, form a continuum of postural responses attributed to delayed but enhanced long loop reflex in degenerative ataxia. It would perfectly explain the stepwise deficit of MFPs in the presence of tremors in MSA-C patients. In other words, MSA-C patients use ankle-generated torques and distal-to-proximal muscle activation for postural correction, which was invalid and inefficient. The 3-Hz tremor presented in MSA was intrinsically the presence of a pathological hip strategy.
Limitations
This study has several limitations. In the study, only anterior-posterior (AP) COP sway was analyzed for several reasons. First, ankle and hip strategies are postural responses primarily identified in the AP direction. Secondly, ataxia patients present more pitch than roll instability [3, 31, 40]. Also, AP stability is an essential predictor of falls among ataxia patients [2]. Finally, the 3 Hz leg tremor is AP postural sway that originates from rhythmic and alternating oscillations of the tibialis anterior and medial gastrocnemius muscles [4, 5].
In the posturography exam, the cut-off value for low, medium, and high-frequency bandwidth is not unanimous across force platforms and studies. In the SPS exam, the low-frequency band is below 0.5 Hz, and the high-frequency band is over 2 Hz. Such a frequency spectra designation differed from the standard proposed by Kapteyn et al. [55]. Still, it was supported by recent studies [37] and the negative correlations between LFP with MFP/HFP in the survey.
Conclusions
In this study, we characterized postural sway patterns of normal stance. We identified MFPs as an effective and easily measurable marker to trace frequency shift and strategy predominance. In SCA, the rigid postural strategy was accounted for by the relative lack of high-frequency physiological sway, which would typically be enhanced by short-loop reflex gain instead of relying on proprioceptive input. In the MSA-C group, medium frequency sway that initiate a hip strategy to restore balance was replaced by a 3-Hz tremor, which adjusts the posture in a pathologically distal to the proximal sequence. It is important to note that these interpretations are speculative and would require further investigation and evidence to confirm. However, they establish a framework for understanding the different postural performances of SCA and MSA-C.
Data Availability
The data that support the finding of this study are available from the corresponding author upon reasonable request.
References
Gatev P, Thomas S, Lou JS, Lim M, Hallett M (1996) Effects of diminished and conflicting sensory information on balance in patients with cerebellar deficits. Mov Disord 11(6):654–664
Ganapathy VS, James TT, Philip M, Kamble N, Bhattacharya A, Dhargave P, Pal PK (2021) Anteroposterior stability: a determinant of gait dysfunction and falls in spinocerebellar ataxia. Ann Indian Acad Neurol 24(4):518–523
Van de Warrenburg BP, Bakker M, Kremer BP, Bloem BR, Allum JH (2005) Trunk sway in patients with spinocerebellar ataxia. Mov Disord 20(8):1006–1013
Li X, Lv X, Liu C, Ye J, Xu Y, Ma G, Li Z, Wang Y (2021) 3-Hz postural tremor in MSA-C and SCA: revisiting an old but underestimated cerebellar sign by posturography. Cerebellum. 20(2):246–253
Li X, Wang Y, Wang Z, Xu Y, Zheng W (2018) 3-Hz postural tremor in multiple system atrophy cerebellar type (MSA-C)-a static posturography study. Neurol Sci 39(1):71–77
Kouzaki M, Masani K (2012) Postural sway during quiet standing is related to physiological tremor and muscle volume in young and elderly adults. Gait Posture 35(1):11–17
Visser JE, Carpenter MG, van der Kooij H, Bloem BR (2008) The clinical utility of posturography. Clin Neurophysiol 119(11):2424–2436
Chiari L, Rocchi L, Cappello A (2002) Stabilometric parameters are affected by anthropometry and foot placement. Clin Biomech (Bristol, Avon) 17(9-10):666–677
Błaszczyk JW, Cieślinska-Swider J, Plewa M, Zahorska-Markiewicz B, Markiewicz A (2009) Effects of excessive body weight on postural control. J Biomech 42(9):1295–1300
Johansson J, Nordström A, Gustafson Y, Westling G, Nordström P (2017) Increased postural sway during quiet stance as a risk factor for prospective falls in community-dwelling elderly individuals. Age Ageing 46(6):964–970
Šarabon N, Kozinc Ž, Marković G (2022) Effects of age, sex and task on postural sway during quiet stance. Gait Posture 92:60–64
Kiers H, van Dieën J, Dekkers H, Wittink H, Vanhees L (2013) A systematic review of the relationship between physical activities in sports or daily life and postural sway in upright stance. Sports Med 43(11):1171–1189
Kouzaki M, Masani K, Akima H, Shirasawa H, Fukuoka H, Kanehisa H, Fukunaga T (2007) Effects of 20-day bed rest with and without strength training on postural sway during quiet standing. Acta Physiol (Oxford) 189(3):279–292
Dault MC, Frank JS (2004) Does practice modify the relationship between postural control and the execution of a secondary task in young and older individuals? Gerontology 50(3):157–164
Golomer E, Dupui P, Bessou P (1994) Spectral frequency analysis of dynamic balance in healthy and injured athletes. Arch Int Physiol Biochim Biophys 102(3):225–229
Davis JR, Campbell AD, Adkin AL, Carpenter MG (2009) The relationship between fear of falling and human postural control. Gait Posture 29(2):275–279
Taguchi K (1978) Spectral analysis of the movement of the center of gravity in vertiginous and ataxic patients. Agressologie 19(B):69–70
Golomer E, Dupui P (2000) Spectral analysis of adult dancers’ sways: sex and interaction vision-proprioception. Int J Neurosci 105(1-4):15–26
Paillard T, Costes-Salon C, Lafont C, Dupui P (2002) Are there differences in postural regulation according to the level of competition in judoists? Br J Sports Med 36(4):304–305
Loram ID, Maganaris CN, Lakie M (2005) Human postural sway results from frequent, ballistic bias impulses by soleus and gastrocnemius. J Physiol 564(Pt 1):295–311
Horak FB, Nashner LM (1986) Central programming of postural movements: adaptation to altered support-surface configurations. J Neurophysiol 55(6):1369–1381
Creath R, Kiemel T, Horak F, Peterka R, Jeka J (2005) A unified view of quiet and perturbed stance: simultaneous co-existing excitable modes. Neurosci Lett 377(2):75–80
Runge CF, Shupert CL, Horak FB, Zajac FE (1999) Ankle and hip postural strategies defined by joint torques. Gait Posture 10(2):161–170
Faraldo-García A, Santos-Pérez S, Crujeiras R, Soto-Varela A (2016) Postural changes associated with ageing on the sensory organization test and the limits of stability in healthy subjects. Auris Nasus Larynx 43(2):149–154
Rosengren KS, Rajendran K, Contakos J, Chuang LL, Peterson M, Doyle R, McAuley E (2007) Changing control strategies during standard assessment using computerized dynamic posturography with older women. Gait Posture 25(2):215–221
Fujimoto C, Kamogashira T, Kinoshita M, Egami N, Sugasawa K, Demura S, Yamasoba T, Iwasaki S (2014) Power spectral analysis of postural sway during foam posturography in patients with peripheral vestibular dysfunction. Otol Neurotol 35(10):e317–e323
Liaw MY, Chen CL, Pei YC, Leong CP, Lau YC (2009) Comparison of the static and dynamic balance performance in young, middle-aged, and elderly healthy people. Chang Gung Med J 32(3):297–304
Hwang S, Tae K, Sohn R, Kim J, Son J, Kim Y (2009) The balance recovery mechanisms against unexpected forward perturbation. Ann Biomed Eng 37(8):1629–1637
Termoz N, Halliday SE, Winter DA, Frank JS, Patla AE, Prince F (2008) The control of upright stance in young, elderly and persons with Parkinson’s disease. Gait Posture 27(3):463–470
Sozzi S, Nardone A, Schieppati M (2021) Specific posture-stabilising effects of vision and touch are revealed by distinct changes of body oscillation frequencies. Front Neurol 12:756984
Baloh RW, Jacobson KM, Beykirch K, Honrubia V (1998) Static and dynamic posturography in patients with vestibular and cerebellar lesions. Arch Neurol 55(5):649–654
Ahmadi SA, Vivar G, Frei J, Nowoshilow S, Bardins S, Brandt T, Krafczyk S (2019) Towards computerized diagnosis of neurological stance disorders: data mining and machine learning of posturography and sway. J Neurol 266(Suppl 1):108–117
Ciesielska J, Lisiński P, Bandosz A, Huber J, Kulczyk A, Lipiec J (2015) Hip strategy alterations in patients with history of low disc herniation and non-specific low back pain measured by surface electromyography and balance platform. Acta Bioeng Biomech 17(3):103–108
Singh NB, Taylor WR, Madigan ML, Nussbaum MA (2012) The spectral content of postural sway during quiet stance: influences of age, vision and somatosensory inputs. J Electromyogr Kinesiol 22(1):131–136
Stambolieva K (2011) Fractal properties of postural sway during quiet stance with changed visual and proprioceptive inputs. J Physiol Sci 61(2):123–130
Bao T, Carender WJ, Kinnaird C, Barone VJ, Peethambaran G, Whitney SL, Kabeto M, Seidler RD, Sienko KH (2018) Effects of long-term balance training with vibrotactile sensory augmentation among community-dwelling healthy older adults: a randomized preliminary study. J Neuroeng Rehabil 15(1):5
Torres-Oviedo G, Ting LH (2007) Muscle synergies characterizing human postural responses. J Neurophysiol 98(4):2144–2156
Wenning GK, Stankovic I, Vignatelli L, Fanciulli A, Calandra-Buonaura G, Seppi K, Palma JA, Meissner WG, Krismer F, Berg D, Cortelli P, Freeman R, Halliday G, Höglinger G, Lang A, Ling H, Litvan I, Low P, Miki Y et al (2022 Jun) The Movement Disorder Society Criteria for the diagnosis of multiple system atrophy. Mov Disord 37(6):1131–1148
Bakdash JZ, Marusich LR (2017) Repeated measures correlation. Front Psychol 8:456
Mauritz KH, Dichgans J, Hufschmidt A (1979) Quantitative analysis of stance in late cortical cerebellar atrophy of the anterior lobe and other forms of cerebellar ataxia. Brain 102(3):461–482
Diener HC, Dichgans J, Bacher M, Guschlbauer B (1984) Characteristic alterations of long-loop “reflexes” in patients with Friedreich’s disease and late atrophy of the cerebellar anterior lobe. J Neurol Neurosurg Psychiatry 47(7):679–685
Horak FB, Diener HC (1994) Cerebellar control of postural scaling and central set in stance. J Neurophysiol 72(2):479–493
Timmann D, Horak FB (1997) Prediction and set-dependent scaling of early postural responses in cerebellar patients. Brain 120(Pt 2):327–337
Grimaldi G, Manto M (2013) Anodal transcranial direct current stimulation (tDCS) decreases the amplitudes of long-latency stretch reflexes in cerebellar ataxia. Ann Biomed Eng 41(11):2437–2447
Oude Nijhuis LB, Hegeman J, Bakker M, Van Meel M, Bloem BR, Allum JH (2008) The influence of knee rigidity on balance corrections: a comparison with responses of cerebellar ataxia patients. Exp Brain Res 187(2):181–191
Ojha A, Alderink G, Rhodes S (2023) Coherence between electromyographic signals of anterior tibialis, soleus, and gastrocnemius during standing balance tasks. Front Hum Neurosci 17:1042758
Gilman S (1969) Fusimotor fiber responses in the decerebellate cat. Brain Res 14(1):218–221
Diener HC, Dichgans J, Bacher M, Guschlbauer B (1984) Improvement of ataxia in alcoholic cerebellar atrophy through alcohol abstinence. J Neurol 231(5):258–262
Sullivan EV, Rose J, Pfefferbaum A (2010) Physiological and focal cerebellar substrates of abnormal postural sway and tremor in alcoholic women. Biol Psychiatry 67(1):44–51
Hirono T, Ikezoe T, Yamagata M, Kato T, Kimura M, Ichihashi N (2021) Relationship between postural sway on an unstable platform and ankle plantar flexor force steadiness in community-dwelling older women. Gait Posture 84:227–231
Loram ID, Maganaris CN, Lakie M (2005) Active, non-spring-like muscle movements in human postural sway: how might paradoxical changes in muscle length be produced? J Physiol 564(Pt 1):281–293
Di Giulio I, Maganaris CN, Baltzopoulos V, Loram ID (2009) The proprioceptive and agonist roles of gastrocnemius, soleus and tibialis anterior muscles in maintaining human upright posture. J Physiol 587(Pt 10):2399–2416
Day JT, Lichtwark GA, Cresswell AG (2013) Tibialis anterior muscle fascicle dynamics adequately represent postural sway during standing balance. J Appl Physiol 115(12):1742–1750
Mauritz KH, Schmitt C, Dichgans J (1981) Delayed and enhanced long latency reflexes as the possible cause of postural tremor in late cerebellar atrophy. Brain 104(Pt 1):97–116
Kapteyn TS, Bles W, Njiokiktjien CJ, Kodde L, Massen CH, Mol JM (1983) Standardization in platform stabilometry being a part of posturography. Agressologie 24(7):321–326
Funding
This study was funded by China’s National Natural Science Foundation (grant 81901396), medical science research grants from Guangdong Province (nos. B2018088 and A2019439), and Science and Technology Program of Guangzhou, China (no. 202102080335).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the ethics committees of the researchers’ institutions.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Liu, C., Ma, G. et al. Three Hertz postural leg tremor impairs posture maintenance in multiple system atrophy-cerebellar type. Neurol Sci 45, 601–612 (2024). https://doi.org/10.1007/s10072-023-07036-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-07036-9