Abstract
Background
Rheumatoid arthritis (RA) increases the risk of cardiovascular disease (CVD), with inflammation playing a key role. Biologic and targeted synthetic drugs used to treat RA can induce systemic immunomodulation and may have pleiotropic effects on vascular function, making it crucial to investigate their impact on CVD risk in RA patients.
Methods
A systematic review of the literature was conducted to investigate the impact of biologic and targeted synthetic treatments approved for RA on various cardiovascular markers, including endothelial function, arterial stiffness, and subclinical atherosclerosis. Our analysis included a search of the MedLine (via PubMed) and Web of Science databases using a pre-determined search strategy. We conducted a narrative synthesis of the included studies due to heterogeneity in study design and outcome measures.
Results
From an initial pool of 647 records, we excluded 327 studies based on their titles and abstracts, and we selected 182 studies for final examination. Ultimately, 58 articles met our inclusion criteria and were included in our systematic review. Our analysis of these studies revealed a positive effect of biologic and targeted synthetic therapies on vascular dysfunction associated with RA. However, the impact of these treatments on subclinical atherosclerosis was inconsistent.
Conclusion
Overall, our systematic review provides important insights into the potential cardiovascular benefits of biologic and targeted synthetic treatments for RA by a still unknown mechanism. These findings can inform clinical practice and contribute to our understanding of their possible effects on early vascular pathology.
Key Points • Great heterogeneity of methods are used to evaluate the endothelial function and arterial stiffness in patients with RA on biologic and targeted synthetic antirheumatic drugs. • Most studies have shown a considerable improvement in endothelial function and arterial stiffness with TNFi, despite some studies reporting only transient or no improvement. • Anakinra and tocilizumab may have a beneficial effect on vascular function and endothelial injury, as indicated by increased FMD, coronary flow reserve, and reduced levels of biomarkers of endothelial function, while the overall impact of JAKi and rituximab remains inconclusive based on the reviewed studies. • To fully comprehend the distinctions between biologic therapies, more long-term, well-designed clinical trials are necessary using a homogeneous methodology. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, extensive scientific research has been devoted to investigating cardiovascular risk, given its major contribution to global mortality [1]. One significant focus has been the link between inflammation and atherosclerosis which was first hypothesized by Virchow in the nineteenth century and later elaborated upon by Ross [2]. Nowadays, clear evidence supports the concept that immune responses are critical in the initiation, progression and destabilization of atherosclerosis [3,4,5]. Excess mortality from cardiovascular diseases is reported in inflammatory arthritides [6]. Rheumatoid arthritis (RA) is the most common autoimmune rheumatic disease, with a prevalence of approximately 1% of adults worldwide [7]. RA is characterized by an increase in cardiovascular disease risk, comparable to that conferred by type 2 diabetes [8].
RA is considered an independent risk factor for cardiovascular disease (CVD) according to guidelines set forth by the European Society of Cardiology. In recognition of this elevated risk, the European Alliance of Associations of Rheumatology (EULAR) recommends that traditional CVD risk scores, which were originally designed for the general population, be adjusted for RA patients. Specifically, these scores should be multiplied by 1.5 to account for the additional impact of non-traditional risk factors, such as the patient's inflammatory status. By adjusting traditional CVD risk scores to account for the impact of RA and related inflammatory factors, clinicians can more accurately predict an RA patient's likelihood of experiencing a cardiovascular event [9, 10].
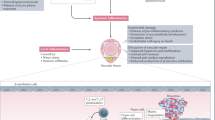
Inflammatory arthropathies and autoimmune rheumatic disorders are associated with a significantly increased risk of cardiovascular morbidity and mortality, which is thought to be driven by a complex interplay between high-grade inflammation and traditional cardiovascular risk factors [11]. Proinflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin 1 (IL-1), and interleukin 6 (IL-6) are known to play a critical role in the development of atherosclerosis, contributing to endothelial dysfunction and the development of premature atherosclerosis [3, 5, 12]. In light of the common pathophysiology underlying both inflammation and atherosclerosis, the mainstay of managing increased cardiovascular risk in patients with inflammatory diseases is achieving sufficient control of disease activity while also adequately addressing traditional cardiovascular risk factors [9].
The endothelium, a thin layer of cells that lines the inner surface of blood vessels, plays a critical role in maintaining vascular homeostasis and regulating arterial tone, coagulation, and smooth muscle cell proliferation. Healthy endothelium is crucial for normal cardiovascular function. In contrast, endothelial dysfunction is characterized by increased expression of adhesion molecules, pro-inflammatory cytokines, pro-thrombotic factors, oxidative stress upregulation, and abnormal vascular tone modulation. Endothelial injury represents an early stage of vascular disorders, and if not addressed, can lead to arterial stiffening, subclinical atherosclerosis, and ultimately, the development of arterial disease. Importantly, endothelial dysfunction, a recognized CVD risk factor, can be measured and is predictive of cardiovascular events in the general population [13]. Therefore, preserving endothelial health is an important goal in the prevention and management of vascular disorders [14].
In the last two decades, the therapeutic options for RA have been expanded with the addition of biologic agents with different mechanisms of action (TNF-α inhibitors [TNFi], IL-1 inhibitors, anti-CD20 monoclonal antibodies, inhibitors of T-lymphocytic co-stimulation, IL-6 inhibitors) and more recently, targeted synthetic drugs (janus kinase inhibitors [JAKi]) [15]. There is a mounting body of evidence that targeted therapies are link to reduction of CVD risk in threated individuals [16,17,18].
We aimed to analyze the available evidence on the potential effects of biologic and targeted synthetic drugs for RA on vascular function. Therefore, a systematic review was conducted to investigate their impact on endothelial function, markers of endothelial injury, arterial stiffness, or subclinical atherosclerosis.
Methods
Search strategy
A comprehensive literature search was conducted up to November 2022 using the Medline (via PubMed) and Web of Science databases. The primary search strategy utilized in Medline consisted of a string of relevant MeSH keywords and subject headings: ("adalimumab" OR "etanercept" OR "infliximab" OR "certolizumab pegol" OR "golimumab" OR “abatacept” OR “anakinra” OR “rituximab” OR “tocilizumab” OR "tofacitinib" OR "baricitinib" OR "upadacitinib") AND (“atherosclerosis” OR “vascular stiffness” OR “Carotid Intima-Media Thickness” OR “cIMT” OR “flow mediated dilat*” OR “FMD” OR “pulse wave velocity” OR “PWV” OR “endothelial dysfunction” OR “endothelial function” OR “forearm blood flow” OR “FBF” OR “peripheral arterial tonometry” OR “cardio-ankle vascular index” OR ”CAVI”). Moreover, to enhance the sensitivity of the search strategy, various relevant keywords were utilized in different combinations for a manual search. Furthermore, the reference lists of the selected articles were reviewed to ensure that no relevant studies were overlooked.
Inclusion criteria and study selection
In order to be eligible for inclusion in the final review, studies were required to satisfy the following predetermined inclusion criteria:
-
1)
Study design: randomized controlled trials (RCTs), quasi-randomized controlled trials (quasi-RCTs) which allocate treatments using methods such as alternation, alternate medical records, or date of birth, cross-sectional studies, prospective cohort studies that measured outcomes before and after an intervention, and meta-analyses.
-
2)
Population: studies involving human subjects.
-
3)
Intervention: treatment with TNF-α and IL-1 inhibitors, tocilizumab, rituximab, abatacept, and JAKi.
-
4)
Outcome: evaluation of the effect of treatment on measures of endothelial function, vascular stiffness, or biochemical markers of endothelial damage and subclinical atherosclerosis.
-
5)
Full-text articles.
Furthermore, we utilized the following exclusion criteria: a) studies published in a language other than English; b) studies that did not report p values for pre-post comparisons.
Data extraction and management
Two independent reviewers (GG and TG) evaluated the titles and abstracts of all the retrieved records to determine whether they met the inclusion criteria for the systematic review. After the initial screening phase, the same two reviewers independently assessed the remaining articles for eligibility based on the predetermined inclusion and exclusion criteria. Any discrepancies between the reviewers were resolved through discussion with two senior reviewers (MD and TS) until a consensus was reached. In instances where the necessary information was unclear, efforts were made to contact the authors of the original reports to obtain further details. The following information was extracted from each study: author names, publication year, study design, statistical method, risk factors (including hazard ratios or odds ratios and their corresponding 95% confidence intervals), as well as the number of cases and controls. Data relating to the impact of biologic and targeted synthetic treatment on endothelial function, vascular inflammation, vascular stiffness, and subclinical atherosclerosis were collected and recorded.
Reporting method
We followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines [http://www.prisma-statement.org/] and the recommendations for comprehensive searches through multiple databases of Gasparyan et al. [19] for preparing our manuscript.
Results
The search strategy yielded a total of 647 records, and no further relevant articles were identified through manual searching of bibliographies. After initial screening of titles and abstracts, 158 duplicate records were excluded, and 327 studies were considered ineligible based on predetermined inclusion and exclusion criteria. Thus, we retrieved the data from the remaining 162 studies. 32 studies examined endothelial function in patients with different inflammatory diseases (ankylosing spondylitis, systemic lupus, psoriasis) other than RA and thus were excluded. 28 studies were excluded because they studied the vascular effect of biologic drugs on non-inflammatory diseases – atherosclerosis, diabetes, heart failure, myocardial infarction, and others. We didn’t also include 15 scientific abstracts and congress posters that fulfilled other inclusion criteria. Other 29 studies were excluded because of wrong outcomes, wrong study design, published not in English, wrong intervention, wrong route of administration, wrong setting, no p values given. Thus, 58 studies were included in the final review. A PRISMA flowchart demonstrating the process of identification, screening, inclusion and exclusion of the studies is presented in Fig 1.
A PRISMA flowchart presenting the process of systematic search and selection of studies on the vascular effects of novel treatments in rheumatoid arthritis. Abbreviations: n – number, JAKi – janus kinase inhibitors, IL-1i – interleukin 1 inhibitors, IL-6i – interleukin 6 receptor inhibitors, TNFi – Tumor necrosis factor alpha inhibitors, WoS – Web of Science.
Tumor necrosis factor-alpha inhibitors
The most commonly prescribed biologic disease-modifying antirheumatic drugs (DMARDs) for RA are TNF-α inhibitors, which encompass a class of five drugs authorized for the treatment of RA: infliximab, adalimumab, etanercept, golimumab, and certolizumab pegol.
In our review, we included 40 studies examining the vascular effects of anti-TNF therapy. Vascular effects of TNFi were studied with different methods: endothelium-dependent (flow-mediated) dilatation of the brachial artery, endothelium-independent (nitro-glycerine induced) vasodilatation, laser-Doppler flowmetry, venous occlusion plethysmography, arterial stiffness assessed by pulse wave velocity, augmentation index and cardio-ankle vascular index (CAVI), common carotid artery intima thickness, dipyridamole Echocardiography and Coronary Flow Reserve, vascular inflammation by 18F-fluorodeoxyglucose positron emission tomography, aortic distensibility by cardiovascular MRI imaging. Studies also assessed biomarkers of endothelial injury including adiponectin, VEGF, ICAM-1, VCAM-1, E-selectin, endothelial progenitor cells (EPC), asymmetric dimethyl arginine (ADMA). An overview of the studies is presented in Table 1.
Out of the 40 studies that were included, 20 studies focused on analyzing the impact of TNF-α inhibitors on endothelial function. Sixteen studies used two primary methods for evaluating endothelial function, which were endothelium-dependent flow-mediated dilation (FMD) and endothelium-independent dilation following sublingual nitroglycerin of the brachial artery. Gonzales-Juanatey et al. [22], Irace et al. [23], and Bosselo et al. [32] found only transient improvement of FMD after treatment with infliximab, Spinelli et al. [50] did not find improvement, but other authors found significant improvement in their cohorts. In a cross-sectional analysis, Cypiene et al. [30], found greater effect of infliximab compared to synthetic DMARD, Tikiz et al. [39] compared etanercept to methotrexate (MTX) and found significant improvement of FMD after anti-TNF compared to MTX. In another study, Gonzales-Juanatey [27] examined patients that were non-responders to infliximab and switched from infliximab to adalimumab. In the observed cohort FMD increased to the same amount as in the control group. Two studies (Hansel [21] et al, Rongen et al. [52]) examined the effect of TNF-α inhibitors on venous occlusion plethysmography after receiving intrabrachial infusions of increasing doses of acetylcholine or a nitrate. Hansel et al examined patients on MTX with low disease activity starting etanercept and did not find improvement in endothelial function. In a substudy to the POEET trial, Roengen examined patients in clinical remission, randomized to stop or continue anti-TNF therapy. In patients who flared after stopping the biologic DMARD responses to acetylcholine and SNP were significantly reduced, but in patients who stopped and did not flare, vasodilator response did not differ between baseline and second visit. Dávida et al. [54] examined the effect of adalimumab on endothelial dysfunction by postocclusive reactive hyperemia (PORCH) tested by laser Doppler flow. They found favorable effects on endothelial function in the adalimumab group compared to the control group. Galaragga et al. [40] measured laser doppler flow after iontophoresis with acetylcholine and sodium nitroprusside on 31 patients treated with a TNF-α inhibitor and 20 patients treated with MTX. The authors found improvement of endothelial function only in patients that had responded to therapy, irrespective of treatment.
We included 13 studies examining the effect of anti-TNF therapy on arterial stiffness and one meta-analysis. The used methods for measurement were pulse wave velocity (PWV) in 11 studies, Augmentation index (AIx) in 10 studies, and CAVI in 1 study. Three studies did not find improvement in PWV after anti-TNF therapy (Komai et al. [29], Kerekes et al. [42], Daïen et al. [47]) and other studies found significant improvement. Interestingly Tam et al. [44] found a reduction in PWV regardless of clinical efficacy in patients treated with MTX+ Infliximab and a superior reduction of PWV compared to MTX monotherapy. Also, Vassilopoulos et al. [51] found statistically significant reduction in PWV both in responders and non-responders to adalimumab.
AIx is a composite measure of arterial stiffness. We included 10 studies that used AIx as an outcome. From them, 3 studies found a statistically significant reduction after treatment with TNFi. In the other studies, there was no change or the reduction was insignificant.
We included one meta-analysis in our review. Vlachopoulos et al. [53] analyzed 14 studies, examining the effect of TNFi on PWV and AIx on 320 patients. Some of the studies incorporate data from patients with different types of inflammatory arthritides and thus, were excluded from our review. Nonetheless, after a sensitivity analysis excluding studies recruiting both RA and seronegative spondylarthritis patients, the authors found a significant decrease in both PWV and AIx.
We included 10 studies on the effect of anti-TNF in carotid intima thickness (cIMT). In 4 studies (Del Porto et al. [31], Kerekes et al. [42], Anghel et al. [57], and Blanken et [58] ) anti-TNF therapy led to a decrease in cIMT, but the other studies did not find any change. We should note that one of the studies [31] that found improvement, included only responders to anti-TNF therapy, while another [58] did not report a p-value for that outcome.
From the included studies, 8 studied different serological markers of endothelial dysfunction and early atherosclerosis. Gonzalez-Gay et al. [25], found a rapid decrease of ICAM-3 and P-selectin, 120 minutes after infusion of infliximab and no improvement of ICAM-1, VCAM-1, or E-selectin. Bosello et al. [32] did not find an effect of anti-TNF-α therapy on ICAM-1, VCAM, VEGF, and E-Selectin levels; however, Klimiuk et al. [37] found a reduction of them in 18 patients treated with etanercept. Furthermore, Komai et al. [29] reported that infliximab increased levels of adiponectin in RA patients. Hjeltnes et al. [46] found a reduction of E-selectin and lipoprotein A but no effect on VCAM-1 and ICAM-1. Four studies used ADMA to measure endothelial dysfunction while 3 of them found a decrease after TNFi therapy [49, 50, 59] and one did not [38]. Ajeganova et al. [41] found that TNFi treatment increases atheroprotective IgM antibodies against phosphorylcholine, In contrast, a decrease of the antibodies were observed with rituximab treatment. One study [59] showed a reduction of homocysteine after treatment, that correlated with cIMT reduction. One study examined serum levels of sulphated glycosaminoglycans and in RA patients treated with TNFi as a novel biomarker of atherosclerosis. They found reduction of heparan sulphate/heparin after treatment associated with reduction of VCAM-1, MCP-1, MMP-9 and ADMA.
Two studies examined the effect of TNF-α inhibition on vascular inflammation measured by 18F-fluorodeoxyglucose-positron emission tomography. Blanken et al. [56] included patients with early RA starting conventional synthetic DMARD therapy and RA patients with established RA starting adalimumab. They reported a reduction of glucose uptake after treatment, regardless of treatment and DAS28 response, but the effect correlated with markers of systemic inflammation (ESR and CRP). The other study from Mäki-Petäjä K et al. [48] examined 17 patients with RA and a control group of patients with stable cardiovascular disease. After 8 weeks of TNFi treatment reduction of 18F-fluorodeoxyglucose uptake was found that correlated with reduction of PWV.
A bolt-on study [55] to a randomised control trial examined aortic distensibility measured by cardiovascular MRI on RA patients randomized to MTX plus etanercept or MTX-only treatment. Anti-inflammatory therapy improved aortic distensibility regardless of the treatment and DAS28 response.
Abatacept
Abatacept is a recombinant fusion protein that modulates the CD80/96-CD28 co-stimulatory signal required for T-cell activation. In our literature review, we identified three studies investigating the vascular effect of abatacept.
Mathieu et al. [60] investigated the effect of abatacept on aortic stiffness measured by PWV on a cohort of 21 RA patients fulfilling the 1987 ACR criteria, which did not respond to at least 2 TNFi or had contraindications for TNFi treatment. Following 6 months of treatment with abatacept, there was a significant rise in PWV levels (9.8±2.9 versus 8.5±3.9 m/second; P=0.02). The alterations in PWV demonstrated a correlation with changes in the Disease Activity Score on 28 joints based on erythrocyte sedimentation rate (r=0.46; P=0.035). The authors inferred that the insufficient reduction in systemic inflammation might have led to the progression of aortic stiffness in their study population. Of the 21 patients, only 6 reached remission during treatment and have no changes of PWV. The other 15 had persisting disease activity and their PWV increased.
In a longitudinal observational study conducted by Provan et al. [61], PWV was evaluated in RA patients before treatment and at 3, 6, and 12 months after receiving abatacept, rituximab, or tocilizumab. A total of 36 patients were recruited for the study, and administered different medications, including abatacept, rituximab, and tocilizumab to 5, 24, and 7 patients, respectively. No statistically significant change of PWV was observed in the 5 patients treated with abatacept. In the tocilizumab group, a statistically significant change of PWV (-0.9 p=0.03) was observed compared to other treatment groups.
Benucci et al. [62] retrospectively observed a group of 45 RA patients treated with abatacept with no known CVD or CV risk factors. They evaluated common carotid intima thickness (ccIMT) and flow-mediated dilatation (FMD) of the brachial artery at baseline and at the 12th month after treatment. During abatacept treatment, ccIMT remained stable and a low statistically insignificant increase of FMD was observed between baseline and at the 12th month. At baseline ccIMT inversely correlated with baseline TNF-α values (p=0.0245). At the end of the 12-month follow-up, a statistically significant correlation was observed between the number of CD3/CD8+ lymphocytes and ccIMT (p=0.0351), while CRP levels showed a significant correlation with FMD (p=0.0075). Furthermore, regression analysis demonstrated that baseline ccIMT and FMD had weak predictive ability for TNF-α (p=0.011) and CRP (p=0.049) at the 12th month.
Interleukin-1 receptor antagonist
Three articles were retrieved from the same author team in our systematic literature review related to anakinra, which is a recombinant human IL-1 receptor antagonist.
First, Ikonomidis et al. [63] studied the effects of anakinra administration on vascular function assessed in 23 RA patients by using FMD, coronary flow reserve, aortic distensibility, systolic, and diastolic (Em) velocity of the mitral annulus, and E to Em ratio (E/Em) using echocardiography, and markers of endothelial injury (malondialdehyde, nitrotyrosine, endothelin-1). The study evaluated the effects in “acute” and “chronic” conditions. A double-blind trial was conducted to study the impact of anakinra in the acute arm, in which a group of patients were randomly assigned to receive either a single subcutaneous injection of anakinra (n=12) or a placebo (n=11), and after 48 hours, they were administered the alternative treatment. In a non-randomized trial conducted on patients with chronic conditions, 23 of them received anakinra while 19 were treated with prednisolone for 30 days, and thereafter, all indicators were evaluated again. In the acute setting, the results of the study showed that anakinra caused an increase in FMD, while placebo resulted in a decrease (P=0.001), whereas nitrate-induced vasodilation remained unchanged (P=0.2). Resting and hyperaemic CF-Vmax, CF-VTI, and CF-VTId increased compared to placebo (P=0.001 for all comparisons). Coronary flow reserve (CFR) and aortic distensibility and strain were greater in anakinra than in placebo (P=0.001). From the biomarkers of endothelial function: a reduction of malondialdehyde, nitrotyrosine, and ET-1 was observed in the treatment group compared to placebo (P=0.001). In the chronic arm analysis, the authors observed a greater percentage increase in FMD and CFR after the use of anakinra compared to prednisolone (P=0.001). Additionally, there was a higher relative increase in all coronary flow indices after anakinra use (P=0.05). Nitrate-induced vasodilation remained unchanged between the two treatment groups. The study also found that aortic distensibility and strain showed a 3-fold increase after anakinra, which was significantly higher than the 7±3% and 11±3% increase observed after prednisolone (P=0.001 for all comparisons). Furthermore, anakinra treatment resulted in a greater percent reduction in malondialdehyde, nitrotyrosine, and endothelin-1 (ET-1) compared to prednisolone (malondialdehyde 33±2% versus 3±2%, P=0.006; nitrotyrosine 50±8% versus 0.5±1%, P=0.006; and ET-1 40±7% versus 22±4%, P=0.04).
A second paper [64] from the same authors observed a cohort of 46 RA patients compared to 23 healthy individuals. Half of the patients received anakinra and the other half did a 5-mg increase in prednisolone dose for 30 days. Study outcomes were myocardial deformation by speckle tracking echocardiography, CFR, FMD nitrotyrosine and malondialdehyde blood levels. In the anakinra group, there was an improvement of myocardial deformation, FMD, CFR, nitrotyrosine and malondialdehyde compared to baseline (p<0.05 for all comparisons). No effect in the prednisolone group was observed on myocardial deformation and worsening of FMD and CFR (p<0.001 for both).
A third paper [65] from the same authors examined the effects of anakinra on RA patients with known coronary atherosclerosis. In a double-blinded crossover trial, 60 patients with CAD and RA and 20 with RA only were randomized to receive a single injection of anakinra or placebo and then the alternative treatment after 48 hours. Compared to individuals without CAD, those with CAD exhibited lower levels of FMD, CFR, and impaired left ventricular function markers, along with significantly elevated levels of IL-1, oxidative stress, and soluble apoptotic markers. Anakinra treatment led to improvements in FMD, CFR-VTId, CFR-VTItotal, and systemic arterial compliance compared to baseline and placebo (all P<0.05). CAD patients had greater relative improvements in CFR, FMD, systemic arterial compliance, and resistance compared to non-CAD patients. Anakinra also improved tissue Doppler and speckle tracking markers of myocardial deformation and twisting. Levels of oxidative stress markers were markedly decreased after anakinra compared to placebo, and the relative decrease of the markers was greater in CAD than in non-CAD patients.
Rituximab
Rituximab is a chimeric monoclonal antibody that depletes CD20 positive B-lymphocytes and their precursors. We included 6 studies of rituximab effect on vascular dysfunction in RA patients. An overview of the studies can be found in Table 2.
Of the 6 described studies, none had a comparable control group and all studies included small numbers of patients. Flow-mediated dilatation of the brachial artery was used in 4 studies and all of them found an increase in FMD after treatment. cIMT was measured in two studies with no effect of therapy. Aortic stiffness measured by PWV and augmentation index were used in two studies. Provan et al found a decrease in PWV after 12 months while Mathieu et al did not find any change. Aix did not change after treatment in both studies.
Tocilizumab
Tocilizumab is a humanized IL-6 receptor-binding protein that binds to soluble and membrane-bound IL-6 receptors. We included 6 studies in our review found in Table 3.
Of the included studies, two were randomized control trials with different treatment arms, while the other 4 were observational cohorts. Three studies measured endothelial dysfunction by brachial FMD and found improvement of FMD after treatment. Arterial stiffness was assessed by 5 studies, 4 of them used PWV and one used cardio-ankle vascular index (CAVI). All studies found a decrease in arterial stiffness after treatment. cIMT was measured by Kume et al on 22 patients on tocilizumab and did not find a significant change after 24 weeks of therapy. Ikonomidis et al measured CFR in two studies and found a decrease after IL-6 treatment. In the two studies, markers of oxidative stress were measured- malondialdehyde (MDA) and protein carbonyls (PCs), which decreased by treatment. The second study by Ikonomidis et al. measured perfused boundary region (PBR) of the sublingual arterial microvessels – an indirect test for endothelial glycocalyx thickness. The authors found a decrease of PBR after treatment, which indicates improvement of endothelial glycocalyx thickness.
Janus kinase inhibitors
JAKi are novel targeted synthetic drugs inhibiting the JAK/STAT intercellular signaling. Three drugs are approved for the treatment of rheumatoid arthritis – tofacitinib, baricitinib, upadacitinib. We included 2 studies of the vascular effect of tofacitinib.
In the first study, Kume at al. (2017) [74] observed a cohort of 48 patients with RA on a stable dose of methotrexate who started treatment with tofacitinib (10mg/daily) due to disease activity (DAS28>3.2). The primary endpoint of the study was the change of cIMT; secondary endpoints were the change of cIMT in patients with atherosclerosis at baseline (cIMT>1.10mm), change of CAVI and AiX@75, change of carotid artery plaque (CAP) score, change of disease activity and lipid profile. After 54 weeks of treatment, no significant change of cIMT was observed (1.09 ± 0.69 and 1.08 ± 0.78 mm, p = 0.82). In contrast, there was a small but statistically significant decrease of cIMT (0.05± 0.026 mm; p < 0.05) in patients with atherosclerosis (n=12) at baseline. CAVI and AIX@75 decreased in the treatment groups (p < 0.01 and p< 0.01 respectively). CAP score did not change at follow-up; serum total cholesterol increased from baseline. The authors discussed that tofacitinib seemed to improve cIMT, CAVI and AIx@75 despite increasing serum lipid levels. Importantly, reducing cIMT in patients with RA who already have high levels of cIMT may help to diminish the extent of atherosclerosis.
Two studies from the same institution [75, 76] observed the same cohort of 30 patients with rheumatoid arthritis starting tofacitinib (randomly assigned to 5mg bid [n=15] and 10mg bid [n=15]) for 12 months. They were assessed at baseline, at 6 and 12 months. Assessment included brachial FMD, common cIMT and aortic PWV, as well as serological markers of early vascular disfunction, oxidative stress and cardiovascular risk: L-arginine, L-citrulline, L-ornithine, inducible nitric oxide synthase (iNOS), asymmetric ADMA and symmetric dimethylarginine (SDMA), L-N-monomethyl-arginine (L-NMMA), cysteine, homocysteine in the first study and lipid analyses (including TC, LDL-C, HDL-C, TG, lipoprotein(a), APOA and APOB), adipokines (adiponectin, chemerin, leptin, adipsin and resistin), myeloperoxidase, thrombospondin-1, paraoxonase 1 for the second study . From the observed cohort 4 patients dropped out, thus 26 patients completed the study. After 6 and 12 months of treatment FMD and PWV did not change, while cIMT slightly increased after 6 months (0.56 ± 0.12 mm; p = 0.05) and 12 months (0.59 ± 0.14 mm; p = 0.002). The first study found an increase of cysteine, homocysteine and methionine after 12 months of treatment in the 10mg bid group (p = 0.028, p = 0.049 and p < 0.001 respectively), but in the 5mg bid only change was seen in methionine (p = 0.002), but no change in homocysteine and cysteine. Authors also found increase of levels of L-arginine (p = 0.004 and p = 0.043), L-ornithine (p = 0.025 and p = 0.119) at 6 and 12 month compared to baseline. L-citrulline increase after 6 months (p = 0.006), but decreased at month 12th (p = 0.023) compared to the 6th month. iNOS levels after 12 months of treatment were significantly higher than those at baseline (p = 0.045) and 6 months (p = 0.020) in all patients. ADMA and SDMA did not alter significantly during JAKi treatment, whereas L-NMMA showed a transient increase at 6 months and a decrease after that. The study found a significant increase in TC, HDL, LDL, APOA, and APOB after treatment (p = 0.007, p = 0.004, p = 0.003, p = 0.001, p = 0.006 respectively). Leptin increased significantly at 12 months (p = 0.003), while chemerin showed a significant decrease after 12 months (p = 0.040). PON1 and MPO showed a numerical decrease after 6 and 12 months respectively, but PON1 significantly decreased after 12 months (p = 0.040) and MPO significantly decreased after 6 months (p = 0.028). TSP-1 significantly increased after 6 months (p = 0.009). Adiponectin, adipsin, and resistin only showed numerical changes. The authors concluded that tofacitinib has a balanced effect on metabolic markers of vascular dysfunction and does not affect endothelial dysfunction and aortic stiffness.
Discussion
The pathophysiology underlying the heightened risk of CVD in RA remains incompletely understood [77], although vascular dysfunction has emerged as a putative mechanism. The development of atherosclerosis is believed to involve endothelial dysfunction as a crucial factor that can be observed both on macrovasculature [78] and microvasculature [79] levels, leading to arterial stiffening and impaired vasodilation, respectively. Endothelial dysfunction is an early functional abnormality and if treated is potentially reversible, making it an interesting treatment target in RA patients [80]. Our systematic literature review reveals that biologic and targeted synthetic antirheumatic drugs have the potential to enhance vascular function among patients with RA. Our systematic review of 40 studies over the past two decades reveals that TNF inhibitors (infliximab, etanercept, and adalimumab) can improve endothelial function, which is often assessed by FMD. FMD measures the response of the endothelium to hypoxia by inducing reactive hyperemia through cuff inflation, which triggers vasodilation by releasing NO. Of the 20 studies that assessed FMD after treatment with TNF inhibitors, 16 reported improvement. Although four studies did not find improvement, three of them showed a transient increase after infliximab infusion, which may be due to the timing of drug administration. Other biologic treatments such as tocilizumab, anakinra, and rituximab also demonstrated improvement in FMD, while abatacept did not. A study on the JAK inhibitor tocilizumab did not show improvement in FMD.
Another method used for endothelial dysfunction measurement is forearm blood flow (FBF) occlusion plethysmography [81]. This is an invasive method in which acetylcholine is injected into the brachial artery and FBF is measured by a strain gauge plethysmograph. It was used in 2 studies for TNFi, but we did not find studies on other drugs using this method. Two other non-invasive measuring methods assessing endothelial dysfunction were also used: laser Doppler flow (LDF) imaging with iontophoresis [82] and endothelial glycocalyx thickness. The latter was evaluated by measuring the perfused boundary region (PBR) by Sidestream Darkfield (SDF) imaging [83]. LDF was used in two studies on TNFi, while PBR was assessed in a single study on IL-6 inhibitor. Positive effects were observed for both drug classes.
Arterial stiffness is a measurement of the elastic properties of the large bore arteries. Abnormal stiffening of the arterial wall evaluated by means of PWV represents a well-established independent predictor of CVD in the general population. The most common measurement method used is pulse wave velocity. This method measures the speed at which a pulse wave travels through the arteries. A faster pulse wave velocity indicates increased arterial stiffness. PWV is measured non-invasively using a device that measures blood pressure at two points on the body. Different points can be used, making three different PWV methods: carotid-femoral PWV (cfPWV) [84], heart-femoral (hfPWV) [85], and brachial-ankle (baPWV) [86]. Another method for arterial stiffness measurement is the CAVI [87]. The principle is based on stiffness parameter β, which is an index obtained from changes in arterial diameter and measured with the pulse in one section by a two-dimensional imaging technique. The β factor is used to calculate CAVI from the PWV and is adjusted for body size making it independent of height and body weight. In our review, we found 9 studies showing improvement of PWV after anti-TNF therapy. IL-6 inhibitors improved PWV in 5 studies. In one small non-controlled study, the JAK inhibitor tofacitinib improved CAVI after 54 weeks of treatment. One study found improvement of PWV in rituximab patients but another study did not find any effect. We found no studies on the effect of anakinra on arterial stiffness and one study on the effect of abatacept, where PWV increased.
Aortic distensibility is a measure of the ability of the aorta to expand and contract in response to changes in blood pressure. Aortic distensibility can be measured using magnetic resonance imaging by assessing changes in the diameter of the aorta in response to changes in blood pressure [88]. A higher value of aortic distensibility indicates that the aorta is more compliant and able to expand and contract in response to changes in blood pressure, while a lower value indicates that the aorta is less compliant and less able to accommodate changes in blood pressure. Aortic distensibility is a surrogate marker for aortic stiffness and its decrease is associated with an increased risk of cardiovascular disease and mortality [89, 90]. We found one bolt-on study to the VEDERA (Very Early vs. Delayed Etanercept in Rheumatoid Arthritis) randomized controlled trial which demonstrated improvement of aortic distensibility after 1 year of treatment with etanercept in patients with early rheumatoid arthritis.
cIMT measurement is an ultrasound technique used to assess the thickness of the inner lining of the carotid artery [91]. The usefulness of cIMT in CVD risk assessment has been consistently confirmed in RA patients [92]. The cIMT is measured as the distance between the lumen-intima and media-adventitia interfaces, typically at the far wall of the carotid artery. cIMT is considered a fixed structural alteration of the arterial wall, in which both inflammatory and non-inflammatory mechanisms play an important role. Results of the effects on cIMT are inconsistent. We included 10 studies on TNF-alpha inhibitors and 4 of them showed a decrease of cIMT after treatment, but the other showed no improvement (and apparently no worsening). The analyzed studies did not find an effect on cIMT with IL-6 inhibitor and rituximab treatment. We did not found data for the other therapies of interest.
Our findings suggest that inflammation in the major arteries may contribute to the vascular complications seen in RA. Increased uptake of radioisotopic glucose measured by PET-CT is predictive of cardiovascular risk in the general population [93] and in patients with inflammatory joint diseases [94, 95]. Two studies were identified that investigated the effects of TNF-alpha inhibition on vascular inflammation using 18F-fluorodeoxyglucose-positron emission tomography. In one of the studies, no significant differences were found between responders and non-responders to anti-inflammatory treatment, and no correlation was observed between changes in RA disease activity markers and vascular inflammation. The study also suggested that synovitis and vascular inflammation may be distinct processes, as treatment responses did not coincide in the joints and the vasculature. In the other study, there was a correlation between decreased fluorodeoxyglucose uptake and a reduction in pulse wave velocity after TNF inhibitor treatment, indicating that vascular inflammation may play a role in the development of arterial stiffness.
Our systematic review included studies that looked at various serological markers for early vascular damage, and the results were inconsistent. One such marker is ADMA, which is a marker of endothelial dysfunction that works by inhibiting the nitric oxide synthases (NOSs) that play a role in the development of endothelial dysfunction. High levels of ADMA can result in reduced production of nitric oxide (NO), which can contribute to the development of cardiovascular disease [96]. Three studies on TNFi demonstrated a decrease in ADMA after treatment and one study on tofacitinib did not show any effect.
sVCAMs are a group of molecules that are shed from the surface of endothelial cells into the bloodstream [97]. They play an important role in the regulation of leukocyte (white blood cell) recruitment to sites of inflammation. sVCAMs are involved in the adhesion and migration of immune cells to the walls of blood vessels, which is a crucial step in the initiation and progression of inflammatory diseases, including atherosclerosis and rheumatoid arthritis [98,99,100]. Although several studies have investigated the impact of TNFi on adhesion molecules, the findings have been inconclusive, and no studies have yet assessed the effects of the other medications of interest.
Strengths and limitations
Although we conducted a thorough and inclusive review of the existing literature, our study has significant limitations, and the results should be interpreted with caution. The primary constraint of our systematic literature review is the inadequate methodological quality of the majority of the studies included. Out of the 58 studies we included in our analysis, only 9 were randomized controlled trials and just 4 studies compared the effects of different biologic therapies on vascular function. In addition, our search strategy did not consider sarilumab, which is an inhibitor of IL-6, because there is insufficient information about its vascular effects to draw meaningful conclusions. The limited number of studies examining off-target effects on vascular repair mechanisms and endothelial damage highlights the need for improved understanding to ensure optimal treatment and personalized care based on individual cardiovascular risk profiles. Although there is a rationale for adverse cardiovascular effects of novel treatments for RA [101], they were outside the scope of this review. To fully comprehend the distinctions between biologic therapies, more long-term, well-designed clinical trials are necessary.
Conclusion
Overall, our systematic review provides important insights into the potential cardiovascular benefits of biologic and targeted synthetic treatments for RA, as those drugs may improve vascular function by a still unknown mechanism. Considering the persistent and chronic nature of RA, the management plan should consider all potential advantages and drawback of immunosuppressive treatments on cardiovascular outcomes. As the most extensively studied, TNF inhibitors have demonstrated efficacy in enhancing endothelial function, reducing arterial stiffness, improving aortic distensibility, and decreasing cIMT, all of which are essential indicators of cardiovascular health in individuals diagnosed with RA. These findings can inform clinical practice, contribute to our understanding of the medications impact on early vascular pathology, and help guide treatment decisions in RA patients with CVR factors. Future studies are warranted to observe potential differences in their effects on CVR.
References
Roth GA, Johnson C, Abajobir A et al (2017) Global, Regional, and National Burden of Cardiovascular Diseases for 10 Causes, 1990 to 2015. J Am Coll Cardiol 70:1–25. https://doi.org/10.1016/j.jacc.2017.04.052
Ross R (1999) Atherosclerosis — An Inflammatory Disease. N EnglJ Med 340:115–126. https://doi.org/10.1056/NEJM199901143400207
Ridker PM (2019) Anticytokine Agents. Circ Res 124:437–450. https://doi.org/10.1161/CIRCRESAHA.118.313129
Tocci G, Goletti D, Marino V et al (2016) Cardiovascular outcomes and tumour necrosis factor antagonists in chronic inflammatory rheumatic disease: a focus on rheumatoid arthritis. Expert Opin Drug Saf 15:55–61. https://doi.org/10.1080/14740338.2016.1218469
Tanaka T, Kishimoto T (2014) The Biology and Medical Implications of Interleukin-6. Cancer Immunol Res 2:288–294. https://doi.org/10.1158/2326-6066.CIR-14-0022
Meyer PW, Anderson R, Ker JA, Ally MT (2018) Rheumatoid arthritis and risk of cardiovascular disease. Cardiovasc J Afr 29:317–321. https://doi.org/10.5830/CVJA-2018-018
Almutairi K, Nossent J, Preen D et al (2021) The global prevalence of rheumatoid arthritis: a meta-analysis based on a systematic review. Rheumatol Int 41:863–877. https://doi.org/10.1007/s00296-020-04731-0
Løgstrup BB, Ellingsen T, Pedersen AB et al (2021) Cardiovascular risk and mortality in rheumatoid arthritis compared with diabetes mellitus and the general population. Rheumatology 60:1400–1409. https://doi.org/10.1093/rheumatology/keaa374
Agca R, Heslinga SC, Rollefstad S et al (2017) EULAR recommendations for cardiovascular disease risk management in patients with rheumatoid arthritis and other forms of inflammatory joint disorders: 2015/2016 update. Ann Rheum Dis 76:17–28. https://doi.org/10.1136/annrheumdis-2016-209775
Visseren FLJ, Mach F, Smulders YM et al (2021) 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J 42:3227–3337. https://doi.org/10.1093/eurheartj/ehab484
Arida A, Protogerou A, Kitas G, Sfikakis P (2018) Systemic Inflammatory Response and Atherosclerosis: The Paradigm of Chronic Inflammatory Rheumatic Diseases. Int J Mol Sci 19:1890. https://doi.org/10.3390/ijms19071890
Ridker PM, Rane M (2021) Interleukin-6 Signaling and Anti-Interleukin-6 Therapeutics in Cardiovascular Disease. Circ Res 128:1728–1746. https://doi.org/10.1161/CIRCRESAHA.121.319077
Murdaca G, Colombo BM, Cagnati P et al (2012) Endothelial dysfunction in rheumatic autoimmune diseases. Atherosclerosis 224:309–317. https://doi.org/10.1016/j.atherosclerosis.2012.05.013
Poredos P, Poredos AV, Gregoric I (2021) Endothelial Dysfunction and Its Clinical Implications. Angiology 72:604–615. https://doi.org/10.1177/0003319720987752
Smolen JS, Landewé RBM, Bijlsma JWJ et al (2020) EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. Ann Rheum Dis 79:685–699. https://doi.org/10.1136/annrheumdis-2019-216655
Xie F, Yun H, Levitan EB et al (2019) Tocilizumab and the Risk of Cardiovascular Disease: Direct Comparison Among Biologic Disease-Modifying Antirheumatic Drugs for Rheumatoid Arthritis Patients. Arthritis Care Res (Hoboken) 71:1004–1018. https://doi.org/10.1002/acr.23737
Singh S, Fumery M, Singh AG et al (2020) Comparative Risk of Cardiovascular Events With Biologic and Synthetic Disease-Modifying Antirheumatic Drugs in Patients With Rheumatoid Arthritis: A Systematic Review and Meta-Analysis. Arthritis Care Res (Hoboken) 72:561–576. https://doi.org/10.1002/acr.23875
Naranjo A, Sokka T, Descalzo MA et al (2008) Cardiovascular disease in patients with rheumatoid arthritis: results from the QUEST-RA study. Arthritis Res Ther 10:R30. https://doi.org/10.1186/ar2383
Gasparyan AY, Ayvazyan L, Blackmore H, Kitas GD (2011) Writing a narrative biomedical review: considerations for authors, peer reviewers, and editors. Rheumatol Int 31:1409–1417. https://doi.org/10.1007/s00296-011-1999-3
Hürlimann D, Forster A, Noll G et al (2002) Anti–Tumor Necrosis Factor-α Treatment Improves Endothelial Function in Patients With Rheumatoid Arthritis. Circulation 106:2184–2187. https://doi.org/10.1161/01.CIR.0000037521.71373.44
Hänsel S, Lässig G, Pistrosch F, Passauer J (2003) Endothelial dysfunction in young patients with long-term rheumatoid arthritis and low disease activity. Atherosclerosis 170:177–180. https://doi.org/10.1016/S0021-9150(03)00281-8
Gonzalez-Juanatey C, Testa A, Garcia-Castelo A et al (2004) Active but transient improvement of endothelial function in rheumatoid arthritis patients undergoing long-term treatment with anti-tumor necrosis factor α antibody. Arthritis Care Res (Hoboken) 51:447–450. https://doi.org/10.1002/art.20407
Irace C, Mancuso G, Fiaschi E et al (2004) Effect of anti TNFalpha therapy on arterial diameter and wall shear stress and HDL cholesterol. Atherosclerosis 177:113–118. https://doi.org/10.1016/j.atherosclerosis.2004.04.031
van Doornum S, McColl G, Wicks IP (2005) Tumour necrosis factor antagonists improve disease activity but not arterial stiffness in rheumatoid arthritis. Rheumatology 44:1428–1432. https://doi.org/10.1093/rheumatology/kei033
Gonzalez-Gay MA, Garcia-Unzueta MT, de Matias JM et al (2006) Influence of anti-TNF-alpha infliximab therapy on adhesion molecules associated with atherogenesis in patients with rheumatoid arthritis. Clin Exp Rheumatol 24:373–379
Bilsborough W, Keen H, Taylor A et al (2006) Anti-tumour necrosis factor-alpha therapy over conventional therapy improves endothelial function in adults with rheumatoid arthritis. Rheumatol Int 26:1125–1131. https://doi.org/10.1007/s00296-006-0147-y
Gonzalez-Juanatey C, Llorca J, Sanchez-Andrade A et al (2006) Short-term adalimumab therapy improves endo-thelial function in patients with rheumatoid arthritis refractory to infliximab. Clin Exp Rheumatol 24:309–312
Mäki-Petäjä KM, Hall FC, Booth AD et al (2006) Rheumatoid Arthritis Is Associated With Increased Aortic Pulse-Wave Velocity, Which Is Reduced by Anti–Tumor Necrosis Factor-α Therapy. Circulation 114:1185–1192. https://doi.org/10.1161/CIRCULATIONAHA.105.601641
Komai N, Morita Y, Sakuta T et al (2007) Anti-tumor necrosis factor therapy increases serum adiponectin levels with the improvement of endothelial dysfunction in patients with rheumatoid arthritis. Mod Rheumatol 17:385–390. https://doi.org/10.1007/s10165-007-0605-8
Cypiene A, Laucevicius A, Venalis A et al (2007) Non-invasive assessment of arterial stiffness indices by applanation tonometry and pulse wave analysis in patients with rheumatoid arthritis treated with TNF-alpha blocker remicade (infliximab). Proc West Pharmacol Soc 50:119–122
del Porto F, Lagana B, Lai S et al (2007) Response to anti-tumour necrosis factor alpha blockade is associated with reduction of carotid intima-media thickness in patients with active rheumatoid arthritis. Rheumatology 46:1111–1115. https://doi.org/10.1093/rheumatology/kem089
Bosello S, Santoliquido A, Zoli A et al (2008) TNF-alpha blockade induces a reversible but transient effect on endothelial dysfunction in patients with long-standing severe rheumatoid arthritis. Clin Rheumatol 27:833–839. https://doi.org/10.1007/s10067-007-0803-y
Wong M, Oakley SP, Young L et al (2009) Infliximab improves vascular stiffness in patients with rheumatoid arthritis. Ann Rheum Dis 68:1277–1284. https://doi.org/10.1136/ard.2007.086157
Sidiropoulos PI, Siakka P, Pagonidis K et al (2009) Sustained improvement of vascular endothelial function during anti-TNFα treatment in rheumatoid arthritis patients. Scand J Rheumatol 38:6–10. https://doi.org/10.1080/03009740802363768
Capria A, de Nardo D, Baffetti FR et al (2010) Long-Term Anti-TNF-α Treatments Reverse the Endothelial Dysfunction in Rheumatoid Arthritis: The Biological Coherence between Synovial and Endothelial Inflammation. Int J Immunopathol Pharmacol 23:255–262. https://doi.org/10.1177/039463201002300123
Galarraga B, Khan F, Kumar P et al (2009) Etanercept improves inflammation-associated arterial stiffness in rheumatoid arthritis. Rheumatology 48:1418–1423. https://doi.org/10.1093/rheumatology/kep251
Klimiuk P, Sierakowski S, Domyslawska I, Chwiecko J (2009) Effect of etanercept on serum levels of soluble cell adhesion molecules (sICAM-1, sVCAM-1, and sE-selectin) and vascular endothelial growth factor in patients with rheumatoid arthritis. Scand J Rheumatol 38:439–444. https://doi.org/10.3109/03009740903079321
Turiel M, Tomasoni L, Sitia S et al (2010) RESEARCH: Effects of Long-Term Disease-Modifying Antirheumatic Drugs on Endothelial Function in Patients with Early Rheumatoid Arthritis. Cardiovasc Ther 28:e53–e64. https://doi.org/10.1111/j.1755-5922.2009.00119.x
Tikiz H, Arslan O, Pirildar T et al (2010) The effect of anti-tumor necrosis factor (TNF)-alpha therapy with etanercept on endothelial functions in patients with rheumatoid arthritis. Anadolu Kardiyoloji Dergisi/The Anatolian Journal of Cardiology 10:98–103. https://doi.org/10.5152/akd.2010.031
Galarraga B, JJF B, Pullar T et al (2010) Clinical Improvement in Rheumatoid Arthritis Is Associated with Healthier Microvascular Function in Patients Who Respond to Antirheumatic Therapy. J Rheumatol 37:521–528. https://doi.org/10.3899/jrheum.090417
Ajeganova S, Fiskesund R, de Faire U et al (2011) Effect of biological therapy on levels of atheroprotective antibodies against phosphorylcholine and apolipoproteins in rheumatoid arthritis - a one year study. Clin Exp Rheumatol 29:942–950
Kerekes G, Soltész P, Szucs G et al (2011) Effects of adalimumab treatment on vascular disease associated with early rheumatoid arthritis. Isr Med Assoc J 13:147–152
Kume K, Amano K, Yamada S et al (2011) Tocilizumab Monotherapy Reduces Arterial Stiffness as Effectively as Etanercept or Adalimumab Monotherapy in Rheumatoid Arthritis: An Open-label Randomized Controlled Trial. J Rheumatol 38:2169–2171. https://doi.org/10.3899/jrheum.110340
Tam L-S, Shang Q, Li EK et al (2012) Infliximab is Associated with Improvement in Arterial Stiffness in Patients with Early Rheumatoid Arthritis — A Randomized Trial. J Rheumatol 39:2267–2275. https://doi.org/10.3899/jrheum.120541
Gonzalez-Juanatey C, Vazquez-Rodriguez TR, Miranda-Filloy JA et al (2012) Anti-TNF-Alpha-Adalimumab Therapy Is Associated with Persistent Improvement of Endothelial Function without Progression of Carotid Intima-Media Wall Thickness in Patients with Rheumatoid Arthritis Refractory to Conventional Therapy. Mediators Inflamm 2012:1–8. https://doi.org/10.1155/2012/674265
Hjeltnes G, Hollan I, Førre O et al (2013) Serum levels of lipoprotein(a) and E-selectin are reduced in rheumatoid arthritis patients treated with methotrexate or methotrexate in combination with TNF-α-inhibitor. Clin Exp Rheumatol 31:415–421
Daïen CI, Fesler P, du Cailar G et al (2013) Etanercept normalises left ventricular mass in patients with rheumatoid arthritis. Ann Rheum Dis 72:881–887. https://doi.org/10.1136/annrheumdis-2012-201489
Mäki-Petäjä KM, Elkhawad M, Cheriyan J et al (2012) Anti-Tumor Necrosis Factor-α Therapy Reduces Aortic Inflammation and Stiffness in Patients With Rheumatoid Arthritis. Circulation 126:2473–2480. https://doi.org/10.1161/CIRCULATIONAHA.112.120410
Spinelli FR, Metere A, Barbati C et al (2013) Effect of Therapeutic Inhibition of TNF on Circulating Endothelial Progenitor Cells in Patients with Rheumatoid Arthritis. Mediators Inflamm 2013:1–8. https://doi.org/10.1155/2013/537539
Spinelli FR, di Franco M, Metere A et al (2014) Decrease of Asymmetric Dimethyl Arginine After Anti-TNF Therapy in Patients with Rheumatoid Arthritis. Drug Dev Res 75:S67–S69. https://doi.org/10.1002/ddr.21200
Vassilopoulos D, Gravos A, Vlachopoulos C et al (2015) Adalimumab decreases aortic stiffness independently of its effect in disease activity in patients with rheumatoid arthritis. Clin Rheumatol 34:359–364. https://doi.org/10.1007/s10067-014-2718-8
Rongen GA, van Ingen I, Kok M et al (2018) Vasodilator function worsens after cessation of tumour necrosis factor inhibitor therapy in patients with rheumatoid arthritis only if a flare occurs. Clin Rheumatol 37:909–916. https://doi.org/10.1007/s10067-017-3961-6
Vlachopoulos C, Gravos A, Georgiopoulos G et al (2018) The effect of TNF-a antagonists on aortic stiffness and wave reflections: a meta-analysis. Clin Rheumatol 37:515–526. https://doi.org/10.1007/s10067-017-3657-y
Dávida L, Pongrácz V, Mohamed EA et al (2020) A prospective, longitudinal monocentric study on laser Doppler imaging of microcirculation: comparison with macrovascular pathophysiology and effect of adalimumab treatment in early rheumatoid arthritis. Rheumatol Int 40:415–424. https://doi.org/10.1007/s00296-019-04503-5
Plein S, Erhayiem B, Fent G et al (2020) Cardiovascular effects of biological versus conventional synthetic disease-modifying antirheumatic drug therapy in treatment-naïve, early rheumatoid arthritis. Ann Rheum Dis 79:1414–1422. https://doi.org/10.1136/annrheumdis-2020-217653
Blanken AB, Agca R, van Sijl AM et al (2021) Arterial wall inflammation in rheumatoid arthritis is reduced by anti-inflammatory treatment. Semin Arthritis Rheum 51:457–463. https://doi.org/10.1016/j.semarthrit.2021.03.008
Anghel D, Sîrbu C, Hoinoiu E-M et al (2021) Influence of anti-TNF therapy and homocysteine level on carotid intima-media thickness in rheumatoid arthritis patients. Exp Ther Med 23:59. https://doi.org/10.3892/etm.2021.10981
Blanken AB, Raadsen R, Agca R et al (2022) Effect of anti-inflammatory therapy on vascular biomarkers for subclinical cardiovascular disease in rheumatoid arthritis patients. Rheumatol Int 43(2):315–322. https://doi.org/10.1007/s00296-022-05226-w
Szeremeta A, Jura-Półtorak A, Zoń-Giebel A et al (2022) TNF-α Inhibitors in Combination with MTX Reduce Circulating Levels of Heparan Sulfate/Heparin and Endothelial Dysfunction Biomarkers (sVCAM-1, MCP-1, MMP-9 and ADMA) in Women with Rheumatoid Arthritis. J Clin Med 11:4213. https://doi.org/10.3390/jcm11144213
Mathieu S, Pereira B, Dubost J-J et al (2012) No significant change in arterial stiffness in RA after 6 months and 1 year of rituximab treatment. Rheumatology 51:1107–1111. https://doi.org/10.1093/rheumatology/kes006
Provan SA, Berg IJ, Hammer HB et al (2015) The Impact of Newer Biological Disease Modifying Anti-Rheumatic Drugs on Cardiovascular Risk Factors: A 12-Month Longitudinal Study in Rheumatoid Arthritis Patients Treated with Rituximab. Abatacept and Tociliziumab. PLoS One 10:e0130709. https://doi.org/10.1371/journal.pone.0130709
Benucci M, Saviola M et al (2013) Factors correlated with improvement of endothelial dysfunction during rituximab therapy in patients with rheumatoid arthritis. Biologics 69. https://doi.org/10.2147/BTT.S39182
Ikonomidis I, Lekakis JP, Nikolaou M et al (2008) Inhibition of Interleukin-1 by Anakinra Improves Vascular and Left Ventricular Function in Patients With Rheumatoid Arthritis. Circulation 117:2662–2669. https://doi.org/10.1161/CIRCULATIONAHA.107.731877
Ikonomidis I, Tzortzis S, Lekakis J et al (2009) Lowering interleukin-1 activity with anakinra improves myocardial deformation in rheumatoid arthritis. Heart 95:1502–1507. https://doi.org/10.1136/hrt.2009.168971
Ikonomidis I, Tzortzis S, Andreadou I et al (2014) Increased Benefit of Interleukin-1 Inhibition on Vascular Function, Myocardial Deformation, and Twisting in Patients With Coronary Artery Disease and Coexisting Rheumatoid Arthritis. Circ Cardiovasc Imaging 7:619–628. https://doi.org/10.1161/CIRCIMAGING.113.001193
Gonzalez-Juanatey C, Llorca J, Vazquez-Rodriguez TR et al (2008) Short-term improvement of endothelial function in rituximab-treated rheumatoid arthritis patients refractory to tumor necrosis factor α blocker therapy. Arthritis Rheum 59:1821–1824. https://doi.org/10.1002/art.24308
Kerekes G, Soltész P, Dér H et al (2009) Effects of rituximab treatment on endothelial dysfunction, carotid atherosclerosis, and lipid profile in rheumatoid arthritis. Clin Rheumatol 28:705–710. https://doi.org/10.1007/s10067-009-1095-1
Hsue PY, Scherzer R, Grunfeld C et al (2014) Depletion of B-Cells With Rituximab Improves Endothelial Function and Reduces Inflammation Among Individuals With Rheumatoid Arthritis. J Am Heart Assoc 3. https://doi.org/10.1161/JAHA.114.001267
Protogerou AD, Zampeli E, Fragiadaki K et al (2011) A pilot study of endothelial dysfunction and aortic stiffness after interleukin-6 receptor inhibition in rheumatoid arthritis. Atherosclerosis 219:734–736. https://doi.org/10.1016/j.atherosclerosis.2011.09.015
McInnes IB, Thompson L, Giles JT et al (2015) Effect of interleukin-6 receptor blockade on surrogates of vascular risk in rheumatoid arthritis: MEASURE, a randomised, placebo-controlled study. Ann Rheum Dis 74:694–702. https://doi.org/10.1136/annrheumdis-2013-204345
Bacchiega BC, Bacchiega AB, Usnayo MJG et al (2017) Interleukin 6 Inhibition and Coronary Artery Disease in a High-Risk Population: A Prospective Community-Based Clinical Study. J Am Heart Assoc 6. https://doi.org/10.1161/JAHA.116.005038
Ikonomidis I, Pavlidis G, Katsimbri P et al (2019) Differential effects of inhibition of interleukin 1 and 6 on myocardial, coronary and vascular function. Clin Res Cardiol 108:1093–1101. https://doi.org/10.1007/s00392-019-01443-9
Ikonomidis I, Pavlidis G, Katsimbri P et al (2020) Tocilizumab improves oxidative stress and endothelial glycocalyx: A mechanism that may explain the effects of biological treatment on COVID-19. Food Chem Toxicol 145:111694. https://doi.org/10.1016/j.fct.2020.111694
Kume K, Amano K, Yamada S et al (2017) Tofacitinib improves atherosclerosis despite up-regulating serum cholesterol in patients with active rheumatoid arthritis: a cohort study. Rheumatol Int 37:2079–2085. https://doi.org/10.1007/s00296-017-3844-9
Soós B, Hamar A, Pusztai A et al (2022) Effects of tofacitinib therapy on arginine and methionine metabolites in association with vascular pathophysiology in rheumatoid arthritis: A metabolomic approach. Front Med (Lausanne) 9. https://doi.org/10.3389/fmed.2022.1011734
Czókolyová M, Hamar A, Pusztai A et al (2022) Effects of One-Year Tofacitinib Therapy on Lipids and Adipokines in Association with Vascular Pathophysiology in Rheumatoid Arthritis. Biomolecules 12:1483. https://doi.org/10.3390/biom12101483
Yuri Gasparyan A, Stavropoulos-Kalinoglou AP, Mikhailidis D et al (2010) The Rationale for Comparative Studies of Accelerated Atherosclerosis in Rheumatic Diseases. Curr Vasc Pharmacol 8:437–449. https://doi.org/10.2174/157016110791330852
Bots ML, Dijk JM, Oren A, Grobbee DE (2002) Carotid intima–media thickness, arterial stiffness and risk of cardiovascular disease. J Hypertens 20:2317–2325. https://doi.org/10.1097/00004872-200212000-00002
Bordy R, Totoson P, Prati C et al (2018) Microvascular endothelial dysfunction in rheumatoid arthritis. Nat Rev Rheumatol 14:404–420. https://doi.org/10.1038/s41584-018-0022-8
Prati C, Demougeot C, Guillot X et al (2014) Endothelial dysfunction in joint disease. Joint Bone Spine 81:386–391. https://doi.org/10.1016/j.jbspin.2014.01.014
Wilkinson IB, Webb DJ (2001) Venous occlusion plethysmography in cardiovascular research: methodology and clinical applications. Br J Clin Pharmacol 52:631–646. https://doi.org/10.1046/j.0306-5251.2001.01495.x
Puissant C, Abraham P, Durand S et al (2013) Reproducibility of Non-Invasive Assessment of Skin Endothelial Function Using Laser Doppler Flowmetry and Laser Speckle Contrast Imaging. PLoS One 8:e61320. https://doi.org/10.1371/journal.pone.0061320
Cerny V, Astapenko D, Burkovskiy I et al (2017) Glycocalyx in vivo measurement. Clin Hemorheol Microcirc 67:499–503. https://doi.org/10.3233/CH-179235
van Bortel LM, Laurent S, Boutouyrie P et al (2012) Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J Hypertens 30:445–448. https://doi.org/10.1097/HJH.0b013e32834fa8b0
Stone K, Fryer S, Faulkner J et al (2021) Acute Changes in Carotid-Femoral Pulse-Wave Velocity Are Tracked by Heart-Femoral Pulse-Wave Velocity. Front Cardiovasc Med 7. https://doi.org/10.3389/fcvm.2020.592834
Sang T, Lv N, Dang A et al (2021) Brachial-ankle pulse wave velocity and prognosis in patients with atherosclerotic cardiovascular disease: a systematic review and meta-analysis. Hypertension Research 44:1175–1185. https://doi.org/10.1038/s41440-021-00678-2
Saiki A, Ohira M, Yamaguchi T et al (2020) New Horizons of Arterial Stiffness Developed Using Cardio-Ankle Vascular Index (CAVI). J Atheroscler Thromb 27:732–748. https://doi.org/10.5551/jat.RV17043
Messroghli DR, Moon JC, Ferreira VM et al (2017) Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: A consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI). J Cardiovasc Magn Reson 19:75. https://doi.org/10.1186/s12968-017-0389-8
Swoboda PP, Erhayiem B, Kan R et al (2018) Cardiovascular magnetic resonance measures of aortic stiffness in asymptomatic patients with type 2 diabetes: association with glycaemic control and clinical outcomes. Cardiovasc Diabetol 17:35. https://doi.org/10.1186/s12933-018-0681-4
Maroules CD, Khera A, Ayers C et al (2014) Cardiovascular outcome associations among cardiovascular magnetic resonance measures of arterial stiffness: the Dallas heart study. J Cardiovasc Magn Reson 16:33. https://doi.org/10.1186/1532-429X-16-33
Bots ML, Hofman A, Grobbee DE (1997) Increased Common Carotid Intima-Media Thickness. Stroke 28:2442–2447. https://doi.org/10.1161/01.STR.28.12.2442
Corrales A, González-Juanatey C, Peiró ME et al (2014) Carotid ultrasound is useful for the cardiovascular risk stratification of patients with rheumatoid arthritis: results of a population-based study. Ann Rheum Dis 73:722–727. https://doi.org/10.1136/annrheumdis-2012-203101
Figueroa AL, Abdelbaky A, Truong QA et al (2013) Measurement of Arterial Activity on Routine FDG PET/CT Images Improves Prediction of Risk of Future CV Events. JACC Cardiovasc Imaging 6:1250–1259. https://doi.org/10.1016/j.jcmg.2013.08.006
Rose S, Sheth NH, Baker JF et al (2013) A comparison of vascular inflammation in psoriasis, rheumatoid arthritis, and healthy subjects by FDG-PET/CT: a pilot study. Am J Cardiovasc Dis 3:273–278
Geraldino-Pardilla L, Zartoshti A, Bag Ozbek A et al (2018) Arterial Inflammation Detected With 18 F-Fluorodeoxyglucose–Positron Emission Tomography in Rheumatoid Arthritis. Arthritis & Rheumatol 70:30–39. https://doi.org/10.1002/art.40345
Dowsett L, Higgins E, Alanazi S et al (2020) ADMA: A Key Player in the Relationship between Vascular Dysfunction and Inflammation in Atherosclerosis. J Clin Med 9:3026. https://doi.org/10.3390/jcm9093026
Blann A (2003) The adhesion molecule P-selectin and cardiovascular disease. Eur Heart J 24:2166–2179. https://doi.org/10.1016/j.ehj.2003.08.021
Littler AJ, Buckley CD, Wordsworth P et al (1997) A distinct profile of six soluble adhesion molecules (ICAM-1, ICAM-3, VCAM-1, E-selectin, L-selectin and P-selectin) in rheumatoid arthritis. Rheumatology 36:164–169. https://doi.org/10.1093/rheumatology/36.2.164
Carter RA, Wicks IP (2001) Vascular cell adhesion molecule 1 (CD106): A multifaceted regulator of joint inflammation. Arthritis Rheum 44:985–994. https://doi.org/10.1002/1529-0131(200105)44:5<985::AID-ANR176>3.0.CO;2-P
Park M, Kulkarni A, Beatty A et al (2015) Soluble endothelial cell selective adhesion molecule and cardiovascular outcomes in patients with stable coronary disease: A report from the Heart and Soul Study. Atherosclerosis 243:546–552. https://doi.org/10.1016/j.atherosclerosis.2015.10.092
Yuri Gasparyan A, Ayvazyan L, Cocco G, Kitas DG (2012) Adverse Cardiovascular Effects of Antirheumatic Drugs: Implications for Clinical Practice and Research. Curr Pharm Des 18:1543–1555. https://doi.org/10.2174/138161212799504759
Disclosure
The authors confirm that they have no conflicts of interest related to this research.
Author information
Authors and Affiliations
Contributions
All authors of this manuscript have made significant contributions to the conception and design of the study, the acquisition and analysis of data, and the interpretation of results. They have all participated in the drafting and critical revision of the manuscript, and have given their final approval for submission to this journal. Additionally, all authors agree to be responsible for all aspects of the work and are willing to be held accountable for any issues that may arise. Finally, all contributing authors have reviewed and approved the final version of the manuscript.
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Part of the Topical Collection entitled ‘Cardiovascular Issues in Rheumatic Diseases’
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gerganov, G., Georgiev, T., Dimova, M. et al. Vascular effects of biologic and targeted synthetic antirheumatic drugs approved for rheumatoid arthritis: a systematic review. Clin Rheumatol 42, 2651–2676 (2023). https://doi.org/10.1007/s10067-023-06587-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-023-06587-8