Abstract
It is of general belief that males are prone to more frequent, more severe manifestations, and less favorable outcome. We evaluated this hypothesis in ophthalmological manifestations (OM) of Behcet's Disease (BD). Visual acuity (VA), anterior uveitis, posterior uveitis (PU), and retinal vasculitis (RV) were checked, according to Ben Ezra, in 1,515 patients with eye lesions. The data at baseline and last visit were compared. Male/female ratio was 1.2 in the BD registry (6,500 patients) and 1.51 for OM patients (Chi2 = 98.962, p < 0.0001). The patients-year-follow-up was 4,987. All parameters improved significantly from the baseline. Mean VA improved from 4.87 to 5.35 for males (p < 0.0001) and from 5.20 to 5.74 for females (p < 0.0001). Difference between males/females at baseline was not significant (p = 0.60). The mean improvement for males/females was statistically non-significant (p = 0.58). Percent improvement of eyes for males/females was 47.1%/48.8% (p = 0.41). Mean PU improved from 1.83 to 0.71 for males (p < 0.0001) and from 1.66 to 0.49 for females (p < 0.0001). Difference between males/females at baseline was significant (p = 0.01). The mean improvement for males/females was statistically non-significant (p = 0.45). Percent improvement of eyes for males/females was 75.4%/81.0% (p = 0.004). Mean RV improved from 2.05 to 1.16 for males (p < 0.0001) and from 1.97 to 0.99 for females (p < 0.0001). Difference between males/females at baseline was not significant (p = 0.42). The mean improvement for males/females was statistically non-significant (p = 0.47). Percent improvement of eyes for males/females was 62%/64.4% (p = 0.33). Male gender is more prone to ocular manifestations. The severity of lesions at baseline was the same for VA and RV. For PU, the difference was statistically significant, but was not clinically relevant. The therapeutic outcome (mean improvement and percent of improved eyes) was the same for all parameters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is of general belief that in Behcet's Disease (BD) male gender favors more major organ involvement, more severe manifestations, and less favorable outcome than the female gender [1–16]. Our previous studies, on a large cohort of patients, in 2002, have shown a gender difference for the occurrence of ophthalmological manifestations (OM), but not the outcome [9]. The aim of this study was to compare the incidence of OM, the severity of OM, and their therapeutic outcome in male and female BD patients.
Materials and methods
Patients
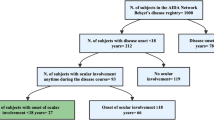
The whole registry of BD patients, from 1975 up to March 2010 was searched for all patients with ophthalmological manifestations. The incidence of eye lesions was calculated in each gender. Then, from the “Treatment Registry of Ophthalmological Manifestation” for BD, All patients were selected for the study. The data of eye lesions were extracted (variety and severity of lesions in, males and females separately, and the therapeutic outcome at the last control). All patients in the “Treatment Registry” had at least one eye needing cytotoxic drugs (active posterior uveitis (PU) and/or retinal vasculitis (RV)) at the baseline. Only eyes that were involved at baseline or during follow-up entered the calculations. Eyes that remained normal during the study were excluded from calculations. All patients except two (99.9%), fulfilled the new International criteria for Behcet's Disease [17, 18]. The ophthalmologic examination, before each follow-up visit, was performed by an ophthalmologist.
Treatment
The first line treatment for ophthalmological manifestations is cytotoxic (cyclophosphamide, azathioprine, methotrexate, chlorambucil) and immunomodulator drugs (cyclosporine), single or in combination, associated with prednisolone (19). For anterior uveitis (AU) alone, usually topical drugs are given by the ophthalmologist. Rarely do they need a short course of prednisolone. In case of posterior uveitis, without evidence of retinal vasculitis (on ophthalmoscopic examination), the patient is treated with methotrexate, azathioprine, chlorambucil, or cyclosporine, in single therapy. Prednisolone as 0.5 mg/kg of body weight is given at the beginning. It is then tapered according to the disappearance of frank improvement of the inflammation. In case of retinal vasculitis, combination of pulse cyclophosphamide and another cytotoxic drug (usually azathioprine, methotrexate, or cyclosporine), is indicated. The first choice is azathioprine [19, 20]. Non-responders to the treatment are switched to another cytotoxic drug or to a combination of cytotoxic drugs and/or immunomodulator.
Follow-up
At baseline and at each ophthalmological visit, the visual acuity (VA), the states of AU, PU, and RV were checked. If a patient was refractory to a cytotoxic drug (always combined with prednisolone), the patients was switched to another cytotoxic drug, or the combination of two cytotoxic drugs. Non-responder or refractory patients are those in whom the Disease Activity Index does not fall dramatically, or remains the same or even increases. Patients are followed up regularly, but the interval depends on the aggressiveness of the lesions and the therapeutic scheme. At the beginning of the treatment, the interval is from 1 to 3 months. Later, it may increase up to 6 months for patients in whom cytotoxic drugs were stopped.
Disease activity indexes
A questionnaire of 106 items (Table 1) was filled by the ophthalmologist. The questionnaire is a list of symptoms and signs that can be detected by ophthalmologic examination in eye lesions of BD. Each item is graded from 0 (normal) to 4+ (maximum impairment). Forty (40) items show the inflammation of different segments of the eye. They are used to calculate the inflammatory index of each of the segments. The remaining of items demonstrates sequels and side effects. They are used as chronicity index. On the questionnaire, items printed in italic represent inflammatory indices, and items in normal font chronicity indices. VA was calculated on a Snellen chart, on a scale of 10 on 10. Disease activity indexes (DAI) was calculated for AU, PU, and RV according to Ben Ezra [21]. Two other activity indexes, the total inflammatory activity index (TIAI) and the total adjusted disease activity index (TADAI) were also calculated [22]. The TIAI calculates the total inflammatory index of both eyes of the patient, showing the overall inflammation. As AU, PU, and RV do not have the same prognosis and not the same impact on the outcome of the eye, a coefficient of aggressiveness was given to each of them. For AU it was 1, for PU it was 2, and for RV it was 3. The TIAI formula is: Right Eye [(AU × 1) + (PU × 2) + (RV × 3)] + Left Eye [(AU × 1) + (PU × 2) + (RV × 3)]. The TADAI adds the calculation of VA, which is both an inflammatory index and a chronicity index, to TIAI. TADAI shows the overall ophthalmological state of the patient, inflammatory and chronicity indexes together. The formula for the calculation of TADAI is: TIAI + Right Eye [(VA − 10) × 2] + left Eye [(VA − 10) × 2]. The chronicity index (CI) was also calculated from the questionnaire of ophthalmology examination by adding together their points (Table 1).
Statistical calculations
The data from baseline and at the last visit, regardless of the treatment they received, were used for the analysis. Student paired t test was used to compare the outcome of VA, PU, RV, TIAI, and TADAI in each gender. Analysis of variance (one way ANOVA) was used to compare the mean inflammatory indexes and the chronicity index between males and females at baseline and the difference between their therapeutic responses. Chi square test (Chi2) was used to compare the number of eyes (for VA, PU, and RV), or the number of patients (for TIAI, TADAI), improved during the treatment, regardless of the amount of their improvement. SPSS software, version 16, was used for statistical analysis.
Results
The latest report of BD from Iran was on 6,500 patients [23]. Among them, 54.9% were males and 45.1% were females. The male to female ratio was 1.2 to 1. Ophthalmologic manifestations were seen more frequently in men (62.4%) than women (50.3%). Male to female ratio was 1.51 to 1. The difference was highly significant (Chi2 = 98.962, p < 0.0001). The Odds ratio of men with BD, versus women, to get ocular lesions is 1.64 (95% CI: 1.40–1.81).
Demographic data
One thousand five hundred and fifteen (1,515) patients (922 males and 593 females) were treated and followed for ocular manifestations, which was up to 20 years for some of them. The patients-year-follow-up was 4.987. The mean follow-up per patient was 3.29 years. In this group, the male/female ratio was 1.56, which was not different from the whole group of patients with a male/female ratio of 1.51.
The mean age of patients was 26.5 years, the standard deviation was 10.2, and the standard error of mean (SEM) was 0.5. For males, the mean age was 26.1 ± 9.6 years (SEM = 0.6). For females, it was 27 ± 10.9 years (SEM = 0.8). The difference between males and females was not significant (independent Student t test was 1.687, p = 0.09). The mean delay in the diagnosis of BD (from the onset of the first symptom) was 5.9 ± 6.0 years. For males it was 5.7 ± 5.7 years and for females 6.2 ± 6.3 years. The difference between males and females was not significant (independent Student t test was 1.597, p = 0.11). The mean duration of the disease was 12.6 ± 9.4 years. For males it was 12.4 ± 9.1 and for females 12.8 ± 9.9. The difference between males and females was not significant (independent Student t test was 0.806, p = 0.42). The mean follow-up was 6.7 ± 7.5 years. For males it was 6.8 ± 7.4 years and for females 6.6 ± 7.6 years. The difference between males and females was not significant (independent Student t test was 0.508, p = 0.61).
Treatment modalities
The treatment used for these patients was oral cyclophosphamide for 41 patients, pulse cyclophosphamide for 536 patients, methotrexate for 638 patients, azathioprine for 161 patients, chlorambucile for 89 patients, cyclosporine for 36 patients, combination of pulse cyclophosphamide and methotrexate for 121 patients, and combination of pulse cyclophosphamide and azathioprine for 241 patients. The total was 1,836 treatment modalities. Some of the patients, as said before, used two or three treatment modalities, but results were calculated for the patient.
Visual acuity
All patients (1,515 patients) had impaired vision, but in 491 patients, only one eye had abnormal vision. Therefore, from 3,030 eyes, 1,521 in males and 1,018 in females, 2,539 eyes, had impaired vision. Calculations were done in this set of eyes. The mean VA at baseline was 4.87 (SD = 4.04) for males and 5.20 (SD = 3.83) for females (Table 2). The difference was not statistically significant; by ANOVA, F was 3.538, and p was 0.60 (Table 3). Male and female had the same amount of visual impairment before the treatment. The mean VA improved to 5.35 for males and to 5.74 for females. The mean improvement was 0.48 for males (t = 6.131, p < 0.0001) and 0.55 for females (t = 5.605, p < 0.0001). The difference between males and females improvement was not statistically significant (F = 0.313, p = 0.58).
For males, from 1,521 eyes on calculation, 716 improved (47.1%, 95% CI: 44.5–49.7%). For females, from 1,018 eyes on calculation, 497 improved (48.8%, 95% CI: 45.7–51.9%; Table 4). By Chi Square test, the difference was not significant (Chi2 = 0.680, p = 0.41). In males, the improvement were as follow: 16.6% of eyes got normal vision, 3.4% were blind and got some vision, 0.4% were blind and got useful vision, 10.6% did not have useful vision, but got a useful vision, and the remaining had useful vision and improved it (16%). In females, the improvement were as follow: 17.4% of eyes got normal vision, 2.8% were blind and got some vision, 0.8% were blind and got useful vision, 12% did not have useful vision, but got a useful vision, and the remaining (15.8%) had useful vision and improved it.
In males, 259 eyes remained unchanged (17%). Among them, 9% were already blind at the baseline. In females, 158 eyes remained unchanged (15.5%). Among them, 6.2% were blind at the baseline.
In males, 546 eyes were aggravated (35.9%). Among them, 8.6% became blind, 5.6% lost their useful vision, and 21.7% decreased their vision. In females, 362 eyes were aggravated (35.6%). Among them, 6.2% became blind, 5.7% lost their useful vision, and 23.7% decreased their vision. There was no statistically significant difference between males and females on the percentage of eyes improved, unchanged, or aggravated, and their subgroups.
In males, from 1,844 eyes, 449 were normal at the baseline; during the progression of the disease, 126 became affected (6.8% of all eyes). In females, from 1,186 eyes, 264 were normal at the baseline; during the progression of the disease, 96 became affected (8.1% of all eyes). The difference between males and females was not significant (Chi2 = 1.692, p = 0.19).
Anterior uveitis
The mean AU at baseline was 2.54 (SD = 2.33) for males and 2.52 (SD = 2.19) for females (Table 2). The difference was not statistically significant (F was 0.00017, and p was 0.99; Table 3). The mean AU improved to 0.70 for males and to 0.58 for females. The mean improvement was 1.84 for males (t = 19.096, p < 0.0001) and 1.93 for females (t = 17.612, p < 0.0001). The difference between males and females improvement was not statistically significant (F = 0.405, p = 0.52). For males, 77.6% of eyes improved, versus 81.1% in females (Table 4). The difference was not significant (Chi2 = 1.095, p = 0.30). In males, 4.5% of eyes kept their baseline data and 17.9% aggravated their inflammatory index. In females, 5% of the eyes kept their baseline data and 13.9% were aggravated.
Posterior uveitis
PU was calculated in 2,332 involved eyes (1,420 in males and 912 in females). The mean PU at baseline was 1.83 (SD = 1.47) for males and 1.66 (SD = 1.38) for females (Table 2). The difference was statistically significant (F was 6.606, and p was 0.01; Table 3). Males had more inflammation in the vitreous than females. The mean PU improved to 0.71 for males and to 0.49 for females. The mean improvement was 1.12 for males (t = 25.898, p < 0.0001) and 1.17 for females (t = 24.235, p < 0.0001). The difference between males and females improvement was not statistically significant (F = 0.576, p = 0.45). For males, 75.4% of eyes improved, versus 81% in females (Table 4). The difference was significant (Chi2 = 8.313, p = 0.004), however, the difference is not clinically relevant. In males, 10.3% of eyes kept their baseline data and 14.2% aggravated their inflammatory index. In females, 9.2% of the eyes remained unchanged and 9.8% were aggravated.
Retinal vasculitis
RV was calculated in 1,916 involved eyes (1,178 in males and 738 in females). The mean RV at baseline was 2.05 (SD = 2.30) for males and 1.97 (SD = 2.41) for females (Table 2). The difference was not statistically significant (F = 0.653, p = 0.42; Table 3). The mean RV improved to 1.16 for males and to 0.99 for females. The mean improvement was 0.89 for males (t = 11.453, p < 0.0001) and 0.98 for females (t = 10.178, p < 0.0001). The difference between males and females improvement was not statistically significant (F = 0.532, p = 0.47). For males, 62% of eyes improved, versus 64.4% in females. The difference was not significant (Chi2 = 0.936, p = 0.33). In males, 10.5% of eyes remained unchanged and 27.4% aggravated their baseline data. While, in females, 10.5% of eyes kept their baseline data and 25.1% deteriorated them.
Total inflammatory activity index (TIAI)
The mean TIAI (Table 2) at baseline was 18.27 (SD = 13.58) for males and 16.90 for females (SD = 14.18). The difference was not significant (F = 2.950, p = 0.09; Table 3). TIAI improved to 8.47 (SD = 11.27) for males (t = 18.583, p < 0.0001) and 6.83 for females (t = 16.051, p < 0.0001). The difference between male and female improvement was not significant (F = 0.114, p = 0.74). In males, 77.1% improved, and in females 80.6% improved. The difference was not significant (Chi2 = 2.533, p = 0.11). In males, 4.6% of eyes kept their baseline data and 18.3% aggravated their inflammatory index. In females, 4.7% of the eyes remained unchanged and 14.7% were aggravated.
Total adjusted disease activity index (TADAI)
The mean TADAI at baseline was 37.79 (SD = 20.91) for males and 35.15 (SD = 21.11) for females (Table 2). The difference was significant (F = 4.180, p = 0.04; Table 3). TADAI improved to 26.51 (SD = 20.65) for males (t = 17.592, p < 0.0001) and 23.16 (SD = 19.19) for females (t = 15.061, p < 0.0001). The difference between male and female improvement was not significant (F = 0.485, p = 0.49). In males, 72.0% improved, and in females 73.6% improved. The difference was not significant (Chi2 = 0.483, p = 0.49). In males, 3.8% of eyes remained unchanged and 27.4% aggravated their baseline data. While, in females, 4.2% of eyes kept their baseline data and 22.1% deteriorated them.
Chronicity index
In males, 16.7% of eyes had a CI at baseline. In females, CI was 18.3%. Females had more chronicity index than males, but the difference was not significant (Chi2 was 0.588, p = 0.44). At the last evaluation, 13.3% of eyes, in males, were added to eyes with CI. For females, it was 13.8%. The difference was not significant (Chi2 = 0.040, p = 0.84).
Discussion
The male prevalence in BD is reported from the majority of countries, all around the world, except from Japan, Korea, Israel, Spain, UK, Sweden, Brazil, and the US [24]. It is of general belief that male gender is more prone to ocular [1, 5, 6, 9–11, 16] neurological [6, 14], and vascular manifestations [6, 8, 13, 15]. They have a more aggressive disease, and they are more resistant to treatment [2–4, 6, 12]. The new data analysis of Behcet's Disease in Iran, in 6,500 cases, confirmed the higher incidence of ocular manifestations in male gender (odds ratio 1.64). Ocular manifestations were reported to occur more in young patients, younger than 25 years old, than older patients [1]. Our data shows that ocular manifestations were seen more in patients of 25 years and older (30.4%, 95% CI: 29.3–31.5%) than in patients younger than 25 years (26.4%, 95% CI: 25.3–27.5%). For males it was 18.1% (95% CI: 17.2–19%) versus 16.1% (95% CI: 15.2–17%). For females it was 12.3% (95% CI: 11.5–13.1%) versus 10.4% (95% CI: 9.7–11.1%).
The analysis of baseline data and their comparison in males and females did not show major difference. Bangs and colleagues [10], like us, did not find any difference between males and females mean age (33.9 ± 9.9 years for males and 33.2 ± 10.4 years for females). For Madanat and colleagues [7] the mean age at onset were 24.15 ± 8.73 years for males and 26.5% ± 1.83 years in females (difference not significant).
For Gurler and colleagues [6], on 2,313 BD patients, the male to female ratio was 2 to 1 (38.1% males, 19.8% females), while in our study, it was 1.56 to 1 (62.4% in men, 50.3% in women). For Madanat and colleagues [7] it was 1.65 to 1. However, for Koh and colleagues [25] it was 1 to 1.15 (females more than males).
Visual acuity was the same, at the baseline, for both genders (4.87 for males, 5.20 for females. P = 0.60). It was the same for anterior uveitis, for retinal vasculitis, and for TIAI (Table 3). However, males had more severe posterior uveitis at the baseline, while the difference, although statistically significant (p = 0.01), was not clinically relevant (males' mean activity index 1.83 and females' 1.66). The same was for TADAI (p = 0.04, males' mean activity index 37.8, females' mean activity index 35.2). For Madanat and colleagues [7] there was no difference between males and females for only anterior uveitis. The difference for posterior uveitis and retinal vasculitis was highly significant (p < 0.001 and p = 0.025).
The response to the treatment was the same in both genders. The mean improvement in VA, PU, RV, TIAI, and TADAI (Table 3), as the percent of improved eyes for VA, RV, TIAI, and TADAI (Table 4) was the same for males and females. There was no difference in chronicity index between males and females.
The weak point and the limitation of the study is the patients' selection, which was not a prospective study. The “Treatment Registry of Ophthalmological Manifestation” of Behcet's patients contain only patients who have active posterior uveitis or retinal vasculitis. Patients with inactive lesions or patients with anterior uveitis alone are not included in the registry. Moreover, patients who did not follow their disease in the BD clinic are not included in the registry. This also explains why the number of reported patients in this study is less than what is expected from the incidence of eye lesions (56.8%) in the last report from Iran [23]. In the same report posterior uveitis and/or retinal vasculitis was only seen in 48.4% of the eyes. The strong point of the study is the large number of patients, the inclusion of all patients of the registry, and the analysis of data by intention to treat, including the data of all patients, even those who stopped their follow-up in the middle of the treatment.
Conclusion
No major differences were seen between males and females in ocular manifestations, except for more inflammatory index of uvea in males than females. They had the same mean age, the same delay in their diagnosis, the same follow-up time, and the same duration of the disease. Males and females were treated the same way, and the treatment outcome was similar, even for major morbidities like blindness.
References
Yazici H, Tuzun Y, Pazarli H et al (1984) Influence of age of onset and patient's sex on the prevalence and severity of manifestations of Behcet's syndrome. Ann Rheum Dis 43:783–789
Yazici H, Tüzün Y, Tanman AB et al (1985) Male patients with Behçet's syndrome have stronger pathergy reactions. Clin Exp Rheumatol 3:137–141
Dilsen N, Konice M, Aral O, Ocal L, Gul A (1993) Risk factors for vital organ involvement in Behcet's Disease. In: Godeau P, Wechsler B (eds) Behcet's Disease. Elsevier Science Publishers B.V, Amsterdam, pp 165–169
Yazici H, Basaran G, Hamuryudan V et al (1996) The ten year mortality in Behcet's syndrome. Br J Rheumatol 35:139–141
Muhaya M, Lightman S, Ikeda E et al (2000) Behçet's disease in Japan and in Great Britain: a comparative study. Ocul Immunol Inflamm 8:141–148
Gurler A, Tursen U, Boyvat A (2000) The evaluation of the clinical findings according to age and sex in 2313 Behcet's patients followed up at the multi-disciplinary Behcet's disease Center in Ankara University School of Medicine. In: Bang D, Lee E, Lee S (eds) Behcet's Disease. Design Mecca Publishing, Seoul, pp 84–88
Madanat W, Fayyad F, Verity D, Zureikat H (2000) Influence of sex on behcet's Disease in Jordan. In: Bang D, Lee E, Lee S (eds) behcet's Disease. Design Mecca Publishing, Seoul, pp 90–93
Ames PR, Steuer A, Pap A, Denman AM (2001) Thrombosis in Behçet's disease: a retrospective survey from a single UK centre. Rheumatology (Oxford) 40:652–655
Davatchi F, Shahram F, Chams H (2003) The influence of gender on the severity and the outcome of ocular lesions in Behçet's disease. Adv Exp Med Biol 528:67–71
Bang D, Oh S, Lee KH, Lee ES, Lee SN (2003) Influence of sex on patients with Behçet's disease in Korea. J Korean Med Sci 18:231–235
Janati K, EL Omari K, Benchiki H, Hamdani M, Lakhdar H (2005) Ocular manifestation of Behçet disease (study of 50 patients consulting in dermatology department). Rev Méd Interne 26:771–776
Ricart JM, Todolí J, Vilata JJ et al (2006) Behçet disease: study of 74 patients. Med Clin (Barc) 127:496–499
Düzgün N, Ateş A, Aydintuğ OT, Demir O, Olmez U (2006) Characteristics of vascular involvement in Behçet's disease. Scand J Rheumatol 35:65–68
Benamour S, Naji T, Alaoui FZ, El-Kabli H, El-Aidouni S (2006) Neurological involvement in Behçet's disease. 154 cases from a cohort of 925 patients and review of the literature. Neurol (Paris) 162:1084–1090
Kwon TW, Park SJ, Kim HK, Yoon HK, Kim GE, Yu B (2008) Surgical treatment result of abdominal aortic aneurysm in Behçet's disease. Eur J Vasc Endovasc Surg 35:173–180
Yang P, Fang W, Meng Q, Ren Y, Xing L, Kijlstra A (2008) Clinical features of chinese patients with Behçet's disease. Ophthalmology 115:312–318
Davatchi F, Sadeghi Abdollahi B, Shahram F et al (2010) Validation of the International Criteria for Behcet's Disease (ICBD) in Iran. Int J Rheum Dis 13:55–60
International Team for the Revision of the International Criteria for Behcet's Disease (2006) Revision of the International Criteria for Behcet's Disease (ICBD). Clin Exp Rheumatol 24(Suppl 42):S14–S15
Davatchi F, Shams H, Shahram F et al (2005) Management of ocular manifestations of Behcet's Disease: outcome with cytotoxic drugs. APLAR J Rheumatol 8:119–123
Davatchi F, Shahram F, Chams-Davatchi C et al (2010) How to deal with Behcet's Disease in daily practice. Int J Rheum Dis 13:105–116
Ben Ezra D, Forrester JV, Nussenblatt RB, Tabbara K, Timonen P (eds). Uveitis scoring system. Sandoz Ltd and Springer Verlag, Berlin Heidelberg.
Davatchi F, Shams H, Shahram F et al (2009) Longitudinal study of chlorambucil in ocular manifestations of Behcet's Disease. Iranian J Ophthalmol 21:3–14
Davatchi F, Shahram F, Chams-Davatchi C et al (2010) Behcet's Disease in Iran: analysis of 6500 cases. Int J Rheum Dis. doi:10.1111_j.1756-185X.2010.01549
Davatchi F, Shahram F, Chams-Davatchi C et al (2010) Behcet's Disease from East to West. Clin Rheumatol 29:823–833
Koh (2000) Ophthalmologic manifestation of Behcet's Disease. In: Bang D, Lee E, Lee S (eds) Behcet's Disease. Design Mecca Publishing, Seoul, pp 90–93
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Davatchi, F., Shahram, F., Shams, H. et al. Gender influence on ocular manifestations and their outcome in Behcet’s Disease. A long-term follow-up of up to 20 years. Clin Rheumatol 30, 541–547 (2011). https://doi.org/10.1007/s10067-010-1574-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-010-1574-4