Abstract
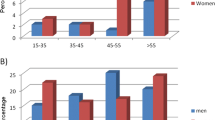
In 200 patients, 143 women and 57 men, with rheumatoid arthritis (RA), age at onset was related to the presence of IgM rheumatoid factor (RF) and antinuclear antibodies (ANA) in serum at presentation. The patients were stratified into bands of age at onset: <40 years, 40– 60 years, and >60 years. In women, the prevalences of ANA (1:160) were 63.0%, 45.5%, and 31.1% in the respective age bands (p=0.002), and the prevalences of IgM RF were 85.2%, 72.7% and 66.4%, respectively (p=0.03). The prevalences of ANA and IgM RF among men were, respectively, 35% and 86% with no association with age at onset. The findings may indicate interactive effects between gender and various pathogenetic factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The presence of autoreactive T-lymphocytes and antibodies classifies rheumatoid arthritis (RA) as an autoimmune disease. Well-known autoantigens, although not specific for RA, are class G immunoglobulins (IgG) and nuclear components resulting in the production of IgM rheumatoid factor (RF) and antinuclear antibodies (ANA), respectively. Inflammatory activity in chronic arthritis may partially depend on an exogenous antigen drive [1], and decreased function of host defense molecules has been shown to increase the risk of immune-mediated complications in patients with phagocyte dysfunction [2]. Recent reports suggest that decreased activity of the innate function of the immune apparatus may increase the susceptibility to RA with younger onset and an erosive course, as illustrated by polymorphisms of the gene encoding for mannose-binding lectin (MBL) [3, 4]. The hypothesis behind the present paper is that mechanisms that result in increased susceptibility to RA and decreased age of onset may also result in an increased prevalence of autoantibodies, which should lead to an association between the two latter in a cohort of RA patients.
Patients and methods
Current outpatients who met the 1987 ACR criteria for RA [5] were identified by means of the patient register of the department. Autoantibodies that had been measured consistently at onset in these patients were ANA and IgM RF. Data on gender, age at onset of physician-observed polyarthritis, and the presence of IgM RF and ANA at onset were retrieved in 200 patients with RA. Determination of IgM RF in serum had been performed by means of nephelometry using the 95th percentile of an unpublished normal material as cut-off value. The presence of IgG ANA in serum diluted 1:160 had been determined by means of an indirect immunofluorescence technique using commercially prepared slides of monolayer HEp-2 cells (ImmunoConcepts, Sacramento, California) as substrate and FITC-labeled rabbit immunoglobulins against human Fcγ (DAKO, Denmark) [6]. The assay methods were stable over the recruitment period. Frequency trends in subsets of the study population were analyzed by means of the χ2test for trends.
Results
The 200 patients included in the study consisted of 143 women and 57 men. The median age and range of age at onset of RA in women and men was 46 years (7–74 years) and 51 years (18–74 years), respectively. The patients were stratified according to age at onset in the following groups: <40 years (n=66), 40– 60 years (n=65), and >60 years (n=69). Table 1 shows the prevalences of the autoantibodies studied for each age band in men and women. The prevalences of ANA, IgM RF and combinations thereof increased with decreasing age at onset in the female patients, but not in the male patients.
Discussion
This study supports the hypothesis that patients with a younger onset of RA have a higher prevalence of certain circulating autoantibodies. Similar findings have been made in patients with Sjögrens syndrome with regard to the presence of anti-SSA/SSB, RF, and the combination of ANA, anti-SSA/SSB and RF [7]. In patients with systemic lupus erythematosus (SLE), the prevalence of anti-DNA was found to increase with younger age of disease onset [8]. Various explanations may be offered for these findings. Serological abnormalities in patients with unspecific signs of rheumatological disease may lead to referral at an earlier point in time, leading to referral bias. Furthermore, the relatively high proportion of RF-positive patients at onset may indicate that using prevalent cases instead of incident cases may have led to a selection bias. The magnitude and relative importance of such biases is, however, difficult to ascertain.
Defects of the innate immune system may both lower host defense mechanisms and cause defective clearing of apoptotic cell material and immune complexes, as seen in MBL and C1q deficiency [9], which may lead to increased levels of various autoantibodies. This may account for the observed association between early onset of disease and increased prevalence of autoantibodies in this study, but none of the statements above explain why this association was only carried by the female RA patients. Female sex increases susceptibility to RA and SLE, particularly in younger persons [10, 11]. Although not directly supported by the data, the findings in this study may indicate an interaction between gender and various factors having an influence on susceptibility and disease modification in RA.
Abbreviations
- ANA:
-
Antinuclear antibodies
- MBL:
-
Mannose-binding lectin
- RA:
-
Rheumatoid arthritis
- RF:
-
Rheumatoid factor
References
Winchester R, Gregersen PK, Merryman P, Dwyer E, Lee S (1988) Hypothesis on the molecular basis of susceptibility to rheumatoid arthritis. Scand J Rheumatol Supplement 76:9–16
Foster CB, Lehrnbecher T, Mol F et al. (1998) Host defense molecule polymorphisms influence the risk for immune-mediated complications in chronic granulomatous disease. J Clin Invest 102:2146–2155
Garred P, Madsen HO, Marquart H et al. (2000) Two edged role of mannose binding lectin in rheumatoid arthritis: a cross sectional study. J Rheumatol 27:26–34
Graudal NA, Madsen HO, Tarp U et al. (2000) The association of variant mannose-binding lectin genotypes with radiographic outcome in rheumatoid arthritis. Arthritis Rheum 43:515–521
Arnett FC, Edworthy SM, Bloch DA et al. (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324
Humbel RL (1993). Detection of antinuclear antibodies by immunofluorescence. In: van Venroij WJ, Maini RN (eds) Manual of biological markers of disease – A2. Kluwer Academic Publishers, Dordrecht, pp 1–16
Haga H-J, Jonsson B (1999) The influence of age on disease manifestations and serological characteristics in primary Sjögrens syndrome. Scand J Rheumatol 28:227–232
Jacobsen S, Petersen J, Ullman S et al. (1998) A multicenter study of systemic lupus erythematosus in 513 Danish patients. I: Disease manifestations and analyses of clinical subsets. Clin Rheumatol 17:468–477
Morgan BP, Walport MJ (1991) Complement deficiency and disease. Immunol Today 12:301–306
Symmons DPM, Barrett EM, Bankhead CR, Scott DGI, Silman AJ (1994) The incidence of rheumatoid arthritis in the United Kingdom: Results from the Norfolk Arthritis Register. Br J Rheumatol 33:735–739
McCarty DJ, Manzi S, Medsger TA, Ramsey-Goldman R, LaPorte RE, Kwoh CK (1995) Incidence of systemic lupus erythematosus. Arthritis Rheum 38:1260–1270
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jacobsen, S. Young age of onset is associated with increased prevalence of circulating IgM rheumatoid factor and antinuclear antibodies at presentation in women with rheumatoid arthritis. Clin Rheumatol 23, 121–122 (2004). https://doi.org/10.1007/s10067-003-0844-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-003-0844-9