Abstract
Mean trophic level (MTL) is one of the most widely used indicators of marine ecosystem health. It usually represents the relative abundance of fished species across a spectrum of TLs. The reality, ubiquity, and causes of a general decline in the MTL of fisheries catch through time, and whether fisheries catch tracks ecosystem level changes, have engendered much attention. However, the consequences of such patterns for broader ecosystem structure and function remain virtually unexplored. Along the Pacific U.S. Coast, previous work has documented fluctuations and a slow increase in ecosystem MTL from 1977 to 2004. Here, we document a decline in the ecosystem MTL of groundfishes in the same ecosystem from 2003 to 2011, the proximate cause of which was a decrease in the biomass of higher TL groundfishes. Using a food web model, we illustrate how these shifts in ecosystem structure may have resulted in short term, positive responses by many lower TL species in the broader ecosystem. In the longer term, the model predicts that initial patterns of prey release may be tempered in part by lagged responses of other higher TL species, such as salmon and seabirds. Although ecosystem functions related to specific groups like piscivores (excluding high-TL groundfishes) changed, aggregate ecosystem functions altered little following the initial reorganization of biomass, probably due to functional redundancy within the predator guild. Efforts to manage and conserve marine ecosystems will benefit from a fuller consideration of the information content contained within, and implied by, fisheries-independent TL indicators.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Managing marine resources has always been challenging, but this task is becoming increasingly difficult as society demands more seafood while also requiring that we act as careful stewards of ocean health. Ecosystem-based management (EBM) is an important approach that emphasizes evaluating trade-offs between the exploitation and conservation of marine species and the ecosystems within which they reside (reviewed by Leslie and McLeod 2007). This emerging paradigm requires that scientists and managers track changes in different components of marine ecosystems and understand the consequences of those changes for ecosystem structure and function more broadly (Levin and others 2009).
Mean trophic level (MTL) is a pervasive and heavily discussed indicator. It provides an integrated view of the organization of trophic structure in marine ecosystems, generally with a specific focus on communities dominated by exploited species (Pauly and Watson 2005; Essington and others 2006; Branch and others 2010; Sethi and others 2010; Stergiou and Tsikliras 2011). The paradigm of “fishing down the food web” (Pauly and others 1998) introduced the idea that industrialized fishing has caused a decline in the biomass of higher TL taxa with the implicit assumption that the structural changes cascade throughout the community as loss of predators reduces top-down forcing. However, MTL estimated from fisheries catch data (catch MTL) may decline for other reasons including the behavior of fishers (for example, increased catch at lower TLs, also known as “fishing through the food web”, Essington and others 2006; Stergiou and Tsikliras 2011) and does not necessarily reflect trophic changes within the communities from which the catch data are derived (ecosystem MTL, Branch and others 2010). Trends in ecosystem MTL, which are better estimated from data sources like fisheries-independent surveys, can be more indicative of current ecosystem status. Nevertheless, ecosystem MTL may still decline due to either a loss of high-TL taxa or an increase in the abundance of low-TL species.
To date, research has focused on the patterns and processes related to variation in catch MTL (Essington and others 2006; Branch and others 2010; Stergiou and Tsikliras 2011), but it has not extended to asking how variation in ecosystem MTL of exploited communities may drive change in the broader ecosystem. For example, what is the significance of a decline in ecosystem MTL of groundfishes for the structure and function of the ecosystem more generally? The point here is not to ask whether the ecosystem MTL of groundfishes tracks other changes in the ecosystem, but to ask how reduced top-down pressure from groundfishes, measured as a decline in ecosystem MTL, might propagate through the rest of the ecosystem. These questions have not been evaluated directly, which is surprising given that predation is known to be a strong structuring force in many marine ecosystems (Heithaus and others 2008).
Indeed, predator removals may have far-reaching consequences as has been shown for other taxa (Daskalov 2002; Pauly and Watson 2005; Baum and Worm 2009; Estes and others 2011). In particular, reduced predator abundance can lead to trophic cascades where impacts propagate “vertically” downward through the food chain (Estes and others 2011), and one might expect that a decline in the abundance of predatory groundfishes would lead to an increase in their prey, at least temporarily. What is not clear is whether such a change would also manifest “horizontal” responses by groundfish competitors (competitive release) and how strong these effects would be in relation to top-down ones (prey release). Competitive release might be likely in systems like the California Current where many taxa, including high-TL fishes, feed on a common and taxonomically somewhat limited prey group (for example, krill and forage fish, Field and others 2006; Smith and others 2011a). That is, if high-TL groundfishes declined in abundance, would other taxa like marine mammals, seabirds and salmon exert increased top-down forcing and control prey populations, or would prey outstrip predators and show large increases in biomass? Large changes in both catch and ecosystem MTL for groundfishes have occurred in the California Current (Branch and others 2010). For example, ecosystem MTL estimated from the US West Coast Triennial Trawl Survey fluctuated substantially from 1977 to 2004, slowly increasing over this time period (Branch and others 2010) demonstrating that it is important to understand how these fluctuations reverberate through the ecosystem.
We use a fisheries-independent data set to (1) examine the trend in ecosystem MTL (here after “MTL”) of groundfishes in the California Current ecosystem during 2003–2011, (2) reveal the underlying processes producing the patterns (that is, changes in the biomass of high-TL vs. low-TL species), and (3) identify the specific species and taxa that contribute to any trends. We then use an ecosystem model to couple this empirical analysis with an exploration of the ecological consequences of the empirical trends in trophic structure of groundfishes for the broader California Current ecosystem focusing on (4) the relative importance of top-down and competitive effects.
Materials and Methods
Mean Trophic Level and Biomass
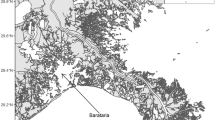
We used data from the West Coast Groundfish Bottom Trawl Survey conducted by the Northwest Fisheries Science Center, U.S. National Marine Fisheries Service (Keller and others 2008) to quantify the trend in ecosystem MTL. This fisheries-independent survey is a depth stratified, random sample that covers approximately the area from the U.S.–Mexico border (32°30′N) north to Cape Flattery, WA (48°10′N) and 50–1,200 m (Figure 1). We used data for all taxa identified to species (312 species and 5,743 trawls) and from the years 2003–2011, as surveys in these years sampled both the shelf and slope, and from 32° to 48°N. Further detail on the survey design and methods can be found in Keller and others (2008).
We calculated ecosystem MTL in two ways, using a simple average and an area-weighted average. In both cases, information on species TL was taken from FishBase.org (Froese and Pauly 2010). First, we took the simple, yearly average of MTL (T y ) as the biomass-weighted average of the TLs of the species caught in a given year y (Jiming 1982; Pauly and others 1998; Pauly and Watson 2005; Branch and others 2010):
where B s,y is the biomass of taxon s in year y and T s is the estimated TL of taxon s.
Although reasonable for estimating MTL of a sample, the above method does not necessarily account for variation in the spatial coverage of depth and latitude strata and variation in sampling effort among these strata for the fisheries-independent bottom trawl survey used here to calculate ecosystem MTL. Therefore, we also calculated an area-weighted ecosystem MTL by first converting data to catch per unit effort (CPUE, kg km−2) and then calculating ecosystem MTL for each haul (T h ) as
where C s,h is the CPUE of species s in haul h. We then binned data into three depth zones—roughly the continental shelf (<200 m), upper slope (201–600 m), and lower slope (>600 m)—based on previous analyses of patterns of assemblage structure and diversity (Horn and Allen 1978; Tolimieri and Levin 2006; Tolimieri 2007; Tolimieri and Anderson 2010). Data were also binned by four geographical regions with northern boundaries at Point Conception (34.4°N), Cape Mendocino (40.4°N), Cape Blanco (42.8°N), and Cape Flattery (here 48°N), which represent known breaks in biogeography (Horn and Allen 1978; Horn and others 2006), assemblage structure (Tolimieri and Levin 2006), and oceanography (GLOBEC 1994). We next calculated the annual MTL (T wt,y ) as the area-weighted mean:
where T h,y is the MTL of haul h in year y and A r is the area of region r expressed as a proportion of the total area (0–1; see below for calculation of the areas of each bin).
For subsequent analyses, species were divided into two trophic bins (T s < 3.5 and T s ≥ 3.5) to determine whether any changes in MTL were due to an increase in the abundance of low-TL fishes or a decrease in the abundance of high-TL fishes. We chose this cut-off because it represents approximately the mid-TL of species of the groundfish assemblage by frequency and by biomass (see “Results”). Setting the cut-off any lower (3.0 or 3.25) would place most fishes into the high-TL grouping. The 3.5 cut-off also places those fishes that are primarily piscivores into the high-TL group (Tremblay-Boyer and others 2011). We next calculated coast-wide biomass for each trophic group in each year to determine how trophic structure changed at a gross scale. We computed the mean CPUE (kg km−2) for each trophic bin, multiplied it by the area (see below) of that bin, and summed across all bins within a year to give total biomass for higher and lower TL species.
The areal extent of each depth × region bin was calculated from the U.S. Coastal Relief Model (http://www.ngdc.noaa.gov/mgg/coastal/crm.html). The native coordinate system of these bathymetry data does not conserve area throughout the study region (for example, a 1° × 1° area in the south is larger than a 1° × 1° area to the north). To correct this problem, we created a regular 0.1° grid over the study area and then re-projected this grid to a cylindrical equal-area projection (units = meters, projection type = 3, longitude of the center of projection = −122°0′0.00″, latitude of the center of projection = 56°30′0.00″, Azimuth = 120.95, and scale factor = 1). The new data layer had the correct area for each 0.1° latitude/longitude grid cell. The total area of a given depth × region bin was calculated by summing the area of the relevant grid cells.
We used linear regression to test for trends in MTL and biomass through time. Because the data were time series, we fit generalized least squares (GLS, using maximum likelihood) models with first-order autocorrelation to calculate the overall model and provide an hypothesis test (Zuur and others 2009). However, GLS models do not produce standard r 2 values, and pseudo-r 2 values are often difficult to interpret. To provide a measure of goodness of fit, we present the squared correlation of the observed versus predicted values from the GLS model. In some cases, we also fit polynomial regressions with year2 as an additional term when the relationship appeared nonlinear. We then used log-likelihood ratio tests and comparison of AIC values to choose the best-fit model.
Ecosystem Modeling
We used an ecosystem model to explore how changes in the biomass of high-TL groundfishes (TL ≥ 3.5) could influence the qualitative responses of other species, along with broader ecosystem structure (abundance of other taxa) and function (trophic representation and bioenergetics processes). This Ecopath with Ecosim (EwE) food web model (Christensen and Pauly 1992; Christensen and others 2005) was parameterized for the northern California Current at the turn of the twenty-first century (Field 2004; Field and others 2006). Our analysis focused on short-term, instantaneous food web responses estimated using Ecopath, and longer term, dynamic food web responses using Ecosim. The Ecopath approach provided insight into direct trophic responses of prey to a change in the biomass of their high-TL groundfish predators, and remained free of the additional parameters and assumptions required of the dynamic Ecosim model (Christensen and others 2005). The Ecosim approach complemented the Ecopath analysis by allowing us to gauge both direct and indirect effects of changes in the biomass of high-TL groundfishes over longer time scales. Although simulated changes in specific high-TL groundfishes species and functional groups followed observed trends, both the instantaneous and dynamic approaches were meant as heuristic exercises to examine if and how these changes would propagate through the ecosystem at large. We did not attempt to account for management decisions, changes in fishing practices, or climate processes—a much larger task and one that is beyond the scope of the current investigation.
Ecopath and ecosim have several important differences (Christensen and others 2005). We highlight the two most germane to our analysis here. First, as noted above, Ecopath allows only for first-order responses of high-TL groundfish prey, whereas Ecosim also allows for second-order responses by groups not linked through direct trophic interactions with high-TL groundfishes. Second, Ecopath assumes a simple linear functional response of predators to increasing prey biomass densities, whereas Ecosim functional responses can be linear or nonlinear, and depend on the time course of trophic interactions. These two differences in particular may lead to varying results between the two methodologies and highlight the value of evaluating both.
Field and others (2006) provide a full list of functional groups and detailed information about the EwE models. Briefly, the model was parameterized using data from stock assessments, survey indices, landings, and diet studies, and provided a reasonable fit to greater than 24 empirical time series spanning the period 1960–2004. Field and others (2006) showed that alternative parameters describing the vulnerability of prey to their predators did not provide a significant improvement in model fit to the empirical data compared with default parameter values. Consequently, we applied the default values for these parameters in our simulation.
We perturbed the EwE models by reducing or augmenting the biomass of the high-TL groundfish taxa in the EwE model and compared the results to an unchanged model. The EwE model includes 15 groundfish species and seven combined groundfish taxa (Table 1). To determine which species contributed to the decrease in MTL (see “Mean trophic level and biomass”), we calculated the area corrected biomass of each of these taxa in the groundfish trawl survey from 2003 to 2011 following the same methodology as that which we used to calculate the biomass of the combined low- and high-TL groundfishes groups above. We then fit linear and polynomial regressions (see above) to each time series to determine whether the taxa showed a significant increase or decrease in biomass from 2003 to 2011. For the high-TL taxa showing significant trends, we used the predicted percentage change in biomass from 2003 to 2011 to parameterize the EwE model by reducing or increasing the biomass of the high-TL groundfishes in the EwE model by the percentage change observed in the trawl survey data. We did not manipulate biomass for low-TL groundfishes. To evaluate the importance of modeling species-specific changes versus a general decline in the biomass of higher TL fishes, we also ran an EwE simulation in which we imposed an across-the-board decrease of 40% on all groundfish taxa of TL (T s ) 3.5 or greater (Appendix A in Electronic Supplementary Material).
Specifically, we gauged shorter term effects on ecosystem structure and function in Ecopath by reducing or augmenting the biomass of high-TL groundfishes, following the percentage changes depicted in Figures 4 and 5 (see “Results”). Other than these perturbations, we held all inputs consistent with the balanced Ecopath model originally published by Field and others (2006) except for the biomass accumulation rate terms, and then re-balanced the model. Solving for biomass accumulation rate terms provided a description of how individual species groups would need to respond to changes in the biomass of high-TL groundfishes to balance the ecosystem-wide bioenergetic budget assumed in the model formulation. With this approach, forced reductions (increases) in predation could only result in a calculated positive (negative) biomass accumulation rate for prey, that is, we could only evaluate first-order effects. We could not, however, test for indirect responses (second-order interactions) by competitors and other species groups using the Ecopath model. Instantaneous responses were standardized to facilitate comparisons among species groups and ecosystem functions, and are reported as differences from the Ecopath baseline model. We tested whether biomass accumulation rate terms were correlated (Spearman rank) with the degree to which prey contributed to high-TL groundfish diets, using diet information contained within Field and others (2006).
We evaluated longer term changes in ecosystem structure and function with Ecosim by simulating the dynamic responses of the food web to forced changes in high-TL groundfish biomass over 10 years. We calculated the mean annual percentage differences in biomasses of food web groups by comparing the results of a baseline simulation with a simulation run with the biomass changes detailed above. Trends and mean annual differences between perturbed and baseline model runs were standardized to values in the first year of the simulations.
For both the instantaneous and dynamic model outputs, we report the responses of individual species groups, as well as trophic groups and ecosystem functions. Trophic groups included herbivores, zooplanktivores, macroinvertivores, non-groundfish piscivores, and scavengers (Table 2). Ecosystem functions included overall rates of production, consumption, respiration, throughput, and net primary production. Throughput describes the sum of all flows of mass or energy that enter and exit the food web during a unit of time.
For the longer term, dynamic simulations, we also analyzed the responses to changes in the biomass of high-TL groundfishes of intermediate and lower TL prey, competitors and other species (Table 2) more loosely connected to high-TL groundfishes. Species composition of prey, competitors, and other groups were chosen for illustrative purposes. Responses, measured as percentage differences in biomass density compared to baseline simulations, were averaged across species for each category.
Results
Empirical Analysis of Change in Ecosystem MTL
From 2003 to 2011 ecosystem MTL (unweighted) declined by approximately 0.13 U from just over 4.04 to slightly less than 3.91. The slope of the relationship was −0.021 U/year (Figure 2)—a rate almost twice the 0.1 U per decade noted as alarming in previous studies (Pauly and others 1998; Branch and others 2010). The absolute drop in ecosystem MTL was just under the 0.15 considered ecologically significant by Essington and others (2006) because it represents a reduction of approximately 50% in the primary production necessary to maintain a given amount of harvest (Pauly and Christensen 1995).
However, although area-weighted ecosystem MTL also declined from 2003 to 2011, the magnitude of the decline was much lower (Figure 2). Area-weighted ecosystem MTL declined from 3.72 to 3.66, a decline of 0.06 points demonstrating that failing to account for sampled area overestimated the magnitude of the decline in MTL. Area-weighted ecosystem MTL was highest in 2004 at 3.72 and lowest in 2009 at 3.64, a difference of 0.08 U. The slope was −0.01 point per year or 0.1 point per decade decline, a decrease in area-weighted MTL comparable to the rate noted as alarming in previous studies as mentioned above but half that of the unweighted estimate.
The decrease in ecosystem MTL of groundfishes may have been caused by loss of high-TL groundfishes and/or increases in the abundance of low-TL species (Pauly and Watson 2005; Essington and others 2006). The approximate mid-TL of the species in the groundfish assemblage by frequency and by biomass was 3.5 (Figure 3), and we defined high-TL groundfishes as those with TL (T s ) of at least 3.5. The biomass of low-TL groundfishes fluctuated somewhat and showed a very modest decline from 2003 to 2011 (Figure 4). However, biomass for high-TL groundfishes declined from 2003 to 2011 by 38% from 1144 × 106 to 709 × 106 kg (Figure 4). That is, a loss of high-TL groundfishes, rather than the expansion of low-TL species, caused the reduction in ecosystem MTL.
For those taxa included in the Ecopath model as individual species, five high-TL species declined in abundance including Pacific hake, lingcod, sablefish, shortspine thornyhead, and widow rockfish (Figure 5). Hake showed the strongest, most linear, and consistent decline dropping by 89% from 2003 to 2011, whereas lingcod and sablefish declined by 70 and 51%, respectively. Widow rockfish showed a nonlinear trend with an initial decline followed by a recent recovery, but it was still around 28% lower in 2011 than in 2003. For shortspine thornyhead, the change was small at only a 9% decrease in biomass. Two high-TL species, arrowtooth flounder and yellowtail rockfish increased in abundance. Arrowtooth flounder increased sharply from 2003 to 2011 by about 71%. For yellowtail, the increase was a recovery following an initial decrease over the first half of the time series.
Biomass trends for the 15 species in the Ecopath model. The title gives the species name and TL from Fishbase.org. Trend lines indicate models with significant linear or nonlinear regressions (P < 0.05). Percentages indicate increases or decreases in biomass for those species with significant trends through time. Arrowtooth arrowtooth flounder, Dover Dover sole, hake Pacific hake, POP Pacific Ocean perch, halibut Pacific halibut, rf rockfish, th thornyhead.
Of the taxa combined into functional groups in the EwE model, dogfishes, shelf rockfish, slope rockfish, small flatfishes, and grenadiers all showed strong nonlinear trends with initial declines followed by stabilization at lower levels or more recent increases, with overall decreases in abundance ranging from 18 to 74% (Figure 6). Skates and grenadiers fluctuated but did not show any trends. Benthic fishes increased by about 22%.
Trends in biomass for the seven combined taxa included in the Ecopath model. Trend lines indicate models with significant linear or nonlinear regressions (P < 0.05). Percentages indicate increases or decreases in biomass for those species with significant trends through time. The number in parentheses is the area-weighted MTL of the species in the group calculated using TLs from Fishbase.org from 2003 to 2011.
Ecosystem Modeling: Predicted Consequences of Changes in Ecosystem MTL
The Ecopath model predicted strongly positive responses of the functional groups directly preyed upon by high-TL groundfishes in the short term (Table 3, middle column). The magnitude of this predicted release from groundfish predation was positively correlated with the degree to which prey contributed to high-TL groundfish diets (Spearman rank correlation = 0.64, P < 0.0001).
In contrast to the shorter term results, dynamic simulations of trophic interactions in the California Current over 10 years revealed three dominant patterns: prey release, competitive release, and no directional change (Figure 7). A subset of the functional groups directly preyed upon by high-TL groundfishes (intermediate TL species such as shrimp, squid, forage fishes, and crabs) showed a persistent positive response (prey release; Table 3). Several functional groups that compete for prey with predatory groundfishes (seabirds, salmon, and albacore tuna) showed an indirect positive response (competitive release; Table 3; Figure 7) that developed more gradually than that of the prey due in part to their slower life histories (lower productivity). Last, several functional groups showed little directional change (Table 3; Figure 7). These weaker responses had two causes. First, the release of intermediate TL prey from predation by high-TL groundfishes tempered or even reversed initial positive responses by low-TL prey (such as krill and pelagic shrimp). Second, some functional groups were simply loosely connected nodes in the food web of high-TL groundfishes (Francis and others 2012), and therefore were relatively insensitive to perturbations directed at higher predators.
Dynamic responses of the California Current food web to a 40% reduction in higher TL groundfishes (TL ≥ 3.5). Predicted differences between 10-year model simulations of a 40% reduction in higher TL groundfishes and baseline trajectories for intermediate and lower TL prey, competitors, and other groups. Species composition of prey, competitors, and other groups were chosen for illustrative purposes. Responses (percentage differences in biomass density) were averaged across species for each category. Intermediate TL prey include forage fish, cephalopods, pandalid shrimp, and crabs; low-TL prey include krill and carnivorous zooplankton; competitors include salmon, albacore tuna, seabirds, harbor seals, and low-TL groundfishes; other groups include phytoplankton, infauna, orcas, and gray whales.
Ecosystem functions related to herbivory and scavenging showed instantaneous increases, and there were also instantaneous decreases in respiration and consumption. However, dynamic simulations revealed that the strong responses of these trophic processes would likely be tempered over time (Table 3, rightmost column). Only piscivory (excluding high-TL groundfishes) and consumption by macroinvertivores (predation on non-planktonic invertebrates) increased in the long term. The increase in piscivory (by non-groundfishes) is indicative of competitive release, while the increase in predation on non-planktonic invertebrates is indicative of intermediate TL prey release, as depicted in Figure 7.
When combined across groups, gross ecosystem functions related to bioenergetic rates (total consumption, total respiration, total throughput, total production, net primary production) did not show persistent long-term change. These functions were robust to the decline in high-TL groundfishes whether we measured shorter- or longer-term responses (Table 3).
Imposing an across-the-board drop in biomass of 40% for all high-TL groundfishes produced similar outcomes for both ecosystem structure and function (Appendix A in Electronic Supplementary Material). This result suggests that MTL provided a broad, overall estimate of predation pressure by groundfishes.
Discussion
The most useful, robust ecosystem indicators are measurable, responsive variables that can be mechanistically related to the status of key ecosystem attributes (Rice and Rochet 2005). The drop in MTL seen here was due to a decrease in the abundance of high-TL groundfishes. The California Current is thought to be influenced from the bottom-up by primary production rates (Ware and Thomson 2005). If top-down forces had little to no effect on the system, a decline in groundfish MTL would only provide information on the abundance of high-TL groundfish species—one component of the ecosystem. However, our modeling analysis and that of others demonstrates the potential importance of top-down drivers in the California Current (Field and others 2006; Halpern and others 2006; Worm and others 2006). Specifically, a decline in the biomass of predatory groundfishes can propagate through the ecosystem driving changes in the abundance of other species—both prey and competitors. It does not appear that the structural changes strongly influenced ecosystem functions in the model at the system-wide scale. Rather, functional redundancy of predators in the system compensated for the loss of predatory groundfishes. Nevertheless, this indicator holds promise for EBM efforts because patterns of ecosystem reorganization are embedded in trends in ecosystem MTL: changes in the biomass of predatory groundfishes may drive structural change in the ecosystem at large.
Ecosystem MTL decreased in an era of reduced fishing pressure on many groundfishes (Hilborn and others 2012). Many depleted groundfish stocks have increased in biomass after substantial reductions in catch at the end of the 1990s (Hilborn and others 2012), and it seems unlikely that the drop in MTL was due primarily to fishing. Instead, the drop in MTL may be linked to a combination of the aging and dying-off of strong 1998- to 1999-year classes for many west coast species and to climate effects (Keller and others 2012). Many high-biomass, high-TL fishes like Pacific hake, dogfish, lingcod, and sablefish had strong year classes around 1999. As this 1999-year class has aged and died off the overall abundance of these species has decreased (investigated in detail by Keller and others 2012). Together, these observations suggest that at a minimum fishing was not the singular driver of the decline in predatory groundfishes. Thus, whereas the structure of the decline was analogous to “fishing down the food web” in that the abundance of high-TL fishes decreased, fishing down was not necessarily the cause, and the decrease in ecosystem MTL may represent natural ecosystem fluctuation.
Irrespective of the cause, large changes in groundfish MTL in the California Current are not anomalous to our study period. Both catch and ecosystem MTL of groundfishes have fluctuated historically (Branch and others 2010). For example, MTL estimated from the US West Coast Triennial Trawl Survey fluctuated substantially from 1977 to 2004 slowly increasing over this time period (Branch and others 2010). However, results from this survey are somewhat difficult to both interpret and compare to the West Coast Groundfish Bottom Trawl Survey because the sampling design of the triennial survey varied through time and because the two surveys differ in methodology (see Supplementary Material in Branch and others 2010; Levin and Schwing 2011). Even in the West Coast Groundfish Bottom Trawl Survey data used in the present study, ecosystem MTL appears to have increased slightly in 2011, and longer term monitoring will be necessary to determine whether the current decline is simply a natural fluctuation or a more persistent ecosystem change. For example, the 2013 stock assessment for Pacific hake suggests strong recruitment (J.T.C. 2013). As Pacific hake are a high-biomass, high-TL species, these changes may push MTL back up. Regardless of the cause of the fluctuations in MTL of groundfishes, it is still important to understand how these changes in predation pressure influence the rest of the ecosystem in order to better inform management decisions (discussed further below).
Trophic cascades and top-down forcing have been demonstrated in many systems (Daskalov 2002; Pauly and Watson 2005; Field and others 2006; Baum and Worm 2009; Estes and others 2011), although the specifics of the responses vary. For example, on the Scotian Shelf and in the Gulf of St. Lawrence, declines in the abundance of piscivorous fishes (primarily cod) due to fishing have lead to prey release and increases in forage fishes like capelin, but the effect can be influenced by the presence of fisheries on forage fishes (Savenkoff and others 2007; Bundy 2005; Bundy and others 2009). These declines in piscivorous fishes can also cascade further through the system. For example, a decrease in high-TL fishes (cod) off the Swedish coast appears to have led to increases in mid-TL fishes like gobies and sticklebacks with concurrent declines in meso-herbivores (amphipods), resulting in the overgrowth of seagrass by filamentous algae (Baden and others 2012). Likewise, otters control urchin densities allowing the development of dense kelp forests, which in turn supports higher densities of kelp-associated fishes (Estes and others 2011).
Like the empirical examples mentioned above, the model simulations we conducted suggest that periodic swings in the abundance of predatory groundfishes on the U.S. west coast can drive structural change in the broader ecosystem. Indeed, our analyses suggested that prey and competitors of species within the exploited groundfish community are likely to demonstrate fluctuations linked to changes in the biomass (as indicated by MTL) of high-TL groundfishes. Understanding and predicting these changes are important to EBM, which aims to identify and resolve trade-offs between different benefits derived from the ocean (Leslie and McLeod 2007). This study directs attention to how trade-offs between so-called ecosystem services can develop. Specifically, our heuristic modeling suggests that a reduction in the abundance of exploited, predatory groundfishes, which might be considered a negative effect for the fishery, is expected to benefit protected species that compete with them (for example, Herrick 2009; Smith and others 2011a). In turn, increases in the abundance of protected species such as salmon and seabirds can improve the delivery of marine ecosystem services related to recreation and tourism. Awareness of trade-offs within and between fisheries and other marine ecosystem services (Worm and others 2006) is the first step toward generating solutions that minimize conflict between different user groups, and will be a cornerstone of successful EBM efforts (Levin and others 2009).
Predictions about the potential for these trade-offs are contingent on at least two preconditions within the California Current ecosystem. First, the positive response of non-groundfish predators to the decline in groundfish MTL would not have been possible were it not for functional redundancy within the predator guild. The California Current food web is characterized by multiple examples of predators with shared prey (in particular, forage fishes and krill (Field and others 2006)). These trophic connections likely buffered the system from exhibiting a strong trophic cascade (Estes and others 2011), and prevented realization of substantial changes in gross ecosystem functions. Taxa like salmon, albacore, and seabird, and trophic groups like piscivores (excluding high-TL groundfishes) all showed persistent increases in the system in response to the removal of high-TL biomass, as did the ecosystem functions directly related to these groups. The insensitivity of the gross ecosystem functions is also partly a result of the assumptions inherent to Ecopath with Ecosim, which is rooted in a bioenergetics budget. In addition, even in the absence of the high-TL groundfish decline, greater than 70% of the energy flow in the model ecosystem is tied up by phytoplankton and detritus groups. As a result, structural changes in the relative abundance of groundfish species would not necessarily be expected to beget large changes in these ecosystem-scale functions.
The predictions are also contingent on the assumptions of the model we used, which has a substantial forage base allowing competitors to capitalize on the decline in predatory groundfishes. This assumption is in line with status quo management of lower TL species, and in particular zero, low or conservative exploitation of forage species (50 CFR Part 660 2009; Smith and others 2011a). However, there is growing interest in developing fisheries for forage species to serve the needs of aquaculture. This study and others suggest that such decisions cannot be made without recognizing the potential for impacts on other parts of the ecosystem and stress the need for EBM approaches (Herrick 2009; Smith and others 2011b).
Our modeling also suggests that MTL should be a useful indicator of top-down forcing in California Current for two reasons. First, and most obvious, the species-specific changes in the biomass of high-TL fishes indicated by declining ecosystem MTL drove structural change in the ecosystem resulting in both prey release and competitive release. Second, we observed highly similar results from an EwE model in which we imposed an across-the-board (non-species-specific) decrease of 40% on all high-TL groundfish species and functional groups (Appendix A in Electronic Supplementary Material). The similarity of the results most likely stems from functional redundancy in the guild of predators—many high-TL groundfishes consume krill and forage fishes (Field 2004; Field and others 2006)—making it less important as to which predator species decrease. The generality of the effect of declining MTL should, therefore, make it a fairly robust indicator of top-down forcing by groundfishes.
EBM brings with it an increasing need for scientific methods to identify mechanistic connections between different components of marine ecosystems. Linking indicators like ecosystem MTL to broad-scale ecosystem structure and function will provide relevant and essential information to socio-political institutions whose goal it is to respond nimbly to a marine environment that is consistently dynamic.
References
50 CFR Part 660. 2009. Fisheries Off West Coast States; Coastal Pelagic Species Fishery; Amendment 12 to the Coastal Pelagic Species Fishery Management Plan.
Baden S, Emanuelsson A, Pihl L, Svensson CJ, Aberg P. 2012. Shift in seagrass food web structure over decades is linked to overfishing. Mar Ecol Prog Ser 451:61–73.
Baum JK, Worm B. 2009. Cascading top-down effects of changing oceanic predator abundances. J Anim Ecol 78:699–714.
Branch TA, Watson R, Fulton EA, Jennings S, McGilliard CR, Pablico GT, Ricard D, Tracey SR. 2010. The trophic fingerprint of marine fisheries. Nature 468:431–5.
Bundy A. 2005. Structure and functioning of the eastern Scotian Shelf ecosystem before and after the collapse of groundfish stocks in the early 1990s. Can J Fish Aquat Sci 62:1453–73.
Bundy A, Heymans JJ, Morissette L, Savenkoff C. 2009. Seals, cod and forage fish: a comparative exploration of variations in the theme of stock collapse and ecosystem change in four Northwest Atlantic ecosystems. Prog Oceanogr 81:188–206.
Christensen V, Pauly D. 1992. Ecopath II—a software for balancing steady-state ecosystem models and calculating network characteristics. Ecol Model 61:169–85.
Christensen V, Walters C, Pauly D. 2005. Ecopath with ecosim: a user’s guide. Vancouver: Fisheries Centre, University of British Columbia.
Daskalov GM. 2002. Overfishing drives a trophic cascade in the Black Sea. Mar Ecol Prog Ser 225:53–63.
Essington TE, Beaudreau AH, Wiedenmann J. 2006. Fishing through marine food webs. Proc Natl Acad Sci USA 103:3171–5.
Estes JA, Terborgh J, Brashares JS, Power ME, Berger J, Bond WJ, Carpenter SR, Essington TE, Holt RD, Jackson JBC, Marquis RJ, Oksanen L, Oksanen T, Paine RT, Pikitch EK, Ripple WJ, Sandin SA, Scheffer M, Schoener TW, Shurin JB, Sinclair ARE, Soule ME, Virtanen R, Wardle DA. 2011. Trophic downgrading of planet earth. Science 333:301–6.
Field JC. 2004. Application of ecosystem-based fishery management approaches in the northern California Current. Seattle: School of Aquatic and Fishery Sciences, University of Washington. p 408.
Field JC, Francis RC, Aydin K. 2006. Top-down modeling and bottom-up dynamics: linking a fisheries-based ecosystem model with climate. Prog Oceanogr 68:238–70.
Francis TB, Scheuerell MD, Brodeur RD, Levin PS, Ruzicka JJ, Tolimieri N, Peterson WT. 2012. Climate shifts the interaction web of a marine plankton community. Global Change Biol 18:2498–508.
Froese R, Pauly D. 2010. FishBase. www.fishbase.org. Accessed July 2010.
Globec U. 1994. Eastern boundary current program: a science plan for the California Current US global ocean ecosystems dynamics, report number 11. Berkeley, CA: US GLOBEC Scientific Steering Coordination Office.
Halpern BS, Cottenie K, Broitman BR. 2006. Strong top-down control in southern California kelp forest ecosystems. Science 312:1230–2.
Hannesson R, Herrick S Jr, Field J. 2009. Ecological and economic considerations in the conservation and management of the Pacific sardine (Sardinops sagax). Can J Fish Aquat Sci 66:859–68.
Heithaus MR, Frid A, Wirsing AJ, Worm B. 2008. Predicting ecological consequences of marine top predator declines. Trends Ecol Evol 23:202–10.
Hilborn R, Stewart IJ, Branch TA, Jensen OP. 2012. Defining trade-offs among conservation, profitability, and food security in the California Current bottom-trawl fishery. Conserv Biol 26:257–66.
Horn MH, Allen LG. 1978. A distributional analysis of California coastal marine fishes. J Biogeogr 5:23–42.
Horn MH, Allen LG, Lea RN. 2006. Biogeography. In: Allen LG, Pondella DJII, Horn MH, Eds. The ecology of marine fishes: California and adjacent waters. Berkley: University of California Press. p 3–25.
J.T.C. 2013. Status of the Pacific hake (whiting) stock in the U.S. and Canadian waters in 2013. In: Hicks AC, Taylor, N, Grandin C, Taylor IG, Cox S, Eds. Prepared for the Joint U.S.-Canada Pacific hake treaty process. p. 190. http://www.nwr.noaa.gov/fisheries/management/whiting/pacific_whiting.html. Accessed 13 Mar 2013.
Jiming Y. 1982. A tentative analysis of the trophic levels of North Sea fish. Mar Ecol Prog Ser 7:247–52.
Keller AA, Horness BH, Fruh EL, Simon VH, Tuttle VJ, Bosley KL, Buchanan JC, Kamikawa DJ, Wallace JR. 2008. The 2005 U.S. West Coast bottom trawl survey of groundfish resources off Washington, Oregon, and California: Estimates of distribution, abundance, and length composition. U.S. Department of Commerce, NOAA Technical Memorandum. NMFS-NWFSC-93. p. 136.
Keller AA, Wallace JR, Horness BH, Hamel OS, Stewart IJ. 2012. Variations in eastern North Pacific demersal fish biomass based on the U.S. west coast groundfish bottom trawl survey (2003–2010). Fish Bull 110:205–22.
Leslie HM, McLeod KL. 2007. Confronting the challenges of implementing marine ecosystem-based management. Front Ecol Environ 5:540–8.
Levin PS, Schwing F. 2011. Technical background for an integrated ecosystem assessment of the California Current: groundfish, salmon, green sturgeon, and ecosystem health. U.S. Deptartment of Commererce, NOAA Technical Memorandum, NMFS-NWFSC-109. p. 330.
Levin PS, Fogarty MJ, Murawski SA, Fluharty D. 2009. Integrated ecosystem assessments: developing the scientific basis for ecosystem-based management of the ocean. PLoS Biol 7:23–8.
Pauly D, Christensen V. 1995. Primary production required to sustain global fisheries. Nature 374:255–7.
Pauly D, Watson R. 2005. Background and interpretation of the ‘Marine Trophic Index’ as a measure of biodiversity. Philos Trans R Soc B Biol Sci 360:415–23.
Pauly D, Christensen V, Dalsgaard J, Froese R, Torres F. 1998. Fishing down marine food webs. Science 279:860–3.
Rice JC, Rochet MJ. 2005. A framework for selecting a suite of indicators for fisheries management. ICES J Mar Sci 62:516–27.
Savenkoff C, Castonguay M, Chabot D, Hammill MO, Bourdages H, Morissette L. 2007. Changes in the northern Gulf of St. Lawrence ecosystem estimated by inverse modelling: evidence of a fishery-induced regime shift? Estuar Coast Shelf Sci 73:711–24.
Sethi SA, Branch TA, Watson R. 2010. Global fishery development patterns are driven by profit but not trophic level. Proc Natl Acad Sci USA 107:12163–7.
Smith ADM, Brown CJ, Bulman CM, Fulton EA, Johnson P, Kaplan IC, Lozano-Montes H, Mackinson S, Marzloff M, Shannon LJ, Shin YJ, Tam J. 2011a. Impacts of fishing low-trophic level species on marine ecosystems. Science 333:1147–50.
Smith LM, Haukos DA, McMurry ST, LaGrange T, Willis D. 2011b. Ecosystem services provided by playas in the High Plains: potential influences of USDA conservation programs. Ecol Appl 21:S82–92.
Stergiou KI, Tsikliras AC. 2011. Fishing down, fishing through and fishing up: fundamental process versus technical details. Mar Ecol Prog Ser 441:295–301.
Tolimieri N. 2007. Patterns in species richness, species density and evenness in groundfish assemblages on the continental slope of the US Pacific coast. Environ Biol Fishes 78:241–56.
Tolimieri N, Anderson MJ. 2010. Taxonomic distinctness of demersal fishes of the California Current: moving beyond simple measures of diversity for marine ecosystem-based management. PLoS ONE 5:e10653.
Tolimieri N, Levin PS. 2006. Assemblage structure of eastern Pacific groundfishes on the U.S. continental slope in relation to physical and environmental variables. Trans Am Fish Soc 135:115–30.
Tremblay-Boyer L, Gascuel D, Watson R, Christensen V, Pauly D. 2011. Modelling the effects of fishing on the biomass of the world’s oceans from 1950 to 2006. Mar Ecol Prog Ser 442:169–85.
Ware DM, Thomson RE. 2005. Bottom-up ecosystem trophic dynamics determine fish production in the northeast Pacific. Science 308:1280–4.
Worm B, Barbier EB, Beaumont N, Duffy JE, Folke C, Halpern BS, Jackson JBC, Lotze HK, Micheli F, Palumbi SR, Sala E, Selkoe KA, Stachowicz JJ, Watson R. 2006. Impacts of biodiversity loss on ocean ecosystem services. Science 314:787–90.
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM. 2009. Mixed effects models and extensions in ecology with R. New York: Springer.
Acknowledgments
Thanks to J. Field for allowing us to analyze his model of the California Current and to C. Harvey, I. Kaplan, T. Branch, T. Essington, and three anonymous reviewers for comments and discussion on the manuscript. Special thanks to the trawl survey staff for data collection and to J. Samurai, B. Wiggins, U. Sada, G. Monmouth, T.V. Bede, G. Money, and T. Lanister.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
NT and JS conceived study, analyzed data, wrote manuscript; VS performed research; BF conducted GIS analyses, wrote manuscript; PL conceived study, wrote manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tolimieri, N., Samhouri, J.F., Simon, V. et al. Linking the Trophic Fingerprint of Groundfishes to Ecosystem Structure and Function in the California Current. Ecosystems 16, 1216–1229 (2013). https://doi.org/10.1007/s10021-013-9680-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-013-9680-1