Abstract
Autism is a hereditary, pervasive neurodevelopmental disorder that starts early in life. The main characteristics of the autism are impairment in social interactions, difficulties in adapting to novel environmental situations and improper reaction to stress. Since the Hypothalamic-Pituitary-Adrenocortical (HPA) axis plays a key role in the response to stress and because the previous research found abnormalities in HPA system, we conducted a study to test several elements of the HPA axis. Because autism is a heritable disorder, autistic subjects were studied as well as their parents. Cortisol circadian rhythm, cortisol daily secretion and its suppression response to dexamethason had been measured from saliva or urine samples of the autistic children and their parents. Cortisol secretion response after ACTH stimulation was done with the autistic children only. The cortisol elevation after ACTH stimulation among the autistic individuals was slower (P = 0.017) than in healthy controls. No differences were found in salivary cortisol circadian rhythm or suppression response, as well as in cortisol daily excretion. These data indicate that, compared to healthy subjects, autistic individuals have fine differences in cortisol response to ACTH stimulation or possibly to other types of stress.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Autism belongs to the group of pervasive developmental disorders that are characterized by a lack of social communication skills, improper language development, limited interests and stereotypical behavior [1, 15]. Despite the fact that it was described 60 years ago, its etiology remains a mystery, which is making autism very challenging from etiological, as well as therapeutical perspective [4, 25, 32, 33]. Many genetic studies [24, 30, 31], hormone and neurotransmitter analysis [10, 26, 28] and studies aiming to find the cause of autism in neurological abnormalities and lesions [2, 12, 19, 22], did not find reasonable explanation for the mechanisms underlining development of autism. The hypothalamic-pituitary-adrenocortical (HPA) axis, as a part of limbic system, deserves special attention, since it is the basis for emotions and social interactions, functions that are affected in autism. There is a plenty of evidence that the limbic system function, together with the HPA axis, has been impaired in autistic children (lack of feelings, miscomprehension of hearing, visual and tactile stimuli, abnormal reaction to almost all stress causing factors) [2, 7, 19, 22]. Furthermore, the HPA axis is mediating response to stress, which is largely modified among autistic individuals [6, 32]. One of the approaches to test the HPA axis is a measurement of its hormones and their subsequent targets, which was used in many studies [8, 29]. Although many studies were indicating about abnormalities in the HPA axis in autistic patients, their findings were not consistent; moreover, they were sometimes contradictory. Some studies found abnormal cortisol or ACTH levels among autistic individuals [3, 14, 21], while others did not found a difference [9, 18, 23, 27, 29], which indicates that more complex studies need to be done.
In our previous study, we found that the cortisol level in plasma was lower, while ACTH was elevated among autistic individuals [14]. Our results hold high statistical significance for observed cortisol difference; therefore, we conducted more profound research of cortisol functioning. In this study we tested: daily rhythm of cortisol secretion, total daily cortisol secretion, cortisol suppression and cortisol stimulation response. Since autism is a heritable disorder, some changes found in autistic individuals should be present in their parents. In order to test this hypothesis, we included parents of autistic individuals in the study.
Methods
Autistic individuals included in the study, were diagnosed according to the DSM-IV and ICD-10 criteria; diagnosis of autism was determined by child psychiatrist. They were examined by child psychologist and tested by Childhood autism rating scale, Stanford-Binet scale and Autistic Diagnostic Interview. Autistic individuals were recruited at the Center for Autism in Zagreb and Split, and they were all males. This study was approved by the Ethical Committee of the Split University Hospital. All the participants (parents for themselves and for children) provided a written informed consent after they received comprehensive information about the nature of the study.
Control group included healthy boys, without psychiatric and endocrinology disorders, matching autistic children regarding their age. Age of autistic individuals was 11.9 ± 2.97 and of control individuals 11.6 ± 4.27; we had nine autistic and seven control individuals. Parents of the control individuals were included in the study and were without psychiatric or endocrine abnormalities, as they reported during an interview. Parents of the control individual were examined by a psychiatrist and were tested by the GHQ-60 test. Parents of the autistic individuals were included into study because they have autistic child. The control individuals for the ACTH test were healthy adult males, since it was difficult to obtain appropriate children for controls.
The salivary cortisol samples for a daily rhythm analysis were collected from nine autistic families (mother, father and autistic son) and seven control families (mother, father and son). The salivary samples were collected 6 times during the day: immediately after awakening, 30 min after awakening, just before lunch, 60 min after lunch, at 6 p.m. and just before going to sleep (around 10 p.m.). The samples were collected and kept until the analysis, as it was described previously [11]. Free cortisol in saliva was determined by saliva cortisol kit (DSL, Texas).
Dexamethason suppression test was performed among the autistic and the control family members, as previously described [5, 13, 20]. Briefly, 1 mg of dexamethason was given “per os”, around 10 p.m. one salivary sample was taken prior to dexamethason treatment and another sample for the cortisol measurement was taken the following morning, just after awakening.
Daily secretion of cortisol was calculated from the cortisol concentration in urine and urine volume, after collecting urine during 24 h period, from the autistic and the control family members. Urinary cortisol concentration was measured using a standard RIA test, (Immuntech, France).
Quick (rapid) ACTH stimulation test was performed on seven autistic individuals and on five control individuals, following the usual procedure [5, 13, 20]. Five salivary samples were collected, one sample prior to the test and at 30, 60, 90, and 120 min after the ACTH application.
For statistical analysis of repeated measures of saliva cortisol, between healthy and autistic children, and also within groups of their mothers and fathers, the Mann-Whitney U test was used. Also, when autistic children were grouped with their parents and compared with control families, similar analysis was performed. For comparison of ACTH stimulation responses, between autistic boys and healthy males, the Mann-Whitney U test was used. Urinary cortisols were compared using T-test for independent variables. For comparison of absolute differences in cortisol levels between different measuring points of saliva cortisol measurement, analysis of variance (ANOVA) test was used.
Results
Important finding of this study is a statistically significant difference in a salivary cortisol levels during the ACTH stimulation test between autistic and normal individuals. The difference is evident at 90 min time point (P = 0.017), but the difference in trend can be observed in all measured time points (Fig. 1).
Cortisol response to rapid ACTH test. Autistic and control subjects were given 250 µg of Tetracosactrine (Synacten®) in intramuscular injection; salivary samples were collected before (time 0), and 30, 60, 90 and 120 min after injection of ACTH. Salivary cortisol concentrations were measured with ELISA assay. Data represents mean and unilateral standard error (bars). *P = 0.017
When the degree of a cortisol increase was measured between 30 and 90 min time points, the difference was even more obvious (P < 0.01), indicating a slower increase of plasma cortisol level among autistic individuals.
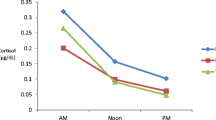
There was no significant difference in measured values of cortisol circadian rhythm among all study subjects (Fig. 2) as well as in the cortisol values after dexamethason suppression test (results not shown).
Daily cortisol secretion curves. Salivary cortisol concentrations were measured and compared between autistic and control children, mothers or fathers of autistic and control children. Also, cortisol concentrations of families with autistic children were compared with control families (mother, father, and child). Cortisol salivary samples: C1- immediately after awakening; C2- 30 min after awakening; C3- just before lunch; C4- 60 min after lunch; C5-6 p.m.; C6-just before going to sleep (around 10 p.m.). Data represents mean and unilateral standard error (bars)
Also, daily cortisol secretion measured via 24 h urine did not differ between autistic individuals and their parents when compared with healthy controls (110 ± 77 nmol/l vs. 70 ± 58 nmol/l; P = 0.26, T-test).
Discussion
In this study, the hypothalamic-pituitary-adrenocortical axis functioning in autistic individuals, their parents and healthy controls was analyzed. Namely, cortisol circadian rhythm, cortisol suppression response (via dexamethason test), cortisol stimulation response (using rapid ACTH test) and daily cortisol secretion (in 24 h urine) were tested.
Our results show that cortisol release following ACTH administration was slower in autistic individuals, when compared with healthy, sex matched individuals. Slow cortisol response to ACTH stimulation among autistic individuals could explain our previous finding of lower cortisol, which was found after venepuncture performed to obtain serum sample [14]. This, slower response of main anti-stress hormone—cortisol, could help explain higher susceptibility of autistic individuals to various stressors [9, 10, 16, 17]. One recent study [3] shows that autistic children responded to social stress with more cortisol release, which is in contrast to our data. Possible explanation for this discrepancy could be that mechanisms involving the interpretation of an emotional response to a social stressor, plays an important role in cortisol release, which is completely lacking in ACTH stimulation test [14, 27]. Although we see slower cortisol release in autistic subjects, finally, after 2 h they reach the same cortisol level as healthy controls. It needs to be noted that parents of autistic children did not undergo the ACTH stimulation test.
In the other part of the study, autistic children and their parents as well as healthy children and they parents were analyzed. In contrast to ACTH stimulation test, by analyzing cortisol circadian rhythm, cortisol suppression response and total daily cortisol secretion, we did not observe any difference. No difference was detected when parents of autistic and non-autistic children were compared. Also, comparison of autistic and healthy children did not show any difference, as well as comparison of autism-affected families with non-affected families. Not seeing a difference in cortisol circadian rhythm, cortisol suppression response and total daily cortisol secretion, could be due to the fact that the difference between parents is not big enough in order to be detected by the methods used in this study. Thus it could be more appropriate to do more sensitive, blood tests for cortisol measurements for comparing parents of autistic and healthy children. However, not seeing difference in cortisol circadian rhythm is in parallel with the same amount of cortisol daily secreted in urine.
In conclusion, we found that autistic individuals have slower cortisol secretion in saliva after ACTH stimulation. That finding should direct some future studies of autism.
References
American Psychiatric Association (1994) Diagnostic and statistical manual of Mental disorders, (4th edn, rev). Author, Washington, DC
Aylward EH, Minshev NJ, Goldstein G, Honeycutt NA, Augustine AM, Yates KO, Barta PE, Pearlson GD (1999) MRI volumes of amygdala and hippocampus in not mentally retarded autistic adolescents and adults. Neurology 53:2145–2150
Corbett BA, Mendosa S, Abdullah M, Wegelin JA, Levine S (2006) Cortisol circadian rhythms and response to stress in children with autism. Psychoneuroendochrinology 31:59–68
Dawson G (2001) The search for autism’s roots. Nature 41:882–884
Harris B, Watkins S, Cook N, Walker RF, Read GF, Riad-Fahmy D (1990) Comparisons of plasma and salivary cortisol determinations for the diagnostic efficacy of the dexametasone suppression test. Biol Psychiatry 27:897–904
Herbert J (1998) Neurosteroids, brain damage, and mental illnes. Exp Gerontol 33:713–727
Hrdlicka M, Dudlova I, Beranova I, Lisy J, Belsan T, Neuwirth J, Komarek V, Faladova L, Havlovicova M, Sedlacek Z, Blatny M, Urbanek T (2005) Subtypes of autism by cluster analysis based on srtuctural MRI data. Eur Child Adolesc Psychiatry 14:138–144
Jacobson L (2005) Hypothalamic-pituitary-adrenocortical axis regulation. Endocrinol Metab Clin North Am 34:271–292
Jansen LM, Gispen-de Wied CC, van der Gaag RJ, ten Hove F, Willemsen-Swinkels SW, Harteveld E, Engeland H (2000) Unresponsivness to psychosocial stress in subgroup of autistic-like children, multiple complex developmental disorder. Psychoneuroendocrinology 25:753–764
Jansen LM, Gispen-de Wied CC, van der Gaag RJ, van Engeland F (2003) Differentiation between autism and multiple complex developmental disorder in response to psychosocial stress. Neuropsychopharmachology 28:582–590
KiessW, Meidert A, Dressendorfer RA, Schriever K, Kessler U, Konig A, Schwarz HP, Strasburger CJ (1995) Salivary cortisol levels throughout childhood and adolescence: relation with age, pubertal stage, and weight. Pediatr Res 37:502–506
Lainhart JE (1997) Developmental abnormalities in autism. Lancet 349:373–374
Lifshitz F (1996) Pediatric endocrinology, (3rd edn). Mercel-Dekkor Inc., New York
Marinovic-Curin J, Terzic J, Bujas-Petkovic Z, Zekan LJ, Marinovic-Terzic I, Marasovic-Susnjara I (2003) Lower cortisol and higher ACTH levels in individuals with autism. J Autism Dev Disord 33:443–448
Mash EJ, Russell AB (2003) Child psychopathology, (2nd edn). The Guilford Press, New York
Matarazzo EB (2002) Treatment of late onset autism as a consequence of probable autoimmune processes related to chronic bacterial infection. World J Biol Psychiatry 3:162–166
Molloy CA, Manning-Courtney P (2003) Prevalence of chronic gastrointestinal symptoms in children with autism and autistic spectrum disorders. Autism 7:165–171
Nir I, Meir D, Zilber N, Knobler H, Hadjez J, Lerner Y (1995) Brief report: circadian melatonin, thyroid-stimulating hormone, prolactin, and cortisol levels in serum of young adults with autism. J Autism Dev Disorder 25:641–654
Palmen SJ, van England H, Hof PR, Shmitz C (2004) Neuropathological findings in autism. Brain 127:2572–2583
Ranke MB (2003) Diagnostics of endocrine function in children and adolescents, (1st edn). Karger, Basel
Richdale AL, Prior MR (1992) Urinary cortisol circadian rhythm in a group of High functioning children with autism. J Autism Dev Disord 22:433–447
Rojas DC, Smith JA, Benkers TL, Camou SL, Reite ML, Rogers SJ (2004) Hippocampus and amygdala volume in parents of children with autistic disorder. Am J Psychiatry 161:2038–2044
Sandman CA, Barron JL, Chiez-DeMet A, DeMet EM (1991) Brief report: plasma beta endorfin and cortisol levels in autistic patients. J Autism Dev Disord 21:83–87
Shao Y, Cuccaro ML, Hauser ER, Raiford KL, Menold M, Wolpert CM, Ravan SA, Elston L, Decena K, Donnelly SL, Abramson RK, Wright HH, Delong GR, Gilbert JR, Pericak-Vance MA (2003) Fine mapping of autistic disorder to chromosome15q11-q13 by use of phenotypic subtypes. Am J Hum Genet 72:539–548
Stokstad E (2001) New hints into the biological basis of autism. Science 294:34–37
Strous RD, Golubchik P, Maayan R, Mozes T, Tuati-Werner D, Weizman A, Spivak B (2005) Lowered DHEA-S plasma levels in adult individuals with autistic disorder. Eur Neuropsychopharmacol 15:305–309
Tordjman S, Anderson GM, McBride PA, Hertzing ME, Snow ME, Hall LM, Thompson SM, Ferrari P, Cohen DJ (1997) Plasma beta-endorfin, adrenocorticotropin hormone, and cortisol in autism. J Child Psychol Psychiatry 38:705–715
Trottier G, Srivastava L, Walker CD (1999) Etiology of infantile autism: a review of recent advances in genetic and neurobiological research. J Psychiatry Neurosci 24:103–115
Vedhara K, Hyde J, Gilchrist ID, Tytherleigh M, Plummer S (2000) Acute stress, memory, attention and cortisol. Psychoneuroendocrinology 25:535–549
Veenstra-van der Weele J, Cook EH (2004) Molecular genetics of autism spectrum disorder. Mol Psychiatry 7:73–78
Wassink TH, Brzustowicz LM, Bartlett CW, Szatmari P (2004) The search of Autism disease genes. Ment Retard Dev Disabil Res Rev 10:272–283
Waterhouse L, Fein D, Modahl C (1996) Neurofunctional mehanisms in autism. Psychol Rev 103:457–489
Wolft S (2004) The history of autism. Eur Child Adolesc Psychiatry 13:201–208
Acknowledgement
We thank to all the children with autism and parents that participated in the study. Also, we are very thankful to the personnel of the «Center for Autism» in Split and Zagreb.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marinović-Ćurin, J., Marinović-Terzić, I., Bujas-Petković, Z. et al. Slower cortisol response during ACTH stimulation test in autistic children. Eur Child Adolesc Psychiatry 17, 39–43 (2008). https://doi.org/10.1007/s00787-007-0632-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-007-0632-1