Abstract
Purpose
The aim of this study was to detect the incidence of bisphosphonate-related osteonecrosis of the jaws (BRONJ) in association with osteoporosis in 2009 in the rural district of Marburg-Biedenkopf, Germany. In addition, the therapeutic regimen of dentists in this area was compared to the treatment guidelines of established international associations.
Methodology
A postal survey including 129 dental offices within the named investigation area was conducted. Additionally, the dentists were asked to contribute to this study during a course of retraining. Ultimately, 107 colleagues participated in this study.
Results
A total of 37 BRONJ cases were revealed, 37.4 % of those were linked to malignant diseases, 62.6 % to osteoporosis. Noticeably, 30.1 % of the BRONJ cases linked to osteoporosis were connected with intravenous application. In total, 62.6 % of all cases were associated with intravenous and 37.4 % with oral application. Considering the estimated number of 1.014 patients using bisphosphonates for osteoporosis treatment in Marburg-Biedenkopf in 2009, the specific incidence of BRONJ could be narrowed down to about 2.27 %. In proportion to the increasing risk potential of the three patient groups, participants conducted fewer surgical interventions themselves but tended to refer patients to colleagues.
Conclusion
This study reveals the incidence of BRONJ in association with osteoporosis as being grossly underrated so far, especially in connection with intravenous bisphosphonate treatment. The therapeutic regimen of the dentists who participated correlated with the established guidelines.
Clinical relevance
The interface between dentistry and medicine may profit from our study’s results which will help to improve interdisciplinary communication. With regard to this, we wish to contribute to an informative discussion since a main focus was to ensure colleagues in their decision making, especially in case of complications after elective dentoalveolar surgery—like implantations. Besides medical there are also economic and political aspects, i.e. the financial responsibility in case of unexpected or inevitable complications, possibly leading to BRONJ, that may become part of future discussions based on this study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since Marx first established a connection between the use of bisphosphonates for malignant bone diseases and the occurrence of necrotic bone in the oral cavity in 2003 [1], numerous studies have been published concerning the disease that soon became known as ‘bisphosphonate-related osteonecrosis of the jaws’ (BRONJ). Until now, the pathogenesis is still not fully understood, the treatment of BRONJ remains problematic and the outcome often unknown.
Although the main indication for the use of bisphosphonates is osteoporosis—in more than 90 % of all prescriptions [2]—most cases of BRONJ are reported in association with malignant diseases such as prostate or breast cancer and multiple myeloma. It is well established that the incidence of BRONJ in association with malignant diseases is much higher [3–5] than in those patients taking the drug because of osteoporosis [6–8]. Assumingly, this aberrant incidence of BRONJ derives from the significant potency of agents like Pamidronate and Zoledronate concerning bone remodelling. These are used as intravenous infusions at regular intervals in cases of malignant disease [9]. Their potency exceeds Alendronate or Etidronate, used orally in the treatment of osteoporosis, by a factor of 5 to 20 [10].
Since the oral application of bisphosphonates as regularly taken pills strictly depends on the patients’ compliance, some authors also favour an intravenous use of zoledronate or ibandronate on a 6 to 12 months’ term with this indication [11, 12]. Although latest studies reveal a potentially higher risk of BRONJ in osteoporosis patients than estimated before [13], there is just one publication dealing with intravenous bisphosphonates for osteoporosis linked to the risk of BRONJ [14]. In August 2007, the Food and Drug Administration approved the intravenous application mode of the agent in the USA [15], which might be the reason why there was only one scientific description by Grbic et al. examining the pathophysiological context. As intravenous use of nitrogen-containing bisphosphonates, especially of zoledronate, is known as important pathogenic factor for BRONJ in patients with malignant diseases [16], one aim of this study was to detect if there might also be a higher risk in patients taking bisphosphonates intravenously for osteoporosis treatment.
As dental treatment could be identified as a risk factor for BRONJ in the past, the main focus concentrated on how patients taking bisphosphonates either for malignant diseases or osteoporosis could be treated by their dentists while at the same time avoiding an increase of BRONJ. In order to adjust and modulate the therapeutic targeting of resident dentists for patients with a risk of BRONJ, several associations developed specific guidelines [17, 18].
These treatment guidelines define the prophylactic management of patients about to begin bisphosphonate treatment—like instructions for good oral hygiene and a comprehensive check-up of present dentures in order to avoid the damage of underlying mucosa. Also, invasive procedures like the removal of non-restorable teeth or implants and a consequent treatment of infection sources, like, for example, inflamed periodontal pockets are considered as prevention indications of BRONJ [17, 18].
Patients who completed or still undergo bisphosphonate treatment need a distinct management when facing dentoalveolar surgery. A prolonged perioperative prophylaxis with antibiotics, an atraumatic approach in the operation with a dismantling of bone edges and a primary covering of the lesion are understood as adequate treatment [17, 18].
Diagnostic imaging techniques like panoramic radiography, computed tomography or magnetic resonance imaging allow to estimate the extent of lesions [19]. Once stated, the disease’s conservative management has shown only little success [20]. Instead, a cautious but complete necrectomy should be followed by a wound closure with mobile, well-vascularized soft tissue cover—a procedure regarded as the international gold standard. In case of extended injury, even extraoral tissues of appropriate donor sites may be useful to gain non-taught covered lesions [21]. Both, systemic antibiotics and sufficient anaesthesia—often generalized—are essential to the treatment of BRONJ. In addition to this, the use of the low-level laser therapy [22] or a fluorescent marking of the necrotic area [23] may be indicated during the intervention [17, 18].
Despite the numerous publications about the disease, only little is known about the context awareness of resident colleagues. Attention is focused mainly on the congruence between scientifically established guidelines and the individual treatment protocols of those taking part in the study. Therefore, another aim of the present study was to identify correlations or discrepancies between the therapeutic skills of resident dentists in the investigation area and the guidelines of international dental associations such as American Association of Oral and Maxillofacial Surgeons (AAOMS) or Deutsche Gesellschaft für Zahn-, Mund- und Kieferheilkunde (DGZMK).
Material and methods
To gain valid data for the evaluation of these questions, we conducted a postal survey in 2009. As investigation area, we chose the administrative district of Marburg-Biedenkopf, a rural region with about 250,000 inhabitants in Hesse, Germany. The center of this district is the city of Marburg with a population of approximately 80,000 people. In 2009, 129 dental offices could be identified within this defined investigation area.
According to the definition of Ruggiero et al. [18], the characteristics of BRONJ were determined with exposed necrotic bone in the oral cavity persisting longer than 8 weeks, the absence of chemotherapy and a continuing or terminated therapy with bisphosphonates.
We developed a questionnaire of 14 parts which enabled us to ascertain how many dentists had dealt with cases of BRONJ during the period given. Positive answers required further details in questions 2 to 7 of the survey, for instance the indication of bisphosphonate treatment or the application mode—with an emphasis on therapeutic management. The interventions ranged from conservative management to invasive actions. Conservative therapy contained the administration of analgetics and antibiotics or the consultation of the bisphosphonate-prescribing physician. With this, a possible discontinuation of the drug application or the prescription of tube feeding can be discussed. Invasive management consisted of necrectomy and lesion cover with mucoperiosteal flaps. Finally, we selected what the therapeutic outcome was. Further information on affected patients as sex or age and about type and dose of administered bisphosphonates was not collected in order to keep the questionnaire focused and easy to answer.
Additionally, we inquired information about the resident dentists’ management of patients who were about to start bisphosphonate use on the one hand and those already facing an oral or intravenous application of the agent on the other hand in questions 8 to 10. We asked for several invasive treatments ranking from simple extractions to osteotomies, implantations and apicoectomies, where mucoperiosteal flaps are designed. In course of the last interventions, the rate of osseous injury exceeds that of extractions. Other procedures we focused on were closed periodontal curettage in contrast to open periodontal surgery. Since both help in reducing intraoral biofilms, they are a regular part of resident dentistry. For a better depiction, positive answers were encoded with ‘1’, negative with ‘0’. We did not evaluate the colleagues’ individual treatment protocols according to the named surgical methods, so discrepancies between different participants in their procedures cannot be excluded here.
For a valid review of the given answers, the participants were asked in the last four parts of the questionnaire if they specialized as orthodontists, oral or maxillofacial surgeons or periodontists, how many patients they treated within a quarter and where their office was located within the investigation area. In order to exclude duplicate answers, only one questionnaire was sent to each office. Alternatively, the form was available online for participation. Matching IP addresses excluded duplication here. Finally, 107 returned forms were included in this study, which is equivalent to a return rate of 82.9 %.
After consulting the Institute of Medical Biometry and Epidemiology at the Philipp University of Marburg for epidemiological and statistical planning, descriptive statistics were chosen for an elaboration of the received data. To extrapolate an approximate incidence of BRONJ in relation with osteoporosis within the investigation area in 2009, we used data from the German Health Report 2009 (GHR) [24], demographic statistics from the Hessian State Office for Statistics (HSOS) [25] and data from the BonEVA study [26].
Results
The rate of participation in this study was 107/129 (82.9 %). Of these, nine (8.4 %) stated that they specialized in ‘Periodontology’, one colleague each was practising as an oral or maxillofacial surgeon (0.9 % each). Twelve out of 107 dentists (11.2 %) were orthodontists, while 84/107 (78.6 %) did not name any specialization.
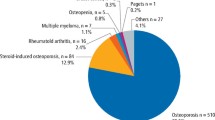
Twenty study participants (18.7 %) reported one or more cases of BRONJ in their surgeries in 2009—altogether 37 cases came to light and were defined as cohort of this study for further analysis (see Table 1). In 23/37 (62.6 %) of these cases, osteoporosis indicated a bisphosphonate treatment, 14/23 (60.9 %) of the affected patients took the drug orally and 9/23 (39.1 %) intravenously. A malignant disease was associated with 14 (37.4 %) of the 37 revealed cases of BRONJ (see Table 1).
The treatment protocol of dentists confronted with BRONJ mostly contained conservative methods. In three cases (8.1 %), a discontinuation of bisphosphonate treatment was prescribed, eight (21.6 %) were treated with antibiotics and five (13.5 %) with analgesics. Study participants consulted the physician who was prescribing bisphosphonates seven times (18.9 %) during their treatment. Nine patients (24.3 %) were recalled in short intervals, and in one case (2.7 %), a feeding tube was required (see Table 2).
Noticeably, only three participating dentists decided to carry out invasive treatment themselves when confronted with BRONJ. Necrectomy was chosen in two cases (5.4 %), while a covering of the lesion with mucoperiosteal flaps was carried out three times (7.1 %), and only one patient with malignant disease was treated with both interventions. None of these three participants described themselves as specialists. Twenty-seven (72.9 %)—i.e. the vast majority of the 37 affected patients—were referred to other dentists by the contributors to this study, and five (13.5 %) underwent inpatient treatment. In addition, all dentists who carried out invasive interventions themselves referred their patients at the same time to other dentists (see Table 2). It was striking that neither the participating oral nor the maxillofacial surgeon reported any case of BRONJ. Assumingly, the non-specialized dentists may have referred their patients directly to the university hospital of the Philipp University of Marburg. Here, further treatment is highly appropriate since the region’s maxillofacial unit enables specific appliances which lead to therapeutic sufficiency.
The treatment outcome recorded 33 cases (89.2 %) of persistent absence from symptoms. When the drug was applied orally, the rate of patients free from symptoms rose to 13/14 (92.9 %); with intravenous administration, it fell slightly to 20/23 (87 %; see Table 2).
For a calculation of the incidence of BRONJ in association with osteoporosis, we used data from the GHR and demographic data of the HSOS. The GHR estimates the lifetime prevalence of osteoporosis as 4.8–24.5 %, depending on sex and age (see Table 3). Data from the HSOS showed that approximately 10,764 people were suffering from osteoporosis during the investigation period (see Table 3). Taking into consideration that only 10 % of those patients also take bisphosphonates, we calculated a number of 1,014 (see Table 3). With regard to the 23 cases of BRONJ based on osteoporosis, reported by the study participants, we defined the specific incidence of this disease at approximately 2.27 %.
In the case of patients who had been scheduled by their dentists for oral restoration before the beginning of a bisphosphonate treatment, 58/107 participants (54.2 %) stated that they would perform extractions, 18/107 (16.8 %) would implant and 43/107 (40.2 %) would carry out osteotomies for tooth removal. When performing periodontal interventions, 59/107 dentists (55.1 %) would refrain from designing mucoperiosteal flaps, whereas 30/107 (28 %) would also choose the open approach. An apicoectomy would be performed, if necessary, by 26/107 participants (24.3 %). Twenty-three out of 107 dentists (21.5 %) would prefer a referral of their patients to colleagues, prior to bisphosphonate therapy (see Fig. 1).
In cases where the patients were on oral bisphosphonates, 27/107 (25.2 %) of the participating dental practitioners would extract destroyed teeth, 8/107 (7.5 %) would even perform osteotomies, if indicated. Three out of 107 participants (2.8 %) are likely to insert dental implants in this group of patients. Periodontal treatment would be performed by 33/107 (30.8 %) dentists without mucoperiosteal surgery, 7/107 (6.5 %) would also use the open approach. Apicoectomies would be chosen by 5/107 participants (4.7 %), while 45 of 107 dentists (42.1 %) would refer patients of this group to other colleagues (see Fig. 1).
Patients with intravenous bisphosphonate therapy would undergo extractions by 6/107 dental practitioners (5.6 %), 2/107 (1.9 %) participants would even use osteotomies. One (0.9 %) colleague would place implants in patients of this category. Periodontal treatment without mucoperiosteal flaps would be performed by 6/107 (5.6 %) study participants. Neither mucoperiosteal surgery for periodontal therapy nor apicoectomies would be performed by any of the dentists. We made the observation that most dentists—first and foremost—chose to refer affected patients to other colleagues. Seventy-nine out of 107 (73.8 %) study participants preferred this method (see Fig. 1). Summarized, for all seven screened interventions in questions 8 to 10, the cumulated results (see “Material and methods” section) tend to ‘0’ with increasing risk potential of bisphosphonate medication, though in case of referrals, the results tend towards ‘1’ (see Fig. 2).
Discussion
Primarily, the high rate of participation in this study clearly shows that BRONJ is a relevant topic for the dental practitioners within the investigation area. The fact that nearly every fifth (18.7 %) practising dentist was confronted with BRONJ in his office during 2009 reinforces this impression.
Strikingly, the category of primary diseases contributed differently to the total number of 37 reported cases, a result which deviated from our expectations. A review of all internationally published cases of BRONJ within the period from January 2003 to September 2009 shows a vast majority of 89 % to be associated with malignant diseases such as prostate or breast cancer and multiple myeloma [27]. In contrast to that publication, the majority of cases were based on osteoporosis in our study. Given the design of the study, duplicate reports of the same cases of BRONJ cannot be excluded completely. However, the results of the survey can be taken as a strong indication that the dentists—if not starting treatment themselves—would have referred their patients to specialized colleagues such as oral or maxillofacial surgeons. Therefore, the fact that these specialists did not report any case of BRONJ greatly reduces the probability of such duplications. These results and the large number of osteoporosis patients suffering from BRONJ, revealed in the present study, indicate an increased incidence of this specific disease. The calculated incidence of about 2.27 % exceeds most of the previously stated rates by far [6–8]. Irrespective of this value, the statistical relevance of our results is strongly supported by the study of Sedghizadeh et al., who even reported an incidence of 4 % in patients taking the drug orally for osteoporosis treatment [13]. Also Otto et al. published statistically reliable data about the number of patients taking bisphosphonates for osteoporosis in absence of other risk factors [28]. Both studies underline the fact that the incidence of BRONJ linked to this prominent non-malignant disease has been underestimated in earlier publications. We found, however, that nearly 40 % of the BRONJ reports were linked to intravenous drug use. Therefore, this study shows for the first time that intravenous bisphosphonate application might be a relevant risk factor for BRONJ in connection with osteoporosis treatment. Still further studies with a focus on bisphosphonate type and dose are needed to verify this thesis.
With an increasing number of osteoporosis patients being treated with intravenous bisphosphonates every 6 to 12 months in order to improve their compliance, the number of patients at risk of BRONJ is likely to rise in the near future. The present study also shows that the participants have a thorough understanding of how to deal with patients already diagnosed with BRONJ or those at risk of this disease. As the guidelines of AAOMS or DGZMK state, the treatment of an established BRONJ—or suspicious lesions—belongs in the hands of oral or maxillofacial surgeons [17, 18]. The fact that only 4/37 (10.8 %) reported cases of BRONJ were treated invasively but 27/37 (72.9 %) were referred to colleagues by the dental practitioners shows fundamental awareness of the difficulty of BRONJ management. Referring their patients to resident specialists or the university hospital, the participants also make sure that affected patients can benefit from centralized knowledge and modern skills. Eventually, they give access to new therapeutic methods which need different preconditions concerning their appliances. One of them is the promising fluorescence-guided necrectomy as described by Pautke et al. [23]. After labelling necrotic bone can get removed in total by clearly identifying its borders.
In cases of conservative management, participating dentists kept strictly to the guidelines of AAOMS or DGZMK when prescribing antibiotics or analgesics as well as consulting the patients’ own physician, in order to clarify the possibility of a discontinuation in bisphosphonate therapy [17, 18].
An invasive treatment protocol—in accordance with the official regulations—should contain both, necrectomy and cover of lesions with mucoperiosteal flaps [17, 18]. This combined approach was only performed by one colleague, which indicates a further need of information on BRONJ treatment.
A well-established consensus in international literature is that dentoalveolar surgery is a risk factor for BRONJ [29]. In particular, extractions or osteotomies are known as possible trigger factors for this disease [30]. Therefore, we attempted to assess the degree of expertise of each resident dentist as well as their awareness of the patients’ exposure to BRONJ prior to and during therapy. This examination of the dentists’ awareness of current developments was made in order to indicate the relevant risk factors of the high incidence of BRONJ stated in association with osteoporosis.
Noticeably, the willingness of the participants to carry out operations decreased in inverse proportion to the increase of complications within the three different patient groups. This observation corresponds to the current guidelines of AAOMS and DGZMK, which postulate a severe indication for such interventions [17, 18]. Consequently, only specialized or experienced dentists should perform such operations. An atraumatic approach with a non-taut cover of the alveole by mucoperiosteal flaps after extractions is seen as adequate management for reducing the risk of BRONJ [17, 18]. Specific forms of dentoalveolar surgery, such as osteotomies, are rarely performed by the participants of this study, probably because of the increased complexity of such interventions. However, according to a prevention strategy by Ferlito et al., the removal of destroyed teeth in bisphosphonate patients seems to be possible—even if osteotomies appear to be inevitable [31].
If patients take bisphosphonates orally or intravenously, participating dentists tend to refrain from all kind of interventions, especially apicoectomies and open periodontal treatment to an even greater extent than in the cases of the extractions and osteotomies stated above. With the exception of one participant who even performed these kinds of interventions in conjunction with bisphosphonate treatment, there is a common tendency to refer affected patients to specialists. This trend corresponds to the established guidelines [17, 18].
When it comes to the possibility of placing implants, the dental practitioners keep to the above stated guidelines. As there are no known contraindications in patients before bisphosphonate treatment, every fifth participant would place implants if necessary. As some studies revealed, implantations are also possible in conjunction with oral bisphosphonate use, if certain precautions are taken [32–34]. Nevertheless, some publications also report unsuccessful implantations which led to BRONJ [35, 36]. We assume that these reports may have corresponded with individual experiences which made the practising colleagues refrain from immediate action because only 2.8 % of the participants conducted implantations in this group of patients. As far as intravenous bisphosphonate use is concerned, the risk of complications in implant therapy is well documented, and according to the guideline consensus, there is a strict contraindication for dental implants in affected patients [17, 18]. Therefore, we would like to stress that the participating dentist, who declared that he would even choose implantation in this group of patients, would be acting with gross negligence—ignoring current medical insights. Since Grötz et al. proposed an algorithm dealing with implantations in bisphosphonate therapy, resident dentists can find support when choosing a suitable therapy [37].
Closed periodontal treatment is one of the most frequently performed interventions within the group of participants. A possible reason for this observation might be the fact that no preparation of mucoperiosteal flaps is necessary, so that more dentists refrain from such surgical interventions. As parodontitis is a well-known risk factor for BRONJ [20, 38], periodontal treatment helps to decrease the grade of inflammation and is therefore a core module in the prevention of this disease [18, 39].
Conclusion
We elaborated an incidence of 2.27 % for BRONJ in association with osteoporosis in the present study, which clearly shows that the risk of BRONJ in osteoporosis patients has been grossly underestimated so far. Although the majority of cases in our study were associated with oral bisphosphonate use, we demonstrated that intravenous application of these agents could be a relevant risk factor in osteoporosis treatment as well. A determined use of this application mode could increase the number of cases of BRONJ in the future. Nevertheless, further studies are needed in order to interpret the determined incidence. A re-evaluation of the risk factors may be helpful here, especially with regard to patients’ exposure to intravenous bisphosphonates.
Additionally, we found a thorough understanding of the causal relationship of BRONJ and dentoalveolar surgery of the resident dentists in the investigation area. There is a common awareness of the pathophysiology of the disease, which leads to proper treatment according to the guidelines of the established associations. The invasiveness of an operation determines whether or not it is conducted by the participants of this survey. With rising risk potential of the three patient groups—before treatment, with oral medication and under intravenous application—resident dentists refrain from performing complex surgical interventions like extractions, osteotomies, implantations or apicoectomies. All named surgical procedures are applied on patients about to begin bisphosphonate therapy, but the number tends towards 0 within the group of patients undergoing intravenous bisphosphonate treatment. However, the number of referrals to other colleagues is inversely proportional to this aspect. Our study revealed that there is some determination to continue therapy regardless of the well-known risk factors of BRONJ. Evidently, continued training in proper procedures in therapeutic management is to be recommended.
References
Marx RE (2003) Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg 61(9):1115–1117
Schwabe U, Ziegler R (2011) Osteoporosemittel. In: Arzneimittelverordnungsreport 2010. Springer, Berlin, pp 739–752
Durie BG, Katz M, Crowley J (2005) Osteonecrosis of the jaw and bisphosphonates. N Engl J Med 353(1):99–102
Dimopoulos MA et al (2006) Osteonecrosis of the jaw in patients with multiple myeloma treated with bisphosphonates: evidence of increased risk after treatment with zoledronic acid. Haematologica 91(7):968–971
Bamias A et al (2005) Osteonecrosis of the jaw in cancer after treatment with bisphosphonates: incidence and risk factors. J Clin Oncol 23(34):8580–8587
Assael LA (2009) Oral bisphosphonates as a cause of bisphosphonate-related osteonecrosis of the jaws: clinical findings, assessment of risks, and preventive strategies. J Oral Maxillofac Surg 67(5 Suppl):35–43
Felsenberg D (2006) Osteonecrosis of the jaw—a potential adverse effect of bisphosphonate treatment. Nat Clin Pract Endocrinol Metab 2(12):662–663
Mavrokokki T et al (2007) Nature and frequency of bisphosphonate-associated osteonecrosis of the jaws in Australia. J Oral Maxillofac Surg 65(3):415–423
Migliorati CA (2008) Intravenous bisphosphonate therapy may lead to osteonecrosis of the jaw in multiple myeloma, breast, and prostate cancer patients. J Evid Based Dent Pract 8(2):93–94
Marx R (2007) Oral & intravenous bisphosphonate-induced osteonecrosis of the jaws (history, prevention, and treatment). Quintessence Publishing Co, Inc., Hanover Park
Bock O, Felsenberg D (2008) Bisphosphonates in the management of postmenopausal osteoporosis—optimizing efficacy in clinical practice. Clin Interv Aging 3(2):279–297
Sewerynek E, Stuss M (2011) The role of i.v. ibandronate administration in osteoporosis therapy. Endokrynol Pol 62(1):51–60
Sedghizadeh PP et al (2009) Oral bisphosphonate use and the prevalence of osteonecrosis of the jaw: an institutional inquiry. J Am Dent Assoc 140(1):61–66
Grbic JT et al (2008) Incidence of osteonecrosis of the jaw in women with postmenopausal osteoporosis in the health outcomes and reduced incidence with zoledronic acid once yearly pivotal fracture trial. J Am Dent Assoc 139(1):32–40
US Food and Drug Administration 2011. http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseaction=Search.Overview&DrugName=BONIVA. Accessed 3 Sept 2012.
Rustemeyer J, Bremerich A (2010) Bisphosphonate-associated osteonecrosis of the jaw: what do we currently know? A survey of knowledge given in the recent literature. Clin Oral Investig 14(1):59–64
Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF) (2012) Bisphosphonat-assoziierte Kiefernekrosen. http://www.awmf.org/uploads/tx_szleitlinien/007-091l_S3_Bisphosphonat-assoziierte_Kiefernekrose_2012-04.pdf. Accessed 3 Sept 2012.
Ruggiero SL et al (2009) American Association of Oral and Maxillofacial Surgeons position paper on bisphosphonate-related osteonecrosis of the jaws—2009 update. J Oral Maxillofac Surg 67(5 Suppl):2–12
Stockmann P et al (2010) Panoramic radiograph, computed tomography or magnetic resonance imaging. Which imaging technique should be preferred in bisphosphonate-associated osteonecrosis of the jaw? A prospective clinical study. Clin Oral Investig 14(3):311–317
Hoff AO et al (2008) Frequency and risk factors associated with osteonecrosis of the jaw in cancer patients treated with intravenous bisphosphonates. J Bone Miner Res 23(6):826–836
Lemound J et al (2012) Bisphosphonate-associated osteonecrosis of the mandible: reliable soft tissue reconstruction using a local myofascial flap. Clin Oral Investig 16(4):1143–1152
Vescovi P et al (2010) Surgical approach with Er:YAG laser on osteonecrosis of the jaws (ONJ) in patients under bisphosphonate therapy (BPT). Lasers Med Sci 25(1):101–113
Pautke C et al (2011) Fluorescence-guided bone resection in bisphosphonate-related osteonecrosis of the jaws: first clinical results of a prospective pilot study. J Oral Maxillofac Surg 69(1):84–91
Gesundheisberichterstattung des Bundes (2009) Prävalenz von Osteoporose. http://www.gbe-bund.de/gbe10/owards.prc_show_pdf?p_id=13742&p_sprache=d&p_uid=&p_aid=&p_lfd_nr=1. Accessed 3 Sept 2012
Hessisches Landesamt für Statistik (2009) Bevölkerung Hessens nach Altersgruppen und Geschlecht. http://www.statistik-hessen.de/themenauswahl/bevoelkerung-gebiet/landesdaten/bevoelkerung-allgemein/bevoelkerung-nach-altersgruppen-und-geschlecht/index.html. Accessed 3 Sept 2012
Haussler B et al (2007) Epidemiology, treatment and costs of osteoporosis in Germany—the BoneEVA Study. Osteoporos Int 18(1):77–84
Filleul O, Crompot E, Saussez S (2010) Bisphosphonate-induced osteonecrosis of the jaw: a review of 2,400 patient cases. J Cancer Res Clin Oncol 136(8):1117–1124
Otto S et al (2011) Osteoporosis and bisphosphonates-related osteonecrosis of the jaw: not just a sporadic coincidence—a multi-centre study. J Craniomaxillofac Surg 39(4):272–277
Marx RE et al (2005) Bisphosphonate-induced exposed bone (osteonecrosis/osteopetrosis) of the jaws: risk factors, recognition, prevention, and treatment. J Oral Maxillofac Surg 63(11):1567–1575
Hoff AO et al (2011) Epidemiology and risk factors for osteonecrosis of the jaw in cancer patients. Ann N Y Acad Sci 1218:47–54
Ferlito S, Liardo C, Puzzo S (2010) Dental extractions in patient treated with intravenous bisphosphonates and risk of osteonecrosis of jaws: presentation of a preventive protocol and case series. Minerva Stomatol 59(11–12):593–601
Ferlito S, Liardo C, Puzzo S (2011) Bisphosponates and dental implants: a case report and a brief review of literature. Minerva Stomatol 60(1–2):75–81
Madrid C, Sanz M (2009) What impact do systemically administrated bisphosphonates have on oral implant therapy? A systematic review. Clin Oral Implants Res 20(Suppl 4):87–95
Mellado-Valero A et al (2010) Implant treatment in patients with osteoporosis. Med Oral Patol Oral Cir Bucal 15(1):e52–e57
Favia G et al (2011) Osteonecrosis of the posterior mandible after implant insertion: a clinical and histological case report. Clin Implant Dent Relat Res 13(1):58–63
Shin EY et al (2010) Implant failure associated with oral bisphosphonate-related osteonecrosis of the jaw. J Periodontal Implant Sci 40(2):90–95
Groetz K et al (2010) Bei welchen Bisphosphonat-Patienten darf ich eigentlich implantieren? Ein systematisches Review. Z Zahnärztl Impl 26(2):153–161
Jadu F et al (2007) A retrospective study assessing the incidence, risk factors and comorbidities of pamidronate-related necrosis of the jaws in multiple myeloma patients. Ann Oncol 18(12):2015–2019
Borromeo GL et al (2011) A review of the clinical implications of bisphosphonates in dentistry. Aust Dent J 56(1):2–9
Conflict of interest
The authors declare no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hansen, P.J., Knitschke, M., Draenert, F.G. et al. Incidence of bisphosphonate-related osteonecrosis of the jaws (BRONJ) in patients taking bisphosphonates for osteoporosis treatment—a grossly underestimated risk?. Clin Oral Invest 17, 1829–1837 (2013). https://doi.org/10.1007/s00784-012-0873-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-012-0873-3