Abstract
Collagen IV accumulation is characteristic of diabetic angiopathy. To test the possible contribution of GH, we studied its effects on collagen IV production by human umbilical vein endothelial cells at 5.5 and 16.7 mmol/l glucose. GH (100 ng/ml) markedly increased collagen IV level in the culture supernatant and in the insoluble extracellular matrix and cell fraction at both glucose concentrations. This stimulating effect of GH was additional to that of high glucose. It was more pronounced on collagen IV than on total protein synthesis. GH increased free latent gelatinase activity slightly at normal and markedly at high glucose. Using GF109203X, a PKC inhibitor, we observed that high glucose, but not GH, activated PKC. These two factors stimulating collagen IV production appear to work through different pathways, favoring an additivity of their effects. This supports the contribution of high plasma GH in diabetic vascular basement membrane thickening.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Irregular thickening of the capillary basement membranes (BM) is characteristic of diabetic microangiopathy. It is associated with an accumulation of type IV collagen (Gubler et al. 1988; Shimomura and Spiro 1987). Retinal (Hayakawa et al. 1990) and glomerular (Danne et al. 1993) capillary endothelial cells (EC) synthesize type IV collagen. An increase in type IV collagen content of the aortic wall was also reported in diabetic macroangiopathy (Rasmussen and Ledet 1993).
Chronic hyperglycemia is recognized as a major factor for the development of microangiopathy. However, good glycemic control does not always prevent progression of microangiopathy and the severity of this progression varies among individuals (Ramsay et al. 1988; The Kroc Collaborative Study Group 1988). This suggests that other factors might be involved in the pathogenesis of diabetic angiopathy. Human growth hormone (GH) might be one of these factors.
The improvement of a severe proliferative retinopathy in a diabetic woman following an accidental hypophysectomy was reported by Poulsen (1953). A link between GH and diabetic retinopathy was suggested by Lundbaek et al. (1970): they insisted on raised plasma levels in juvenile diabetes and on the persistence of abnormal GH response to exercise even in well-controlled diabetics. Hypophysectomy could reverse or slow down the progression of proliferative retinopathy (Kohner et al. 1976). This relationship was often suggested but has been the subject of controversy (Ballintine et al. 1981; Rabin et al. 1984). Rymaszewski et al. (1991) showed that GH stimulated proliferation of human retinal microvascular EC in vitro, but only in the presence of serum. The effect of GH on EC is not only proliferative. Ledet and Heickendorff (1985) reported increased synthesis and decreased degradation of arterial BM-like material secreted in rabbit aortic myomedial cell cultures, when GH was added.
The effects of GH on cell metabolism are known to be mediated principally by tyrosine kinase activation, through JAK2 associated to its receptor, but also by other signaling pathways (Brooks and Waters 2010; Lanning and Carter-Su 2006). Protein kinase C (PKC) activation appeared to be involved in GH action on isolated rat hepatocytes, adipocytes, proadipocytes and lymphoma cells (Clarkson et al. 1995; Gaur et al. 1996; Horn et al. 1994; Nivet et al. 1993; Smal and De Meyts 1989).
We had previously shown that the increase in type IV collagen production by human EC cultured in high glucose was linked to PKC activation since it was prevented by a specific PKC inhibitor (PKCI) (Grigorova-Borsos et al. 1996).
The aim of this study was to answer the following questions:
-
does GH act on type IV collagen production by human EC in culture?
-
does high glucose concentration modify this possible effect of GH?
-
is an activation of PKC implicated in the eventual effects of GH, as it was shown to be implicated in the effects of high glucose (Grigorova-Borsos et al. 1996)? If it is not the case, this would allow additivity of GH and high glucose effects mediated by different pathways.
We used recombinant hGH and human umbilical vein EC (HUVEC), known to secrete type IV collagen into a BM-like matrix in culture (Jaffe et al. 1976; Kramer et al. 1985). We measured the soluble type IV collagen secreted into the culture supernatant and the insoluble type IV collagen accumulated in the extracellular matrix and cell fraction. For comparison we evaluated total protein synthesis by determining proline incorporation in the proteins of the supernatant and in the matrix and cell fractions. Besides, we studied gelatinase activity (GA) in the culture supernatant. We had shown indeed by gelatin zymography that HUVEC produced almost exclusively a 72 kDa gelatinase (Grigorova-Borsos et al. 1996), which is known to degrade type IV collagen (Woessner 1991). In order to compare the contribution of PKC activation in the effects of high glucose and GH on type IV collagen metabolism in HUVEC, we treated EC by a specific PKC inhibitor (PKCI), bisindolylmaleimide (GF109203X). This PKCI, which targets the ATP-binding site of the catalytic domain, was reported to equally inhibit the four PKC isotypes tested—α, β1, β2 and γ—in vitro, displaying no activity against protein kinases other than PKC (Toullec et al. 1991); it was later shown to inhibit PKC∂ (Wu et al. 2010). HUVEC are known to express α, β1, β2, ε and ζ PKC isotypes (Mattila et al. 1994) and also ∂ isoform (Holmes et al. 2010). Activation of PKC isoforms α, β1, β2 and ∂ has been implicated in endothelial cell dysfunction in diabetes through an increased synthesis of their activator diacylglycerol consecutive to inhibition of GAPDH dehydrogenase (Du et al. 2003; Geraldes and King 2010).
Materials and methods
HUVEC cultures
HUVEC were isolated and cultured by the procedure described by Jaffe et al. (1976) for the study of BM synthesis, slightly modified (Bakillah et al. 1996). All experiments were performed in primary cultures, after the beginning of confluence which had been achieved in 199 culture medium (M199, 1 ml/well) with 10% human serum (HS).
Effectors and treatment
The effectors were added at the beginning of confluence for a 3-day treatment either (i) in 1 ml serum-free M199 (containing 5.5 mmol/l glucose) supplemented with 3 g/l lactalbumin hydrolysate (Gibco, Uxbridge, England) and 35 mg/l human serum albumin (Sigma, St Louis, Missouri) as serum substitute, or (ii) in 1 ml M199 with 5% HS. These two culture media were compared since we had observed that the EC produce more type IV collagen in serum-free medium than in medium with serum, at the standard 5.5 mmol/l or the high 16.7 mmol/l glucose concentration (Grigorova-Borsos et al. 1996). Besides, when HS was used, the amount of immunoreactive type IV collagen present in serum had to be subtracted in the ELISA (Grigorova-Borsos et al. 1996). GA could only be measured in serum-free medium because of the inhibitors present in HS.
d-Glucose (Sigma) was added to the culture medium for the high glucose condition, at a final level of 16.7 mmol/l.
Recombinant human growth hormone (Maxomat, CHOAY, France) was dissolved in distilled water (2 IU/ml or 0.74 mg/ml containing 22 mg/ml alanine). Then it was diluted in the culture medium to obtain 1, 10 and 100 ng/ml final hGH concentrations. GH solutions (20 μl) were added daily to the culture supernatants during 3 days.
GF109203X (a gift from J. Kirilovsky, Glaxo, France) was first dissolved in dimethyl sulfoxide at 5 mmol/l, then diluted in M199. The final 2.5 μmol/l concentration used was chosen as the most efficient on type IV collagen overproduction at high glucose concentration, with minimal cytotoxicity (Grigorova-Borsos et al. 1996).
Extracellular and cellular fractions
Culture supernatants were collected at 3-day intervals and frozen at −20°C until used. The cells were counted with a Malassez hematocytometer. Collagen was quantitatively extracted from the insoluble extracellular matrix and cell fraction according to Kramer et al. (1985) with slight modifications (Bakillah et al. 1996).
Type IV collagen assay
Type IV collagen was determined by a two-step ELISA developed in our laboratory (Bakillah et al. 1996). When 5% HS was used in the culture medium, the amount of immunoreactive type IV collagen present in serum was subtracted.
[14C]Proline incorporation into the total proteins synthesized by EC
[14C]Proline incorporation into the total proteins present either in the culture supernatant or in the insoluble extracellular matrix and cell protein fraction was measured after 3-day incubation with 0.2 μCi/ml [14C]proline, as previously described (Grigorova-Borsos et al. 1996).
Gelatinase activity
GA was determined in serum-free M199 according to Grigorova-Borsos et al. (1996) with and without previous dissociation from tissue inhibitors of matrix proteinases (TIMPs), evaluating either total or only free GA. Immediately active gelatinase was measured without APMA activation. Latent GA was disclosed only after APMA activation.
Expression of results and statistical analysis
The values presented are generally means of four culture wells ± SEM. Results concerning type IV collagen or total proteins are generally expressed per well (or ml), corresponding to the same number of cells present at the beginning of the treatment. When the number of cells varied after treatment, it was also of interest to evaluate the protein produced per 104 cells present at the end of the treatment. Comparison of two means was done by the Student t test or the non-parametric Mann–Whitney’s U test. Multiple comparisons were carried out by one-way, two-way or three-way analysis of variance, followed by the Bonferroni Student’s t test using the residual variance. The significance threshold retained was p < 0.05.
Results
GH and glucose levels in the culture medium
After 3 days of culture in a serum-free medium, the GH concentration measured by immunoradiometric assay was found to be 77, 8 and 0.68 ng/ml in media where 100, 10 and 1 ng/ml had been added, respectively, every day. In the experiments where HS was added to culture medium, it contained a negligible amount of GH (0.1 ng/ml, before dilution).
After 3 days of culture in a serum-free medium, the glucose concentrations measured by the glucose oxidase procedure were found to be 16.5 ± 0.5 and 3.9 ± 0.1 mmol/l for the 16.7 and 5.5 mmol/l starting levels, respectively.
Effect of GH and glucose on HUVEC number: dose–response study
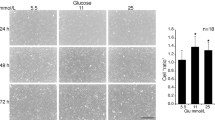
Figure 1a shows the effect of increasing concentrations of GH on the number of cells at day 11 of culture, without serum (Experiment I). At 5.5 mmol/l glucose, the cell number increased moderately with GH concentration: a significant regression of cell number to log (GH) was observed (r = 0.51, p = 0.03); at 100 ng/ml GH, the increase was of 13.5% (p = 0.05). At 16.7 mmol/l glucose, no significant effect of GH on the cell number was observed (r = 0.34; p = 0.20).
Effect of different concentrations of hGH on HUVEC proliferation (a) and on type IV collagen secreted in the culture supernatant (b) and accumulated in the matrix and cells fraction (c), studied in Experiment I. GH was added daily from day 8 to 11 to the culture supernatant containing either 5.5 mmol/l (white squares) or 16.7 mmol/l (black squares) glucose, without HS. Each point represents the mean of 4 wells ± SEM. Statistical comparisons were carried out by two-way analysis of variance. Effect of GH (100 ng/ml) at 5.5 mmol/l glucose: p < 0.05 for a; p < 0.001 for b and c. Effect of GH (100 ng/ml) at 16.7 mmol/l glucose: p < 0.001 for b and c. Effect of glucose (between 5.5 and 16.7 mmol/l glucose) for a p = 0.08, p = 0.06, p = 0.06 and p < 0.05 at 0, 1, 10 and 100 ng/ml GH, respectively; for b p = 0.10 in the absence of GH, p < 0.01 at 100 ng/ml GH; for c p = 0.10 in the absence of GH, p < 0.001 at 100 ng/ml GH
High glucose decreased moderately the number of cells at the different GH concentrations at day 11 without serum: the decreases were, respectively, of −9% (p < 0.30), −15% (p < 0.05), −15% (p < 0.05) and −17% (p < 0.02) at 0, 1, 10 and 100 ng/ml GH, respectively.
Effect of GH and glucose on type IV collagen production: dose–response study
In the absence of serum, GH increased the amount of type IV collagen secreted into the culture medium or accumulated in the extracellular matrix and cells, only at the highest concentration of 100 ng/ml (Fig. 1b, c). At the standard 5.5 mmol/l glucose concentration, GH increased type IV collagen produced per well by 51% (p < 0.001) and 112% (p < 0.001) in the culture supernatant and in the matrix and cell fraction, respectively. At the high 16.7 mmol/l glucose concentration, GH increased type IV collagen produced per well by 61% (p < 0.001) and 148% (p < 0.001), respectively. If collagen production was expressed per 104 cells, the increases were +33% (p < 0.05) and +87% (p < 0.001), in the culture supernatant and in the matrix and cell fraction, respectively, at 5.5 mmol/l glucose; they reached +60% (p < 0.001) and +135% (p < 0.001) in the culture supernatant and in the matrix and cell fraction, respectively, at 16.7 mmol/l glucose.
In the absence of GH, high glucose increased moderately but insignificantly the amount of type IV collagen secreted into the culture medium or accumulated in the extracellular matrix and cells (+18%, p = 0.10 and +32%, p = 0.10, respectively, when expressed per well; +29%, p < 0.10 and +44%, p < 0.20, respectively, when expressed per 104 cells. In contrast, at 100 ng/ml of GH, high glucose increased very significantly the amount of type IV collagen measured per well in the culture supernatant (+26%, p < 0.01) and in the matrix and cell fraction (+ 54%, p < 0.001), also when expressed per 104 cells (+49%, p < 0.001 and +82%, p < 0.001, respectively).
Thus the effects of GH and glucose appeared to be additional. Further experiments were conducted only with the effective 100 ng/ml GH concentration. In Experiment II (Fig. 2) carried out without serum, in the presence and absence of 100 ng/ml GH, the additivity of high glucose and GH effects was confirmed particularly clearly when expressing these effects as absolute changes in total type IV collagen content (culture medium supernatant plus extracellular matrix and cells, see Fig. 2c): pure high glucose effect (in the absence of GH) was an increase of 31 ± 7 ng/well (p < 0.001), pure GH effect (in the absence of glucose) was +30 ± 6 ng/well (p < 0.001), combined high glucose and GH effect was +54 ± 1 ng/well (p < 0;02 vs. high glucose alone; p < 0;02 vs. GH alone).
Effects of hGH (100 ng/ml), glucose and GF190203X, a PKCI, on type IV collagen levels in the supernatant (a) or in the matrix and cell fraction (b) of HUVEC cultures in Experiment II. The change in total type IV collagen content presented in (c) has been calculated by adding individual a and b values, then subtracting the control total content found at 5.5 mM glucose, in the absence of GH and PKCI. The cells were incubated for 3 days (day 8–11) in the presence of 5.5 or 16.7 mmol/1 glucose, without (white rectangles) or with (hatched rectangles) GH 100 ng/ml, in the absence of serum. Each result is the mean of 4 wells ± SEM. Statistical comparisons of the changes in total type IV collagen content were carried out by three-way analysis of variance. Effect of GH: F = 33.3, p < 0.001; comparisons between GH-treated and untreated cells: at 5.5 mmol/1 glucose, p < 0.005 and p = 0.05 in the absence or in the presence of PKCI, respectively; at 16.7 mmol/1 glucose: p < 0.02 and p < 0.005 in the absence or in the presence of PKCI, respectively. Effect of glucose: F = 13.4, p = 0.001; comparisons between 5.5 and 16.7 mmol/1 glucose: p < 0.001 without GH and PKCI, p < 0.02 with GH and without PKCI. Effect of the PKCI: F = 5.68, p = 0.025; comparisons between PKCI-treated and untreated cells: NS at 5.5 mol/1 glucose, p < 0.01 at 16.7 mmol/1 glucose without GH, p < 0.05 at 16.7 mmol/1 glucose with GH. Significant interaction was found only between PKCI and glucose (F = 7.68, p = 0.01). The numbers of cells in this experiment were: 11.72 ± 0.43, 12.32 ± 0.69, 11.62 ± 1.00, and 11.57 ± 0.75 × 104/well for 5.5 mmol/1 glucose, 5.5 mmol/1 glucose + GH, 5.5 mmol/1 glucose + PKCI and 5.5 mmol/1 glucose + GH + PKCI, respectively; 9.90 ± 0.73, 10.20 ± 0.69, 10.62 ± 0.48 and 9.55 ± 0.67 × 104/well for 16.7 mmol/1 glucose, 16.7 mmol/1 glucose + GH, 16.7 mmol/1 glucose + PKCI and 16.7 mmol/1 glucose + GH + PKCI, respectively, with a significant glucose effect (F = 12, p < 0.002), without GH or PKCI effects
Effect of GH on total protein synthesis: dose–response study
This study was carried out during Experiment I, in the absence of serum (Table 1).
Proline incorporation into the total proteins present in the culture supernatant
At 5.5 mmol/l glucose, the different GH concentrations tested had no effect on proline incorporation into the total proteins present in the culture supernatant. At 16.7 mmol/l glucose, GH increased proline incorporation into the total proteins present in the culture supernatant only at the highest level of 100 ng/ml: +20% (p < 0.001) when expressed par dpm/well or +14% p < 0.001) when expressed per dpm/104 cells.
Proline incorporation into the total proteins of the insoluble matrix and cell fraction
At 5.5 mmol/l glucose, a very significant dose effect of GH was observed for proline incorporation into the total proteins of the insoluble matrix and cell fraction: the increases in proline incorporation were +39% (p < 0.05), +57% (p < 0.01) and +71% (p < 0.001) at 1, 10 and 100 ng/ml, respectively, when expressed in dpm/well; +36% (p = 0.05), +42% (p < 0.05) and +51% (p < 0.02) at 1, 10 and 100 ng/ml, respectively, when expressed in dpm/104 cells. At 16.7 mmol/l glucose, without GH, a marked high glucose-induced increase in proline incorporation was observed: +53% (p < 0.01) when expressed in dpm/well or +67% (p < 0.005) when expressed in dpm/104 cells. At 16.7 mmol/l glucose, an effect of the hormone was found only at 100 ng/ml: +25% (p < 0.05) when expressed in dpm/well, +18% (p = 0.10) when expressed in dpm/104 cells.
Proline incorporation into the total proteins present in the whole culture well (supernatant plus insoluble matrix and cell fraction)
Effect of GH Significant increases in total protein synthesis were observed only at 100 ng/ml GH at 5.5 mmol/l glucose (+20.4%, p < 0.01) and 16.7 mmol/l glucose (+22.0%, p < 0.001) when expressed in dpm/well.
High glucose effect In the absence of GH, an insignificant increase of 9.0% in dpm/well was observed; at 100 ng/ml GH, a significant increase of 10.4% was noted (p < 0.05), showing an additivity of 100 ng/ml GH and high glucose effects of 30.8% (10.4% + 20.4%).
Influence of human serum on the effects of GH
Normal HS contains immunoreactive type IV collagen antigens. In the study presented here, M199 with 5% HS contained 26 ng/ml type IV collagen. This amount was subtracted from the levels measured by ELISA in the culture supernatants with HS. Besides, HS brought a negligible amount of GH (0.1 ng/ml before dilution). In order to determine whether the presence of serum influences the effects of GH, the cells were grown in the presence or absence of 5% HS, with or without 100 ng/ml of GH, at 5.5 mmol/l glucose, from day 8 to day 11. This study was simultaneous and complementary to Experiment I described in Fig. 1; Tables 1, 2.
The numbers of cells were increased in the presence of HS, with or without GH, when compared with the respective control wells lacking serum (p < 0.001) with or without GH. No stimulatory effect of GH on cell proliferation was observed in the presence of HS (Table 2).
No significant stimulatory effect of GH on type IV collagen production was observed in the presence of HS. In contrast, in the absence of HS, type IV collagen levels were increased in the culture medium and in the matrix and cells, more markedly in the presence of GH (p always <0.005) than in the absence of GH (p only significant when expressed per 104 cells.
No effect of GH on total protein synthesis was seen in the presence of HS.
Effect of GH and glucose on gelatinase activity of the culture supernatant
No effect of GH was observed on total gelatinase activity (after dissociation of TIMPs and APMA activation), either at 5.5 or 16.7 mmol/l glucose.
In contrast, the hormone increased the free latent GA (measured before dissociation of TIMPs and after APMA activation), slightly at 5.5 mmol/l glucose (+25%, p = 0.10) but very markedly at 16.7 mmol/l glucose (+108%, p < 0.001) (Fig. 3). In the absence of GH, the free latent GA was moderately diminished by high glucose alone (from 1.8 to 1.0 U/ml; p = 0.07), in agreement with our previous results which had shown a decrease of free GA from 1.8 to 1.2 U/ml (Grigorova-Borsos et al. 1996).
Effect of hGH (100 ng/ml), glucose and the PKCI GF190203X on free gelatinase activity measured after APMA activation, in the supernatant of HUVEC cultures in Experiment II. The cells were incubated for 3 days (day 8–11) in the presence of 5.5 or 16.7 mmol/l glucose, without or with GH 100 ng/ml, in the absence of serum. Each result is the mean of 4 wells ± SEM. Statistical comparisons were carried out by three-way analysis of variance. Effect of GH at 16.7 mmol/l glucose without PKCI: p < 0.001. Effects of glucose (between 5.5 and 16.7 mmol/l glucose): p = 0.07 without GH and PKCI; p < 0.001 with GH and PKCI. Effects of the PKCI: p < 0.001 at 5.5 mol/l glucose with GH and p < 0.001 at 16.7 mmol/l glucose without GH. The numbers of cells are the same as in Fig. 2
Before activation by APMA, immediately active free GA was very low, barely detectable as previously, and was not modified by glucose or GH.
Effect of GF109203X on type IV collagen production in the presence of GH and glucose
In the absence of GH, PKCI had no effect on type IV collagen level in the culture supernatant or in the matrix and cell fraction at 5.5 mmol/l glucose, in Experiment II (Fig. 2); it reduced the total increase in total type IV collagen production induced by high glucose from +31.5 to +5.8 ng per well (p < 0.01).
In the presence of GH and 5.5 mmol/l glucose, the PKCI did not significantly correct the increase in total type IV collagen production induced by the hormone (+25.0 vs. +29.9 ng/well). In the presence of GH and 16.7 mmol/l glucose, there was only a partial effect of the PKCI on the additive increase in total type IV collagen production, which was diminished from +52.5 to +34.9 ng/well (p = 0.07).
For what concerns the number of cells, a significant decrease by glucose alone was observed and a slight but insignificant increase by GH alone; no significant effect of PKCI was found (see legend of Fig. 2).
Effect of PKCI on free latent gelatinase activity in the presence of GH and glucose
In the absence of GH, the PKCI corrected the decrease in free latent GA induced by high glucose (p < 0.001) (Fig. 3). In the presence of GH and 5.5 mmol/l glucose, the PKCI diminished free latent GA (p < 0.001), in contrast with the slight stimulatory hormone effect. In the presence of GH and 16.7 mmol/l glucose, there was no effect of the PKCI on the increase in free latent GA induced by GH, but the final free latent GA was already normalized by GH and no further effect of the inhibitor was found.
Discussion
We investigated the effect of GH on the metabolism of type IV collagen synthesized by HUVEC in culture for a 3-day period at the beginning of confluence. GH at 100 ng/ml markedly stimulated type IV collagen production by cells cultured in serum-free medium containing either normal or high glucose concentrations. The stimulatory effects of GH and high glucose on type IV collagen production appeared additional. GH increased type IV collagen level in both culture supernatant fraction and matrix and cell fraction. GH stimulatory effect was more pronounced on type IV collagen production than on total protein synthesis by HUVEC in culture (+51 vs. +3% increase per well in the supernatant; +112 vs. +71% in the matrix and cell fraction, at 5.5 mmol/l glucose; besides at 16.7 mmol/l glucose, the increase was of 61 vs. 20% per well in the supernatant; 148 vs. 25% in the matrix and cell fraction). Free latent GA, unlinked to TIMPs, was increased by GH in the presence of high glucose. This was either linked to the general stimulation of protein synthesis, or to a secondary reaction to the accumulation of type IV collagen production. Besides, total latent GA was not modified. This suggests a lower stimulation by GH of TIMP production than on collagenase production.
A moderate proliferative effect of GH at 100 ng/ml was observed on HUVEC in a serum-free medium at 5.5 mmol/l glucose concentration on day 11 in Experiment I (+13.5%, Fig. 1), whereas in the presence of 5% serum, no significant proliferative effect of GH was observed (+4%, Table 2). The slight proliferative effect of GH observed on HUVEC in a serum-free medium may have been disclosed under the limiting condition related to the absence of serum. Rymaszewski et al. (1991) found no proliferative effect of GH, added to concentrations as high as 200 ng/ml, on HUVEC cultured either with or without serum, but observed a proliferative effect on human retinal EC with serum only. The small variations in GH proliferative effect could depend on the cell culture confluence degree (Ohlsson et al. 1992; Rymaszewski et al. 1991). The subconfluent stage of our cultures could have favored some detachment of cells while deprived of serum, thus allowing the GH proliferative effect on the remaining layer. In serum-free medium cultures with 16.7 mmol/l glucose, GH had no proliferative effect on HUVEC, as high glucose antiproliferative effect probably became predominating.
The GH concentration in the culture medium effective in markedly stimulating type IV collagen synthesis by HUVEC was 100 ng/ml. The hormone was added every 24 h. This period corresponds approximately to the half-life of recombinant GH deduced from the hormone levels that we found in vitro after 3 days of culture (see “Results”), whereas the plasmatic half-life of recombinant hGH, Maxomat, in vivo is much shorter, 2.9 h (Vidal 2009). The absence of significant effect at 1 or 10 ng GH/ml and the demonstration of a significant effect at 100 ng GH/ml are suggestive of a contribution of GH in type IV collagen synthesis in diabetic angiopathy and apparently not in normal state. Indeed it has been reported that maximal diurnal GH peaks varied between 13 and 18 ng/ml in normal subjects, between 25 and 43 ng/ml in type 1 diabetic patients (Hansen and Johansen 1970). After 20 min exercise, GH level did not vary in normal subjects, whereas it increased to 45.9 ± 10.1 (M ± SEM) with an upper range of 82.8 ng/ml (M + 2SD) in newly diagnosed juvenile diabetic patients (Hansen 1970). In another study, after 25-min exercise, GH level reached 22 ± 7 ng/ml in patients with type 1 diabetes of short duration and without retinopathy, versus 44 ± 8, with an upper range of 83 ng/ml, in patients with type 1 diabetes of short duration associated with retinopathy and autonomic neuropathy (Sundkvist et al. 1984).
Whether human serum should be included in culture media in order to show a metabolic effect of GH was a matter of controversy (Rymaszewski et al. 1991). Therefore, we first chose to carry out HUVEC cultures with and without serum. No effect of GH was observed either on type IV collagen and total protein synthesis or on HUVEC proliferation in culture medium with 5% HS, at 5.5 mmol/l glucose. This lack of GH effect might be due to the presence of serum components which could interact with GH, its receptor or the transduction pathway signals within cells. In culture medium without serum and without GH, serum deprivation per se already resulted in an increase in type IV collagen synthesis by HUVEC. The lack of serum factors regulating collagen metabolism could be an explanation: for instance, soluble immunoreactive type IV collagen antigens present in serum or other circulating matrix antigens, might inhibit collagen synthesis by binding to integrins. Indeed, it has been shown that cells grown on insoluble extracellular matrix secrete less collagen than cells grown on plastic (Tseng et al. 1983). On the other hand, type IV collagen-soluble fragments from the NC1 domain of the alpha 3 (IV) chain have been reported to possess an inhibitory potential on another cell metabolic activity, namely 92 kDa gelatinase release by polymorphonuclear leukocytes in culture (Monboisse et al. 1994). These different considerations incited us to use serum-free medium in our main experiments.
Autophagy has been described in cells maintained in serum-deficient media (Motyl et al. 2007). It is possible that some degree of autophagy existed here in the endothelial cells when cultured in normal glucose without serum during 3 days after the beginning of confluence; however, we found that type IV collagen production was higher in normal glucose without serum than with 5% serum. In fact, serum substitute was added, containing 3 g/l lactalbumin hydrolysate and 35 mg/l human serum albumin; the free amino acid content of lactalbumin hydrolysate corresponds roughly to the quantity of amino acids of the proteins present in 5% serum, but the substitute lacks the various serum hormones and growth factors. High glucose should inhibit autophagy by supplying the cells with more energetic substrates. GH was reported to inhibit autophagy via mTOR (Motyl et al. 2007).
We showed that GH had its own stimulating effect on type IV collagen synthesis by HUVEC at 100 ng/ml in the serum-free culture medium at 5.5 or 16.7 mmol/l glucose; this effect was in addition to the stimulating effect on type IV collagen synthesis resulting from the lack of serum and from the high glucose concentration. A stimulating effect of GH on type I procollagen metabolism has been reported in cultures of rabbit aortic myomedial cells (Ledet and Vuust 1980). These results and ours, obtained in vitro, suggest that GH can influence the metabolism of different types of vascular collagens. Several factors might then modulate this role of GH: particularly the GH receptor number influenced by high GH and glucose levels. Nilsson et al. (1990) showed that in rat epiphyseal chondrocyte cultures, the synthesis of GH receptors was increased by GH, in a time and dose-dependent way. In rat hepatocyte cultures, with a glucose-poor medium, Niimi et al. (1991) observed a decrease in the number of GH receptors; this effect disappeared when glucose was added. The marked effect of GH on type IV collagen production that we observed at 16.7 mmol/l glucose could be explained by an increase in the number of GH receptors in the presence of high levels of GH and glucose. The stimulating effect of high GH level on type IV collagen metabolism of EC could contribute in the thickening of capillary BM or aortic wall and in the increase in serum levels of type IV collagen or 7S collagen observed in diabetic patients (Hogemann et al. 1986; Matsumoto et al. 1990).
Concerning the role of PKC activation in the alterations of type IV collagen metabolism induced by GH or glucose, we observed that PKCI GF109203X reduced the increase in total type IV collagen induced by high glucose, but not that induced by GH. Our results suggest that high glucose, but not GH, activates the PKC pathway (Table 3).
In high glucose alone PKC activation may be explained by increased formation of diacylglycerol. PKC signaling may activate transcription of genes through phosphorylation of transcription regulatory proteins such as Regulator of Calcineurin 1.4 which controls nuclear factor NFAT (in endothelial cells, Holmes et al. 2010) or Activation Transcription Factor-2 which controls c-jun (in F9 cells, Yamasaki et al. 2009) or possibly Special AT-rich Binding protein 1 (in T cells, Kumar et al. 2006).
GH in normal glucose has been reported to increase PKC activity in hepatocytes or adipocytes, but it appeared here inactive on PKC in HUVEC. GH in normal glucose induces in HUVEC only PKCI-insensitive signaling which must implicate GHR. The GHR signaling is known to implicate essentially the classical JAK2 Tyr kinase-dependent STAT pathway (Brooks and Waters 2010). The GHR JAK2 Tyr kinase-independent signaling—which includes Src/ERK but also PKC (Brooks and Waters 2010)—seems not to be implicated.
Combination of high glucose and GH induces an overproduction of collagen by two additive mechanisms shown here: PKC activation and PKCI-insensitive signaling.
For what concerns GA, only total free GA was increased by GH in vitro. Thus GH does not increase type IV collagen production by lowering its degradation. In GH-deficient patients, however, GH replacement was recently reported to lower plasma 92 kDA gelatinase but to a lesser extent 72 kDA gelatinase (Randeva et al. 2004); the latter is the gelatinase predominating in EC supernatant (Grigorova-Borsos et al. 1996).
The role of GH in diabetic glomerulopathy has been suggested by the study of bGH or bGH antagonist transgenic mice treated with streptozotocin (Chen et al. 1995). The diabetic bGH transgenic mice had much more severe glomerulosclerosis than nontransgenic diabetic controls. The diabetic bGH antagonist transgenic mice were protected from diabetes-induced nephropathy. Besides in the diabetic mice with severe glomerulosclerosis, increase in mRNAs of alpha1 IV procollagen and of 72 kDa collagenase was found in the total glomerulus (Esposito et al. 1996). In another study, diabetic rats from a genetic GH/IGF-I deficient dwarf strain showed less albuminuria and glomerular hypertrophy than diabetic GH/IGF-I intact rats (Gronbaek et al. 1997).
In conclusion, we have shown that GH can induce a marked increase in type IV collagen production in HUVEC cultures, additive to the effect of high glucose concentration. High glucose, but not GH, appears to activate the PKC pathway. Our results support the hypothesis of a GH contribution in diabetic vascular BM thickening by enhancing the synthesis of type IV collagen in EC.
Abbreviations
- APMA:
-
p Aminophenylmercuric acetate
- BM:
-
Basement membrane
- EC:
-
Endothelial cells
- GH:
-
Growth hormone
- hGH:
-
Human GH
- bGH:
-
Bovine GH
- HS:
-
Human serum
- HUVEC:
-
Human umbilical vein EC
- JAK2:
-
Janus kinase 2
- M199:
-
199 culture medium
- PKC:
-
Protein kinase C
- PKCI:
-
PKC inhibitor
References
Bakillah A, Grigorova-Borsos AM, Guillot R, Urios P, Sternberg M (1996) Effect of an aldose reductase inhibitor on type IV collagen production by human endothelial cells cultured in high glucose. Diabetologia 39:641–648
Ballintine EJ, Foxman S, Gorden P, Roth J (1981) Rarity of diabetic retinopathy in patients with acromegaly. Arch Intern Med 141:1625–1627
Brooks AJ, Waters MJ (2010) The growth hormone receptor: mechanism of activation and clinical implications. Nat Rev Endocrinol 6:515–525
Chen NY, Chen WY, Bellush L, Yang CW, Striker LJ, Striker GE, Kopchick JJ (1995) Effects of streptozotocin treatment in growth hormone (GH) and GH antagonist transgenic mice. Endocrinology 136:660–667
Clarkson RW, Chen CM, Harrison S, Wells C, Muscat GE, Waters MJ (1995) Early responses of trans-activating factors to growth hormone in preadipocytes: differential regulation of CCAAT enhancer-binding protein-beta (C/EBP beta) and C/EBP delta. Mol Endocrinol 9:108–120
Danne T, Spiro MJ, Spiro RG (1993) Effect of high glucose on type IV collagen production by cultured glomerular epithelial, endothelial, and mesangial cells. Diabetes 42:170–177
Du X, Matsumura T, Edelstein D, Rossetti L, Zsengeller Z, Szabo C, Brownlee M (2003) Inhibition of GAPDH activity by poly(ADP-ribose) polymerase activates three major pathways of hyperglycemic damage in endothelial cells. J Cin Invest 112:1049–1057
Esposito C, Liu ZH, Striker GE, Phillips C, Chen NY, Chen WY, Kopchick JJ, Striker LJ (1996) Inhibition of diabetic nephropathy by a GH antagonist: a molecular analysis. Kidney Int 50:506–514
Gaur S, Yamaguchi H, Goodman HM (1996) Growth hormone increases calcium uptake in rat fat cells by a mechanism dependent on protein kinase C. Am J Physiol 270:C1485–C1492
Geraldes P, King GL (2010) Activation of protein kinase C isoforms and its impact on diabetic complications. Circ Res 106:1319–1331
Grigorova-Borsos A-M, Bakillah A, Urios P, Leblond V, Guillot R, Sternberg M (1996) Production of type IV collagen and 72-kDa gelatinase by human endothelial cells cultured in high glucose. Effects of a protein kinase C inhibitor, GF 109203X. Biochem Cell Biol 74:659–667
Gronbaek H, Volmers P, Bjørn SF, Østerby R, Ørskof H, Flyvberg A (1997) Effect of GH/IGF-I deficiency on long-term renal changes and urinary albumin excretion in diabetic dwarf rats. Am J Physiol 272:E918–E924
Gubler M, Noel L, Mounier F, Gros F, Wieslander J (1988) Immunohistochemical, study of extracellular matrix components in diabetic glomerulosclerosis. In: Gubler M, Sternberg M (eds) Progress in basement membrane research. Renal and related aspects in health and disease. John Libbey, London, pp 201–204
Hansen AP (1970) Abnormal serum growth hormone response to exercise in juvenile diabetics. J Clin Invest 49:1467–1478
Hansen AP, Johansen K (1970) Diurnal patterns of blood glucose, serum free fatty acids, insulin, glucagon and growth hormone in normals and juvenile diabetics. Diabetologia 6:27–33
Hayakawa K, Walker L, Constable IJ (1990) The effect of glucose and insulin concentration on type IV collagen biosynthesis by bovine retinal capillary endothelial cells in vitro. Jpn J Ophthalmol 34:463–471
Hogemann B, Balleisen L, Rauterberg J, Voss B, Gerlach U (1986) Basement membrane components (7S collagen, laminin P1) are increased in sera of diabetics and activate platelets in vitro. Haemostasis 16:428–432
Holmes K, Chapman E, See V, Cross MJ (2010) VEGF stimulates RCAN1.4 expression in endothelial cells via a pathway requiring Ca 2+/calcineurin and protein kinase C-δ. PloS One 5:e11435
Horn S, Cohen R, Gertler A (1994) Regulation of heat-shock protein (hsp70) gene expression by hGH and IL2 in rat Nb2 lymphoma cells. Mol Cell Endocrinol 105:139–146
Jaffe EA, Minick CR, Adelman B, Becker CG, Nachman R (1976) Synthesis of basement membrane collagen by cultured human endothelial cells. J Exp Med 144:209–225
Kohner EM, Hamilton AM, Joplin GF, Fraser TR (1976) Florid diabetic retinopathy and its response to treatment by photocoagulation or pituitary ablation. Diabetes 25:104–110
Kramer RH, Fuh GM, Karasek MA (1985) Type IV collagen synthesis by cultured human microvascular endothelial cells and its deposition into the subendothelial basement membrane. Biochemistry 24:7423–7430
Kumar PP, Purbey PK, Sinha CK, Notani D, Limaye A, Jayani RS, Galande S (2006) Phosphorylation of SATB1, a global gene regulator, acts as a molecular switch regulating its transcriptional activity in vivo. Molecular Cell 22:231–243
Lanning NJ, Carter-Su C (2006) Recent advances in growth hormone signaling. Rev Endocr Metab Disord 7:225–235
Ledet T, Heickendorff L (1985) Growth hormone effect on accumulation of arterial basement membrane-like material studied on rabbit aortic myomedial cell cultures. Diabetologia 28:922–927
Ledet T, Vuust J (1980) Arterial procollagen type I, type III, and fibronectin: effects of diabetic serum, glucose, insulin, ketone, and growth hormone studied on rabbit aortic myomedial cell cultures. Diabetes 29:964–970
Lundbaek K, Christensen NJ, Jensen VA, Johansen K, Olsen TS, Hansen AP, Orskov H, Osterby R (1970) Diabetes, diabetic angiopathy and growth hormone. Lancet 2:131–133
Matsumoto E, Matsumoto G, Ooshima A, Kikuoka H, Bessho H, Miyamura K, Nanjo K (1990) Serum type IV collagen concentrations in diabetic patients with microangiopathy as determined by enzyme immunoassay with monoclonal antibodies. Diabetes 39:885–890
Mattila P, Majuri ML, Tiisala S, Renkonen R (1994) Expression of six protein kinase C isotypes in endothelial cells. Life Sci 16:1253–1260
Monboisse JC, Garnotel R, Bellon G, Ohno N, Perreau C, Borel JP, Kefalides NA (1994) The alpha 3 chain of type IV collagen prevents activation of human polymorphonuclear leukocytes. J Biol Chem 269:25475–25482
Motyl T, Gajewska M, Zarzynska J, Sobolewska A, Gajkowska B (2007) Regulation of autophagy in bovine mammary epithelial cells. Autophagy 3:484–486
Niimi S, Hayakawa T, Tanaka A, Ichihara A (1991) Glucose regulation of growth hormone receptors in primary cultured rat hepatocytes. Endocrinology 129:2734–2739
Nilsson A, Carlsson B, Mathews L, Isaksson OG (1990) Growth hormone regulation of the growth hormone receptor mRNA in cultured rat epiphyseal chondrocytes. Mol Cell Endocrinol 70:237–246
Nivet V, Clot JP, Do XT, Barrault V, Prelot M, Durand D (1993) Evidence that growth hormone stimulates protein kinase C activity in isolated rat hepatocytes. Metabolism 42:1291–1295
Ohlsson C, Nilsson A, Isaksson OG, Lindahl A (1992) Effect of growth hormone and insulin-like growth factor-I on DNA synthesis and matrix production in rat epiphyseal chondrocytes in monolayer culture. J Endocrinol 133:291–300
Poulsen JE (1953) Recovery from retinopathy in a case of diabetes with Simmonds’ disease. Diabetes 2:7–12
Rabin D, Bloomgarden ZT, Feman SS, Davis TQ (1984) Development of diabetic complications despite the absence of growth hormone in a patient with post-pancreatectomy diabetes. N Engl J Med 310:837–839
Ramsay RC, Goetz FC, Sutherland DE, Mauer SM, Robison LL, Cantrill HL, Knobloch WH, Najarian JS (1988) Progression of diabetic retinopathy after pancreas transplantation for insulin-dependent diabetes mellitus. N Engl J Med 318:208–214
Randeva HS, Lewandowski KC, Komorowski J, Murray RD, O’Callaghan CJ, Hillhouse EW, Stepien H, Shalet SM (2004) Growth hormone replacement decreases plasma levels of matrix metalloproteinases (2 and 9) and vascular endothelial growth factor in growth hormone-deficient individuals. Circulation 109:2405–2410
Rasmussen LM, Ledet T (1993) Aortic collagen alterations in human diabetes mellitus. Changes in basement membrane collagen content and in the susceptibility of total collagen to cyanogen bromide solubilisation. Diabetologia 36:445–453
Rymaszewski Z, Cohen RM, Chomczynski P (1991) Human growth hormone stimulates proliferation of human retinal microvascular endothelial cells in vitro. Proc Natl Acad Sci USA 88:617–621
Shimomura H, Spiro RG (1987) Studies on macromolecular components of human glomerular basement membrane and alterations in diabetes. Decreased levels of heparan sulfate proteoglycan and laminin. Diabetes 36:374–381
Smal J, De Meyts P (1989) Sphingosine, an inhibitor of protein kinase C, suppresses the insulin-like effects of growth hormone in rat adipocytes. Proc Natl Acad Sci USA 86:4705–4709
Sundkvist G, Almer L, Lilja B, Pandolfi M (1984) Growth hormone and endothelial function during exercise in diabetics with and without retinopathy. Acta Med Scand 215:55–61
The Kroc Collaborative Study Group (1988) Diabetic retinopathy after two years of intensified insulin treatment. Follow-up of the Kroc collaborative study. JAMA 260:37–41
Toullec D, Pianetti P, Coste H, Bellevergue P, Grand-Perret T, Ajakane M, Baudet V, Boissin P, Boursier E, Loriolle F, Duhamel L, Charon D, Kirilovsky J et al (1991) The bisindolylmaleimide GF 109203X is a potent and selective inhibitor of protein kinase C. J Biol Chem 266:15771–15781
Tseng SC, Savion N, Gospodarowicz D, Stern R (1983) Modulation of collagen synthesis by a growth factor and by the extracellular matrix : comparison of cellular response to two different stimuli. J Cell Biol 97:803–809
Vidal (2009) Dictionnaire des spécialités pharmaceutiques, éditions du Vidal, Issy-les Moulineaux, France
Woessner JF (1991) Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J 5:2145–2154
Wu HM, Schally AV, Cheng JC, Zarandi M, Varga J, Leung PC (2010) Growth hormone-releasing hormone antagonist induces apoptosis of human endometrial cancer cells through PCCδ-mediated activation of p53/p21. Cancer Lett 298:16–25
Yamasaki T, Takahashi A, Pan J, Yamaguchi N, Yokoyama KK (2009) Phosphorylation of activation transcription factor-2 at serine 121 by protein kinase C controls c-Jun-mediated activation of transcription. J Biol Chem 284:8567–8581
Acknowledgments
This work was supported by the University of Paris V and VI and the “Fondation pour la Recherche Médicale”. We thank Dr H. Cohen and colleagues (Maternité, Hôpital International de l’Université de Paris) for providing us with umbilical cords; Pr C. Roussel, Pr J.P. Bisson and colleagues (Département de Biologie de la Reproduction, Faculté de Médecine des Saints-Pères) for cell culture facilities; Dr D. Hartmann (Institut Pasteur, Lyon) for providing us with anti-type IV collagen antiserum and various antigens; J. Kirilovsky (Glaxo) for his generous gift of GF109203X; Choay Laboratories for providing us with hGH; G.Mozère and G. Hirbec for technical help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bakillah, A., Guillot, R., Urios, P. et al. Stimulating effect of growth hormone on type IV collagen production by endothelial cells cultured in normal and high glucose. Amino Acids 43, 697–707 (2012). https://doi.org/10.1007/s00726-011-1118-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-011-1118-5