Abstract
Focusing on drug discovery non-proteinogenic amino acids have often been used as important building blocks for construction of compound libraries in the filed of combinatorial chemistry and chemical biology. Highly homogeneous l-mimosine, α-amino-β-(3-hydoxy-4-oxo-1,4-dihydropyridin-1-yl)-propanoic acid, a non-proteinogenic amino acid, has been successfully isolated and purified on an industrial scale from wild leaves of Leucaena (Leucaena leucocephala de Wit) which is a widely distributed legume in Okinawa, a sub-tropical island in Japan. Optical purity determinations used for quality control have been established through diastereomer formation. Physico-chemical properties and biological properties of purified mimosine have been clarified. Mimosine is sparingly soluble in water and organic solvents but can be dissolved in aqueous alkaline solution. The tyrosinase pathway is of particular interest in the cosmetic field, since mimosine is an analog of tyrosine. Thus the present purified mimosine have been tested in tyrosinase inhibitory assays. The IC50 for tyrosinase inhibitory activity of purified Mim was compared with kojic acid. Mimosine shows significant inhibition of melanin production in murine melanoma cells. The derivatization of mimosine has been investigated with a focus on its use in conventional peptide syntheses to generate mimosyl peptides. N-(9-Fluorenylmethoxycarbonyloxy)-mimosine and resin-bound mimosine for solid-phase syntheses have also been performed. Highly homogeneous Mim is a useful material for the development of functional cosmetics or active pharmaceutical ingredients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A non-proteinogenic amino acid, l-mimosine (Mim), α-amino-β-(3-hydoxy-4-oxo-1,4-dihydropyridin-1-yl)-propanoic acid, involving a hydroxyketone group, which has received attention in cancer therapeutics, is found in young leaves and mature seeds of a tropical legume, Leucaena (Leucaena leucocephala de Wit), which has a high protein content and is widely distributed in Okinawa island (wild, not cultivated). Thus, it is easy to collect leaves as starting material; although this plant cannot be used for cattle feed as it is believed to be toxic. Achiral chemical synthesis of mimosine (d- and l-mixture) has been reported almost half century ago (Adams and Johnson 1949). Over the last few decades combinatorial chemistry and chemical biology have undergone rapid development for the discovery of useful functional compounds, especially drugs. Hence, non-proteinogenic amino acids are useful building blocks, and numerous chiral compounds have been produced. Naturally occurring materials are often beneficial because of their defined chirality. In the case, the source (raw material) is abundant and purification on a large scale is not complicated, these materials promise the development of useful compounds such as pesticides, functional cosmetics and/or drugs. In fact, the anti-tumor effect (DeWys and Hal 1973) and the inhibitory effect (Chang et al. 1999) of Mim have been reported, and show promise in drug development. Additionally, recently it has been confirmed that Mim has strong allelopathic properties, characteristic of the legume trees, and that can be used for pesticides (Xuan et al. 2006). The tyrosinase inhibitory activities of Mim are also of interest in the cosmetic field for whitening. However, the confined supply of Mim and limited knowledge of its properties have restricted its use and potential for future industrial applications. The present paper describes the preparative scale extraction and purification of l-mimosine from leaves of Okinawa’s Leucaena in a simple and low cost manner without causing environmental damage.
Materials and methods
General
The major reagent for peptide chemistry, amino acid derivatives, solid supports, HBTU and HOBt were from HiPep Laboratories (Kyoto, Japan). N-(9-Fluorenylmethoxycarbonyloxy) succinimide ester (Fmoc-OSu, Orpegen, Heidelberg, Germany), triisopropyl silane (TIS, Sigma-Aldrich, Tokyo, Japan), tert-butyldimethylsilyl chloride (TBDMS-Cl, Sigma-Aldrich, Tokyo), Di-tert-butyl dicarbonate (Boc2O, Kokusan Chemicals, Tokyo), 4,4′-dimethoxytrityl chloride (DMT-Cl, Wako Pure Chemical Industries, Osaka, Japan), 1,2-ethanedithiol (EDT, Nacalai Tesque, Kyoto) and acetic anhydride (Nacalai Tesque, Kyoto) were used as-received. Other reagents and solvents used for derivatization of Mim, except chiral analyses, peptide syntheses, deprotection and cleavage were purchased from Nacalai Tesque, which were used as received. Ethanol, ammonium hydroxide, hydrochloric acid and sodium hydroxide were purchased from Wako. Amberlite IR120 H was purchased from GFS Chemicals Inc. (OH, USA). 2,3,4,6-Tetra-O-acetyl-β-d-glucopyranosyl isothiocyanate (GITC) was from Tokyo Chemical Industry (Tokyo). Mushroom tyrosinase, 3,4-dihydroxy-l-phenylalanine (l-DOPA) and kojic acid were purchased from Sigma-Aldrich (Tokyo, Japan). Other reagents and solvents used for the tyrosinase inhibition assay were obtained from Nacalai Tesque. Water was prepared by a Milli-Q apparatus (Millipore, Tokyo). Peptide-chain assembly was performed on an automated synthesizer, PSSM-8 (Shimadzu, Kyoto, Japan) or a manual synthesizer, PetiSyzer® (HiPep Laboratories), which was used also in larger scale syntheses, especially for Fmoc-Mim loading onto resins. Analyses by reversed-phase (RP)-HPLC were carried out using LC-10A System (Shimadzu) and on-line LCMS analyses were performed on an Agilent 1100 with ion-trap mass spectrometer (HCT ultra, Bruker Daltonics Yokohama, Japan) or LC-20A with electrospray ionization (ESI) mass spectrometer (LCMS-2010EV, Shimadzu). Analytical RP-HPLC columns used were HiPep-Cadenza (3.0 id × 150 mm, HiPep Laboratories, Kyoto), HiPep-Intrada (3.0 id × 150 mm, HiPep Laboratories) or POROS-R2 (4.6 id × 50 mm, Applied Biosystems, Tokyo, Japan). Preparative purification was carried out using a HiPep-Intrada (20 id × 250 mm, HiPep Labs.) with LC-8A System (Shimadzu). Optical rotation was measured by a P-1030 Polarimeter (JASCO, Tokyo). UV spectrophotometer used was V-570 (JASCO). The 1H NMR spectra were recorded on an AM-300 AVANCE II NMR spectrometer (Bruker, Yokohama, Japan). FTICR-MS of purified Mim was measured on an Apex-Q94e with Apollo II source (Bruker-Daltonics, Yokohama).
Extraction and isolation of Mim from Leucaena leucocephala
Fresh Leucaena leaves (20 kg) were collected and added to boiling water (100 L), allowed to stand for 10 min, then cooled down to ambient temperature and filtered. To the resulting filtrate cation-exchange resin (10 kg, Amberlite IR120 H) was added and allowed to stand overnight. The resin was washed by immersion in 80% ethanol (20 L) for 12 h and distilled water (20 L) for 2 h. Absorbed Mim was eluted from the resin with 2 M ammonia solutions (40 L) for 6 h, and then active charcoal (5 g) was added and stirred for 10 min. After removal of charcoal, the filtrate was concentrated to ca 1.5 L under reduced pressure. Hydrochloric acid (6 M) was added to this solution to give a pH 4.5–5.0, and Mim was precipitated at 4°C overnight. The resulting crude Mim was recrystallized from 6 M sodium hydroxide (1.8 L) and 6 M hydrochloric (pH 4.5–5.0), and then allowed to stand at 4°C overnight to give purified Mim (100 g from an initial 20 kg of fresh Leucaena leaves). MS m/z: calcd. for C8H10N2O4 199.18 ([M+H]+), found 199.07. mp. 218–220°C, 1H NMR (300 MHz, D2O) δ 7.61–7.57 (br m, 2H), 6.64–6.5 (br m, 1H), 4.44–4.27 (br m, 2H), 4.12 (m, 1H) (Supplementary sheet). [α] 20.2D = –23.2 (c = 2.11 mg/1.5 mL, H2O), Ref. mp 235–236°C, [α] 22D = −21 (C = 4.9 mg/2.5 mL, H2O) (1).
Chiral analysis of Mim
Purified Mim from Leucaena was derivatized by GITC to give the diastereomers. Saturated mimosine in water (5 μL), 10 mM GITC acetonitrile solution (5 μL) and saturated solution of sodium hydrogen carbonate (5 μL) were combined. After 30 min the mixture was diluted (10×) in water and analyzed by LCMS using HiPep-Intrada column (HiPep Laboratories). MS m/z: calcd for C23H29N3O13S 588.6 ([M+H]+) found 588.0.
Tyrosinase inhibitory assay
l-DOPA and inhibitor candidates were dissolved in 50 mM sodium phosphate buffer, pH 6.8, as a stock solution. These solutions (25 μL), tyrosinase solution (2U/μL in 50 mM sodium phosphate buffer, pH 6.8, 12.5 μL) and 50 mM sodium phosphate buffer, pH 6.8 (112.5 μL) were mixed. To this 1.25 mM l-DOPA (100 μL) was added and mixed. The resulting solution was transferred immediately to a cell, and UV-absorbance at 475 nm was measured every second up to 1 min. The kinetic data were fitted to a sigmoidal curve, hence 100% was no inhibition. IC50 (concentration at 50% inhibition) was calculated (triple replications).
Synthesis of Fmoc-protected Mim
Mim (10 g, 50 mmol) and sodium carbonate (10 g, 100 mmol) were dissolved in water (150 mL). To this Fmoc-OSu (25 g, 75 mmol) in dioxane (150 mL) was added and stirred at ambient temperatures for 6 h. The reaction mixture was diluted with 0.1 M sodium carbonate solution (600 mL) and further stirred for 6 h. The resulting mixture was filtered, and the filtrate was washed with ethyl acetate (500 mL × 2). The aqueous layer was neutralized with 1 M HCl and the resulting precipitate was collected by filtration, washed with water (1 L) and dried (22.1 g). The crude material was recrystallized from hot isopropanol (14.8 g, 70%). The purified material was analyzed by LCMS (column: POROS-R2, eluent A = 0.1% TFA–water; eluent B = 0.1% TFA–90% acetonitrile; flow rate: 0.3 mL/min). MS m/z: calcd for C23H20N2O6 421.4 ([M+H]+), found: 421.1([M+H]+). 1H NMR (300 MHz, DMSO-d 6 ) δ 8.13–7.85 (m, 4H), 7.60–7.59 (br m, 2H), 7.44–7.29 (m, 4H), 7.03–7.00 (br m, 1H), 4.69 (br s, 1H), 4.55 (br s, 1H), 4.37–4.18 (m, 4H) (Ref Supplementary sheet). Optical rotation: [α] 23.4D = –3.09 (C = 2.34 mg/1.5 mL, methanol). mp. 181–182°C.
Side chain protection of Fmoc-Mim
Mim (18 mg, 42 μmol) and imidazole (11 mg, 168 μmol) were dissolved in DCM:DMF (1:1 v/v). To this was added TBDMS-Cl (13 mg, 84 μmol) on an ice bath. The resulting mixture was stirred overnight at 45°C. The LCMS analysis indicated the formation of the desired N-α-Fmoc-O-tert-butyldimetylsilyl-l-mimosine, although significant amounts of by-products were found. Introduction of other groups was also attempted such as reaction with Boc2O in sodium carbonate, DMTr-Cl with DIEA, acetic anhydride with pyridine, but the desired products were not obtained.
Stability of Mim in conditions used in Fmoc-tBu based peptide syntheses
Mim-Tyr as a model dipeptide was synthesized, which can be easily detected by LCMS using RP HPLC column. The conventionally solid-phase synthesised Fmoc-Mim-Tyr(tBu) was immobilized on a 2-chlorotrityl resin. The resulting dipeptide resin was treated with a mixture of 2% piperidine and 2% DBU in DMF (v/v) or 20% piperidine in DMF, followed by cleavage using the mild conditions of AcOH:TFE:DCM (1:1:8 v/v). The liberated dipeptide was analyzed by the above LCMS to confirm Mim-Tyr(tBu) (MS m/z calcd for C21H27N3O6 418.5 ([M+H]+), found: 418.2). The above resin-bound Mim-Tyr(tBu) was cleaved with TIS:water:TFA (2.5:2.5:95 v/v) or TIS:water:EDT:TFA (2.5:2.5:3:92 v/v). Mim-Tyr was confirmed as the major component by LCMS (MS m/z: calcd for C17H19N3O6 362.4 ([M+H]+), found: 362.1). The above Mim-Tyr(tBu)-2-Cl-Trt-resin was treated with acetic anhydride in pyridine (1:1 v/v) for 1 h and cleaved using TIS:water:TFA (2.5:2.5:95 v/v). Ac-Mim-Tyr as the major product was confirmed by LCMS (MS m/z calcd for C19H21N3O7 404.4 ([M + H]+), found: 404.1). Side chain acetylated Mim-dipeptide was also found as a minor component. A tripeptide, Mim-Phe-Lys(Alloc)-NH2, as a model peptide was assembled. This was treated with catalytic amounts of [(C6H5)3P]4Pd with phenyl silane (10-fold excess) in DCM for 30 min at ambient temperatures. The resulting material was analyzed by LCMS. None of the desired Mim-Phe-Lys-NH2 was found, instead the 2,4-dihydroxy pyridine derivative of the side chain of Mim was formed and decomposed to a complicated mixture of products.
Preparation of resin bound Mim
Fmoc-Mim-OH 8.4 g (20 mmol) was dissolved in dried DCM (100 mL) and DIEA (13.7 mL, 80 mmol). This solution was added dropwise to 2-chlorotrityl chloride resin (100–200 mesh, 1% divinylbenzene, substitution rate 0.98 mmol/g) 20 g (20 mmol) on an ice bath and stirred for 60 min at r.t. The whole procedure was carried out under a nitrogen atmosphere. The resin was washed three times with DCM/methanol/DIEA (17/2/1), DCM, DMF, MeOH and tert-butyl methyl ether, respectively, and dried in vacuo (26.8 g). An aliquot of resins (ca. 10 mg) was quantified by a spectrophotometric method (Bernatowicz et al. 1989; Meienhofer et al. 1979) and indicated that the substitution of mimosine was 0.46 mmol/g.
Results and discussion
Extraction and purification of Mim from natural resources
Previously, Mim in wild Leucaena leaves was eluted into water and separated using ultra filtration (Tawata 1994), although this method has not been used for industrial production of Mim. As Leucaena leaves may contain mimosinase (Personal communication; Fukuta, Ryukyu University), we have employed an extraction method using hot water. Fresh leaves were allowed to stand in boiling water to avoid enzymatic degradation. After removal of leaves, a cation-exchange resin was added to the resulting liquor to absorb Mim. After washing the resin with 80% ethanol to remove chlorophyll and other soluble organic compounds followed by water, Mim was eluted with aqueous ammonia, treated with charcoal and then concentrated under reduced pressure at room temperature. The residue was acidified with hydrochloric acid (pH 4.5–5.0) and allowed to stand at 4°C overnight. The precipitate was collected and recrystallized from aqueous ammonia with hydrochloric acid to give a high quality Mim that shows the expected mass spectrum.
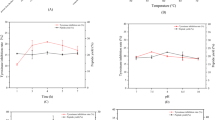
Mim content in Leucaena has been quantified using HPLC (Hongo et al. 1987; Puchala et al. 1996), although Mim was eluted almost at the void position and thus the purity could not be determined. Hongo et al. calculated Mim content based on leaf dry weight, and the actual content of fresh leaves was not established. IR or UV spectroscopic methods had been proposed (Prasad 1995; Lalitha et al. 1993), but the extract from Leucaena contains several compounds which influence absorbance and give overlapping signals. Thus, the previous reports were not reliable and the precise content of the extracts could not be ascertained. We have isolated Mim continuously for 6 months during the summer season and confirmed the actual amounts of purified material. As indicated in Fig. 1 the amounts are actual weights purified from freshly collected leaves, where even mechanical loss has been taken into account. It appears that Mim is produced in the largest amounts under strong sunlight at the highest daily temperatures, and in 6 months allowed the production of 1 kg from a monthly extract of ca. 40–60 kg leaves.
Characterization of Mim
The solubility of Mim, required for derivatization and bioassays, is summarized for saturated solutions in Table 1. Mim is sparingly soluble in water, as well as in particular organic solvents even at elevated temperatures, but can be dissolved in aqueous alkaline solution. Mim is not stable in strong acids. The Fourier transform ion cyclotron resonance (FTICR) mass spectrometry gives molecular masses at a very high resolution and accuracy. The purified Mim prepared here has a protonated molecular ion [M+H]+ at 199.07136, and elemental composition C8H11N2O4 that agrees with the theoretical composition (Ref Supplementary sheet).
An octadodecyl silica column with reversed-phase HPLC is often used for purification and characterization of biomolecules, although Mim is not retained on commercially available columns and elutes near the void. Moreover, Mim is absorbed on normal-phase silica columns, and may interact with silanol groups that generate broadened profiles (data not shown). In fact, during isolation and purification we have employed alkaline conditions, which may cause racemization, thus chiral analysis is indispensable, although it has not yet been reported. Chiral analyses of natural amino acids by enantiomeric labeling are usually employed (Frank et al. 1978), and analyses of non-natural amino acids by this method have also been reported (Nokihara and Gerhardt 2001). However, Mim is not stable under the derivatization conditions used (data not shown). While the chiral column for separation of amino acids, Crownpak® CR(+), is commercially available (URL: http://www.daicelchiral.com), we have attempted to use this column for the determination of the chirality of Mim. Unfortunately the elution position is very near that of l-alanine (Fig. 2), which is a major degradation product of Mim and thus quantitative analysis is not possible.
As chiral determination is indispensable for bio-medical applications, we have developed a pre-column derivatization method and generated diasteromers using GITC (Fig. 3). The reaction products were analyzed using a recently developed reverse-phase column (HiPep-Intrada: http://www.hipep.jp) combined with on-line mass spectrometry. Racemized Mim has been prepared from the purified Mim according to Adams and Johnson. Determination of d- and l-Mim could be realized as shown in Fig. 4, and it has been confirmed that the purified Mim produced here was a racemization-free compound.
HPLC profiles and MS spectra of GITC-Mim. Left upper: GITC-Mim (A) from the present work. Left lower GITC-Mim from racemized Mim (B,C) prepared by treatment overnight in boiling water. Peak (D) is unreacted GITC. Column: HiPep-Intrada (3.0 id × 150 mm) temp. 25°C, Eluent: A = 0.1% trifluoroacetic acid (TFA), B = 0.1% TFA in 90% acetonitrile, A/B 99/01 to 69/31 in 30 min, flow rate 0.3 mL/min, UV: 210 nm. Right: MS spectra of peaks B and C. MS m/z: Both calculated for C23H29N3O13S, 588.6 ([M+H]+), found 588.0)
Biological properties of purified Mim
Both the tyrosinase pathway and the melanogenesis are of particular interest in the dermatological field. Tyrosinase (EC 1.14.18.1) is a copper containing enzyme, which catalyzes the oxidation of monophenols, o-diphenols and o-quinones, and is known to play a key role in melanin biosynthesis. Mim, as well as kojic acid, are known tyrosinase inhibitors (Cabanes et al. 1987). Different IC50 values for tyrosinase inhibitory activity of Mim and kojic acid have been reported (Ahmed et al. 2004; Khan et al. 2006), although the purity of Mim used in these experiments was not defined. Hence, we have tested our purified Mim for tyrosinase inhibitory effects and inhibition of melanin production. The IC50 for tyrosinase inhibitory activity of our purified Mim was 41.3 μM, and that of kojic acid was 18.2 μM (Fig. 5).
Derivatization of Mim and syntheses of Mim containing peptides
Amino and/or carboxyl termini and eventually side chain functional groups of Mim should be protected for introduction of Mim in the peptide chain. As the amino-protecting group Fmoc-, Trt-, Boc- and Z-group are generally used, as well as the acetyl group which is useful for N-terminal amino acid residues. Hence, we have attempted the introduction of these protecting groups using conventional methods. Fmoc-, Boc- and Ac-groups were introduced not only at the amino group, but also at the side chain hydroxyl group of Mim. However, side chain Fmoc- and Boc-groups were unstable, and underwent deprotection during further treatment. Introduction of a Trt-, Z- and acetyl group in Mim caused decomposition of the side chain, and/or formation of significant byproducts, thus these are not practical derivatizations (data not shown). In the case of the Fmoc-derivatization (Fig. 6), the side chain Fmoc-group of bis-Fmoc-Mim 5 was easily removed by washing with sodium carbonate solution to give Fmoc-Mim 4 in a practical yield, although careful treatment was required as small amounts of Fmoc–Ala were produced at the same time. These could be easily removed by washing with isopropanol or recrystallization from hot isopropanol. HPLC-profile of the purified Fmoc-Mim is shown in Fig. 7. On the other hand, after the introduction of the Boc-group removal of the side chain Boc-group was not easy, and the compound undergoes slow degradation, which caused difficulties not only with quality control but also introduction of bis-Boc-Mim in the peptide chain generates unknown components. Thus, it is clear that Fmoc-Mim is the most suitable starting material for peptide syntheses. Side chain protection of the hydroxyl group of Mim with tert-butyldimethylsilyl (TBDMS) and dimethoxytrityl (DMTr) groups was not possible due to steric hindrance.
Deprotection and cleavage conditions were also carefully investigated using model peptides and confirmed stability of the side chain functional group of Mim. These included repeated treatment with piperidine solution for removal of the Fmoc-group and cleavage using trifluoroacetic acid with scavengers. As a model dipeptide, Fmoc-Mim-Tyr(tBu) was constructed on the chlorotrityl chloride polystyrene resin (2-Cl-Trt-resin) by coupling Fmoc-Mim with 1-hydroxybenzotriazole (HOBt): 2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethylaminium hexafluorophosphate (HBTU): N,N-diisopropylethylamine (DIEA) (1:1:2 eq) on Tyr(tBu)-2-Cl-Trt-resin with single, double or triple coupling conditions, as introduction of Fmoc-Mim was more difficult than simple Fmoc amino acids. After coupling an aliquot of beads was washed, dried and cleaved under mild conditions with a cocktail of acetic acid: trifluoroethanol (TFE) and dichloromethane (DCM) (1:1:8 v/v). The liberated peptide was characterized by HPLC, and the coupling efficiency was calculated from the amount of unreacted Tyr(tBu) present. Figure 8 summarizes the coupling efficiency of Fmoc-Mim. Surprisingly, the result indicates that an excess of Fmoc-Mim as acyl component is not favorable, but triple coupling of a three-fold excess gave an optimal yield of the desired product. No significant difference has been observed between 20% piperidine in DMF and 2% 1,8-diazabicyclo[5.4.0]-7-undecene 2-methoxy-2-methylpropane (DBU) with 2% piperidine conventionally used for the removal of the Fmoc group. Applicability of the cleavage cocktail used for the conventional solid-phase syntheses was sufficient. Thus, both TIS/water/TFA (2.5/2.5/95 v/v) and TIS/water/EDT/TFA (2.5/2.5/2.5/92.5 v/v) did not generate significant degradation of the side chain of Mim, especially as thiol that is often used as a cation scavenger does not affect Mim. The stability of the Mim-side chain was assessed mainly from the formation of the corresponding Ala-derivatives. The Alloc-group has been used for useful protection in orthogonal syntheses or preparation of branched peptides based on the εLys residue. Therefore the stability of Mim under Alloc-removal conditions was also investigated. Mim-Phe-Lys(Alloc)-NH2 was synthesized and analyzed by LC–MS after the cleavage procedure. Alloc-removal was carried out using [(C6H5)3P]4Pd in the presence of C6H5SiH3 (10 eq.) for 30 min, and resulted in a di-hydroxypyridine ring in Mim that decomposed within 30 min. The same peptide without the use of the Alloc-removal procedure, and the same peptide without the Alloc-group were used as controls. Thus, Mim does not survive the Alloc removal conditions. Both peptidyl resins were cleaved with TIS/water/TFA (5/5/90 v/v). Significant amounts of peaks with 24 larger masses have been obtained, and small amounts of Ala containing components were found. Compared to peptides consisting of only proteinogenic amino acids, Mim-peptides showed more peaks in their HPLC-profiles. Boc-Mim can be used, although the presence of the bis Boc derivative generates complications for interpretation of HPLC-profiles, since the side chain O-Boc is unstable. In conclusion, Fmoc-Mim without side chain protection can be used successfully for solid-phase peptide syntheses, while other protecting groups are not suitable for either temporary or permanent protection. For production of C-terminally Mim containing peptides, Mim was easily introduced on to 2-Cl-Trt-resin in the conventional manner, since peptides immobilized on this resin can be cleaved under mild conditions. The loading amounts were adjusted to 0.45–0.60 mmol/g resin.
High quality l-mimosine had been produced from leaves of Okinawa’s Leucaena in a low cost manner without causing environmental damage. In fact the Leucaena plant grows rapidly and during the summer season the same amount of leaves can be cropped at 1–2 monthly intervals from the same plants while in winter the same is possible at 4–5 monthly intervals. Seeds also contain Mim, although seeds must be ground and throughput is lower. Quality control methods, especially to check the optical purity of purified Mim, have been established. The Mim produced here has been derivatized for use in conventional peptide synthesis as a building block focusing on discovery of novel functional materials as ingredients for pharmaceuticals or cosmetics.
Conclusion
Isolation and characterization of Mim have been performed on an industrial scale. As Leucaena is a widely distributed legume in Okinawa Prefecture, southern Japan, the source of Mim is quite sustainable. Focusing on applications of this unique non-proteinogenic amino acid as a building block in peptides, Fmoc-Mim and resin-bound Mim for solid-phase syntheses have also been performed. Highly homogeneous Mim is a useful material for the development of functional cosmetics or active pharmaceutical ingredients.
Abbreviations
- Mim:
-
Mimosine
References
Adams R, Johnson JL (1949) Leucenol. VI. A total synthesis. J Am Chem Soc 71:705–708
Ahmed VU, Ullah F, Hussain J, Farooq U, Zubair M, Kahn MTH, Choudhary MI (2004) Tyrosinase inhibitors from rhododendron collettianum and their structure activity relationship (SAR) studies. Chem Pharm Bull 52:1458–1461
Bernatowicz MS, Daniels SB, Köester HA (1989) Comparison of acid labile linkage agents for the synthesis of peptide C-terminal amides. Tetrahedron Lett 30:4645–4648
Cabanes J, García-Cánovas F, Tudela J, Lozano JA, García-Carmona F (1987) L-Mimosine a slow-binding inhibitor of mushroom tyrosinase. Phytochemistry 26:917–919
Chang HC, Lee TH, Chuang LY, Yen MH, Hung WC (1999) Inhibitory effect of mimosine on proliferation of human lung cancer cells is mediated by multiple mechanisms. Cancer Lett 145:1–8
DeWys WD, Hal TC (1973) Anti-tumor effect of the amino acid mimosine. Europ J Cancer 9:281–283
Frank H, Nicholson GJ, Bayer E (1978) Enantiomer labeling, a method for the quantitative analysis of amino acids. J Chromatogr 167:187–196
Hongo F, Kawashima Y, Tawata S, Sunagawa K, Moromizato S (1987) Studies on chemical composition and mimosine content of Leucaena leucocephala de Wit. The Sci bull Coll Agric, Univ Ryukyu 34:51–57
Khan KM, Maharvi GM, Khan MTH, Shaikh AJ, Perveen S, Begumb S, Choudharya MI (2006) Tetraketones: a new class of tyrosinase inhibitors. Bioorg Med Chem 14:344–351
Lalitha K, Vargheese CM, Balasubramanian N (1993) Spectrophotometric determination of mimosine and 3-hydroxy-4-(1H)-pyridone-the toxic principles of Leucaena leucocephala. Anal Biochem 13:57–62
Meienhofer J, Waki M, Heimer EP, Lambros TJ, Makofske RC, Chang C-D (1979) Solid phase synthesis without repetitive acidolysis: preparation of leucyl-alanyl-glycyl-valine using 9-fluorenylmethyloxycarbonylamino acids. Int J Peptide Protein Res 13:35–45
Nokihara K, Gerhardt J (2001) Development of an improved automated gas chromatographic chiral analysis system: application to non natural amino acids and natural protein hydrolysates. Chirality 13:431–434
Prasad NMV (1995) Analysis of Leucaena mimosine, Acacia tannins and total phenols by near infrared reflectance spectroscopy. Biomass Bioenergy 8:203–205
Puchala R, Davis JJ, Sahlu T (1996) Determination of mimosine and 3, 4-dihydroxypyridine in milk and plasma of goats. J Chromatogr B 685:375–378
Tawata S (1994) Purification method of mimosine for leaves and stems of Leucaena leucocephala de Wit. Agric Chem (in Japanese) 41:18–22
Xuan TD, Elzaawely AA, Deba F, Fukuta M, Tawata S (2006) Mimosine in Leucaena as a potent bio-herbicide. Agron Sustain 26:89–97
Acknowledgments
Authors thank to Dr. Shin-kichi Tawata, Ryukyu University, for the helpful discussion regarding the isolation of Mim, and Yukiko Kodama, Naeko Miyazato, Dr. Farah Deba, Noriko Ono, and Midori Miyajima, HiPep Laboratories for their excellent assistance. Dr. Victor Wray, HZI Braunschweig Germany, is thanked for discussion and linguistic advice. A part of the present work was funded by the Regional Resource Utilization R&D Programs, Ministry of Economy, Trade and Industry, Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Nokihara, K., Hirata, A., Sogon, T. et al. Preparative scale isolation, purification and derivatization of mimosine, a non-proteinogenic amino acid. Amino Acids 43, 475–482 (2012). https://doi.org/10.1007/s00726-011-1104-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-011-1104-y