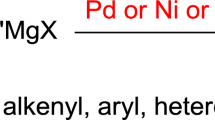Abstract
The formation of the carbon–carbon bond is a fundamental reaction in modern organic synthesis, which is mediated by transition-metal catalysts. Over the past 30 years, the carbon–carbon coupling reactions, particularly the Kumada-Tamao-Corriu coupling reaction, have been dramatically expanded providing a simple methodology for synthetic organic chemistry. So, this review highlights several recent and major developments of carbon–carbon bond-forming reactions by alkylation, alkenylation, arylation, and heteroarylation of Grignard compounds using different catalysts and ligands.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transition-metal catalysis [1] can forge carbon–carbon bonds between functionalized and sensitive substrates which provide new opportunities [2], predominantly in total synthesis, medicinal and chemistry process, as well as in nanotechnology and chemical biology [3]. It is almost impossible nowadays to find an issue of a journal in the field of organic or organometallic chemistry without a donation dealing with some aspect of cross-coupling reactions [4].
The names of Heck [5], Suzuki [6], Stille [7], Sonogashira [8], Negishi [9], and Hiyama [10] are distinguished for their contribution to this chemistry [11] and among them the Kumada coupling has a predominant place which was reported in 1972 by Tamao et al. [12]. In the same year, Corriu independently reported the reaction of β-bromostyrene with phenylmagnesium bromide to afford trans-stilbene in the presence of Ni(acac)2 as catalyst [13]. It is a protocol between an alkyl or aryl Grignard reagent and an aryl, alkyl, or vinyl halocarbon catalyzed based on nickel or palladium catalyst [14]. Palladium and nickel phosphine complexes accompanied by different ligands catalyze the selective cross-coupling reactions of Grignard reagents. This process has opened up the possibility of preparing a large variety of organic compounds by C–C bond formation especially from different organic halides and there have been reported numerous characteristic features of the reaction, such as asymmetric synthesis [15], stereochemical aspects [16], and heterocycle coupling [17].
Cross-coupling reactions such as Kumada-Tamao-Corriu coupling, Suzuki–Miyaura coupling, Negishi coupling, Stille coupling, Hiyama coupling have been overviewed by A. C. Frisch and M. Beller specially for non-activated alkyl halides [18]. Also, a mechanistic perspective of nickel-catalyzed cross-coupling of non-activated alkyl halides was investigated [19]. But now, in the following sections, we discuss the application of different ligands and catalysts to form different carbon–carbon bonds by Kumada-Tamao-Corriu coupling reaction to synthesize organic compounds.
Alkylation of Grignard compounds via alkyl halides
Alkyl halides are non-activated substrates for metal catalyzed C–C coupling reactions due to their reluctance to undergo oxidative addition, and also metal alkyl intermediates are prone to unproductive β-hydride eliminations. However, significant advance as shown in the following paragraphs has been achieved in this field over the recent years to construct C–C bonds by cross-coupling reactions of alkyl halides and Grignard reagents using a nickel, palladium, or iron catalyst.
Yang et al. [20] have used a range of different catalysts and ligands for coupling of inactivated alkyl bromides or iodides with Grignard reagents 1 in Kumada-Tamao-Corriu coupling conditions. Palladium acetate in the presence of a phosphine ligand experimentally was not an effective catalyst. However, electron-donating trialkylphosphine ligands may accelerate the oxidative addition of low-valent palladium aliphatic carbon-halogen cross-couplings, but better performance was observed by using triarylphosphine ligands compared to trialkylphosphine ligands. It was shown that the cross-coupling reaction may be a reductive elimination-controlled process. Thus, Pd2(dba)3 and Ph3P appear to be essential for the catalytic process of Scheme 1.
A mild palladium-catalyzed Kumada-Tamao-Corriu coupling of secondary benzylic bromides 3 with aryl and alkenyl Grignard reagents has been developed by Lopez-Perez et al. [21]. In this protocol a catalytic system capable of minimizing the competitive β-hydride elimination pathway was tested by using Pd(CH3CN)2Cl2 as catalyst and a collection of various ligands (Scheme 2). As a result, bidentate ligands with a large bite angle such as dppf (96º) and, especially, xantphos (5) (111º) turned out to be superb ligands due to their lower reaction time, high yields, and minimized β-hydride elimination. In addition, no transformation was observed when the catalyst loading was reduced to 1 mol %.
Non-activated and functionalized alkyl halides 7 (alkyl halides containing ester, amide, ether, thioether, alcohol, pyrrole, indole, furan, nitrile, conjugated enone) coupled to alkyl, aryl, or heteroaryl Grignard reagents 8 catalyzed by a nickel(II) pincer complex [(MeNN2)NiCl] (6) in a Kumada-Tamao-Corriu cross-coupling reaction (Scheme 3) [22].
A structure–activity relationship has been studied that improved catalysts for Kumada-Tamao-Corriu cross-coupling of secondary alkyl halides such as a series of new nickel(II) complexes 10 including those with tridentate pincer bis(amino)amide ligands (RN2N) and those with bidentate mixed amino-amide ligands (RNN) [23]. Analogous pincer complexes such as 10a and 10b were not efficient due to their inability to activate alkyl halides. The five-coordinate complex 10f was moderately active. Also, the square-planar complexes 10e and 10c were active. But, complex [(HNN)Ni(2,4-lutidine)Cl] (10e) specially was the most efficient catalyst. No significant isomerization products were formed using these catalysts, unlike pincer complex 10a (Scheme 4).
Primary and secondary alkyl halides (RX), depending on the nature of the R group, undergo sp3–sp3 Kumada-Tamao-Corriu coupling with alkyl Grignard reagents by using an iron catalyst (Scheme 5) [24].
Alkyl halides containing ester, amide, ether, acetal, nitrile, thioether, and heterocyclic groups have undergone Kumada-Tamao-Corriu cross-coupling reaction with alkyl Grignard reagents by using [(MeNN2)NiII-Cl] complex 15 as nickel catalyst. The functional group of alkyl halides did not participate in reaction with Grignard reagents (Scheme 6) [25].
Other alkylation of Grignard compounds
Much attention has also been paid to the activation of the relatively stable but commonly encountered C–O bonds in direct functionalizations by Grignard reagents.
Ni-catalyzed alkylation of Grignard compounds via sp3 C–O bond activation in ethers was reported as a Kumada-Tamao-Corriu cross-coupling reaction. With dppe as the ligand, the methylation totally failed. The desired product 17 was isolated in good yield when dppf was applied as the ligand. However NiCl2 and hydrated NiCl2 were not very efficient. Finally, Ni(acac)2 showed a better catalytic ability in the presence of bidentated ligands other than dppe in this coupling reaction (Scheme 7) [26].
An effect of 1,3-butadienes was reported in the nickel-catalyzed cross-coupling reaction of Grignard reagents with alkyl halides and tosylates. The desired product was prepared in high yields by using 1,3-butadiene as an additive instead of phosphine ligands (Scheme 8) [27]. Cross-coupling reactions of organozinc reagents with alkyl halides catalyzed by nickel were also performed using the same protocol [28].
Alkenylation of Grignard compounds via alkyl halides
Electrophiles such as alkenyls have undergone useful Kumada-Tamao-Corriu couplings to emerge as powerful tools in synthetic organic chemistry for the construction of carbon–carbon bonds.
An interesting 3-step sequence for the preparation of exomethylene-type allylsilane 20 from aldehyde via 1,1-dibromo-1-alkene 18 has been investigated. 1,1-Dibromo-1-alkene 18 can be derived from the corresponding aldehyde. Then, the Pd-catalyzed tandem Kumada-Tamao-Corriu cross-coupling reaction of 18 was performed with an excess of trimethysilylmethyl Grignard reagent to produce compound 19 in good to excellent yields. In the process, PPh3 was added to a mixture of Pd(OAc)2 and 18 in THF, stirring for 10 min at room temperature. Me3SiCH2MgCl was added at 0 °C, stirring for 2–12 h at room temperature. After quenching and purification, the desired bisallylsilane 19 was obtained. Finally, protodesilylation of bisallylsilanes 19 by using PPTS provided 2-(trimethylsilylmethyl)-1-alkenes 20 selectively (Scheme 9) [29].
Lithium triarylmagnesiates 22 are rapidly prepared by electron-rich aryl bromides 21, which undergo nickel-catalyzed Kumada-Tamao-Corriu reactions with a diversity of aryl and alkenyl bromides, chlorides, tosylates, and triflates (Scheme 10) [30]. A variety of catalysts were evaluated. As reported by Kumada-type, the observed reactivity of nickel-phosphine catalysts followed this order: NiCl2(dppp) > NiCl2(dppe) > NiCl2(PPh3)2 [31]. In addition, when Pd catalysts were employed, isolated yields were low, and no reaction was observed with Co(acac)2.
High yields of α-fluorostyryls 26 and α-fluorothiophenes 27 have been achieved by a Kumada-Tamao-Corriu cross-coupling reaction (Scheme 11). This reaction is useful in the synthesis of other fluorovinylsubstituted heterocycles of biological attention or in the industrial scale manufacture of fluorostyryl monomers or polymer chemistry [32].
The first stereospecific Mn-catalyzed coupling reaction of aryl Grignard reagents with various alkenyl halides such as 28 was reported by Cahiez et al. (Scheme 12). In this protocol stereospecific coupling was obtained except in the case of the (Z)-3-(2-bromoethenyl)pyridine due to the slow reaction rate of the reductive elimination step. Indeed, Mn(II) species are commonly stable at room temperature [33]. This manganese-catalyzed coupling reaction represents a more environmentally friendly alternative to nickel- or palladium-catalyzed reactions.
Other alkenylation of Grignard compounds
As shown in Scheme 13, vinyl sulfides 30 undergo Kumada-Tamao-Corriu cross-coupling to prepare 1,1-disubstituted olefins 31. Vinyl sulfides were afforded by regioselective catalytic alkyne hydrothiolation of both aromatic and aliphatic alkynes using TpRh(PPh3)2 as catalyst. Then, cross-coupling reactions of alkyl vinyl sulfides with various Grignard reagents (alkyl, vinyl, and aryl) were performed using 10 mol % NiCl2(PPh3)2 as catalyst [34].
The 1,1-Disubstituted alkenes 33 were prepared by Kumada-Tamao-Corriu cross-coupling reaction of cyclic and acyclic vinyl phosphates 32 and magnesium reagents (Scheme 14) [35]. The reactions proceed by simple palladium salts such as PdCl2, without the necessity of phosphine ligands [36].
Cross-coupling of alkenyl/aryl carboxylates 34 with Grignard reagent 35 was performed via C–O bond activation by using FeCl2 as catalyst under mild conditions. Other catalysts were not useful in this protocol. Ligands such as heterocyclic NHC carbenes 37-39 improved the yield, while lower yields were obtained by using other mono- or bidentated phosphine and nitrogen ligands (Scheme 15) [37].
Arylation of Grignard compounds via aryl halides
Aryl halide is another choice of organic precursors in Kumada-Tamao-Corriu cross-coupling reactions. With great efforts from different research groups in this field, significant achievements have been made to synthesize a broad range of organic compounds.
Compatibility of strongly basic carbon-center reagents like Grignard reagents with phosphonium ILs (phosILs) were reported first by Ramnial et al. (Scheme 16) [38]. The NHCs 41 produced in [P66610 +][C9H19COO−] in conjunction with Ni(cod)2 (cod = 1,5-cyclooctadiene) can successfully catalyze Kumada-Tamao-Corriu reactions [39].
Use of 3-Bromo-1-(trifluoromethyl)benzene (43) or its derivatives coupled with methylmagnesium chloride or ethyl magnesium chloride in the presence of NiCl2/xantphos to prepare 3-methyl- or 3-ethyl-1-(trifluoromethyl)benzenes 44 resulted in good yields (Scheme 17). Also, 3-Chloro- and 3-bromo-1-(trifluoromethyl)benzene caused different behaviors in Kumada’s coupling reaction with NiCl2(dppe). As demonstrated in Scheme 17, the reaction between 3-bromo-1-(trifluoromethyl)benzene and ethyl magnesium bromide caused 3-ethyl-1-(trifluoromethyl)benzene in good yield, with a low level of biaryls. Parameters such as the nature of solvent, increase of temperature, and the nature of the metal catalyst did not affect the cross-coupling versus homocoupling of alkylation reaction. Therefore, the effect of nickel ligand was investigated. Finally, the best result was achieved by xantphos [4,5-bis(diphenylphosphino)-9,9-dimethylxanthene] as the nickel ligand [40].
Isopropyl iodide accelerated Kumada-Tamao-Corriu cross-coupling and led to a rapid radical catalysis reaction of a wide range of functionalized aryl- and heteroarylmagnesium reagents 46 with aryl bromides (Scheme 18) [41]. Thus, functionalized biphenyl was obtained by the cross-coupling within 5 min at 25 °C as a single product in high yield.
Arylation of Grignard compounds via aryl bromides
A palladium- and nickel-phosphinous acid catalyst performed a Kumada-Tamao-Corriu reaction with sterically hindered substrates under mild reaction conditions. Multifunctional biaryls 50 were formed in 87–98 % yield even when electron-rich aryl chlorides were used. Also, the reaction ensued with aryl iodides at −20 °C (Scheme 19) [42].
The Kumada-Tamao-Corriu cross-coupling reactions of tertiary alkylmagnesium halide and aryl bromide 51 using nickel as catalyst have been developed. For this cross-coupling reaction, NHCs were identified as the most promising ligand candidates. Thus, the effect of steric and electronic perturbations of the NHC backbone was examined. Increased formation of reduction product was observed, when new sources of NiCl2 were employed alongside ligand L 3 (Scheme 20). Aryl triflates, vinyl chlorides, and vinyl bromides as the electrophilic component also would undergo the same procedure [43].
Kumada-Tamao-Corriu coupling of bromobenzoic acid derivatives 53 with the acetaldehyde enolate synthon (1,3-dioxolan-2-ylmethyl)magnesium bromide (54) proceeded under Pd catalysis with alkoxide additives to display an ortho-directing effect of the carboxylate functionality (Scheme 21) [44].
An applicable cross-coupling reaction of 2-pyridyl organomagnesium reagents 57 was performed by using alkyl-substituted secondary phosphine oxides preligand 58 (Scheme 22) [45]. A catalyst derived from more sterically hindered aryl-substituted preligand proved to be less efficient. Consequently, optimal result was obtained with sterically demanding, non-hygroscopic preligand 58.
Arylation of Grignard compounds via aryl chlorides
A new route to synthesize multisubstituted benzene derivatives was reported by Ishikawa et al. Ortho-selective cross-coupling of dichlorobenzene derivatives 60 was achieved by using Pd2(dba)3 and PCy3 and substrates with directing groups such as OH, CH2OH, NH2, NHAc, and NHBoc. The mechanism of selectivity remains unknown. Use of hydroxylated terphenylphosphines (61) enhanced the reactions of dichlorophenol and dichloroaniline (Scheme 23) [46].
The functionalized linear acene 64 was achieved by using Kumada-Tamao-Corriu cross-coupling of polychlorinated acene 63 containing two ortho substituents with Grignard reagents at low temperature (Scheme 24). Tetramethyltetracene (64) was generated in excellent yield (98 %) by adding MeMgBr (3 equiv per chlorine) to a mixture of 5,6,11,12-tetrachlorotetracene (63) and PEPPSI-IPr (65) at room temperature in 1,4-dioxane. A brief solvent screen confirmed that dioxane gave better results. Lower yields were achieved by using more traditional cross-coupling catalysts such as Pd(OAc)2 with X-Phos and Ru-Phos ligands. Using iron and nickel cross-coupling systems led to the uneffective decomposition of product to the starting material, resulting in no desired product formation. So, treatment of polychlorinated acenes with the PEPPSI-IPr catalyst (65) and MeMgBr produce distorted octamethylnaphthalene [47].
Also, 1,5-Dimethylanthracene (67) was achieved in a moderate yield of 51 % by the reaction of 1,5-dichloroanthracene (66) with methylmagnesium chloride using nickel catalysts [48]. There are only a few examples for synthesis of compound 67 in the literature [49, 50] because of producing a mixture of 1,5- and 1,8-dimethylanthracene in the synthesis of these compounds. The reported procedures afforded a mixture of 1,5- and 1,8-dimethyl analogues, which has to be purified by crystallization. This problem was solved by the Kumada-Tamao-Corriu coupling reaction as illustrated in Scheme 25.
Arylation of Grignard compounds via aryl iodides
A Pd-catalyzed Kumada-Tamao-Corriu coupling was performed at temperatures ranging from −20 to −65 °C using polyfunctionalized Grignard reagents 69-71 to synthesize a broad spectrum of valuable compounds, including heterocyclic biaryls and polyfluoro biaryls (Scheme 26) [51].
Thienyl Grignard 74 and either 1,3- or 1,3,5-bromo- or iodobenzenes 75 were coupled to form meta-substituted thienylbenzenes in a Kumada-Tamao-Corriu reaction catalyzed by PdCl2(dppf) (Scheme 27) [52]. Iodo-substituted benzenes gave the trisubstituted product under mild synthetic conditions (rt for 48 h) whereas bromo-substituted benzenes only gave mono- and disubstituted products under heating overnight (60 °C).
Other arylation of Grignard compounds
A nickel-catalyzed Kumada-Tamao-Corriu coupling of diaryl sulfates 79 afforded the products 81 which caused a new synthetic procedure for constructing biaryls and showed the potential of using sulfates as a new kind of capable, green electrophiles in different cross-coupling reactions (Scheme 28) [53]. Ni-catalysts with different ligands were screened in this Kumada-Tamao-Corriu cross-coupling reaction, and PCy3 was proved to be the best one. In addition, other monodentate and bidentate phosphine ligands, such as PPh3, dppe, dppp, and dppf, were not efficient. However, some common catalysts of Kumada-Tamao-Corriu couplings such as Pd, Fe, and Co complexes, entirely failed. Finally, a good yield of desired product 81 was obtained in the presence of Ni(PCy3)2Cl2 and the additional PCy3 in Et2O after 2 h.
Low reactive aryl tosylates 82 have been coupled with aryl Grignard reagents 83 in the presence of 0.1–1.0 mol % Pd(dba)2 and PPF-t-Bu in toluene at either room temperature or 80 °C to produce the respective biaryls 84 in good yields (Scheme 29) [54].
Kumada-Tamao-Corriu cross-coupling reactions of magnesium phenolate 85 with Grignard reagents 86 to construct biaryl scaffolds 87 were afforded by Shi et al. This study suggested that MO− species could be a useful leaving group for different transformations. Screening of the reaction conditions indicated that nonpolar solvents, a 3:1 mixture of toluene and diisopropyl ether (DIPE), were suitable for this transformation. This solvent system in the presence of PCy3 as a ligand showed the highest reactivity for this transformation (Scheme 30) [55].
Grignard reagents have been utilized as the coupling partners in direct cross-coupling of C–H bonds through cobalt catalysis. This was the first regioselective C–H transformation of benzo[h]quinolone (88) and phenylpyridine derivatives with Grignard reagents at room temperature that has a significant place in the field. In the absence of catalyst, direct nucleophilic attack by Grignard reagents at the 2-position of pyridine derivatives occurred due to decrease in the reactivity of RMgBr. Grignard reagents in a solution of THF, TMEDA, and 2,3-dichlorobutane were added to a Schlenk tube containing benzo[h]quinoline and Co(acac)3. The reaction mixture was stirred at rt for 48 h to produce 89 (Scheme 31) [56].
Heteroarylation of Grignard compounds
4-Allylisoindoline (92) has been prepared by a practical, cost-effective Kumada-Tamao-Corriu coupling reaction (Scheme 32). Olefin isomerization as a side reaction was suppressed by examination of various reaction parameters. The first catalyst screen for this reaction with allylmagnesium reagents 91 is reported by Zacuto et al. [57]. A comparison between Pd sources of catalysts and readily available ligands led to the selection of (neopentyl)(t-Bu)2P·HBF4 as the best one.
Room temperature Kumada-Tamao-Corriu cross-coupling of unactivated aryl chlorides, vinyl chlorides, and heteroaryl chlorides 93 catalyzed by N-heterocylic carbene-based nickel(II) complexes 96-99 is reported by Zhenxing Xi et al. (Scheme 33). The stabilization effect of the NHC ligands provides high catalytic activities of these nickel complexes [58].
The significance of azine-containing biaryls 102 in pharmaceutical and material chemistry has impelled many methods such as Kumada-Tamao-Corriu coupling for their synthesis [59]. Kumada et al. [60] reported the use of 3-pyridylmagnesium chloride (100) in palladium-catalyzed Kumada-Tamao-Corriu couplings of aryl iodides. However, use of a Ni(acac)2/dppe catalyst was found to be optimal with aryl bromide substrates (Scheme 34). Only heterocyclic aryl bromide and chlorides are reported with 34–76 % yield.
A selective method for the synthesis of dihexyl-2,2′-bithiophenes 107, 108 starting from 2-bromo-3-alkylthiophene 103 or 2-bromo-4-alkylthiophene 104 using catalytic amounts of Ni(dppp)Cl2 was reported [61]. Then 107 and 108 were obtained as head-to-tail and tail-to-tail regioisomers in high yields and excellent selectivity using a Kumada-Tamao-Corriu cross-coupling reaction (Scheme 35).
Synthesis of (S)-macrostomine (116) has been skilled in five steps (Scheme 36). One step was a Kumada-Tamao-Corriu cross-coupling reaction on a 1-chloroisoquinoline intermediate 114 that provided the plant alkaloid [62]. The appropriate catalyst and optimization of conditions have been investigated. Consequently, a ligand-free reaction with Ni(acac)2 as catalyst was selected to afford the enantiopure natural product 116 in 63 % yield. Actually, the enantiopure natural product was obtained by separation of a diastereomeric mixture of 113.
After halogenations of 3-arylimidazo[1,5-a]pyridines, Kumada-Tamao-Corriu cross-coupling of the produced 1-iodo-3-arylimidazo[1,5-a]pyridines 117 and aryl Grignard reagents was performed to prepare 1,3-diarylated imidazo[1,5-a]pyridines 118 (Scheme 37). Aryl Grignard reagent was added dropwise to a solution of 1-iodo-3-arylimidazopyridine 117 and Ni(dppp)Cl2 in THF at 0 °C under Ar atmosphere. After addition of Grignard reagent, the reaction mixture was stirred at room temperature to produce the desired product [63].
Phosphatyloxy-substituted heterocycles 119 undergo Kumada-Tamao-Corriu cross-coupling. This conversion proceeded most efficiently in the attendance of Pd2dba3/CyPF-t-Bu mixture. These heterocycles 120 were synthesized via migration/cycloisomerization approach by Schwier et al. [64] (Scheme 38).
Mild room temperature palladium-catalyzed C3-arylation of 2(1H)-pyrazinones 121 is reported via a desulfitative Kumada-Tamao-Corriu cross-coupling reaction (Scheme 39). In most cases, the desired cross-coupled products provide good to excellent yields [65]. First Fe(acac)3 or Ni-(OAc)2/PPh3 were examined as catalyst for the desulfitative cross-coupling. But an extensive amount of aryl–aryl homocoupling product was produced rather than the corresponding cross-coupled product. Thus, Pd was utilized as Pd(dba)2 with TFP (tri-2-furylphosphine) as ligand and THF as solvent.
Asymmetric Kumada-Tamao-Corriu cross-coupling reactions
Catalytic asymmetric Kumada-Tamao-Corriu carbon–carbon bond formation is one of the most significant reactions in synthetic organic chemistry. The development of new chiral ligands for use in this field has continued to undergo rapid growth.
An asymmetric Kumada-Tamao-Corriu reaction of alkyl electrophiles is reported by Lou et al. (Scheme 40). In this protocol, racemic α-bromoketones 123 coupled to aryl Grignard reagents [66]. Consequently, readily available bis(oxazolines) are shown for the first time to be effective ligands for cross-couplings of alkyl electrophiles.
Using Pd2(dba)3·CHCl3 as catalyst and N-aryl axially chiral mimetic-type ligands 128a-128d afforded good enantioselectivities in an asymmetric Kumada-Tamao-Corriu cross-coupling reaction (Scheme 41) [67]. Also, other palladium and nickel complexes have been investigated. Thus, Pd2(dba)3·CHCl3 is the most effective complex. The ligands possessing a pendant oxygen group exhibited faster reaction rate than ligands possessing a pendant nitrogen group. Ligands possessing the pendant nitrogen substituent as a strong coordinating group perform the internal coordination of the pendant nitrogen group to palladium (129). Such a problem was solved by using a piperidine based ligand, because coordination of the pendant nitrogen group to the internal palladium seemed to be torsionally unfavorable.
Conclusions
This review highlights recent developments of carbon–carbon bond formation of Kumada-Tamao-Corriu cross-coupling reactions. Several selected examples of alkylation, alkenylation, arylation, and heteroarylation of Grignard compounds and some examples of asymmetric Kumada-Tamao-Corriu cross-coupling reaction using various catalysts and ligands are discussed. Although many advances dealing with the Kumada-Tamao-Corriu coupling reaction have been made in the last few years, it will persist to be a fast-moving topic for the next several years. We hope this review will create strong interest among the wide-ranging readership of this journal.
References
Kulkarni AA, Daugulis O (2009) Synthesis 24:4087
Zapf A (2003) Angew Chem Int Ed 42:5394
Nicolaou KC, Bulger PG, Sarlah D (2005) Angew Chem Int Ed 44:4442
Marion N, Nolan SP (2008) Acc Chem Res 41:1440
Heck RF, Nolley JP (1972) J Org Chem 37:2320
Miyaura N, Yamada K, Suzuki AA (1979) Tetrahedron Lett 20:3437
Milstein D, Stille JK (1979) J Am Chem Soc 101:4992
Sonogashira K, Tohda Y, Hagihara N (1975) Tetrahedron Lett 16:4467
Baba S, Negishi E (1976) J Am Chem Soc 98:6729
Hatanaka Y, Hiyama T (1988) J Org Chem 53:918
Li CJ (2003) Angew Chem Int Ed 42:4856
Tamao K, Sumitani K, Kumada M (1972) J Am Chem Soc 94:4374
Corriu RJP, Masse JP (1972) J Chem Soc Chem Commun 144
Tamao K (2002) J Organomet Chem 653:23
Kiso Y, Tamao K, Miyake N, Yamamoto K, Kumada M (1974) Tetrahedron Lett 15:3
Zembayashi M, Tamao K, Kumada M (1975) Tetrahedron Lett 16:1719
Thorsett ED, Stermitz FR (1973) J Heterocycl Chem 10:243
Frisch AC, Beller M (2005) Angew Chem Int Ed 44:674
Hu X (2011) Chem Sci 2:1867
Yang LM, Huang LF, Luh TY (2004) Org Lett 6:1461
López-Pérez A, Adrio J, Carretero JC (2009) Org Lett 11:5514
Vechorkin O, Proust V, Hu X (2009) J Am Chem Soc 131:9756
Ren P, Vechorkin O, Von Allmen K, Scopelliti R, Hu X (2011) J Am Chem Soc 133:7084
Dongol KG, Koh H, Sau M, Chai CLL (2007) Adv Synth Catal 349:1015
Vechorkin O, Hu X (2009) Angew Chem Int Ed 48:2937
Guan BT, Xiang SK, Wang BQ, Sun ZP, Wang Y, Zhao KQ, Shi ZJ (2008) J Am Chem Soc 130:3268
Terao J, Watanabe H, Ikumi A, Kuniyasu H, Kambe N (2002) J Am Chem Soc 124:4222
Terao J, Todo H, Watanabe H, Ikumi A, Kambe N (2004) Angew Chem Int Ed 43:6180
Uenishi J, Iwamoto T, Ohmi M (2007) Tetrahedron Lett 48:1237
Lau SYW, Hughes G, O’Shea PD, Davies IW (2007) Org Lett 9:2239
Tamao K, Sumitani M, Kumada M (1972) J Am Chem Soc 94:4374
Qiu J, Gyorokos A, Tarasow TM, Guiles J (2008) J Org Chem 73:9775
Cahiez G, Gager O, Lecomte F (2008) Org Lett 10:5255
Sabarre A, Love J (2008) Org Lett 10:3941
Yu DG, Li BJ, Shi ZJ (2010) Acc Chem Res 43:1486
Gauthier D, Beckendorf S, Gøgsig TM, Lindhardt AT, Skrydstrup T (2009) J Org Chem 74:3536
Li BJ, Xu L, Wu ZH, Guan BT, Sun CL, Wang BQ, Shi ZJ (2009) J Am Chem Soc 131:14656
Ramnial T, Taylor SA, Bender ML, Gorodetsky B, Lee PTK, Dickie DA, McCollum BM, Pye CC, Walsby CJ, Clyburne JAC (2008) J Org Chem 73:801
Liu Y, Wang SS, Liu W, Wan QX, Wu HH, Gao GH (2009) Curr Org Chem 13:1322
Roques N, Saint-Jalmes L (2006) Tetrahedron Lett 47:3375
Manolikakes G, Knochel P (2009) Angew Chem Int Ed 48:205
Wolf C, Xu H (2008) J Org Chem 73:162
Joshi-Pangu A, Wang CY, Biscoe MR (2011) J Am Chem Soc 133:8478
Houpis IN, Van Hoeck JP, Tilstam U (2007) Synlett 2179
Ackermann L, Potukuchi HK, Kapdi AR, Schulzke C (2010) Chem Eur J 16:3300
Ishikawa S, Manabe K (2007) Org Lett 9:5593
Yagodkin E, Douglas CJ (2010) Tetrahedron Lett 51:3037
Bringmann S, Ahmed SA, Hartmann R, Mattay J (2011) Synthesis 2291
Clar E, Mackay CC (1972) Tetrahedron 28:5049
Cristol SJ, Caspar ML (1967) J Org Chem 33:2020
Martin R, Buchwald SL (2007) J Am Chem Soc 129:3844
Cornacchio ALP, Price JT, Jennings MC, McDonald R, Staroverov VN, Jones ND (2009) J Org Chem 74:530
Guan BT, Lu XY, Zheng Y, Yu DG, Wu T, Li KN, Li BJ, Shi ZJ (2010) Org Lett 12:396
Roy AH, Hartwig JF (2003) J Am Chem Soc 125:8704
Yu DG, Li BJ, Zheng SF, Guan BT, Wang BQ, Shi ZJ (2010) Angew Chem Int Ed 49:4566
Li B, Wu ZH, Gu YF, Sun CL, Wang BQ, Shi ZJ (2011) Angew Chem Int Ed 50:1109
Zacuto MJ, Shultz CS, Journet M (2011) Org Process Res Dev 15:158
Xi Z, Liu B, Chen W (2008) J Org Chem 73:3954
Campeau LC, Fagnou K (2007) Chem Soc Rev 36:1058
Tamao K, Kodama S, Nakajima I, Kumada M, Minato A, Suzuki K (1982) Tetrahedron 38:3347
El-Shehawy AA, Abdo NI, El-Barbary AA, Lee JS (2010) Tetrahedron Lett 51:4526
Enamorado MF, Ondachi PW, Comins DL (2010) Org Lett 12:4513
Shibahara F, Yamaguchi E, Kitagawa A, Imai A, Murai T (2009) Tetrahedron 65:5062
Schwier T, Sromek AW, Yap DML, Chernyak D, Gevorgyan V (2007) J Am Chem Soc 129:9868
Mehta VP, Modha SG, Van Der Eycken E (2009) J Org Chem 74:6870
Lou S, Fu GC (2010) J Am Chem Soc 132:1264
Horibe H, Fukuda Y, Kondo K, Okuno H, Murakami Y, Aoyama T (2004) Tetrahedron 60:10701
Acknowledgments
We are grateful to the Alzahra Research Council for partial financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heravi, M.M., Hajiabbasi, P. Recent advances in Kumada-Tamao-Corriu cross-coupling reaction catalyzed by different ligands. Monatsh Chem 143, 1575–1592 (2012). https://doi.org/10.1007/s00706-012-0838-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-012-0838-x