Abstract
Aerosol (PM10) samples were collected and its precursor gases, i.e., NH3, NO, NO2, and SO2 measured over Bay of Bengal (BoB) during winter months of December 2008 to January 2009 to understand the relationship between particular matter (PM) and precursor gases. The observations were done under the winter phase of Integrated Campaign on Aerosols, gases and Radiation Budget (W_ICARB). The distribution of water-soluble inorganic ionic composition (WSIC) and its interaction with precursor gases over BoB are reported in present case. Average atmospheric concentration of NH3, NO, NO2, and SO2 were recorded as 4.78 ± 1.68, 1.89 ± 1.26, 0.31 ± 0.14, and 0.80 ± 0.30 μg m−3, whereas WSIC component of PM10, i.e., NH4 +, SO4 2−, NO3 −, and Cl− were recorded as 1.96 ± 1.66, 8.68 ± 3.75, 1.92 ± 1.75, and 2.48 ± 0.78 μg m−3, respectively. In the present case, abundance of nss-SO4 2− in the particulate matter is recorded as 18 %. It suggests the possibility of long-range transport as well as marine biogenic origin. Higher SO4 2−/(SO2 + SO4 2−) equivalent molar ratio during the campaign indicates the gas-to-particle conversion with great efficiency over the study region.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Atmospheric NH3 is the reduced form of nitrogen which plays important role in various environmental concerns. In the atmosphere and ocean, NH3 and its ionized form NH4 + are ubiquitous. Naturally and anthropogenically produced NH x (NH3 + NH4 +) are transported through the atmosphere and generally their concentration in air decreases as the distance from land increases. It has been suggested that in pre-industrial times the oceans were probably a net source of NH x of the continents (Duce et al. 1991), but this is not the case today (Sutton et al. 1995, 2000). NH x is produced in surface water by the biological reduction of nitrate (either directly or via the degradation of biologically synthesized organic nitrogenous material). In solution, NH x is partitioned between NH4 + and NH3 according to equilibrium thermodynamics: the proportion of NH x that occurs as NH3 (depending on pH, temperature and ionic strength of the medium) is available for emission to the atmosphere (Aneja et al. 2001). NH3 is also emitted to the atmosphere by plants, animals, and its environments, by soil micro-organisms and by various industrial and agricultural processes, including the direct volatilization of solid NH4NO3 salts and fertilizers (Sutton et al. 2000; Li et al. 2006; Sharma et al. 2010a, b). There is also evidence of volcanic source of NH x to the atmosphere (Uematsu et al. 2004) and of substantial NH3 emissions from seabird and colonies (Blackall et al. 2007; Theobald et al. 2006).
Atmospheric NH3 plays an important role not only in formation of secondary aerosols while combining with atmospheric acid gases (sulfuric acid, nitric acid, and hydrochloric acid) but also contributes to adverse health effects, mainly respiratory diseases (Warneck 1988) and climate change. Sulfuric acid (H2SO4) and nitric acid (HNO3) are the major acid gases in the atmosphere that occur from oxidation of SO2 and NO x , respectively. These acid gases are neutralized by NH3 in the atmosphere and thereby forming NH4HSO4 and (NH4)2SO4 and NH4NO3 aerosols, respectively.
Gas to particle conversion may be accomplished by condensation which depends on water vapor and concentration of acid gases in the atmosphere (Stelson and Seinfeld 1982; Seinfeld 1986). The formation of aerosols over the continent occurs either through various natural processes or due to anthropogenic activities. On the other hand, oceanic aerosols are mostly of natural origin produced in situ by action of wind on the ocean surface and phytoplankton activity by producing di-methyl sulfide (Charlson et al. 1987). Hence, the sources and chemical composition of continental aerosols may be different from the oceanic aerosols. However, the experimental results have revealed the presence of continental aerosols in oceanic environment (Garrett and Hobbs 1995; Krishnamurti et al. 1998; Rajeev et al. 2000) and presence of oceanic aerosols in the continental environment (Carmichael et al. 1997; Teinila et al. 2000).
The ‘Integrated Campaign for Aerosols, gases and Radiation Budget (ICARB)’ initiated under the Geosphere Biosphere Programme of Indian Space Research Organization (ISRO-GBP) has the major objective of mapping aerosols and trace gases over Bay of Bengal (BoB), identifying transport mechanisms and studying their impacts on regional radiative forcing. The W_ICARB was conducted during the winter period of December 27, 2008 to January 30, 2009. For the first time, ship-based measurements were carried out with vast longitudinal coverage over BoB, including southern and eastern parts of this oceanic region (Aastar and Nair 2010). During this period of study (northern winter), the synoptic wind was northeasterly (Asnani 1993) favouring an outflow of continental airmass over to BoB and Indian Ocean region. The atmospheric NH3, NO, and NO2 have been monitored precisely over BoB on chemiluminescence method. In the present study, we have estimated the concentration and distribution of WSIC of PM10, trace gases and their interaction over BoB during W_ICARB campaign. The aerosol samples were collected simultaneously along with the measurement of trace gases (NH3, NO, NO2. and SO2) on board Sagar Kanya over BoB.
2 Experimental setup and cruise track of W_ICARB
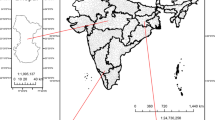
Measurement of PM10 and trace gases (NH3, NO, NO2, SO2) were done over BoB under W_ICARB campaign during 27 December 2008 to 30 January 2009 onboard Sagar Kanya (SK-254) cruise as per scheduled track given in Fig. 1. The cruise tracks covered the region ranging from 21°N to 3.5°N latitude and 76.3°E to 98°E longitudes over BoB. The cruise movements started from 27 December 2008 from Chennai and ended at Kochi (9.96°N, 76.3°E), India on 31 January 2009. The ship halt time is also indicated in the figure by open circles. Sampling inlets of all analysers, particle sampler and the portable weather station were stationed at same height (about 11 m from sea surface) and same location of the deck of the ship. These instruments were operated continuously for almost 1 month during winter (28 December 2008 to 25 January 2009).
2.1 Sampling of PM10 and analysis
PM10 samples were collected by Particle Sampler (Envirotech, APM 460NL) having flow rate 1.12 m3 min−1 (accuracy ±2 %) placed on the deck of the ship at about 11 m height from sea level. Samples were collected on Quartz Microfibre filter (QM-A) papers (20 × 25 cm2) on 18 h basis (1200–0600 hours). The collected samples (n = 28) were stored in polythene bags and kept in a desiccator till analysis. The samples were brought to the laboratory for further extraction and analysis.
For the extraction, a 5 × 5 cm2 size portion was cut from QM-A filter papers and transferred into 50 ml deionized water. The water-soluble fraction was extracted using ultrasonic system for 60 min. Cations (Na+, K+, NH4 +, Ca2+, and Mg2+) and anions (Cl−, F−, SO4 2− and NO3 −) were determined using Ion Chromatograph (DIONEX ICS-3000). The analytical conditions employed for the separation anion and cation were an anion separation column (IonPac AS11-HC, 4 × 250 mm) with a guard column (IonPac AG11-HC, 4 × 50 mm) and a cation separation column (IonPac CS17-HC, 4 × 250 mm) with a guard column (IonPac CG17-HC, 4 × 50 mm). Post-column eluent suppression was carried out using self-regenerating suppressor ASRS-300 and CSRS-300 (4 mm) for anion and cation analysis, respectively. Eluents used for the separation of anion and cation were 20 mM NaOH (50 % w/w) and 5 mM MSA at the flow rate of 1.5 and 1 ml/min, respectively. Suppressed conductivity detection was used to monitor the eluted analysts. The IC system was fitted with a 25-μL sample loop that was used to introduce the sample manually. Working standards were prepared from stock standard solutions procured from DIONEX, USA. All of the standard solutions were filtered using 0.45 μm nylon membrane filters (Millipore) and degassed by ultrasonication. Several blank filters were also analyzed for cations and anions. The analytical errors (repeatability) were estimated to be 3 % based on triplicate (n = 3) analysis.
2.2 Measurement of trace gases
Concentration of atmospheric NH3 was measured continuously over BoB during W_ICARB using NH3-analyzer (Model: CLD88CYp, M/s. ECO Physics AG, Switzerland) operating on chemiluminescence method. In this analyzer two catalytic converters of different characteristics allow sequential detection of NO x and NO x-amines by converting them into NO. Concentration of atmospheric NH3 was calculated from the difference between NO x and NO x-amine (NH3 = NO x-amine − NO x ). The measurement ranges of NH3 analyzer vary automatically from 0–5 to 0–5,000 ppb (accuracy ±0.050 ppb). NO and NO2 were measured continuously using NO x -analyzer (Model: CLD88p, M/s. ECO Physics AG, Switzerland) with photo catalytic converter (Model: PLC860, M/s. ECO Physics AG, Switzerland) also operating based on chemiluminescence method. The estimation ranges of NOx-analyzer vary from 0–5 to 0–5,000 ppb (accuracy ±0.050 ppb). The response time of these analyzers are less than 1 s with a signal noise of 1 % of measured value and the estimation efficiency is >90 %. The zero air calibration of these analyzers was done using Pure Air Generator (Model: PAG-003, M/s. ECO Physics AG, Switzerland) having air pollutant elimination capacity <0.010 ppb. The span calibrations of these analyzers were calibrated and validated using NIST-USA traceable certified NO gas (250 ppb ± 2.5 %, M/s Spectra Gases Inc., USA). The analyzers were shown less than ±1 % error during calibration. Zero and span calibrations (before and after the measurement) of these analyzers were done for a week time to get the reproducible values. Atmospheric SO2 was measured continuously using calibrated SO2-analyzer (Model: APSA 360A, M/s. Horiba Ltd, Japan) at the same location. The lowest detection level is 0.50 ppb at a flow rate of 0.8 l min−1. The instrument was calibrated every day using in-built calibrator for zero and span (permutation tube).
The gas analysers were used to record the atmospheric NH3, NO, NO2, and SO2 concentration at every 1-min interval throughout the study period. The concentrations of all the above trace gases were converted into μg m−3 by respective factors (0.71 for NH3, 1.25 for NO, 1.91 for NO2 and 2.87 for SO2) of gases to keep uniformity of the results. Statistical analysis of all the data sets collected during the study period was performed using standard recommended methods. Meteorological parameters such as temperature (accuracy ±1 °C), relative humidity (accuracy ±2 %), wind direction (accuracy ±3°), and wind speed (accuracy ±2 % of full scale) were recorded at hourly intervals using calibrated portable weather station.
The Sea-Viewing Wide-Field Sensor (SeaWiFS) maps are also used to understand the temporal and spatial variability in the ocean and also to know how chlorophyll variability may be related to other fundamental biogeochemical measurements (phytoplankton chlorophyll) over BoB during W_ICARB.
3 Results and discussion
3.1 Water-soluble inorganic ions composition
Abundance of PM10 over BoB was observed during the study with range from 22.5 to 107.2 μg m−3 and an average value of 55.9 ± 26.3 μg m−3. Typically average concentration of PM10 in northern BoB (64.4 ± 26.9 μg m−3) is observed higher than in eastern, south-eastern and southern (46.62 ± 11.95 μg m−3) part of BoB. A positive latitudinal gradient (2.23) is also noticed in PM10 mass concentration from south to north BoB. The highest value of 107.2 μg m−3 observed in the north BOB at around 18.4°N 87.1°E 88.1 μg m−3 in the south-eastern part of BoB around 8.22°N 91.7°E indicated a continental influence of pollution over BoB. Table 1 shows the mass concentration (μg m−3) of different water-soluble inorganic ions (Na+, K+, NH4 +, Mg2+, Ca2+, F−, Cl−, NO3 −, and SO4 2−) observed over the BoB during W_ICARB. Average concentration of anion (3.92 μg m−3), and cation (1.99 μg m−3) is recorded to be ~10 % of total aerosol loading (PM10).
WSIC (NH4 +, SO4 2−, NO3 −, Cl−, F−, Na+, K+, Ca2+ and, Mg2+) of marine aerosol constitutes of non-sea-salt fraction (nss) and sea salt fraction (ssf). Percentages non-sea-salt fraction (nss) and sea salt fraction (ssf) of the WSIC concentration are estimated to understand the contribution of major ions other than those from the marine sources (Keene et al. 1986) using the relation,
where, X is components, r is ratio X/Na+ in sea water and the values of r were taken as 1.8, 0.25, 0.04, 0.04, and 0.12 for Cl−, SO4 2−, K+, Ca2+ and Mg2+, respectively (Duce et al. 1983, 1986; Keene et al. 1986).
Figure 2a shows spatial distribution of WSIC (NH4 +, SO4 2−, NO3 −, Cl−, K+, Ca2+) over BoB region. The variation in the concentration of nss-SO4 2− (range 1.16–31.23 μg m−3, average 9.21 ± 6.96 μg m−3), and nss-K+ (range 0.16–4.41 μg m−3, average 1.25 ± 0.95 μg m−3) was noticed. Highest concentration of nss-K+ (average 1.65 ± 1.22 μg m−3) was noticed over northern BoB as compared with eastern, south-eastern, and southern BoB (average 0.95 ± 0.44 μg m−3), indicating the existence of nss components due to continental outflow, specifically over the north BoB. Similar observations were also reported by Reddy et al. (2008) over Arabian Sea and BoB during ICARB campaign 2006.
Particulate NH4 + concentration shows maximum value (5.49 μg m−3) near Kolkata region, the north-eastern coast of BoB. This resembles the earlier observation of Biswas et al. (2005) who reported NH3 exchange near at Sunderban region. A very low value of NH4 + is encountered in the middle and southern parts of BoB with decreasing latitudinal pattern. Gupta et al. (2003) have shown earlier that when NH3/NH4 + >1, nature of aerosol is basic and transformation of gaseous NH3 into particulate NH4 + is less. In the present study, the average ratio of NH3/NH4 + >1 near the coastal region suggests weaker transformation from NH3 into NH4 +. Gaseous NH3 normally attains equilibrium forming particulate NH4 + in the presence of NO3 − and SO4 2−. Gaseous NH3 converting to NH4 + may react with SO4 2−, forming fine particle aerosol and may turn into coarse aerosol particle. On the other hand, tendency of gaseous NH3 to condense into ammonium nitrate depends on relative humidity (RH) and temperature (Seinfeld and Pandis 2006).
High concentration of NO3 − (6.0–6.5 μg m−3) is reported at two regions over BoB: one is near Chennai and another one is near Kolkata. The presence of such high NO3 − could be due to the reduced deposition, high NOx emission from ship (Lawrence et al. 1999), and the presence of marine-related species as well as availability of Na+ sufficient concentration of NH4 + being not available to neutralize particulate NO3 −. Several studies have also reported that high concentration NO3 − of marine region is observed mainly in the coarse mode (Athanasopoulou et al. 2008; Bardouki et al. 2003; Eleftheriadis et al. 1998a, b). Since Chennai and Kolkata region is highly dense area of shipping routes, significant portion of NO x converting to HNO3, may react with sea salt aerosol (SSA) fraction in the form of NaNO3 (Huebert et al. 1996; Mcinnes et al. 1994).
Like NH4 +, high concentration of SO4 2− is noticed near coastal region of Chennai and Kolkata. In the middle of BoB and southern BoB, SO4 2− is comparatively low as compared with coastal area. Spatial distribution of particulate SO4 2− may be dependent on various factors such as relative amounts of gaseous NH3 and H2SO4, long-range transport of nss components and emissions from sea salt component. The nss component of SO4 2− may be linked to its biological origin, anthropogenic origin, and natural gypsum. Availability of anthropogenic component of SO4 2− and NO3 − may be interpreted from the deficit of Cl−. Normally, particulate SO4 2− and NO3 − increase if the Cl− deficit is assumed to be solely due to HCl displacement via reactions involving strong mineral acids such as H2SO4 and HNO3 aerosol (Mcinnes et al. 1994; Sturges and Barrie 1988).
In the present case, the Cl− deficit varies from 32.3 to 82.2 % with an average value of 55.4 % over BoB. The large Cl− deficit over BoB indicates the dominance of finer aerosols (anthropogenic input) suggesting the role of particulate SO4 2− and NO3 −. Cl− deficit also reflects the affinity of sulfate ion towards Na+ over NH4 +; this could be the region of existence of NH3/NH4 + >1. This observation is also supported by relatively high abundance of nss-SO4 2−, which is one of the main neutralizing species of NH4 + particulate.
From the present study, it is so far observed that the primary aerosol Mg2+, Cl−, and Na+ are originated from sea salt as their ratio represent standard sea salt ratios (Reddy et al. 1986). It is also observed that 16–18 % fraction of the major ions SO4 2−, Ca2+, K+, etc. are of sea salt origin only. Major fractions of SO4 2−, Ca2+, and K+ are identified as nss components and might ++be contributed from other sources like anthropogenic or crustal influences. To understand the anthropogenic contribution or crustal influences, possible chemical formulation among nss-SO4 2−, nss-Ca2+, nss-K+, NH4 +, and NO3 − could be identified by using ternary plots in the latter part of discussion. Possible transformation of precursor gases (NH3, NO, NO2, and SO2) into WSIC as well as influences of “on-the way” conversion will be discussed later.
3.2 Trace gases (NH3, NO, NO2, SO2)
The concentration of trace gases, i.e., NH3, NO, NO2, and SO2 were also measured over BoB during W_ICARB campaign. The average values of aerosol precursor gases with day and night average values are summarized in Table 2.
Figure 2b represents the spatial distribution of trace gases (NH3, NO, SO2) over BoB region. The average concentration of atmospheric NH3 during campaign is recorded as 4.78 ± 1.68 μg m−3 with a range of 0.23–14.34 μg m−3. Concentration of ambient NH3 gas in the present study is almost one order higher than the earlier reported values over central Indian Ocean as well as BoB (Norman and Leck 2005; Schafer et al. 1993). Norman and Leck (2005) have reported NH3 of the order of 0.05–0.2 nmol m−3 (0.0085–0.0034 μg m−3), whereas Gibb et al. (1999) have reported 10–20 nmol m−3 (0.17–0.34 μg m−3) in the central Indian Ocean and 2.5–5.6 nmol m−3 (0.043–0.095 μg m−3) over coastal Arabian Sea, and 0.4–1.8 nmol m−3 (0.007–0.031 μg m−3) over remote Arabian Sea. Schafer et al. (1993) have reported the concentration of NH3 gas over BoB of the order of 14.29–29.29 nmol m−3 (0.243–0.500 μg m−3). Johnson et al. (2008a, b) have reviewed the measurement of ambient NH3 gases along with sea water NHx (NH3 + NH4 +) over Pacific, Atlantic, and Indian Ocean. Table 3 summarizes the results on concentration of ambient NH3 over ocean reported by different researchers.
A strong positive west–east (zonal) gradient (0.18) is observed in ambient NH3 concentration (6.0–9.0 μg m−3) over the area close to the west coast of BoB as compared with other parts of the BoB (Fig. 2). It is to be noted that agricultural activities (Sharma et al. 2010a, b), livestock, biomass burning, and transport might contribute to the emission of large amount of NH3 (Sutton et al. 1995, 2000). Khemani et al. (1987) reported the concentration of NH3 in coastal region of BoB of the order of 1.4–2.9 μg m−3. Carmichael et al. (2003) also reported of high NH3 concentration (6.4–7.1 μg m−3) at two sites (Bhubaneswar and Berhampur) of western coast of BoB. West–east positive gradient observed in the western coast of BoB could be due to transport of NH3 locally. Since we do not have any record of dissolved NH3 in seawater and NH4 + in the sea water, it is difficult to comment on comparative quantification of biogenic oceanic source of NH3 due to phytoplankton and sea birds as compared with anthropogenic activities. Norman and Leck (2005) have reported distribution of NH3 with the range of 1.1–3.2 nmol m−3 (0.019–0.054 μg m−3) in the marine boundary layer over Atlantic and South Indian Ocean during Cruise99. They have also connected peak value of NH3 to biomass combustion and dust sources on the African continent. Compared with these data, it may be concluded that NH3 concentration over BoB is quite high. Since lifetime of ambient NH3 gas is short (1–3 days), particularly in the humid oceanic atmosphere, conversion to particulate NH4 + is supposed to be very fast (30 % h−1). In the middle region of BoB and southern part of BoB, ambient NH3 is of the order of 1–2 μg m−3.
Observation of nitric oxide (NO) over BoB is not reported so far except over Indian Ocean (Naja et al. 1999; Rhoads et al. 1997). The reported value over Arabian Sea and North Indian Ocean varies in the range of 0.06–0.19 μg m−3. In the present study, average concentration of NO was of the order of 1.89 μg m−3 with day/night ratio 0.98, one order higher than the reported value over Arabian Sea. Spatial distribution of NO shows two peaks in the southern part of BoB with low value in the coastal region and middle of BoB. The peak value of NO resembles the peak of SST and RH. Due to short lifetime, source of NO in the southern BoB is obviously marine rather than continental transport. The conversion from gaseous NO to particulate NO3 − is unlikely in the southern BoB.
Observation of in situ NO2 over Indian Ocean, particularly BoB, is very limited and only supported by satellite observations (Kunhikrishnan et al. 2004; Franke et al. 2009). They showed that the central Indian Ocean in the southern hemisphere is not always as pristine as observed earlier during the winter monsoon period, but is polluted during the monsoon transition periods by pollution plumes from Africa and South-east Asia. Generally, the most polluted region is the BoB, which is influenced by Indian and South-east Asian outflow during most of the year and China during part of the year. In the present study, average concentration of NO2 was recorded as 0.31 μg m−3 with a day night ratio 0.79. Concentration of NO2 is very low and very often it is observed to be below the lower detection limit (LDL ±0.04 μg m−3) of the instrument at few locations over BoB. Correlations of these trace gases with meteorological parameters are summarized in Table 4 indicating the relative role of occurrence of gas–particle partitioning.
Similarly, measurement of SO2 over Indian Ocean is also very limited (Reiner et al. 2001; Shon et al. 2001; Putaud et al. 1992) and mostly done over southern Indian Ocean. Using aircraft data, Reiner et al. (2001) have reported SO2 profile with surface concentration of the order of 0.63 μg m−3 within the area of 8.1–8.3°N and 69.7–70.1°E. They have reported an elevated layer with mixing ratio >1.9 μg m−3 just above 2,000 m altitude. In the present study, average concentration of SO2 was recorded as 0.80 μg m−3 with a day night ratio of 1.15. Since its lifetime is few days, transport of continental SO2 to BoB, which is surrounded by dense polluted areas, changes the concentration depending on the wind pattern. In the eastern coastal region, gaseous SO2 shows low value and it seems that gas-phase of SO2 has been immediately converted to SO4 2− in the region. Spatial distribution of chemical composition of aerosol and its precursor gases (NH3, NO, NO2 and SO2) indicates the equilibrium between gaseous component and aerosol composition have occurred.
3.3 Relation between WSIC of aerosol and trace gases
Figure 3 shows the pie chart of nss-SO4 2−, Ca2+, K+, NH4 +, and NO3 − . Abundance of SO4 2− to total aerosol (PM10) loading was highest (~18.7 %). Sea salt also contributes to the concentration of a few ions like SO4 2−, Cl−, Na+, K+, and Ca2+ in the ambient atmosphere of BoB. Non sea salt fraction of SO4 2− accounts for 58 % of the total nss ions and ~16.0 % of the total aerosol loading.
Gas-to-particle conversion in the atmosphere involved complex aqueous and gaseous chemical process. Formation of (NH4)2SO4 and NH4NO3 depend on the concentration of NH3, acid precursors i.e., SO2, NO x and meteorological conditions. Hence, in a system consisting of NH3, H2SO4, HNO3, and water droplets, the composition of aerosols in the atmosphere may be influenced by the characteristics of the experimental sites. For gas-to-particle conversion by NH4 +, it is essential to have NH3-rich environment and to capture this process the constituents of aerosols must be analyzed in a meaningful manner by computing the equivalent ratios of NH4 +/SO4 2−, NH4 +/NO3 −, and NH4 +/(SO4 2− + NO3 − + Cl−). Table 5 shows the ratio and equivalent molar ratio of the measured concentrations of various particulates. The molar ratio of SO4 2−/(SO2 + SO4 2−), called the S ratio, is defined as the ratio of particulate SO4 2− to total sulfur (S; SO2 + SO4 2−). The ratio is used as an indicator of air mass age, chemical conversion efficiency, and deposition. Higher SO4 2−/(SO2 + SO4 2−) equivalent molar ratio during W_ICARB indicates that gas-to-particle conversion occurrs with greatest efficiency during the winter over BoB. Similar results were also reported by Baek and Aneja, (2004) at a commercial hog farm in North Carolina, USA. The relationship between particulates and NH3 were analyzed and summarized in Table 4. Each particulates species has a good correlation with NH4 + (NH4 + vs. SO4 2−, r 2 = 0.676; NH4 + vs. NO3 −, r 2 = 0.867) and NH3 (NH3 vs. SO4 2−, r 2 = 0.353; NH3 vs. NO3 −, r 2 = 0.537). The assimilation of HNO3 into NH4 + aerosols depends on the amount of H2SO4 used in neutralization with NH3. Thus the sample containing higher SO4 2− and lower NO3 − concentration (Table 1) could be representative of aerosols transported over a long distance or precursors from a continental air mass, whereas samples containing lower SO4 2− and higher NO3 − concentration could be representative of local area biogenic NO x emission leading to NO3 − or marine masses having high Cl− concentrations. Non-significant correlation of Cl− with NH3 and NH4 + indicates the very less conversion of chloride aerosol over BoB compared with sulfate and nitrate aerosols.
Higher correlation of NH3 with NO3 − in contrast with lower correlation of SO4 2− suggests the presence of relatively coarse mode of aerosol over BoB. Once H2SO4 is formed through oxidation of SO2 then in the presence of high NH3 it reacts with H2SO4 to form (NH4)2SO4 confining to fine particles. In order to understand the aerosol chemistry related to SO4 2−, we have plotted triangle diagram (ternary plot) using the combination of six major water-soluble ions, i.e., SO4 2−, Ca2, K+ and NH4 +, and NO3 − (Fig. 4). We would like to discuss the dominance of three different types of aerosol particle mixture of neutralized SO4 2−, by K+, Ca2+, NH4 +, three ternary plots of K+, SO4 2−, NO3 −, Ca2+, SO4 2−, NO3 − and NH4 +, SO4 2−, and NO3 −. Among three ternary plots, it is clear that neutralization of SO4 2− is most likely dominated by K+ and Na+. The present analysis is not conclusive as fraction of SO4 2− (~28 %) must be neutralized by some other cations even if it is assumed that complete portion of nss-Ca2+, nss-K+, and nss-NH4 + have neutralized SO4 2−. K+, Ca2+, NH4 + represents three indicators of three different types of sources, i.e., biomass burning, dust aerosol, and biogenic sources. Since aerosol containing SO4 2− along with K+, Ca2+, and NH4 + shows the combination of three types of aerosol suggesting that fine dust particle (Ca2+) traveling longer distance might have accumulated the signature of biomass burning as well as biogenic aerosol. Isentropic backward trajectory shows that air mass originated from Rajasthan desert area and traveled through IGP area before reaching to BoB.
3.4 Role of meteorological parameters
Spatial distribution of sea surface temperature (SST), ambient temperature, and RH are plotted over BoB (Fig. 5). North-to-south positive gradient in ambient temperature and SST is noticed with peak value in the southern of BoB, whereas spatial distribution of RH shows zonal gradient. Influences of ambient temperature and RH on aerosol could be identified in nitrate formation (Seinfeld and Pandis 2006). The RH dependence is discontinuous, i.e., below deliquescence relative humidity (DRH). There is no dependence of the equilibrium constant on RH, whereas above the DRH the equilibrium constant decreases rapidly with RH shifting the equilibrium strongly towards the aerosol phase. We have calculated the equilibrium constant (K p) for the humidity below the DRH using van’t Hoff equation.
Above the DRH, the tendency to form aerosol nitrate will be greater. If ratio (RRH) of RH and DRH is <1, NH4NO3 exits as solid, whereas, if RRH is >1, NH4NO3 is in solution form.
Particulate NO3 − shows the negative correlation (r 2 = −0.67) with ambient temperature during campaign (Table 4). The reason is that the diurnal variation of HNO3 is anti correlated with decline in temperature. Connected with the nocturnal rise in RH is a growth in the liquid water content with PM, which increases the absorbing capacity of aerosol with regard to gaseous HNO3 and H2SO4 under higher RH (Baek and Aneja 2004). Because NH3 is a major factor in neutralization of these acid gases, RH can affect the NH4 + concentration by neutralization of acid gases’ aerosol fraction. From the analysis of RH effect, NH4 + concentration was characterized for observations in which RH exceeded 60 %. The neutralization of NH3 by HNO3 and H2SO4 was enhanced at RH greater than 60 %, as a function of the increased absorbing capacity of the aerosols (Baek and Aneja 2004).
Concentration of SO2 in the marine boundary layer is primarily controlled by continental transport and marine biogenic sources. Gaseous SO2 shows higher value in the eastern coast of BoB suggesting the continental influence, whereas high value in the southern part of BoB, marine biogenic origin. Higher ambient temperature (~27 °C) and SST (~28 °C) with high RH in southern BoB suggest that air–sea interaction of gaseous SO2 might have been produced, but it could not be converted to SO4 2− immediately. We do not have measurement of marine biogenic sources (e.g., DMS, COS and MSA) over the study region; however, Shenoy et al. (2000) have reported high value of dimethyl sulfonic acid (DMS) value over south BoB during BOBMEX period in the monsoon period. Johansen et al. (1999) have also reported that when sea surface temperature is above 20 °C, ocean may emit SO2 which might be converted to SO4 2−. Chlorophyll value obtained from SeaWiFS may provide information of biological activity in BoB (Fig. 6). SeaWiFS chlorophyll map during December 2008 in the east coast of BoB (near Kolkata and Yangon) shows the presence of high concentration of chlorophyll (~10 mg m−3) indicating high biological activity, which in turn may release SO2 into the atmosphere. The presence of large amounts of SO2 in this region during campaign when ship mostly traveled close to the coastal region indicates the possibility of biological origin. Similarly, in the large west coast of BoB (near Chennai to Bhubaneswar), a moderate concentration of chlorophyll (0.3–1.0 mg m−3) is noticed, whereas middle of BoB and south BOB show low concentration of chlorophyll (0.03–0.1 mg m−3). During January 2009, when the ship mostly had traveled in the middle and south of BoB, east and west coast of BoB had maintained the similar biological activity, whereas middle and south of BoB have developed some biological activity with the presence of moderate range of chlorophyll (0.3–1.0 mg m−3). The possibility of moderate range of biological activity in the middle and south of BoB may explain the observed ambient SO2, when the probability of continental transport of SO2 to the south BoB could be less. Measurement of MSA in this region could have given better insight into the linkage between biological activity and ambient SO2.
In order to identify the strength of sources for precursor gases and water-soluble ions, PSCF analysis was performed (Poirot and Wishinshi 1986; Poissant 1999; Polissar et al. 2001) using concentration of chemical species (trace gases and ions) and 7 days’ backward trajectory calculated using the HYbrid Single Particle Lagrangian Integrated Trajectory (HYSPLIT) model and GDAS meteorological data (Draxler and Rolph 2003). The basis of PSCF is that if a source is located at (i,j), an air parcel back trajectory passing through that location indicates that material from the source can be collected and transported along the trajectory to the receptor site. Since the sampling of aerosol commenced around 7.30 a.m. on the sampling date and extended up to 6.30 a.m. in next day, for each sample, four back trajectories at four different points were calculated at 00, 06, 12, and 18 UTC. In this study, the geophysical region covered by the trajectories was divided into 1 × 1 grid cells. PSCF is defined as
where, n ij denotes the total number of trajectories passed through the cell (i,j) and m ij is the number of times the source concentration was high when the trajectories passed the cell (i,j) (Malm et al. 1986). The criteria for determining m ij were either the 75th or 90th percentile highest source contributions, depending on the structure of the source contribution time series for species. A cell (i,j) with a PSCF value close to one indicates a probable source location. For trace gases, e.g., SO2, NH3 we have calculated the PSCF using its hourly averaged value. Figure 7 shows PSCF analysis, which is being calculated for four major ions i.e., SO4 2−, NO3 −, Ca2+, and NH4 +. PSCF analysis shows that Ca2+ has originated mostly in Indian subcontinent extending up to western region of India in addition to coastal region near Bhubaneswar. Particulate NH4 + was transported as continental pollutant close to coastal India and extended up to central BoB region. Like NH4 +, transport path of particulate SO4 2− and NO3 − also follows the route but maxima is close the coastal Kolkata region. PSCF analysis shows that continental pollution containing water-soluble ions, i.e., Ca2+, NH4 +, SO4 2−, and NO3 − has been transported up to central BoB. PSCF analysis of SO2 shows two different regimes: one from East Asia and another from Indian subcontinent extending up to mid of Arabian Sea. Plumes from East Asia containing large amounts of SO2 have reached Central of Bengal. Of course, coastal region of BoB may contribute to the origin of SO2 due to biological activity in ocean during December 2008 and January 2009. Variation of SO2 over wide area of BoB is balanced by in situ conversion of aerosol and long-range transport, whereas that of NH3, NO, NO2 is mostly local. PSCF analysis has suggested that nss fraction of particulate ion has transported substantially from Indian subcontinent rather than East Asia.
4 Conclusion
Prominent latitudinal and longitudinal variations of the trace gases were observed along with PM10 over BoB. WSIC of PM10 comprised of 28 % of total aerosol. SO4 2− contributed 18 % of total aerosol loading. Out of nss fraction, SO4 2− contributing 58 % represented the major ionic composition. The average atmospheric concentration of NH3, NO, NO2, and SO2 were recorded as 4.78 ± 1.78, 1.89 ± 1.26, 0.31 ± 0.14, and 0.80 ± 0.30 μg/m3, respectively, with a range of 0.23–14.34, 0.21–9.80, 0.10–1.00, and 0.35–3.53 μg/m3, respectively. Coastal region of BoB reported large concentration of NH3, NO/NO2, and SO2 suggesting the role of continental influence in addition to biogenic sources particularly near Kolkata and Chennai region. The presence of large amounts of WSIC, particularly SO4 2− and measurement of ambient SO2 over BoB suggests the possible role of gas to particle conversation. Higher SO4 2−/(SO2 + SO4 2−) ratio during the study indicating that gas-to-particle conversion is occurring with great efficiency. PSCF analysis of nss-NH4 +, nss-SO4 2−, SO2, nss-Ca2+, nss-K+, and 7 days’ backward trajectories suggest that PM10 containing nss-K+, nss-Ca2+, nss-SO4 2− originated from desert area and traveled through Indo-Gangetic Plain (IGP) before reaching BoB. Aerosol over BoB could be mixture of mineral dust biomass burning and biogenic aerosol. PSCF of SO2 and backward trajectories suggest that it might have been transported from Indian subcontinent as well as from East Asia.
References
Aastar GI, Nair PR (2010) Spatial distribution of near surface CO over bay of Bengal during winter: role of transport. J Atmos Solar Terr Phys 75:1241–1250
Aneja VP, Roelle PA, Murray GC, Southerland J, Erisman JW, Fowler D, Asman WAH, Patni N (2001) Atmospheric nitrogen compounds II: emissions, transport, transformation, deposition and assessment. Atmos Environ 35:1903–1911
Asnani GC (1993) Tropical Meteorology, Indian Institute of Tropical Meteorology, Pashan, Pune
Athanasopoulou E, Tombrou M, Pandis SN, Russell AG (2008) The role of Sea-salt emission and heterogeneous chemistry in the air quality of polluted coastal areas. Atmos Chem Phys 8:5755–5769
Ayers GP, Gras JL (1980) Ammonia gas concentration over the Southern Ocean. Nature 284:539–540
Baek BH, Aneja VP (2004) Measurement and analysis of the relation between ammonia, acid gases and fine particles in eastern North Carolina. J Air Waste Manag Assoc 54:623–633
Bardouki H, Liakakou H, Economou C, Sciare J, Smolik J, Zdimal V, Eleftheriadis K, Lazaridis M, Dye C, Mihalopoulos N (2003) Chemical composition of size-resolved atmospheric aerosols in the eastern Mediterranean during summer and winter. Atmos Environ 37:195–208
Biswas H, Chatterjee A, Mukhopadhya SK, De TK, Sen S, Jana TK (2005) Estimation of ammonia exchange at the land–ocean boundary condition of Sundarban mangrove northeast coast of Bay of Bengal, India. Atmos Environ 39:4489–4499
Blackall TD, Wilson LJ, Theobald MR, Milford C, Nemitz E, Bull J, Bacon PJ, Hamer KC, Wanless S, Sutton MA (2007) Ammonia emission from seabird colonies. Geophys Res Lett 34:L10801. doi:10.1029/2006GL028928
Carmichael GR, Hong MS, Ueda H, Chen LL, Murano K, Park JK, Lee H, Kim Y, Kang C, Shim S (1997) Aerosol composition at Cheju Island, Korea. J Geophys Res 102:6047–6061
Carmichael GR, Ferm M, Thongboonchoo N, Woo JH, Chan LY, Murano K, Viet PH, Mossberg C, Bala R, Boonjawat J, Upatum P, Mohan M, Adhikary SP, Shrestha AB, Pinaar JJ, Brunke EB, Chen T, Jie T, Guoan D, Peng LC, Dhiharto S, Harjanto H, Jose AM, Kimani W, Kirouane A, Lacaus J-P, Richard S, Barturen O, Cerda JC, Athayde A, Tavares T, Cotrina JS, Bilici E (2003) Measurements of sulfur dioxide, ozone and ammonia concentration in Asia, Africa and South America using passive samplers. Atmos Environ 37:1293–1308
Charlson RJ, Lovelock JE, Andreae MO, Warren SG (1987) Oceanic phytoplankton, atmospheric sulfur, cloud albedo and climate. Nature 326:655–661
Draxler RR, Rolph GD (2003) HYSPLIT (HYbrid Single-Particle Lagrangian Integrated Trajectory) Model access via NOAA ARL READY Website (http://www.arl.noaa.gov/ready/hysplit4.html). NOAA Air Resources Laboratory, Silver Spring
Duce RA, Arimoto R, Ray BJ, Unni CK, Harder PJ (1983) Atmospheric trace elements at Enewetak Atoll I, concentrations, sources and temporal variability. J Geophys Res 88:5321–5342
Duce RA, Liss PS, Merrill JT, Atlas EL, Menard PB, Hicks BB, Miller JM, Prospero JM, Arimoto R, Church TM, Ellis W, Galloway JN, Hansen L, Jickells TD, Knap AH, Reinhardt KH, Schneider B, Soudine A, Tokos JJ, Keene WC, Pszenny AP, Vogt R, Galloway JN, Hawley ME (1986) Sea salt corrections and interpretations of constituent ratios in marine precipitation. J Geophys Res 91:6647–6658
Duce RA, Liss PS, Merrill JT, Atlas EL, Buat-Menard P, Hicks BB (1991) The atmospheric input of trace species to the world ocean. Global Biogeochem Cycles 5(3):193–259
Eleftheriadis K, Balis ZI, Manalis N (1998a) Atmospheric aerosol and gaseous species in Athens, Greece. Atmos Environ 32:2183–2191
Eleftheriadis K, Chung MC, Colbeck I (1998b) Atmospheric aerosol formation over Athens. J Aerosol Sci 29:S25–S26
Franke K, Richter A, Bovensmann H, Eyring V, J¨ockel P, Hoor P, Burrows JP (2009) Ship emitted NO2 in the Indian Ocean: comparison of model results with satellite data. Atmos Chem Phys 9:7289–7301
Garrett TJ, Hobbs PV (1995) Long-range transport of continental aerosols over the Atlantic Ocean and their effects on cloud structure. J Atmos Sci 56:2977–2984
Gibb SW, Mantouura RFC, Liss PS (1999) Ocean–atmosphere exchange and speciation of ammonia and methylamines in the region of the NW Arabian Sea. Global Biogeochem Cycles 13:161–178
Gupta A, Kumar R, Maharaj KK, Srivastava SS (2003) Measurement of NO2, HNO3, NH3 and SO2 and related particulate matter at a rural site in Rampur, India. Atmos Environ 37:4837–4846
Huebert BJ, Zhuang L, Howell S, Noone K, Noone B (1996) Sulfate, nitrate, methanesulfonate, chloride, ammonium and sodium measurements from ship, island and aircraft during the Atlantic Stratocumulus Transition Experiment/Marine Aerosol Gas Exchange. J Geophys Res 101:4413–4423
Johansen AM, Siefert RL, Hoffmann MR (1999) Chemical characterization of ambient aerosol collected during the southwest-monsoon and inter-monsoon seasons over the Arabian Sea: anions and cations. J Geophys Res 104:26325–26347
Johnson GC, Purkey SG, Toole JM (2008a) Reduced Antarctic meridional overturning circulation reaches the North Atlantic Ocean. Geophys Res Lett 35:L22601. doi:10.1029/2008GL035619
Johnson M, Liss P, Bell T, Lesworth T, Baker A, Hind AJ, Farhana B, Jickells T, Woodward M, Gibb S (2008b) Field observations of the ocean–atmosphere exchange of ammonia: fundamental importance of temperature as revealed by a comparison of high and low latitudes. Global Biogeochem Cycles 22:GB1019
Keene WC, Pszenny AAP, Galloway JN, Hawley ME (1986) Sea-salt corrections and interpretation of constituent ratios in marine precipitation. J Geophys Res 91:6647–6658
Khemani LT, Momin GA, Singh G (1987) Variation in trace gases concentrations in different environments in India. PAGEOPH 125:151–158
Krishnamurti TN, Jha B, Prospero JM, Jayaraman A, Ramanathan V (1998) Aerosol and pollutant transport and their impact on radiative forcing over the tropical Indian Ocean during the January–February 1986 pre-INDOEX cruise. Tellus 50B:521–542
Kunhikrishnan T, Lawrence MG, von-Kuhlmann R, Richter A, Ladstätter-Weißenmayer A, Burrows JP (2004) Analysis of tropospheric NO x over Asia using the model of atmospheric transport and chemistry (MATCH-MPIC) and GOME-satellite observations. Atmos Environ 38(4): 581–596
Lawrence MG, Crutzen PJ, Rasch PJ, Eaton PE, Mahowald NM (1999) A model for studies of tropospheric chemistry photochemistry: description, global distribution and evaluation. J Geophys Res 104:26245–26277
Lebel PJ, Hoell JM, Levine JS, Vay SA (1985) Aircraft measurements of ammonia and nitric acid in the lower troposphere. Geophys Res Lett 12:401–404
Li Y, Schwab JJ, Demerjian KL (2006) Measurement of ambient ammonia using a tunable diode laser absorption spectrometer: characteristics of ambient ammonia emissions in an urban area of New York City. J Geophys Res 111(D10). doi:10.1029/2005JD006275
Malm WC, Johnson CE, Bresch JF (1986) Application of principal component analysis for purposes of identifying source–receptor relationships in receptor methods for source apportionment. In: Pace TG (ed) Air Pollution Control Association, Pittsburgh, p 127–148
Mcinnes LM, Covert DS, Quinn PK, Germani MS (1994) Measurements of chloride depletion and sulfur enrichment in individual sea-salt particles collected from the remote marine boundary layer. J Geophys Res 99:8257–8268
McKee CM (2001) Biogeochemical cycles of ammonia and dimethylsulphide in the marine environment, University of East Anglia, Norwich
Naja M, Shyam L, Modh KS, Chand D (1999) Variabilities in O3, NO, CO and CH4 over Indian Ocean during winter. Curr Sci 76:931–937
Norman M, Leck C (2005) Distribution of marine boundary layer ammonia over the Atlantic and Indian Ocean during the Aerosols 99 cruise. J Geophys Res 110:D16302
Poirot RL, Wishinshi PR (1986) Visibility, sulfate, and air mass history associated with the summertime aerosol in northern Vermont. Atmos Environ 20:1457–1469
Poissant L (1999) Potential sources of atmospheric total gaseous mercury in the St. Lawrence River Valley. Atmos Environ 33:2537–2547
Polissar AV, Hopke PK, Harris JM (2001) Source regions for atmospheric aerosol measured at Barrow, Alaska. Environ Sci Technol 35:4214–4226
Putaud JP, Mihalopoulous N, Nguyen BC, Campin JM, Beloviso S (1992) Seasonal variation of atmospheric sulfur dioxide and dimethylsulfide concentrations at Amsterdam Island in the southern Indian Ocean. J Atmos Chem 15:117–131
Qunin PK, Charlson RJ, Bates TS, Johnson JE, Covert DS, Charlson RJ (1990) Interactions between the sulfur and reduced nitrogen cycles over the central Pacific Ocean. J Geophys Res 95(16):405–416
Rajeev K, Ramanathan V, Meywerk J (2000) Regional aerosol distribution and its long-range transport over the Indian Ocean. J Geophys Res 105:2029–2043
Reddy LAK, Kulshrestha UC, Satyanarayana J, Kulshrestha MJ, Seinfeld JH (1986) Atmospheric chemistry and physics of air pollution. Wiley, New York, pp 337–378
Reddy LAK, Kulshrestha UC, Satyanarayana J, Kulshrestha MJ, Moorthy KK (2008) Chemical characteristics of PM10 aerosols and airmass trajectories over Bay of Bengal and Arabian Sea during ICARB. J Earth Syst Sci 117:345–352
Reiner T, Sprung D, Jost C, Gabriel R, Mayol-Bracero OL, Andreae MO, Campos TL Shetter RE (2001) Chemical characterization of pollution layers over the tropical Indian Ocean: signatures of emissions of from biomass and fossil fuel burning. J Geophys Res 106(D22):28497–28510
Rhoads KP, Dickerson RR, Kelley P, Carsey T, Farmer M, Savie D, Prospero J (1997) The comparison of the troposphere over the Indian Ocean during the monsoonal season. J Geophys Res 102(15):18981–18995
Schafer P, Kreilen H, Muller M, Gravenhorst G (1993) Cycling of inorganic nitrogen compounds between atmosphere and ocean in tropical area of south east Asia. SCOPE/UNEP 76:19–36
Seinfeld JH (1986) Air pollution. John Wiley, New York
Seinfeld JH, Pandis SN (2006) Atmospheric chemistry and physics: from air pollution to climate change, 2nd edn. John Wiley, New York, p 1203
Sharma SK, Datta A, Saud T, Mandal TK, Ahammed YN, Arya BC, Tiwari MK (2010a) Study on concentration of ambient NH3 and interactions with some other ambient trace gases. Environ Monit Asses 162:225–235
Sharma SK, Datta A, Saud T, Saxena M, Mandal TK, Ahammed YN, Arya BC (2010b) Seasonal variability of ambient NH3, NO, NO2 and SO2 over Delhi. J Environ Sci 22(7):1023–1028
Shenoy DM, Kumar DM, Sarma VVSS (2000) Control of dimethyl sulphide in the Bay of Bengal during BOBMEX-Pilot cruise 1998. Earth Planet Sci 109:279–283
Shon ZH, Davis D, Chen G, Grodzinsky G, Bandy A, Thornton D, Sandholm S, Bradshaw J, Stickel R, Chameides W, Kok G, Russell L, Mauldin L, Tanner D, Eisele F (2001) Evaluation of the DMS flux and its conversion to SO2 over the southern ocean. Atmos Environ 35:159–172
Stelson AW, Seinfeld JH (1982) Relative humidity and pH dependence of the vapour pressure of ammonium nitrate-nitric acid and solutions at 25 degree Celsius. Atmos Environ 16:993–1000
Sturges WT, Barrie LA (1988) Chlorine, bromine and iodine in Arctic aerosols. Atmos Environ 22(6):1179–1194
Sutton MA, Place CJ, Eagar M, Fowler D, Smith RL (1995) Assessment of the magnitude of ammonia emissions in the United Kingdom. Atmos Environ 29:1393–1411
Sutton MA, Dragostis U, Tang YS, Flower D (2000) Ammonia emissions from non-agricultural sources in the UK. Atmos Environ 34:855–869
Teinila K, Kerminen VM, Hillamo R (2000) Study of size-segregated aerosol chemistry in the Antarctic atmosphere. J Geophys Res 105:3893–3904
Theobald MR, Crittenden PD, Hunt AP, Tang YS, Dragosits U, Sutton MA (2006) Ammonia emission from Cape fur seal colony, Cape Cross, Namibia. Geophys Res Lett 33:L03812. doi:10.1029/2005GL024384
Uematsu M, Torantini M, Kajino M, Narita Y, Senga Y, Kimoto T (2004) Enrichment of primary production in the western North Pacific caused by the eruption of Miyake-jima volcano. Geophys Res Lett 31:L06106. doi:10.1029/2003GL018790
Warneck P (1988) Chemistry of the natural atmosphere. International Geophysics, vol 41. Academic Press, New York, pp 313–326, 422–483
Zhuang L, Huebert BJ (1996) Lagrangian analysis of the total ammonia budget during Atlantic stratocumulus transition experiment/marine aerosol and gas exchange. J Geophys Res 101:4341–4350
Acknowledgments
The authors are thankful to Director and Head, RASD, CSIR-National Physical Laboratory, New Delhi, India, for their constant encouragement and support. The authors are also grateful to Director, Bose Institute, Kolkata, for agreeing to participate in the campaign. The authors acknowledge Indian Space Research Organization, Department of Space (ISRO/DOS) for financial support for participation in W_ICARB. One of the authors, T. Saud is thankful to CSIR for providing Senior Research Fellowship (SRF). AKS, SKG and SR thank Department of Science & Technology for support under IRHPA scheme.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: J. Fasullo
Rights and permissions
About this article
Cite this article
Sharma, S.K., Singh, A.K., Saud, T. et al. Study on water-soluble ionic composition of PM10 and related trace gases over Bay of Bengal during W_ICARB campaign. Meteorol Atmos Phys 118, 37–51 (2012). https://doi.org/10.1007/s00703-012-0204-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00703-012-0204-x