Abstract
Despite extensive research over the last decades the clinical significance of white matter lesions (WMLs) is still a matter of debate. Here, we review current knowledge of the correlation between WMLs and cognitive functioning as well as their predictive value for future stroke, dementia, and functional decline in activities of daily living. There is clear evidence that age-related WMLs relate to all of these outcomes on a group level, but the inter-individual variability is high. The association between WMLs and clinical phenotypes exists particularly for early confluent to confluent changes, which are ischaemic in aetiology and progress quickly over time. One reason for the variability of the relationship between WMLs and clinic on an individual level is probably the complexity of the association. Numerous factors such as cognitive reserve, concomitant loss of brain volume, and ultrastructural changes have been identified as mediators between white matter damage and clinical findings, and need to be incorporated in the consideration of WMLs as visible markers of these detrimental processes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence of white matter lesions (WMLs) in the general population is common. It increases steadily with age and the majority of subjects above the age of 75 years are affected (de Groot et al. 2000). WMLs have been consistently related to vascular risk factors (Longstreth et al. 1996; Schmidt et al. 2000); but despite extensive research, their clinical importance is still debated. Thus, it is not surprising that clinicians often struggle to explain to their patients the implications of these MRI findings. Here, we summarize current results on the association between WMLs and cognitive functioning, and discuss their role as predictors of stroke and disability. We address the issue of different lesion types and their relation to clinical findings, and seek to explain the large variability in the clinical presentation of individual subjects with WMLs.
WML types
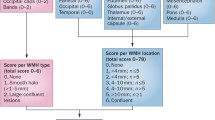
White matter lesions are best visualized by fluid-attenuated inversion recovery (FLAIR) sequences, which have the advantage of suppressing cerebrospinal fluid (CSF) signal and thus allow for high contrast lesion delineation even at periventricular or perisulcal locations. FLAIR also enables a simple distinction between WML and lacunes as the core of lacunes is typically of CSF-like appearance. Most MRI-rating scales distinguish between WMLs in periventricular and deep/subcortical regions, and grade the respective changes separately (Kapeller et al. 2003). Both regions contain not only small vessel disease-related but also non-ischaemic changes (Fazekas et al. 1991, 1993). In the periventricular area, non-ischaemic changes are also called “caps”, “lining”, and “bands” or a “halo” of high T2 signal (Fig. 1). They reflect only mild pathology in the 5–10 mm zone directly adjacent to the ventricles. Disruption of the ependymal lining, myelin pallor, and some subependymal astrogliosis are histopathological correlates. In the deep and/or subcortical white matter, one can distinguish between “punctate”, “early confluent”, and “confluent” WMLs. Per definition “punctate” WMLs are single lesions <10 mm and/or areas of “grouped” lesions <20 mm in any diameter; “early confluent” WMLs are hyperintense lesions 10–20 mm linked by no more than “connecting bridges” or single lesions >20 mm in any diameter; while “confluent” WMLs represent widespread hyperintense areas of >20 mm in any diameter which show clear confluency between abnormalities. Early confluent abnormalities tend to progress to confluent lesions over time.
Punctate changes can result from a number of often non-ischaemic aetiologies including widening of perivascular spaces or even heterotopia of ganglionic cells (Fazekas et al. 1991, 1993). In contrast, early confluent and confluent changes in the deep but also in the periventricular white matter are clearly related to small vessel disease. They mostly relate to extensive demyelination and axonal loss, which may or may not contain small cavities of tissue destruction (Fig. 2).
In line with these pathological findings, the Austrian Stroke Prevention Study has shown that punctate lesions remain stable over time, while participants with a baseline finding of early confluent or confluent changes showed a remarkably rapid increase in lesion volume (Schmidt et al. 2003). Almost two-thirds of study participants with early confluent and all subjects with confluent lesions demonstrated progression beyond measurement error over 6 years, while this was seen in none of the subjects with a normal baseline MRI scan and in only 14% of those with punctate foci. Clinical correlates are in keeping with these data as all studies consistently demonstrated an association between progression of leukoaraiosis and cognitive decline, with individuals who had coalescent white matter damage declining the most (Garde et al. 2005; Schmidt et al. 2005; van den et al. Heuvel 2006).
Therefore, histopathological, radiological, and clinical evidence clearly indicates that punctate WMLs are probably of mixed origin and rather benign, while early confluent and confluent WMLs are ischaemic, progressive, and thus malignant. Progression of WMLs was also demonstrated in subjects with dementia of various aetiologies but from these small studies no indication can be derived for an influential effect on the dementing illness per se. Progression rates were again influenced primarily by the severity of lesions at baseline rather than by the type of dementia or cognitive decline (Burton et al. 2006).
A recent investigation showed that progression of leukoaraiosis occurs throughout the brain but with some regional variation. The increase in deep WML volume was greater than that seen in the periventricular white matter. Frontal lesions progressed the most and occipital lesions the least (Sachdev et al. 2007).
Cognitive impairment, cognitive decline, and dementia
There is ample evidence that WMLs are associated with deficits in executive function, often affecting activities of daily living, which result from erroneous goal formation, planning, and organization. Abstract reasoning is also commonly affected, but patients have only mild memory deficits with slight recognition and cueing difficulties. A summary of cross-sectional studies on this topic conducted over the last decade is given in Table 1. As can be seen from this table, the studies were done in both community- and hospital-based cohorts with largely varying sample sizes and neuropsychological test batteries. Besides these differences, some cognitive impairment has been reported in all but one small investigation (Ross et al. 2005), and effects on cognition were seen in community-dwelling subjects as well as in individuals suffering from mild cognitive impairment and Alzheimer’s disease.
Importantly, several studies in population-based samples reported WMLs to be related not only to cognitive impairment but also to cognitive decline (Prins et al. 2005; Schmidt et al. 2005). Similar results have also been described in high-risk populations, with executive dysfunction and speed of mental processing declining the most (Longstreth et al. 2005; Kramer et al. 2007; Van Dijk et al. 2008).
A recent meta-analysis showed WMLs to independently predict dementia in the general population, and this was seen for both incident vascular dementia and Alzheimer’s disease (Debette and Markus 2010). However, the relation to incident Alzheimer’s disease was driven by a single large population-based study (Kuller et al. 2003), whereas smaller studies in patients with mild cognitive impairment did not identify significant associations (Debette and Markus 2010).
When summarizing the findings of previous investigations, WMLs seem to have subtle but noticeable cognitive consequences, which very likely result from frontal–subcortical circuit involvement (Schmidt et al. 2006). First evidence from functional MRI indirectly supports this notion (Linortner et al. 2010). However, so far, the mechanisms by which white matter damage predicts dementia have not been fully determined, although interactive effects with Alzheimer pathology are likely to play an important role (Zlokovic 2005).
Another suggested mechanism responsible for the association between WMLs and incident dementia is amyloid angiopathy, which is frequently seen as part of the spectrum of Alzheimer pathology. In such cases, it is likely atrophy precedes the development of WMLs rather than follows it. We investigated this question in a small subgroup of a study on the relationship between the longitudinal change in WMLs and brain atrophy, and their interactive effects on cognition (Schmidt et al. 2005). A small subgroup of study participants had no brain atrophy but extensive white matter changes at baseline. This subgroup experienced a substantially greater loss of brain volume during subsequent years than their counterparts without white matter foci. This supports the view that WML progression precedes loss in parenchymal volume in non-demented elderly subjects, but longitudinal data in larger samples are needed to better understand the temporal relationship between subcortical small vessel disease, cerebral atrophy. and their effects on cognitive functioning.
Incident stroke
White matter lesions have been repeatedly found to predict future strokes in high-risk groups but also in the general population (Wong et al. 2002; Vermeer et al. 2003; Kuller et al. 2004; Fu et al. 2005; Naka et al. 2006). In all studies, the association of WMLs with stroke remained significant after adjustment for vascular risk factors, which suggests either that WMLs reflect the overall effect of uncontrolled vascular risk factors better than the mere presence or absence of each individual factor or that other, yet unknown, factors play a role in the association between WMLs and stroke.
Functional decline in activities of daily living
The LADIS study has shown that despite independent everyday-life activities at baseline, subjects with WMLs developed functional impairment in activities of daily living at 1 year (Inzitari et al. 2007) and even more pronounced at three years follow-up (Inzitari et al. 2009). The study used Lawton and Brody’s (1969) instrumental activities of daily living scale and defined the change from a score of 0 or 1 at baseline to a score of 2 or more as the primary endpoint. These findings are important as they clearly demonstrate that deficits related to WMLs are not only detectable by demanding neuropsychological test batteries but also affect daily-life functions.
Clinical correlates of WMLs on the individual level
The high prevalence of WMLs in older populations and the large inter-individual variability of the clinical presentation of subjects with such abnormalities make clinicians uncertain whether WMLs are actually important and how to explain their clinical meaning to patients. In a study by Garde and co-workers (2000), WMLs only partly accounted for the inter-individual differences of age-related cognitive decline. Although cognitive dysfunction related to WMLs is most likely influenced by their distribution in strategically relevant brain regions and their volume (Desmond 2002), it is still unclear which locations are crucial and if there is a threshold of lesion volume which consistently leads to substantial cognitive impairment or dementia.
In a study on mixed dementia, Jellinger and Attems (2007) described that a vascular lesion volume smaller than 10 ml in manifest AD does not further influence cognitive impairment, unless strategically located infarcts occur. Their data are in favour of a complex interplay between location and volume influencing the cognitive phenotype of individuals with white matter disease. Another factor that is likely to be responsible for inter-individual variability of the cognitive consequences of WMLs is cognitive reserve, an issue that has remained virtually unappreciated in research on small vessel disease, so far. There is only one study suggesting that complex cognitive leisure activity exerts interactive effects with WML load on cognitive functioning in the general population (Saczynski et al. 2008).
WMLs, ultrastructural changes, and cognition
Diffusion tensor imaging (DTI) and magnetization transfer imaging (MTI) are currently the two most promising techniques for a more direct assessment of the composition and integrity of white matter structures. DTI and MTI have been used as tools to explain impaired cognition in individuals with WMLs beyond what can be expected from lesion volumetry alone (Fig. 3).
a Fluid attenuation inversion recovery, b mean diffusivity, c fractional anisotropy and d directional colour-coded fractional anisotropy images of a 66-year-old male subject at 3T. Maps b–d are derived from a diffusion tensor imaging (DTI) sequence using 12 diffusion gradient directions. DTI allows to quantitatively assess the mean water diffusivity at each voxel (b) as well as fractional anisotropy values as a measure of tissue integrity (c). By colour-coding, the principle Eigen-vector of the DTI data the principal fibre direction can be visualized (d) (e.g. fibres of the corticospinal tract running in z-direction shown in blue, fibres of the corpus callosum in the x–y direction shown in red). Arrows indicate confluent and punctate white matter lesions in the posterior and frontal white matter, respectively (a). Mean diffusivity in white matter lesions is increased probably due to demyelination (b) and this is paralleled by a loss of fractional anisotropy values as an indicator of impaired tissue integrity. Different shades of gray in c represent different magnitude of altered tissue integrity, while d indicates the predominant direction of fibres both in lesions and normal-appearing brain tissue
It is hypothesized that, in subjects with age-related WMLs, an increase in the apparent diffusion coefficient occurs not only in areas of T2 hyperintensity but also in normal-appearing white matter. In line with this assumption, it was shown that mean diffusivity of normal-appearing white matter is more closely related to clinical deficits than the volume of visible white matter damage. Two studies (Vernooij et al. 2009; Schmidt et al. 2010) corrected the association between diffusion imaging variables and cognitive functioning for both WML load and brain atrophy, and consistently found that diffusivity in normal-appearing white matter remained more strongly related to cognition than lesional volume and global brain atrophy.
Less data are available for MTI. Wong et al. (1995) found lower magnetization transfer ratio (MTR) values in periventricular brain regions compared with normal white matter. Our own group described the MTR of WMLs to be significantly lower than that of normal-appearing white matter and found a significant decrease with increasing lesion severity. The MTR of normal-appearing white matter was not different between subjects with very few and extensive WMLs. WML volume was associated with lower normal-appearing white matter MTR of the frontal lobes, but the MTR in frontal normal-appearing white matter was only related to fine motor dexterity but not to cognitive performance (Fazekas et al. 2005). These data are contrasted by work from Hanyu et al. (1999) who found correlations between the MTR values of age-related WMLs and cognitive functioning. It is important to emphasize that the pathological correlates of altered diffusivity and MTI measures are not yet fully determined. There is evidence that fractional anisotropy and mean diffusivity correlate directly with the amount of myelin in the white matter and to a lesser extent also with axonal count (Schmierer et al. 2007). Nonetheless, there is a need for more studies exploring the ultrastructural correlates of changes in the apparent diffusion coefficient and MTR to gain more insight into what histopathological abnormalities in the normal-appearing white matter can be picked up by either of the two methods. At this point, DTI appears to be more sensitive for tissue changes within WMLs and more robust for multicenter settings (Ropele et al. 2009).
Conclusions and future directions
Early confluent to confluent WMLs are related to cognitive decline, stroke, and functional decline in activities of daily living. Large, probably multi-centric collaborative efforts are needed to fully determine the threshold of WML load and strategically relevant locations that unequivocally lead to the different clinical phenotypes related to WMLs on a group level. Currently, the reasons for large inter-individual variability in the clinical presentation of subjects with WMLs are widely undetermined. Ultrastructural tissue changes that obviously occur in normal-appearing white matter in association with WMLs and other consequences of small vessel disease are at least partly responsible for inter-individual differences in the clinical picture in subjects with similar WML load. Histopathological post-mortem correlations together with the application of new imaging techniques in animal models of small vessel disease, which at this point are only sparsely available, are likely to increase our understanding of the whole spectrum of WML-related brain damage.
Abbreviations
- AD:
-
Alzheimer’s disease
- ADAS:
-
Alzheimer’s disease assessment scale
- ARIC:
-
Atherosclerosis risk in communities
- DM II:
-
Diabetes mellitus type 2
- CDR:
-
Clinical dementia rating (scale)
- CASCADE:
-
Cardiovascular determinants of dementia (study)
- LADIS:
-
Leukoaraiosis and disability (study)
- MCI:
-
Mild cognitive impairment
- MEMO:
-
Memory and morbidity in Augsburg elderly (study)
- MMSE:
-
Mini mental state examination
- MRI:
-
Magnetic resonance imaging
- PROSPER:
-
Prospective study of Pravastatin in the elderly at risk
- SMART-MR:
-
Second manifestations of ARTerial disease-MR substudy
- WMH:
-
White matter hyperintensities
- WML:
-
White matter lesions
References
Au R, Massaro JM, Wolf PA, Young ME, Beiser A, Seshadri S, D’Agostino RB, DeCarli C (2006) Association of white matter hyperintensity volume with decreased cognitive functioning: the Framingham Heart Study. Arch Neurol 63(2):246–250
Baune BT, Roesler A, Knecht S, Berger K (2009) Single and combined effects of cerebral white matter lesions and lacunar infarctions on cognitive function in an elderly population. J Gerontol A Biol Sci Med Sci 64(1):118–124
Bracco L, Piccini C, Moretti M, Mascalchi M, Sforza A, Nacmias B, Cellini E, Bagnoli S, Sorbi S (2005) Alzheimer’s disease: role of size and location of white matter changes in determining cognitive deficits. Dement Geriatr Cogn Disord 20(6):358–366 Epub 2005 Sep 26
Burns JM, Church JA, Johnson DK, Xiong C, Marcus D, Fotenos AF, Snyder AZ, Morris JC, Buckner RL (2005) White matter lesions are prevalent but differentially related with cognition in aging and early Alzheimer disease. Arch Neurol 62(12):1870–1876
Burton EJ, McKeith IG, Burn DJ, Firbank MJ, O’Brien JT (2006) Progression of white matter hyperintensities in Alzheimer disease, dementia with Lewy bodies, and Parkinson disease dementia: a comparison with normal aging. Am J Geriatr Psychiatry 14:842–849
Chen YF, Wang H, Chu Y, Huang YC, Su MY (2006) Regional quantification of white matter hyperintensity in normal aging, mild cognitive impairment, and Alzheimer’s disease. Dement Geriatr Cogn Disord 22(2):177–184
de Groot JC, de Leeuw FE, Oudkerk M, van Gijn J, Hofman A, Jolles J, Breteler MM (2000) Cerebral white matter lesions and cognitive function: the Rotterdam Scan Study. Ann Neurol 47(2):145–151
Debette S, Markus HS (2010) The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ 341:c3666
Debette S, Bombois S, Bruandet A, Delbeuck X, Lepoittevin S, Delmaire C, Leys D, Pasquier F (2007) Subcortical hyperintensities are associated with cognitive decline in patients with mild cognitive impairment. Stroke 38(11):2924–2930
Delano-Wood L, Abeles N, Sacco JM, Wierenga CE, Horne NR, Bozoki A (2008) Regional white matter pathology in mild cognitive impairment: differential influence of lesion type on neuropsychological functioning. Stroke 39(3):794–799
Desmond DW (2002) Cognition and white matter lesions. Cerebrovasc Dis 13(suppl 2):53–57
Fazekas F, Kleinert R, Offenbacher H, Payer F, Schmidt R, Kleinert G, Radner H, Lechner H (1991) The morphologic correlate of incidental punctate MR white matter hyperintensities. AJNR Am J Neuroradiol 12:915–921
Fazekas F, Kleinert R, Offenbacher H, Schmidt R, Kleinert G, Payer F, Radner H, Lechner H (1993) Pathologic correlates of incidental MRI white matter signal hyperintensities. Neurology 43(9):1683–1689
Fazekas F, Ropele S, Enzinger C, Gorani F, Seewann A, Petrovic K, Schmidt R (2005) MTI of white matter hyperintensities. Brain 128(Pt 12):2926–2932
Fu JH, Lu CZ, Hong Z, Dong Q, Luo Y, Wong KS (2005) Extent of white matter lesions is related to acute subcortical infarcts and predicts further stroke risk in patients with first ever ischaemic stroke. J Neurol Neurosurg Psychiatry 76:793–796
Garde E, Mortensen EL, Krabbe K, Rostrup E, Larsson HB (2000) Relation between age-related decline in intelligence and cerebral white-matter hyperintensities in healthy octogenarians: a longitudinal study. Lancet 356(9230):628–634
Garde E, Lykke Mortensen E, Rostrup E, Paulson OB (2005) Decline in intelligence is associated with progression in white matter hyperintensity volume. J Neurol Neurosurg Psychiatry 76:1289–1291
Geerlings MI, Appelman AP, Vincken KL, Mali WP, van der Graaf Y, SMART Study Group (2009) Association of white matter lesions and lacunar infarcts with executive functioning: the SMART-MR study. Am J Epidemiol 170(9):1147–1155
Hanyu H, Asano T, Sakurai H, Iwamoto T, Takasaki M, Shindo H, Abe K (1999) Magnetization transfer ratio in cerebral white matter lesions of Binswanger’s disease. J Neurol Sci 166(2):85–90
Heo JH, Lee ST, Chu K, Park HJ, Shim JY, Kim M (2009) White matter hyperintensities and cognitive dysfunction in Alzheimer disease. J Geriatr Psychiatry Neurol 22(3):207–212
Inzitari D, Simoni M, Pracucci G, Poggesi A, Basile AM, Chabriat H, Erkinjuntti T, Fazekas F, Ferro JM, Hennerici M, Langhorne P, O’Brien J, Barkhof F, Visser MC, Wahlund LO, Waldemar G, Wallin A, Pantoni L, LADIS Study Group (2007) Risk of rapid global functional decline in elderly patients with severe cerebral age-related white matter changes: the LADIS study. Arch Intern Med 167(1):81–88
Inzitari D, Pracucci G, Poggesi A, Carlucci G, Barkhof F, Chabriat H, Erkinjuntti T, Fazekas F, Ferro JM, Hennerici M, Langhorne P, O’Brien J, Scheltens P, Visser MC, Wahlund LO, Waldemar G, Wallin A, Pantoni L, LADIS Study Group (2009) Changes in white matter as determinant of global functional decline in older independent outpatients: three year follow-up of LADIS (leukoaraiosis and disability) study cohort. BMJ 339:b2477. doi:10.1136/bmj.b2477
Jellinger KA, Attems J (2007) Neuropathological evaluation of mixed dementia. J Neurol Sci 257(1–2):80–87
Kapeller P, Barber R, Vermeulen RJ, Adèr H, Scheltens P, Freidl W, Almkvist O, Moretti M, del Ser T, Vaghfeldt P, Enzinger C, Barkhof F, Inzitari D, Erkinjunti T, Schmidt R, Fazekas F, European Task Force of Age Related White Matter Changes (2003) Visual rating of age-related white matter changes on magnetic resonance imaging: scale comparison, interrater agreement, and correlations with quantitative measurements. Stroke 34(2):441–445
Koga H, Takashima Y, Murakawa R, Uchino A, Yuzuriha T, Yao H (2009) Cognitive consequences of multiple lacunes and leukoaraiosis as vascular cognitive impairment in community-dwelling elderly individuals. J Stroke Cerebrovasc Dis 18(1):32–37
Kramer JH, Mungas D, Reed BR, Wetzel ME, Burnett MM, Miller BL et al (2007) Longitudinal MRI and cognitive change in healthy elderly. Neuropsychology 21:412–418
Kuller LH, Lopez OL, Newman A, Beauchamp NJ, Burke G, Dulberg C et al (2003) Risk factors for dementia in the cardiovascular health cognition study. Neuroepidemiology 22:13–22
Kuller LH, Longstreth WT Jr, Arnold AM, Bernick C, Bryan RN, Beauchamp NJ Jr (2004) White matter hyperintensity on cranial magnetic resonance imaging: a predictor of stroke. Stroke 35:1821–1825
Lawton MP, Brody EM (1969) Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist 9:179–186
Linortner P, Fazekas F, Schmidt R, Ropele S, Pendl B, Petrovic K, Loitfelder M, Neuper C, Enzinger C (2010) White matter hyperintensities alter functional organization of the motor system. Neurobiol Aging (Epub ahead of print)
Longstreth WT Jr, Manolio TA, Arnold A, Burke GL, Bryan N, Jungreis CA, Enright PL, O’Leary D, Fried L (1996) Clinical correlates of white matter findings on cranial magnetic resonance imaging of 3301 elderly people: The Cardiovascular Health Study. Stroke 27(8):1274–1282
Longstreth WT Jr, Arnold AM, Beauchamp NJ Jr, Manolio TA, Lefkowitz D, Jungreis C et al (2005) Incidence, manifestations, and predictors of worsening white matter on serial cranial magnetic resonance imaging in the elderly: the Cardiovascular Health Study. Stroke 36:56–61
Mosley TH Jr, Knopman DS, Catellier DJ, Bryan N, Hutchinson RG, Grothues CA, Folsom AR, Cooper LS, Burke GL, Liao D, Szklo M (2005) Cerebral MRI findings and cognitive functioning: the Atherosclerosis Risk in Communities study. Neurology 64(12):2056–2062
Muller M, Appelman AP, van der Graaf Y, Vincken KL, Mali WP, Geerlings MI (2009) Brain atrophy and cognition: interaction with cerebrovascular pathology? Neurobiol Aging (Epub ahead of print)
Mungas D, Reed BR, Farias ST, Decarli C (2009) Age and education effects on relationships of cognitive test scores with brain structure in demographically diverse older persons. Psychol Aging 24(1):116–128
Naka H, Nomura E, Takahashi T, Wakabayashi S, Mimori Y, Kajikawa H et al (2006) Combinations of the presence or absence of cerebral microbleeds and advanced white matter hyperintensity as predictors of subsequent stroke types. AJNR Am J Neuroradiol 27:830–835
Pohjasvaara TI, Jokinen H, Ylikoski R, Kalska H, Mäntylä R, Kaste M, Erkinjuntti T (2007) White matter lesions are related to impaired instrumental activities of daily living poststroke. J Stroke Cerebrovasc Dis 16(6):251–258
Price CC, Jefferson AL, Merino JG, Heilman KM, Libon DJ (2005) Subcortical vascular dementia: integrating neuropsychological and neuroradiologic data. Neurology 65(3):376–382
Prins ND, van Dijk EJ, den Heijer T, Vermeer SE, Jolles J, Koudstaal PJ, Hofman A, Breteler MM (2005) Cerebral small-vessel disease and decline in information processing speed, executive function and memory. Brain 128:2034–2041
Rabbitt P, Mogapi O, Scott M, Thacker N, Lowe C, Horan M, Pendleton N, Jackson A, Lunn D (2007) Effects of global atrophy, white matter lesions, and cerebral blood flow on age-related changes in speed, memory, intelligence, vocabulary, and frontal function. Neuropsychology 21(6):684–695
Ropele S, Seewann A, Gouw AA, van der Flier WM, Schmidt R, Pantoni L, Inzitari D, Erkinjuntti T, Scheltens P, Wahlund LO, Waldemar G, Chabriat H, Ferro J, Hennerici M, O’Brien J, Wallin A, Langhorne P, Visser MC, Barkhof F, Fazekas F (2009) Quantitation of brain tissue changes associated with white matter hyperintensities by diffusion-weighted and magnetization transfer imaging: the LADIS (Leukoaraiosis and Disability in the Elderly) study. J Magn Reson Imaging 29(2):268–274
Ross ED, Hansel SL, Orbelo DM, Monnot M (2005) Relationship of leukoaraiosis to cognitive decline and cognitive aging. Cogn Behav Neurol 18(2):89–97
Sachdev PS, Wen W, Christensen H, Jorm AF (2005) White matter hyperintensities are related to physical disability and poor motor function. J Neurol Neurosurg Psychiatry 76(3):362–367
Sachdev P, Wen W, Chen X, Brodaty H (2007) Progression of white matter hyperintensities in elderly individuals over 3 years. Neurology 68:214–222
Saczynski JS, Jonsdottir MK, Sigurdsson S, Eiriksdottir G, Jonsson PV, Garcia ME, Kjartansson O, van Buchem MA, Gudnason V, Launer LJ (2008) White matter lesions and cognitive performance: the role of cognitively complex leisure activity. J Gerontol A Biol Sci Med Sci 63(8):848–854
Schmidt R, Schmidt H, Fazekas F (2000) Vascular risk factors in dementia. J Neurology 247:81–87
Schmidt R, Enzinger C, Ropele S, Schmidt H, Fazekas F, Austrian Stroke Prevention Study (2003) Progression of cerebral white matter lesions: 6-year results of the Austrian Stroke Prevention Study. Lancet 361:2046–2048
Schmidt R, Ropele S, Enzinger C, Petrovic K, Smith S, Schmidt H, Matthews P, Fazekas F (2005) White matter lesion progression, brain atrophy, and cognitive decline: the Austrian stroke prevention study. Ann Neurol 58:610–616
Schmidt R, Enzinger C, Ropele S, Schmidt H, Fazekas F (2006) Subcortical vascular cognitive impairment: similarities and differences with multiple sclerosis. J Neurol Sci 245(1–2):3–7
Schmidt R, Ropele S, Ferro J, Madureira S, Verdelho A, Petrovic K, Gouw A, van der Flier WM, Enzinger C, Pantoni L, Inzitari D, Erkinjuntti T, Scheltens P, Wahlund LO, Waldemar G, Rostrup E, Wallin A, Barkhof F, Fazekas F, on behalf of the LADIS study group (2010) Diffusion-weighted imaging and cognition in the leukoariosis and disability in the elderly study. Stroke 41(5):e402–e408
Schmierer K, Wheeler-Kingshott CA, Boulby PA, Scaravilli F, Altmann DR, Barker GJ, Tofts PS, Miller DH (2007) Diffusion tensor imaging of post mortem multiple sclerosis brain. Neuroimage 35(2):467–477
Shenkin SD, Bastin ME, Macgillivray TJ, Deary IJ, Starr JM, Rivers CS, Wardlaw JM (2005) Cognitive correlates of cerebral white matter lesions and water diffusion tensor parameters in community-dwelling older people. Cerebrovasc Dis 20(5):310–318
Söderlund H, Nilsson LG, Berger K, Breteler MM, Dufouil C, Fuhrer R, Giampaoli S, Hofman A, Pajak A, de Ridder M, Sans S, Schmidt R, Launer LJ (2006) Cerebral changes on MRI and cognitive function: the CASCADE study. Neurobiol Aging 27(1):16–23
Sonohara K, Kozaki K, Akishita M, Nagai K, Hasegawa H, Kuzuya M, Yokote K, Toba K (2008) White matter lesions as a feature of cognitive impairment, low vitality and other symptoms of geriatric syndrome in the elderly. Geriatr Gerontol Int 8(2):93–100
Stenset V, Hofoss D, Berstad AE, Negaard A, Gjerstad L, Fladby T (2008) White matter lesion subtypes and cognitive deficits in patients with memory impairment. Dement Geriatr Cogn Disord 26(5):424–431
Tiehuis AM, Vincken KL, Mali WP, Kappelle LJ, Anbeek P, Algra A, Biessels GJ (2008) Automated and visual scoring methods of cerebral white matter hyperintensities: relation with age and cognitive function. Cerebrovasc Dis 25(1–2):59–66
van den Heuvel DM, ten Dam VH, de Craen AJ, Admiraal-Behloul F, Olofsen H, Bollen EL, Jolles J, Murray HM, Blauw GJ, Westendorp RG, van Buchem MA (2006) Increase in periventricular white matter hyperintensities parallels decline in mental processing speed in a non-demented elderly population. J Neurol Neurosurg Psychiatry 77(2):149–153
van der Flier WM, van Straaten EC, Barkhof F, Ferro JM, Pantoni L, Basile AM, Inzitari D, Erkinjuntti T, Wahlund LO, Rostrup E, Schmidt R, Fazekas F, Scheltens P, LADIS study group (2005) Medial temporal lobe atrophy and white matter hyperintensities are associated with mild cognitive deficits in non-disabled elderly people: the LADIS study. J Neurol Neurosurg Psychiatry 76(11):1497–1500
Van Dijk EJ, Prins ND, Vrooman HA, Hofman A, Koudstaal PJ, Breteler MM (2008) Progression of cerebral small vessel disease in relation to risk factors and cognitive consequences: Rotterdam Scan study. Stroke 39:2712–2719
van Harten B, Oosterman J, Muslimovic D, van Loon BJ, Scheltens P, Weinstein HC (2007) Cognitive impairment and MRI correlates in the elderly patients with type 2 diabetes mellitus. Age Ageing 36(2):164–170
Vermeer SE, Hollander M, van Dijk EJ, Hofman A, Koudstaal PJ, Breteler MM (2003) Silent brain infarcts and white matter lesions increase stroke risk in the general population: the Rotterdam Scan Study. Stroke 34:1126–1129
Vernooij MW, Ikram MA, Vrooman HA, Wielopolski PA, Krestin GP, Hofman A et al. (2009) White matter microstructural integrity and cognitive function in a general elderly population. Arch Gen Psychiatry 66:545–553
Wong KT, Grossman RI, Boorstein JM, Lexa FJ, McGowan JC (1995) Magnetization transfer imaging of periventricular hyperintense white matter in the elderly. AJNR Am J Neuroradiol 16(2):253–258
Wong TY, Klein R, Sharrett AR, Couper DJ, Klein BE, Liao DP et al (2002) Cerebral white matter lesions, retinopathy, and incident clinical stroke. JAMA 288:67–74
Zhou G, Ren S, Chen N, Duan L, Zhang Z, Fang S, Zhao W (2008) Cerebral white matter lesions and cognitive function in a non-demented Chinese veteran cohort. J Int Med Res 36(1):115–122
Zlokovic BV (2005) Neurovascular mechanisms of Alzheimer’s neurodegeneration. Trends Neurosci 28:202–208
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is dedicated to Professor Kurt Jellinger in admiration of his outstanding research achievements in the field of dementia and in appreciation of his long-standing support for our research group.
Rights and permissions
About this article
Cite this article
Schmidt, R., Grazer, A., Enzinger, C. et al. MRI-detected white matter lesions: do they really matter?. J Neural Transm 118, 673–681 (2011). https://doi.org/10.1007/s00702-011-0594-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-011-0594-9