Abstract
Background
Combined bypass, including direct and indirect procedures, has been recognized as the maximal revascularization to prevent further hemorrhagic or ischemic stroke in adult moyamoya disease (MMD). It is also important to consider cosmetic aspects when planning combined bypass for MMD. However, there are few reports that have described the cosmetic considerations in bypass surgery for MMD.
Methods
We demonstrate our surgical techniques aimed at achieving extended revascularization as well as excellent cosmetic outcomes with figures and video.
Conclusion
Our combined bypass procedures which focus on achieving maximal cosmetic results are effective methods that require no special instruments or techniques.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Relevant surgical anatomy
Combined bypass, including direct superficial temporal artery (STA)-middle cerebral artery (MCA) anastomosis with an indirect procedure, achieves maximal revascularization in order to prevent further ischemic or hemorrhagic stroke in adult moyamoya disease (MMD) [4]. It is also important to consider the cosmetic aspects when planning combined bypass for MMD. Combined bypass requires STA as the donor vessel for direct anastomosis and subcutaneous tissues as the source of indirect bypass, which may result in loss of blood supply to the scalp. Understanding the anatomy and blood supply of subcutaneous tissues, including the scalp, galea aponeurotica, temporal fascia, and pericranium, is a key factor in performing combined bypass while achieving good cosmetic results.
Description of the technique
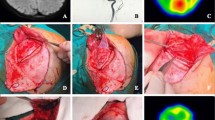
Our basic bypass procedure for adult MMD is a combined bypass procedure that involves one or two direct anastomoses [1]. Hair is not shaved, unless, the patient requests that the head be shaved. The patient is placed in the supine position, and the head position is fixed with rotation to the contralateral side at 60 to 90° using a vacuum-stiffened beanbag pillow. The caliber and course of STA are evaluated by preoperative digital subtraction angiography, and a scalp incision is designed to be semicircular and contain at least one STA branch. If there is severe hemodynamic impairment in the anterior cerebral artery (ACA) region, the medial side of the scalp incision is made near the midline, containing both STA parietal and frontal branches. The incision line, except around the STA edge, is anesthetized with 0.5% lidocaine containing diluted epinephrine. The scalp is incised with a scalpel immediately below the galea aponeurotica. Hemostasis of the scalp is achieved by a bipolar coagulation for only bleeding from the edges of the scalp arteries which run above the galea. Excessive electrocoagulation may result in alopecia around the incision line. The scalp clamps are used for hemostasis of oozing from the subcutaneous fatty tissue. The scalp is reflected inferiorly, and the subcutaneous tissues beneath the galea must be completely preserved at this moment (Fig. 1a). One or two STA branches are dissected under a microscope with a minimal incision of the surrounding tissues using cutting bipolar forceps (Cutting Edge; Fujita Medical Instruments Co. Ltd., Tokyo, Japan). The following process is an important part of our combined cosmetic bypass procedure. After making the tunnel through the temporal muscle that allows penetration of the donor STA, the temporal fascia is incised just above the scalp fold and carefully dissected antero-superiorly with complete preservation of the temporal muscle. The temporal fascia continues to the pericranium above the superior temporal line, and further upward dissection of the temporal fascia/pericranium is conducted. The prepared antero-superiorly pedicled temporal fascia/pericranium is used for indirect bypass in a later stage. The temporal muscle itself is dissected using the retrograde dissection technique [6] to prevent muscle atrophy and is reflected inferiorly (Fig. 1b). The extent of craniotomy is determined to optimize the cerebral blood flow in the affected hemisphere in each case; usually, a large fronto-temporo-parietal craniotomy would be necessary to achieve maximal revascularization. The dura mater is incised while preserving the main branch of the middle meningeal artery. After opening the dura, an appropriate MCA cortical branch (M4 segment), usually running in the suprasylvian MCA region, is identified and is prepared as an anastomotic recipient. The donor STA is cut and transferred to the surgical field through the temporal muscle tunnel, and then the cutoff edge of the STA is trimmed. A safe and reliable anastomotic procedure is quite important in direct or combined bypass surgery for MMD, because the recipient vessel diameter is smaller and the vessel wall is more fragile in MMD than in other conditions. Interrupted STA-MCA end-to-side anastomosis is usually performed using 10-0 or 11-0 monofilament suture. When performing a double-barrel anastomosis, another STA branch is anastomosed to the ACA cortical branch or another M4 branch. After completion of the anastomotic procedure, the dura mater is widely incised and is inverted to the brain surface (Fig. 1c). Then, the field of craniotomy is entirely covered by a superior pedicled temporal fascia/pericranium flap, and an indirect procedure is completed (Fig. 1d). We named this indirect bypass procedure as inversed flap encephalo-duro-pericranium-synangiosis. A bone flap is placed (Fig. 1e) and fixed using absorbable fixtures (CranioFix® absorbable; Aesculap AG, Tuttlingen, Germany). The temporal muscle is sutured to the pericranium edge. Note that the pedicled tissues are entirely preserved just under the scalp incision line (Fig. 1f), contributing to the preservation of blood supply of the scalp and prevention of wound infection. For the scalp closure, the galea suturing technique is employed; only the galea is sutured using 3-0 absorbable sutures in order to maintain the blood supply of subcutaneous tissues, and the outer layer of the scalp is then closed with skin staples [8].
When the scalp is incised and reflected, the subgaleal tissues are kept completely intact (a). The temporal fascia/pericranium (F/P) flap that is used for a later indirect procedure is dissected upwardly (green arrow), and the temporal muscle (TM) is reflected inferiorly (red arrow) (b). After completion of the anastomosis, the dura mater is widely incised and is inverted to the brain surface (c). The bone window is widely covered by a superior pedicled F/P flap as an indirect procedure (arrow) (d). The bone flap is then placed and fixed mainly using absorbable fixtures (e). The TM is sutured to the pericranium in a primary fashion. Note that the pedicled tissues are entirely preserved just under the scalp incision line (f). f/p temporal fascia/pericranium flap, tm, temporal muscle
Indications
We consider bypass surgery in adult MMD as a prevention of cerebral ischemia for ischemic hemisphere or for the hemisphere with severe hemodynamic impairment (CVR less than 0%) and as a prevention of repeat hemorrhagic stroke for the hemorrhagic hemisphere. The presenting method can be adopted for all MMD patients because it does not require any special instruments or techniques. In this method, the temporal muscle is not used for indirect procedures and is completely preserved outside the bone. Especially in adult cases, using the temporal muscle itself for indirect procedures may result in a mass effect on the brain because of acute swelling [5] and often causes depression deformity of the temporal region. Thus, adult MMD cases are considered the best candidates for this procedure.
Limitations
It has been reported that the use of the temporal muscle itself for indirect procedures may lead to better angiogenesis rather than the dura or fascia alone [7]. However, no study has answered the question of which indirect bypass procedure has the best neovascularization potential [2]. The potential for angiogenesis using this method compared to other reported indirect procedures should be further investigated.
How to avoid complications
It is highly desirable to maintain normocapnia with normotension during bypass surgery for MMD because perioperative ischemic complications in MMD may often be caused by perioperative hypo- and hypercapnia or hypoperfusion rather than surgery itself [3].
Specific perioperative considerations
Oral antiplatelet therapy (aspirin 100mg daily) is suspended 2 days prior to surgery, and resumed 24 h after surgery. To prevent hyperperfusion syndrome, strict systolic blood pressure control (110–140 mmHg) and intravenous administration of edaravone (30mg twice daily) are continued until postoperative day seven [9].
Specific information for the patient
The surgical wound is left open and uncovered. The subcutaneous drain is removed on the first day, and hair washing is allowed from the third day after surgery. The skin staplers are removed on the seventh postoperative day, as in the other craniotomy surgeries.
References
Egashira Y, Yamauchi K, Enomoto Y, Nakayama N, Yoshimura S, Iwama T (2017) Disruption of cortical arterial network is associated with the severity of transient neurologic events after direct bypass surgery in adult moyamoya disease. World Neurosurg 100:311–315. https://doi.org/10.1016/j.wneu.2017.01.039
Houkin K, Kuroda S, Ishikawa T, Abe H (2000) Neovascularization (angiogenesis) after revascularization in moyamoya disease. Which technique is most useful for moyamoya disease? Acta Neurochir (Wien) 142:269–276. https://doi.org/10.1007/s007010050035
Iwama T, Hashimoto N, Yonekawa Y (1996) The relevance of hemodynamic factors to perioperative ischemic complications in childhood moyamoya disease. Neurosurgery 38:1120–1125; discussion 1125-1126. https://doi.org/10.1097/00006123-199606000-00011
Nguyen VN, Motiwala M, Elarjani T, Moore KA, Miller LE, Barats M, Goyal N, Elijovich L, Klimo P, Hoit DA, Arthur AS, Morcos JJ, Khan NR (2022) Direct, indirect, and combined extracranial-to-intracranial bypass for adult moyamoya disease: an updated systematic review and meta-analysis. Stroke. https://doi.org/10.1161/STROKEAHA.122.039584
Noguchi K, Aoki T, Orito K, Kajiwara S, Fujimori K, Morioka M (2018) Novel indirect revascularization technique with preservation of temporal muscle function for moyamoya disease encephalo-duro-fascio-arterio-pericranial-synangiosis: a case series and technical note. World Neurosurg 120:168–175. https://doi.org/10.1016/j.wneu.2018.08.171
Oikawa S, Mizuno M, Muraoka S, Kobayashi S (1996) Retrograde dissection of the temporalis muscle preventing muscle atrophy for pterional craniotomy. Technical note. J Neurosurg 84:297–299. https://doi.org/10.3171/jns.1996.84.2.0297
Rennert RC, Budohoski KP, Grandhi R, Couldwell WT (2022) Combined direct and indirect superficial temporal artery-to-middle cerebral artery bypass with a hinged bone flap: how I do it. Acta Neurochir (Wien) 164:3203–3208. https://doi.org/10.1007/s00701-022-05393-6
Suetake K, Uede T, Momota H, Oka S, Piao H, Minamida Y, Hashi K (1999) Usefulness of galea suturing method for scalp closure. No Shinkei Geka 27:427–430
Uchino H, Nakayama N, Kazumata K, Kuroda S, Houkin K (2016) Edaravone reduces hyperperfusion-related neurological deficits in adult moyamoya disease: historical control study. Stroke 47:1930–1932. https://doi.org/10.1161/STROKEAHA.116.013304
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by YE, YE, and KK. The first draft of the manuscript was written by YE, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Official approval from the institutional review board is waived for case presentation. All procedures employed in the present study are in accordance with the ethical standards of the Gifu University Hospital and with the 1964 Helsinki Declaration and its later amendments. The patient consented to the publication of his case in the present study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available online.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Egashira, Y., Enomoto, Y., Kano, K. et al. How I do it: combined bypass for adult moyamoya disease with maximal consideration of cosmetic aspects. Acta Neurochir 165, 2073–2076 (2023). https://doi.org/10.1007/s00701-023-05604-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-023-05604-8