Abstract
Background
It is currently unclear if there are subsets of patients undergoing transsphenoidal surgery (TSS) in which intraoperative high-field magnetic resonance imaging (3T-iMRI) is particularly advantageous. We aimed to investigate whether a radiological grading scale predicts the utility of 3T-iMRI in pituitary adenoma (PA) TSS.
Methods
From a prospective registry, patients who underwent endoscopic TSS for PA using 3T-iMRI were identified. Adenomas were graded using the Zurich Pituitary Score (ZPS). We assessed improvement after 3T-iMRI in terms of gross total resection (GTR), residual volume (RV), and extent of resection (EOR).
Results
Among 95 patients, rates of conversion to GTR after 3T-iMRI decreased steadily from 33% for grade I to 0% for grade IV adenomas, with a statistically significant conversion rate only for grade I (p = 0.008) and grade II (p < 0.001). All grade I adenomas were completely resected after 3T-iMRI. Median RV change was statistically significant for grades I to III, but not for grade IV (p = 0.625). EOR improvement ranged from a median change of 0.0% (IQR 0.0–4.5%) for grade I to 4.4% (IQR 0.0–9.0%) for grade IV, with a significant improvement only for grades I to III (p < 0.05).
Conclusions
Interestingly, this study shows that clinical utility of 3T-iMRI is highest in the more “simple” adenomas (ZPS grades I–II) than for the more “complex” ones (ZPS grade III–IV). Grade I adenomas are amenable to GTR if 3T-iMRI is implemented. In grade III adenomas, EOR and RV can be improved to clinically relevant levels. Conversely, in grade IV adenomas, 3T-iMRI may be of limited use.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In recent decades, endonasal transsphenoidal surgery (TSS) has established itself as the gold standard technique for surgical treatment of pituitary adenomas (PA)[1,2,3,4,5] With both endoscopic and microscopic techniques, high rates of gross total resection (GTR) can be achieved in a relatively safe procedure with minimal morbidity and mortality [1, 6,7,8,9].
Several factors are known to influence the likelihood of GTR, including adenoma and sellar morphology [3, 9,10,11,12,13], diameter, and volume [13, 14], as well as invasion into the cavernous sinus space (CSS) [8, 9, 11] and sellar dura [15]. Some of these factors have been compounded into classification schemes, such as the Knosp classification [9, 11] and the Zurich Pituitary Score (ZPS) [13]. Both demonstrate a strong correlation with the likelihood of GTR [9, 13]. There is some evidence that the Knosp classification may be more suitable for prediction of residual tumor in the CSS [8, 9, 13], while the ZPS relates more strongly to volumetric extent of resection (EOR) and residual tumor volume (RV) and has excellent interrater agreement [9, 13, 16].
In patients harboring hormone-secreting adenomas, especially, but not exclusively, GTR is the primary surgical goal [17], since residual tumor volume is associated with postoperative morbidity and mortality [17,18,19,20]. Preservation and if possible restoration of endocrinological and neurological function and nasal quality of life [2, 21,22,23,24] are further treatment goals. Intraoperative high-field magnetic resonance imaging (3T-iMRI) is an assistive technique which has been reported to improve GTR, RV, and EOR by several authors [25,26,27,28,29,30,31,32,33,34,35,36,37].
The utility of intraoperative MRI for pituitary surgery is highly debated in the neurosurgical community. Several authors keep that the reported improvement in GTR, RV, and EOR rate would be the result of the more conservative attitude of those surgeons who, knowing they will perform 3T-iMRI, tend to be less aggressive in the surgical phase prior to imaging [38]. Other authors recommend not to use intraoperative imaging due to its alleged high incidence of false positives [39]. Indeed, despite its clear benefits, 3T-iMRI is costly, prolongs operative time, and is not always available. Understanding better whether 3T-iMRI is going to be of value in a particular patient has the potential to improve patient counseling, surgical decision-making, cost-effectiveness, and outcome prediction.
The purpose of this study was to investigate whether simple pre-operative imaging grading systems can predict the utility of 3T-iMRI in patients undergoing endoscopic TSS for PA. Specific interest was set to test the common assumption that intraoperative imaging leads to measurable benefits particularly in higher-grade PAs.
Materials and methods
Patients
A consecutive series of patients that underwent 3T-iMRI-assisted endoscopic TSS for PA performed by two senior neurosurgeons (L.R. and C.S.) at the Department of Neurosurgery of our institution was evaluated. From October 2012 onwards, all patients undergoing TSS for a PA were treated according to the same protocol with clinical and radiological data collected in a prospective registry [34]. A mononostril approach was used in all patients in this series. Inclusion criteria were the availability of pre-, intra-, and 3-month postoperative imaging. Patients undergoing transcranial or combined procedures, as well as those in which the initial surgical goal was decompression only, were excluded. 3T-iMRI was carried out routinely, unless medically contraindicated [40]. This manuscript was compiled according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement [41]. The registry was approved by our institutional committee (KEK St-V-Nr 2015-0142), and data were treated according to the Declaration of Helsinki.
Outcome measures
Primary endpoint was the rate of conversion to GTR after 3T-iMRI, defined as the percentage of patients who underwent further resection because of 3T-iMRI, eventually reaching GTR.
All patients underwent pre-, intra-, and 3-month postoperative volumetric contrast-enhanced MRI (Siemens 3-T Skyra VD13) at a field strength of 3 T. Extent of resection (EOR) was measured on the 3-month postoperative MRI and was calculated as the percentagewise reduction of RV to baseline tumor volume on preoperative MRI. GTR was defined as an EOR of 100%. Adenoma morphology was graded according to the ZPS [13] as well as the modified Knosp classification [9]. Each adenoma was manually contoured on source volumetric sequences to allow subsequent 3D rendering and volumetric measurement (iPlan Cranial, BrainLab). The utility of 3T-iMRI was assessed as the difference in outcome measures between the endoscopic result (3T-iMRI) and the final result (3-month MRI).
Statistical analysis
Continuous data are given as mean ± standard deviation (SD), medians (interquartile ranges (IQR)), and ranges, whereas categorical data are presented as numbers (percentages). Since no missing data was observed for the analyzed variables and the included patients, there was no need for imputation. Comparisons of paired continuous data were made using the exact version of Wilcoxon’s signed-rank test [42], while McNemar’s test was used on dichotomous data. Trends over ZPS grades were statistically tested for. The Cochrane-Armitage test was applied for dichotomous variables, and the exact version of the Jonckheere-Terpstra test, based on 10,000 permutations, was applied for continuous variables. A p ≤ 0.05 was considered statistically significant. All analyses were carried out in R version 3.5.1 (The R Foundation for Statistical Computing, Vienna, Austria) [43]. The statistical code is provided as Supplementary Methods 1.
Results
Patient population
A total of 95 patients underwent TSS using 3T-iMRI and fit criteria for inclusion (Table 1). There was no missing data. Most patients (68%) harbored non-functioning adenomas (NFPA). Adenoma morphology corresponded most commonly to ZPS grade II (49%), Hardy grade II (33%), and Knosp grade 2 (29%).
Gross total resection
Overall, GTR conversion rates increased from an endoscopic 44% to a final 72% (Table 2). When stratified by ZPS grade, endoscopic GTR rates were 67%, 43%, 25%, and 0% for ZPS I to IV, respectively (Fig. 1). On 3-month follow-up imaging, we observed GTR in 100%, 74%, 38%, and 0% of patients, respectively (p < 0.001). Rates of conversion to GTR after 3T-iMRI were significantly higher in low-ZPS-grade patients (Fig. 2), decreasing steadily from 33% for grade I to 0% for grade IV adenomas. Statistically significant conversion rates were seen only for grade I (p = 0.008) and grade II (p < 0.001).
Residual tumor volume
The median preoperative tumor volume was 3.1 (IQR 1.17 to 5.93) cm3, which decreased to a median intraoperative RV of 0.08 (IQR 0.0 to 0.4) cm3 and a median overall final RV of 0.0 (0.0 to 0.08, p < 0.001) cm3 (Table 3). Median reduction of RV per ZPS grade was statistically significant for grade I (− 0.0, IQR − 0.06 to 0.0, p = 0.004) and grade II (0.0, IQR − 0.32 to 0.0, p < 0.001), as well as grade III (− 0.15, IQR − 0.42 to 0.0, p = 0.001), but not for grade IV (− 0.35, IQR − 0.55 to 0.0, p = 0.625) adenomas (Fig. 3). The decreasing trend in RV change reached statistical significance (p = 0.008).
Residual tumor volumes (RV) as measured a on intraoperative magnetic resonance imaging (MRI) and b on the 3-month follow-up MRI, stratified by the Zurich Pituitary Score. Values are provided in cubic centimeters. An extreme outlier (grade III) presenting with an intraoperative RV of 14.55 cm3 is not depicted in order to preserve scale
Extent of resection
Overall, EOR increased from an endoscopic median of 97.8% (IQR 90.2 to 100%) to a final 100% (IQR 99.0 to 100%) at 3 months postoperatively (Table 4). When stratified by ZPS grade, median endoscopic GTR rates were 100%, 97.1%, 95.9%, and 80.5% for ZPS I to IV, respectively (Supplementary Figure 1). On 3-month follow-up imaging, we observed median EORs of 100%, 100%, 99.0%, and 89.5% respectively. Rates of improvement in EOR after 3T-iMRI were significant only for grades I to III (all p < 0.05) although the trend in EOR change failed to reach statistical significance (p = 0.085).
Discussion
In a prospective registry of patients operated for PA, we investigated whether subsets of patients exist in which 3T-iMRI is most valuable as an intraoperative assistive technique and conversely such in which it is of little added value. Our approach was to identify these subsets of patients by grading adenoma morphology and invasion into adjacent structures according to the ZPS which had shown to be effective in predicting GTR, EOR, and RV [13]. Our data demonstrate that there are differences in the utility of intraoperative imaging when patients are stratified by this radiological classification. In particular, GTR can virtually always be achieved in ZPS grade I adenomas when 3T-iMRI is utilized. Conversely, the benefit in grade IV adenomas is inconsequential, since it is likely that no improvement in GTR rate can be achieved, while the change in EOR and RV is not clinically relevant. For medium-grade adenomas, intraoperative imaging may be useful, as both EOR and RV can be improved to satisfactory levels, although no statistically significant benefit in GTR was proven in our series.
In the modern era, maximization of tumor resection is possible with minimal morbidity and mortality [2, 23, 26, 37]. In the quest for maximal resection, several adjuvant techniques have been introduced, among them endoscopy and intraoperative imaging [39]. Although class I evidence is lacking, such tools are reported by several centers to be beneficial for reaching higher GTR rates and are thus seeing increased use [27].
Currently, only few centers have implemented high-field intraoperative MRI in their routine clinical practice. This may be partially explained by the inherent costs and prolonged operative time [27, 28]. Furthermore, 3T-iMRI is not always feasible, either for logistic or for medical reasons. Little is known on the specific utility of 3T-iMRI in certain morphological subsets of pituitary adenomas. Paľa et al. found that in 28 patients who underwent endoscopic TSS, GTR increased by 6.6% for Knosp grade 0–2 adenomas and by 15.4% for Knosp grade 3–4 adenomas after 1.5T-iMRI [32]. They conclude that the added value of intraoperative imaging increases with the complexity of the treated tumor. Apart from this data, the analysis of our prospective registry is the only currently available report dedicated to this research question.
Our analysis indicates that the added value of 3T-iMRI may be greatest in low-grade adenomas. Although this may seem paradoxical at first, it reflects our clinical experience in using routine 3T-iMRI for PA in TSS. Intrasellar remnants are technically always removable. However, even in experienced hands, they may be missed by endoscopic inspection, particularly if they are very small (see Table 3). The use of high-field intraoperative (io)-MRI guarantees that such tiny rests do not go overlooked. Intracavernous remnants on the contrary will not be resectable even detected on io-MRI. Because ZPS grade I and II adenomas are unlikely to infiltrate laterally into the CSS and because in these cases, the surgical corridor allows full access to the entire extent of the tumor [10, 13], these adenomas should in theory always be amenable to GTR. In other words, a missed intrasellar residual, particularly in ZPS grade I, should be considered as a gross surgical failure. In our series, the benefit of 3T-iMRI in ZPS grade I and II adenomas was evident, increasing the rate of GTR from 51% intraoperatively to 84% at 3 months and leading to GTR in 100% of grade I adenomas. To a lesser extent, GTR in grade III tumors increased from 25 to 38%, without statistical significance. Still, in grade III adenomas, we demonstrated a statistically significant improvement in both EOR and RV, both reaching excellent levels (see Table 3). High EOR and low RV have been repeatedly proven to be associated with improved outcome, particularly for functioning adenomas [44,45,46]. Thus, one could say that particularly in grade I and II adenomas, the use of high-field intraoperative imaging shows the highest benefit, since GTR should always be the desired outcome (Fig. 4).
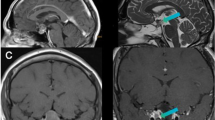
Illustrative case. Preoperatively, a large macroadenoma with a Zurich Pituitary Score of III was seen (a). On intraoperative magnetic resonance imaging (b), residual tumor tissue was detected intrasellarly and invading into the left cavernous sinus space, which was fully resected after intraoperative imaging (c)
Pituitary adenomas encasing the internal carotid artery (grade IV) did not appear to benefit in the same way. Intuitively, one might be inclined to think that especially in very complex and invasive tumors, the utility of intraoperative imaging may be the highest [32]. In our series, 3T-iMRI did not lead to conversion to GTR in any of the grade IV cases, and only marginal improvements in RV and EOR were observed. While this improvement probably did not reach statistical significance due to low statistical power, clinical significance is not given because 3T-iMRI failed to elevate both EOR and RV to clinically relevant levels, i.e., to a negligibly low final RV. This is illustrated in Figs. 3 and 4. In addition, this finding is corroborated by case series both with and without high-field intraoperative MRI, which all report an extremely low rate of GTR in adenomas encasing the internal carotid artery [8, 9, 13, 35].
However, other reasons for which intraoperative imaging may be useful in more complex PA must be taken into account but are not the purpose of this report. For example, in adenomas extending into the suprasellar space, the surgeon may opt for 3T-iMRI to monitor the position of the diaphragma sellae to reduce the risk of intraoperative CSF leaks and postoperative fistulas or to image a specific part of the tumor compressing the optic chiasm, such as reported by Zaidi et al. [37].
Limitations
Even though data were obtained from a prospective registry, this was a retrospective analysis. Thus, selection bias is possible, albeit inclusion criteria were clearly defined and the majority of patients in the registry were eligible for inclusion. All data used in this analysis stem from the same center, possibly limiting the generalizability of our findings. However, the baseline characteristics of the included patients, as well as the surgical results, are in line with the available literature. As expected, the number of grade IV adenomas was low, limiting the pairwise statistical analysis in power.
Conclusions
Using data from a prospective registry of TSS for PA, we demonstrate that there are subsets of patients in which the use of 3T-iMRI is particularly advantageous. These subsets can be described by the ZPS. The clinical utility of 3T-iMRI is probably most noticeable in ZPS grade I–III adenomas. In particular, grade I adenomas were always amenable to GTR if 3T-iMRI is implemented. Conversely, in grade IV adenomas, 3T-iMRI may be of limited use, because it does not appear that its use leads to clinically relevant improvements in surgical outcome. Our findings warrant validation in external populations.
References
Barker FG, Klibanski A, Swearingen B (2003) Transsphenoidal surgery for pituitary tumors in the United States, 1996-2000: mortality, morbidity, and the effects of hospital and surgeon volume. J Clin Endocrinol Metab 88(10):4709–4719
Fatemi N, Dusick JR, Mattozo C, McArthur DL, Cohan P, Boscardin J, Wang C, Swerdloff RS, Kelly DF (2008) Pituitary hormonal loss and recovery after transsphenoidal adenoma removal. Neurosurgery 63(4):709–719
Hardy J, Vezina JL (1976) Transsphenoidal neurosurgery of intracranial neoplasm. Adv Neurol 15:261–273
Mehta GU, Oldfield EH (2012) Prevention of intraoperative cerebrospinal fluid leaks by lumbar cerebrospinal fluid drainage during surgery for pituitary macroadenomas. J Neurosurg 116(6):1299–1303
Przybylowski CJ, Dallapiazza RF, Williams BJ, Pomeraniec IJ, Xu Z, Payne SC, Laws ER, Jane JA (2016) Primary versus revision transsphenoidal resection for nonfunctioning pituitary macroadenomas: matched cohort study. J Neurosurg 126(3):889–896
Agam MS, Wedemeyer MA, Wrobel B, Weiss MH, Carmichael JD, Zada G (2018) Complications associated with microscopic and endoscopic transsphenoidal pituitary surgery: experience of 1153 consecutive cases treated at a single tertiary care pituitary center. J Neurosurg 1–8. https://doi.org/10.3171/2017.12.JNS172318
Chen C-J, Ironside N, Pomeraniec IJ, Chivukula S, Buell TJ, Ding D, Taylor DG, Dallapiazza RF, Lee C-C, Bergsneider M (2017) Microsurgical versus endoscopic transsphenoidal resection for acromegaly: a systematic review of outcomes and complications. Acta Neurochir 159(11):2193–2207
Dhandapani S, Singh H, Negm HM, Cohen S, Anand VK, Schwartz TH (2016) Cavernous sinus invasion in pituitary adenomas: systematic review and pooled data meta-analysis of radiologic criteria and comparison of endoscopic and microscopic surgery. World Neurosurg 96:36–46
Micko ASG, Wöhrer A, Wolfsberger S, Knosp E (2015) Invasion of the cavernous sinus space in pituitary adenomas: endoscopic verification and its correlation with an MRI-based classification. J Neurosurg 122(4):803–811
Elhadi AM, Hardesty DA, Zaidi HA, Kalani MYS, Nakaji P, White WL, Preul MC, Little AS (2015) Evaluation of surgical freedom for microscopic and endoscopic transsphenoidal approaches to the sella. Neurosurgery 11(Suppl 2):69–78 discussion 78-79
Knosp E, Steiner E, Kitz K, Matula C (1993) Pituitary adenomas with invasion of the cavernous sinus space: a magnetic resonance imaging classification compared with surgical findings. Neurosurgery 33(4):610–617 discussion 617-618
Serra C, Maldaner N, Muscas G, Staartjes V, Pangalu A, Holzmann D, Soyka M, Schmid C, Regli L (2017) The changing sella: internal carotid artery shift during transsphenoidal pituitary surgery. Pituitary:1–7
Serra C, Staartjes VE, Maldaner N, Muscas G, Akeret K, Holzmann D, Soyka MB, Schmid C, Regli L (2018) Predicting extent of resection in transsphenoidal surgery for pituitary adenoma. Acta Neurochir. https://doi.org/10.1007/s00701-018-3690-x
Hofstetter CP, Nanaszko MJ, Mubita LL, Tsiouris J, Anand VK, Schwartz TH (2012) Volumetric classification of pituitary macroadenomas predicts outcome and morbidity following endoscopic endonasal transsphenoidal surgery. Pituitary 15(3):450–463
Meij BP, Lopes M-BS, Ellegala DB, Alden TD, Laws ER (2002) The long-term significance of microscopic dural invasion in 354 patients with pituitary adenomas treated with transsphenoidal surgery. J Neurosurg 96(2):195–208
Mooney MA, Hardesty DA, Sheehy JP, Bird R, Chapple K, White WL, Little AS (2016) Interrater and intrarater reliability of the Knosp scale for pituitary adenoma grading. J Neurosurg 126(5):1714–1719
Grabowski MM, Recinos PF, Nowacki AS, Schroeder JL, Angelov L, Barnett GH, Vogelbaum MA (2014) Residual tumor volume versus extent of resection: predictors of survival after surgery for glioblastoma. J Neurosurg 121(5):1115–1123
Jahangiri A, Wagner J, Han SW et al (2014) Morbidity of repeat transsphenoidal surgery assessed in more than 1000 operations. J Neurosurg 121(1):67–74
Negm HM, Al-Mahfoudh R, Pai M, Singh H, Cohen S, Dhandapani S, Anand VK, Schwartz TH (2016) Reoperative endoscopic endonasal surgery for residual or recurrent pituitary adenomas. J Neurosurg:1–12
Sughrue ME, Chang EF, Gabriel RA, Aghi MK, Blevins LS (2011) Excess mortality for patients with residual disease following resection of pituitary adenomas. Pituitary 14(3):276–283
Nelson AT, Tucker HSG, Becker DP (1984) Residual anterior pituitary function following transsphenoidal resection of pituitary macroadenomas. J Neurosurg 61(3):577–580
Soyka MB, Serra C, Regli L, Meier E, Holzmann D (2017) Long-term olfactory outcome after nasoseptal flap reconstructions in midline skull base surgery. Am J Rhinol Allergy 31(5):334–337
Staartjes VE, Stricker S, Muscas G, Maldaner N, Holzmann D, Burkhardt J-K, Seifert B, Schmid C, Serra C, Regli L (2018) Intraoperative unfolding and postoperative pruning of the pituitary gland after transsphenoidal surgery for pituitary adenoma: a volumetric and endocrinological evaluation. Endocrine. https://doi.org/10.1007/s12020-018-1758-2
Webb SM, Rigla M, Wägner A, Oliver B, Bartumeus F (1999) Recovery of hypopituitarism after neurosurgical treatment of pituitary adenomas. J Clin Endocrinol Metab 84(10):3696–3700
Berkmann S, Schlaffer S, Nimsky C, Fahlbusch R, Buchfelder M (2014) Follow-up and long-term outcome of nonfunctioning pituitary adenoma operated by transsphenoidal surgery with intraoperative high-field magnetic resonance imaging. Acta Neurochir 156(12):2233–2243
Buchfelder M, Schlaffer S-M (2012) Intraoperative magnetic resonance imaging during surgery for pituitary adenomas: pros and cons. Endocrine 42(3):483–495
Chittiboina P (2017) iMRI during transsphenoidal surgery (TSS). Neurosurg Clin N Am 28(4):499–512
Coburger J, König R, Seitz K, Bäzner U, Wirtz CR, Hlavac M (2014) Determining the utility of intraoperative magnetic resonance imaging for transsphenoidal surgery: a retrospective study. J Neurosurg 120(2):346–356
Fomekong E, Duprez T, Docquier M-A, Ntsambi G, Maiter D, Raftopoulos C (2014) Intraoperative 3T MRI for pituitary macroadenoma resection: initial experience in 73 consecutive patients. Clin Neurol Neurosurg 126:143–149
Netuka D, Masopust V, Belšán T, Kramář F, Beneš V (2011) One year experience with 3.0 T intraoperative MRI in pituitary surgery. Acta Neurochir Suppl 109:157–159
Nimsky C, von Keller B, Ganslandt O, Fahlbusch R (2006) Intraoperative high-field magnetic resonance imaging in transsphenoidal surgery of hormonally inactive pituitary macroadenomas. Neurosurgery 59(1):105–114
Paľa A, Knoll A, Brand C, Etzrodt-Walter G, Coburger J, Wirtz CR, Hlaváč M (2017) The value of intraoperative MRI in endoscopic and microsurgical transsphenoidal pituitary adenoma resection. World Neurosurg. https://doi.org/10.1016/j.wneu.2017.02.132
Pamir MN (2011) 3 T ioMRI: the Istanbul experience. Acta Neurochir Suppl 109:131–137
Serra C, Burkhardt J-K, Esposito G, Bozinov O, Pangalu A, Valavanis A, Holzmann D, Schmid C, Regli L (2016) Pituitary surgery and volumetric assessment of extent of resection: a paradigm shift in the use of intraoperative magnetic resonance imaging. Neurosurg Focus 40(3):E17
Sylvester PT, Evans JA, Zipfel GJ et al (2015) Combined high-field intraoperative magnetic resonance imaging and endoscopy increase extent of resection and progression-free survival for pituitary adenomas. Pituitary 18(1):72–85
Szerlip NJ, Zhang Y-C, Placantonakis DG, Goldman M, Colevas KB, Rubin DG, Kobylarz EJ, Karimi S, Girotra M, Tabar V (2011) Transsphenoidal resection of sellar tumors using high-field intraoperative magnetic resonance imaging. Skull Base 21(4):223–232
Zaidi HA, De Los RK, Barkhoudarian G, Litvack ZN, Bi WL, Rincon-Torroella J, Mukundan S, Dunn IF, Laws ER (2016) The utility of high-resolution intraoperative MRI in endoscopic transsphenoidal surgery for pituitary macroadenomas: early experience in the advanced multimodality image guided operating suite. Neurosurg Focus 40(3):E18
Schwartz TH, Stieg PE, Anand VK (2006) Endoscopic transsphenoidal pituitary surgery with intraoperative magnetic resonance imaging. Oper Neurosurg 58(suppl_1):ONS-44-ONS-51
Kuo JS, Barkhoudarian G, Farrell CJ, Bodach ME, Tumialan LM, Oyesiku NM, Litvack Z, Zada G, Patil CG, Aghi MK (2016) Congress of Neurological Surgeons systematic review and evidence-based guideline on surgical techniques and technologies for the management of patients with nonfunctioning pituitary adenomas. Neurosurgery 79(4):E536–E538
Stienen MN, Fierstra J, Pangalu A, Regli L, Bozinov O (2018) The Zurich checklist for safety in the intraoperative magnetic resonance imaging suite: technical note. Oper Neurosurg Hagerstown Md. https://doi.org/10.1093/ons/opy205
Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, Initiative STROBE (2007) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335(7624):806–808
Streitberg B, Röhmel J (1986) Exact distributions for permutation and rank tests: an introduction to some recently published algorithms. Stat Softw Newsl 12(1):10–17
Core Team R (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Colao A, Attanasio R, Pivonello R, Cappabianca P, Cavallo LM, Lasio G, Lodrini A, Lombardi G, Cozzi R (2006) Partial surgical removal of growth hormone-secreting pituitary tumors enhances the response to somatostatin analogs in acromegaly. J Clin Endocrinol Metab 91(1):85–92
O’Sullivan EP, Woods C, Glynn N, Behan LA, Crowley R, O’Kelly P, Smith D, Thompson CJ, Agha A (2009) The natural history of surgically treated but radiotherapy-naïve nonfunctioning pituitary adenomas. Clin Endocrinol 71(5):709–714
Schwyzer L, Starke RM, Jane JA, Oldfield EH (2014) Percent reduction of growth hormone levels correlates closely with percent resected tumor volume in acromegaly. J Neurosurg 122(4):798–802
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Cantonal Ethics Committee Zürich, KEK St-V-Nr 2015-0142) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Pituitaries
Electronic supplementary material
701_2019_4018_MOESM1_ESM.r
Supplementary Methods 1 Supplementary Content 1. R Code for the statistical analysis and figure rendering. The code was executed in R Version 3.5.1 (The R Foundation for Statistical Computing, Vienna, Austria) on a machine running Windows 10 (Microsoft Corp., Redmond, WA, USA). (R 17 kb)

Supplementary Figure 1
Extent of resection (EOR) in percent as measured (A) on intraoperative magnetic resonance imaging (MRI) and (B) on the 3-month follow-up MRI, stratified by Zurich Pituitary Score. An extreme outlier (Grade II) presenting with an intraoperative EOR of 19.6% is not depicted in order to preserve scale. (JPG 8296 kb)
Rights and permissions
About this article
Cite this article
Staartjes, V.E., Serra, C., Maldaner, N. et al. The Zurich Pituitary Score predicts utility of intraoperative high-field magnetic resonance imaging in transsphenoidal pituitary adenoma surgery. Acta Neurochir 161, 2107–2115 (2019). https://doi.org/10.1007/s00701-019-04018-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-019-04018-9