Abstract
Purpose
Radical oxygen species produced after injury counteracts antioxidant activity and frequently causes severe oxidative stress for the tissues. Alpha-lipoic acid is a powerful metabolic antioxidant with immunomodulatory effects which provides neuroprotection. The aim of this study is to investigate the neuroprotective and anti-apoptotic effects of alpha-lipoic acid on spinal cord ischemia–reperfusion.
Methods
Twenty-four adult, male, New Zealand rabbits were divided into sham (n = 8), control (n = 8), and treatment groups (n = 8). The abdominal aorta was clamped for 30 min by an aneurysm clip, approximately 1 cm below the renal artery and 1 cm above the iliac bifurcation in control and treatment groups. Only laparotomy was performed in the sham group. Twenty-five cubic centimeters of saline in control group and 100 mg/kg lipoic acid were administered intraperitoneally in the treatment group after closure of the incision. The animals were killed 48 h later. Spinal cord segments between L2 and S1 were harvested for analysis. Levels of nitric oxide, glutathione, malondialdehyde, advanced oxidation protein products, and superoxide dismutase were analyzed as markers of oxidative stress and inflammation. Caspase-3 activity was analyzed to detect the effect of lipoic acid on apoptosis.
Results
In all measured parameters of oxidative stress, administration of lipoic acid significantly demonstrated favorable effects. Both plasma and tissue levels of nitric oxide, glutathione, malondialdehyde, and advanced oxidation protein products significantly changed in favor of antioxidant activity. There was no significant difference between the plasma superoxide dismutase levels of the groups. Histopathological evaluation of the tissues also demonstrated significant decrease in cellular degeneration and infiltration parameters after lipoic acid administration. However, lipoic acid has no effect on caspase-3 activity.
Conclusions
Although further studies considering different dose regimens and time intervals are required, the results of the present study prove that alpha-lipoic acid has favorable effects on experimental spinal cord ischemia–reperfusion injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal cord injury (SCI) has traumatic and nontraumatic origin and often leads to catastrophic dysfunction and disability. The pathophysiologic mechanisms that underlie hypoxic/ischemic injury to the spinal cord have not been totally explained yet. The neurological damage at the time of insult is called “primary injury”. Activation of endogenous substances enhances the secondary damage in spinal cord injury. Inflammation and free radical formation play an important role in pathological mechanisms involved in secondary damage [4, 17, 19, 20, 31, 46]. Ischemic injury in neural tissue is aggravated by reperfusion, resulting in lipid peroxidation and fiber degeneration, progressive loss of motor neurons accompanied with a steady decline of motor function [14, 40]. Glutamate-mediated excitotoxicity, formation of reactive oxygen species (ROS), and lipid peroxidation are prominent events thought to contribute to neuronal dysfunction and cell loss following traumatic and ischemic injury to the central nervous system (CNS). Both types of injury have been shown to result in increased glutamate release, the sustained activation of glutamate receptors, and the increased accumulation of calcium (Ca+2). There is direct evidence that activation of glutamate receptors and the Ca+2 influx induces the formation of ROS, superoxide anion, and hydrogen peroxide [3, 22].
alpha-Lipoic acid (LA) is a neuroprotective metabolic antioxidant with proven immunomodulatory effects [5]. LA is both hydrophilic and lipophilic which is a unique feature among antioxidants. For this reason, it is referred as “antioxidant of antioxidants” [61]. LA potently suppresses clinical and pathological diseases in the animal model of multiple sclerosis, autoimmune encephalomyelitis, ischemia–reperfusion (I/R) injury of heart [57, 58], experimental diabetic neuropathy [9, 45], and peripheral nerves and brain [10, 12, 43, 48, 68]. LA has also immunomodulatory effect in CNS by inhibiting T cell migration and increasing cyclic adenosine monophosphate [12, 55]. However, effectiveness of LA in ischemia–reperfusion injury in spinal cord has not been investigated yet. The aim of this study is to investigate the neuroprotective and anti-apoptotic effects of alpha-lipoic acid on an experimental spinal cord ischemia–reperfusion injury model.
Materials and method
This study was performed in Gazi University School of Medicine, Experimental Research Center. The experimental protocol was evaluated and approved by the Ethics Review Committee of Gazi University School of Medicine. All animals received humane care, in compliance with the “Principles of Laboratory Animal Care” formulated by the National Society for Medical Research and the “Guide for the Care and Use of Laboratory Animals” prepared by the Institute of Laboratory Animal Resources and published by the National Institute of Health (NIH publication no. 85-23, revised 1986).
Totally, 24 adult male New Zealand rabbits weighing between 2.5 and 3 kg were divided into sham (n = 8), control (n = 8), and treatment groups (n = 8). The animals were kept at optimal (18–21°C) room temperature and fed with standard diet. A 12-h light–dark cycle was implemented. Free access to food and water was allowed.
The animals were anesthetized by intramuscular injection of 70 mg/kg ketamine (Ketalar, Parke-Davis, Eczacıbaşı, Istanbul, Turkey) and 5 mg/kg xylazine (Rompun, Bayer, Istanbul, Turkey) and allowed breath spontaneously. Body temperatures were measured by rectal thermometry and maintained at 37°C with a heating pad. The arterial pressure and heart rate were monitored continually. The spinal cord ischemia–reperfusion model described by Erten et al. [19] was used. Animals were placed in supine position. After sterile preparation, a 10-cm midline incision was made and the abdominal aorta was exposed through a transperitoneal approach. One hundred fifty units per kilogram heparin was administered through intravenous route 5 min before clamping for anticoagulation. The aorta was cross clamped at two sites by using two aneurysm clips of 70 g closing force (Yasargil FE 721, Aesculap, Germany). The occlusion sites were approximately 1 cm below the renal artery and 1 cm above the bifurcation and performed under surgical microscope. Cross clamp time was 30 min. At the end of the occlusion period, the clips were removed and restoration of blood flow was visually verified. The incision was closed in layers. Only laparotomy was performed in the sham group, and cross clamping of aorta for 30 min was performed in the treatment groups. Additionally, control group animals received 25 cc of normal saline, and the treatment group animals received 100 mg/kg lipoic acid (Thioctacid, Gen İlaç, Ankara, Turkey) intraperitoneally after closure of the incisions.
Free access to food and water was allowed 2 h after operation. Credé’s maneuver was performed to animals with neurogenic bladder at least two times a day. The animals were killed 48 h after operation by injection of pentobarbital (200 mg/kg). Ten-cubic centimeter blood sample was taken from the heart for biochemical analysis in plasma. Spinal cord segment between L2 and S1 were quickly harvested after total laminectomies from T12 to S1 and cutting the nerve roots. Spinal cord segments between L2 and L5 were removed for biochemical and histopathological analysis.
Levels of nitric oxide (NO), glutathione (GSH), malondialdehyde (MDA), advanced oxidation protein products (AOPP), and superoxide dismutase (SOD) were analyzed in spinal cord and plasma.
Histopathology and immunohistochemistry
Formalin-fixed paraffin-embedded tissue sections of 5 µ were incubated for one night at 37°C and for 1 h at 60°C. Xylol applications (15 min) were performed twice. The slides were then laid in 96% absolute alcohol and 80% ethanol for 10 min, followed by distilled water, twice for 5 min. They were boiled in high temperature microwave oven in 10% citrate buffer for caspase 3 immunoperoxidase. After 20 min at room temperature, the tissue was encircled with a pap-pen (hydrophobic pen). After washing with distilled water and phosphate-buffered saline (PBS), hydrogen peroxide was added dropwise. After washing with PBS, ultra V block (Cat. # TA-125-UB Thermo Fisher Scientific, Fremont, CA, 94539, USA) was applied. After application of primary antibody (dilution ratio 1/200, Cat # RB-1197, Labvision/NeoMarkers Corporation, Fremont CA, USA) for 1 h, the samples were washed with PBS and a post-PBS level was applied (biotinylated goat antipolyvalent and streptavidin peroxidase, respectively). After rewashing with PBS, the specimens were placed in 10 min 3-amino, 9-ethylcarbazole (Cat. # TA-125-HA Thermo Fisher Scientific, Fremont, CA, 94539, USA) chromogen. Finally, the counterstain with Mayer’s hematoxylin was performed for 5 min. All slides were evaluated with Leica DMI 4000 B light microscope (Leica, Wetzlar, Germany).
To prevent interindividual bias, all tissues were evaluated by the same histologist (N.L.), who was blinded to the origin of the samples.
Axonal damage, neuronal degeneration, and astrocyte and microglia infiltration were analyzed for histopathological changes. Axonal damage was graded as G0 (normal), G1 (mild swelling and vacuolization in axons), and G2 (severe swelling and vacuolization in axons). The intensity of degenerated neurons was calculated in each field. Microglia and astrocyte infiltration was graded as +1, +2, +3, and +4 according to their intensity in each field.
The relative intensity of immunoreactivity staining was assessed quantitatively as previously described by McCarty et al. [41], taking into account both the intensity and the distribution of a specific staining. A value of histological score (HSCORE) was derived from the sum of the percentages of positively stained epithelial cells multiplied by the weighted intensity of staining. HSCORE = Σ Pi (I + 1), where “I” represents staining intensity (0 = no expression, 1 = mild, 2 = moderate, and 3 = intense) and Pi is the percentage of stained cells for each intensity [8].
Biochemical analyzes
AOPP levels were measured by a spectrophotometric method (Shimadzu UV 1601 spectrophotometer, Shimadzu, Tokyo, Japan) in the presence of potassium iodide at 340 nm [67] and calibrated with chloramine-T solutions. AOPP levels were expressed in micromoles chloramine-T equivalents per liter.
Lipid peroxidation was estimated using the thiobarbituric acid reactive substances (TBARS) test as described previously [11, 70, 71]. Briefly, TBARS formation was quantitated using 1, 1, 3, 3-tetraethoxypropane as standard, and the absorbances of the TBARS were read at 532 nm using Shimadzu UV 1601 spectrophotometer, Shimadzu, Tokyo, Japan.
The NO levels were estimated by the method of Miranda et al. and Taskiran et al. [42, 64]. Samples were deproteinized with 0.3 M NaOH and 5% (w/v) ZnSO4, centrifuged, and supernatants were used for the assays. After loading the plate with samples (100 μl) at room temperature, addition of vanadium III chloride (VCl3; 100 μl) to each well was rapidly followed by addition of Griess reagents, sulfanilamide (50 μl), and N-(1-naphtyl)ethylenediamine dihydrochloride (50 μl). After incubation (usually 30–45 min), samples were measured at 540 nm using an ELISA reader.
The SOD activity measurements were carried out by inhibiting the SOD activity by nitro blue tetrazolium reduction. Xanthine–xanthine oxidase was used as a superoxide generator, and 1 IU was defined as the quantity of SOD required to produce 50% inhibition [63].
Protein levels were determined by a spectrophotometric method (Shimadzu UV 1601 spectrophotometer, Shimadzu, Tokyo, Japan) using bovine serum albumin as the standard [39].
The tissue GSH levels were determined by a modified Ellman method [2]. Briefly, after centrifugation at 3,000 rpm for 10 min, 0.5 ml of supernatant was added to the 2 ml of 0.3 M Na2HPO4.2H2O solution. Next, 0.2 ml solution of dithiobisnitrobenzoic acid solution (0.4 mg/ml in 1% sodium citrate) was added, and the absorbance at 412 nm was measured immediately after mixing. The GSH levels were calculated using an extinction coefficient of 13,000 mol−1 cm−1.
The plasma GSH levels were determined as R-SH [37]; 0.5 ml of each sample was mixed with 1 ml of a solution containing 100 mM Tris-HCl, pH 8.2, 1% sodium dodecyl sulfate, and 2 mM EDTA. The mixture was incubated for 5 min at 25°C and centrifuged to remove any precipitate. 5,5-Dithiobis (2-nitrobenzoic acid) 0.3 mM was then added to each reaction volume and incubated for 15 min at 37°C. The absorbance of each sample was determined at 412 nm. The R-SH levels were calculated assuming a molar extinction coefficient of 13,000 mol−1 cm−1 at 412 nm.
Statistical analysis
Data collected during the experiment were analyzed using SPSS 15 for Windows. Kruskal–Wallis variance test was used for nonparametric data to compare differences among groups. When analysis of variance showed significance, Mann–Whitney U test was applied to determine the difference. Histopathological data was compared using Levene’s test. The data were expressed as mean ± standard deviation, and probability value less than 0.05 was accepted as statistically significant.
Results
Biochemical analysis
Tissue changes
Tissue NO level was 31.20 ± 1.7 in the sham group, and induction of the ischemia–reperfusion injury significantly increased this level to 57.01 ± 14.06 in the control group (p = 0.004). Administration of LA resulted in significant decrease in the tissue NO level of the treatment group to a value of 30.21 ± 3.56 (p = 0.004).
Tissue MDA level was 3.20 ± 0.80 in the sham group and induction of the ischemia–reperfusion injury significantly increased this level to 6.61 ± 1.65 in the control group (p = 0.010). Administration of LA resulted in significant decrease in the tissue MDA level of the treatment group to a value of 3.41 ± 0.64 (p = 0.008).
Tissue GSH level was 1.17 ± 0.33 in the sham group and induction of the ischemia–reperfusion injury significantly decreased this level to 0.45 ± 0.21 in the control group (p = 0.006). Administration of LA resulted in significant increase in the tissue GSH level of the treatment group to a value of 1.21 ± 0.27 (p = 0.004).
Tissue AOPP level was 4.75 ± 1.59 in the sham group, and induction of the ischemia–reperfusion injury significantly increased this level to 11.05 ± 1.34 in the control group (p = 0.004). Administration of LA resulted in significant decrease in the tissue AOPP level of the treatment group to a value of 4.19 ± 0.90 (p = 0.004).
Tissue SOD level was 20.62 ± 2.94 in the sham group, and induction of the ischemia–reperfusion injury significantly decreased this level to 16.77 ± 1.80 in the control group (p = 0.016). Administration of LA resulted in significant increase in the tissue SOD level of the treatment group to a value of 21.30 ± 1.39 (p = 0.004).
Plasma changes
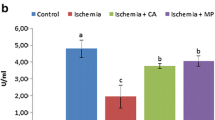
Plasma NO level was 46.85 ± 5.24 in the sham group, and induction of the ischemia–reperfusion injury significantly increased this level to 63.49 ± 5.63 in the control group (p = 0.004). Administration of LA resulted in significant decrease in the plasma NO level of the treatment group to a value of 45.26 ± 7.69 (p = 0.004).
Plasma MDA level was 4.14 ± 0.64 in the sham group, and induction of the ischemia–reperfusion injury significantly increased this level to 5.51 ± 0.70 in the control group (p = 0.008). Administration of LA resulted in significant decrease in the plasma MDA level of the treatment group to a value of 4.16 ± 1.15 (p = 0.025).
Plasma GSH level was 454.97 ± 35.70 in the sham group, and induction of the ischemia–reperfusion injury significantly decreased this level to 373.94 ± 29.99 in the control group (p = 0.008). Administration of LA resulted in significant increase in the plasma GSH level of the treatment group to a value of 454.79 ± 9.97 (p = 0.004).
Plasma AOPP level was 128.76 ± 43.10 in the sham group, and induction of the ischemia–reperfusion injury significantly increased this level to 343.76 ± 36.15 in the control group (p = 0.004). Administration of LA resulted in significant decrease in the plasma AOPP level of the treatment group to a value of 145.43 ± 27.21 (p = 0.004).
Plasma SOD level was measured as 1.07 ± 0.13, 0.87 ± 0.20, and 1.09 ± 0.20 in the sham, control, and treatments groups, respectively. There was no statistically significant difference between groups (p = 0.278). Bar graphs in Figs. 1 and 2 demonstrate the summary of the presented results.
Bar graph showing the plasma levels of NO, MDA, GSH, AOPP, and SOD for sham, control, and treatment groups. Bars represent mean levels and the error bars represent ±2 standard deviation (*p < 0.05 treatment vs control group; Ψp > 0.05; there were no significant difference between groups regarding plasma SOD levels)
Histopathological evaluation
The histological structure of the specimens from the sham-operated group was normal and the I/R injury in the control group significantly destroyed this structure. However, axonal damage, neuronal degeneration, and glial cell infiltration parameters were significantly lower in the LA received group compared to the control group (p < 0.001).
On the other hand, while the I/R injury significantly increased caspase 3 immunoreactivity (p < 0.001; sham vs control) administration of LA did not significantly decrease caspase activity (p = 0.149). Figure 3 demonstrates both hematoxylin and eosin stained tissue samples and caspase 3 immunoreactivity in tissue samples. Table 1 demonstrates scores derived after histopathological evaluation.
Histopathology of spinal cord. Hematoxylin and eosin staining of spinal cord tissue from the three groups: sham group (a), control group (c), and the treatment (lipoic acid) group (e). Figure demonstrate normal structure of spinal cord (a), neuronal degeneration, axonal separation (asterisk), and infiltration of astrocyte–microglia (c, e). Caspase 3 immunoperoxidase staining of spinal cords from three groups: sham group (b), control group (d), and the treatment (lipoic acid) group (f). No immunoreactivity in control-1 group whereas strong (d) and moderate (e) immunoreactivity in neurons and neuroglial cell in control-2 and lipoic acid group
Discussion
Oxidative stress is known to contribute to the pathogenesis of neurodegeneration in various CNS disorders. The findings that antioxidants improve neurological function after traumatic CNS injury in animals provide further evidence that oxidative stress has a deleterious effect on neuronal cells [21, 69].
In spinal cord injury models, oxidative stress counteracts antioxidant activity and is attributed to neurodegeneration in neurons, whereas constitutive antioxidants protect the neural tissue from oxidative stress. Reperfusion occurs in the first few days following SCI, which exacerbates the oxidative stress and the damage [20]. Oxidative stress initiates lipid peroxidation cascades that lead to the damage of highly vulnerable cell membranes during the first few days after injury [13, 30].
Although several endogenous antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, and catalase can detoxify ROS, the overproduction of the latter during the reperfusion of the ischemic segment of spinal cord can cause oxidative stress [62]. The first event at the cellular level during ischemia is the depolarization and the consequent opening of voltage-depended ion channels, glutamate release, and Ca++ influx [24]. Ca++ influx initiates several damaging actions including mitochondrial dysfunction, activation of mitochondrial and cytoplasmic nitric oxide synthetase, and production of NO. Activation of phospholipase A2 initiates arachidonic acid metabolism which is then converted by cyclooxygenase to a number of deleterious prostanoids and by lipoxygenases which is mediating axonal damage in SCI [14, 23, 28, 36].
The central nervous system possesses high levels of polyunsaturated lipids and metabolic rate with low antioxidant enzyme activity when compared to other tissues. This exacerbates the damage from free radical formation [32].
Therapeutic approaches which limit oxidative stress may be potentially beneficial in several neurological diseases [31]. In experimental SCI models, corticosteroids, lazaroids, gangliosides, calcium channel blockers, free radical scavengers, magnesium, sodium channel blockers, cyclosporin A, opioid receptor antagonists, NMDA receptor antagonists, and thyrotropin-releasing hormone have shown promise [1, 6, 18, 34, 52]. NMDA and non-NMDA receptor antagonists have demonstrated neuroprotective properties both in vitro and in vivo, but their major adverse effects limit their clinical use [6, 34, 52].
Lang-Lazdunski et al. have shown that MgSO4 and riluzole provided significant neuroprotection without any adverse effects in rabbit model of spinal cord ischemia [38].
Pharmacological measures to prevent spinal cord injury have been pursued, and these include hypothermia, anesthetic agents, ion channel blockers, anti-inflammatory drugs, immune modulators, and free radical scavengers [4, 18].
Only methylprednisolone has been shown to provide benefit in large clinical trials. Although this statement is not generally accepted by many centers who prefer not to use this agent in clinical grounds, methylprednisolone is almost the only agent which is used in neuroprotection studies as a comparative agent. For this reason, we preferred to use it in order to compare the results gathered after LA administration. Its neuroprotective effect has been attributed to an inhibitory effect on lipid peroxidation [7, 13, 23–25, 54], and although methylprednisolone reduces MDA levels within the first 12 h, it has no effect on the second lipid peroxidation [13].
An effective treatment strategy to reduce or reverse the permanent effects of SCI remains elusive. Lipid peroxidation is one of the most important and damaging effects of free radicals following SCI and a key mechanism in oxidative stress [1, 3, 23–25]. For this reason, in this study, we examined the effectiveness of LA on oxidative stress of spinal cord ischemia–reperfusion in rabbits. LA and its reduced form, dihydrolipoic acid (DHA), are potent biological antioxidants [43]. These compounds are capable of scavenging hydroxyl radicals and singlet oxygen. Hydroxyl radicals, singlet oxygen, peroxynitrite, and hypochlorous acid are scavenged by LA, and hydroxyl radicals, superoxide, peroxyl radical, peroxynitrite, nitric oxide, and hypochlorous acid are scavenged by DHA. Apart from its direct antioxidant effect, there is no question that LA treatment boosts cellular GSH levels both in vivo and in vitro and maintains GSH during times of oxidative insult [61]. They also act as a chelator of transition metals such as Cu+, Mn+, and Zn+ [5, 43]. LA is soluble in water as well as in fats which is a unique feature among antioxidants. For this reason, it is called an “antioxidant of antioxidants” [61]. In mammals, LA is synthesized do novo in mitochondria by lipoic acid synthase, but it can be absorbed from diet. LA is a necessary cofactor for mitochondrial α-ketoacid dehydrogenases and thus serves a critical role in energy metabolism. It is still unknown whether de novo synthesis is enough to supply all requirements for LA or dietary intake is also at least conditionally necessary [59]. There is no established upper limit for LA consumption in humans; however, safe levels for acute oral LA intake have been defined in animals. At a higher chronic dose (180 mg/kg), body weight gain and food consumption were decreased in rats and dogs [47]. LA is taken up and reduced in cells and tissues to dihydrolipoate, which is also exported to the extracellular medium; hence, protection is afforded to both intracellular and extracellular environments. LA also crosses blood–brain barrier and enters the brain and cerebrospinal fluid [61]. Thus, it seems an ideal substance in the treatment of oxidative brain and neural disorders involving free radical processes.
CNS damage due to stroke, cardiac arrest, hemorrhage, or head injury is a result of sudden reoxygenation of tissues (reperfusion) after a period of hypoxia. Administration of LA in animal models of ischemia–reperfusion alleviated effects of reperfusion. Level of ROS in brain cells decreases, extent of damage was reduced, and survival time of animals was longer in comparison with the control group [61].
Examination of current research about LA reveals protective effects of these compounds in cerebral ischemia–reperfusion, excitotoxic amino acid brain injury, mitochondrial dysfunction, diabetes and diabetic neuropathy, and other causes of acute or chronic damage to brain and neural tissue [47].
Prehn et al. demonstrated that, in focal ischemia model in mice, dihydrolipoate reduced the size of the infarct [50]. Cao and Phillis also observed a protective effect of LA against ischemia–reperfusion injury in the Mongolian gerbil model which offers the advantage of more complete ischemia [10].
LA pretreatment also almost completely abolished ischemia–reperfusion-induced losses of glutathione in the brain and dramatically decreases lipid peroxidation in the brain [50].
Elevated levels of extracellular glutamate are responsible for neuronal damage and degeneration in brain disorders [53]. LA was shown to protect C6 glial cells from glutamate-induced cytotoxicity. Low concentration of LA has been shown to bypass the adverse effects of elevated extracellular glutamate [26].
LA potently suppresses clinical and pathological disease in the animal model of multiple sclerosis and autoimmune encephalomyelitis, by inhibiting the migration of pathogenic T cells to the spinal cord. The mechanism is unknown [55].
The present study evaluated the biochemical markers of oxidative stress. NO, AOPP, MDA, GSH, and SOD are agents that work for or against oxidative stress which are used to measure the severity of the stress. Mammalian cells have developed antioxidant defense systems to prevent oxidative damage and to allow survival in an aerobic environment. These systems consist of nonenzymatic antioxidants with low molecular weights (vitamins A and E, beta-carotene, uric acid) and of enzymes such as SOD, catalase, glutathione peroxidase, and glutathione reductase [3, 5, 17, 18, 23, 28, 35, 51, 56, 60, 62, 63, 67].
Nitric oxide is a gas that induces macrophage cytotoxicity and vessel dilation and is involved in neural signaling. It is a unique molecule involved in many physiological processes in the CNS. Studies have demonstrated its both protective and detrimental effects in several diseases affecting the CNS. It has been demonstrated that lower NO concentrations play important roles in physiological processes, whereas larger amounts increase oxidative stress. After the SCI, inducible NO synthase is immediately expressed in the spinal cord. Excessive NO production has cytotoxic effects and induces neuronal apoptosis, secondary neural degeneration, and neuronal dysfunction [15, 27, 33, 65]. Our results demonstrated that while the I/R injury significantly increased the NO level both in the tissue and the plasma, administration of LA significantly decreased these levels.
As a marker of protein oxidation [67], levels of AOPP significantly increased after the I/R injury both in plasma and the spinal cord tissue. This increase in oxidative protein products is the proof of the increase in levels of the free radicals generated [60]. Researches have demonstrated that levels of AOPP are correlated with the levels of MDA, suggesting that AOPP acts as an oxidative stress mediator [49]. On the other hand, AOPP is not only a marker of oxidative stress but also acts as inflammatory mediator [67]. The significant decrease in the AOPP levels after administration of the LA in this study proves the antioxidant effects of this molecule.
SOD is the primary defense system against the reactive oxygen metabolites [66]. Results of our study demonstrated significant increase in levels of tissue SOD after administration of LA. However, this significant increase could not be observed in levels of SOD in the plasma.
GSH is an antioxidant which protects cells from free radicals [66]. The decrease in the level of GSH increases susceptibility to oxidative damage. As in other markers, compared to the control group, LA significantly increased GSH levels of the animals in the treatment group.
The central nervous system consists of largely of lipids and is damaged easily by free radical induced lipid peroxidation [56]. Lipid peroxidation is recognized as one of the main pathophysiological mechanisms involved in secondary damage [16] and MDA, which is formed from the breakdown of polyunsaturated fatty acids, serves as an important and reliable index for determining the extent of the peroxidation reaction [29, 44, 51]. In fact, most efforts aiming neuroprotection after spinal cord injury are intended to counteract early lipid peroxidation [16]. The results of our study demonstrate significant effects of LA in lowering MDA levels both in plasma and the neural tissue.
Histopathological evaluation includes neuronal and axonal damage and microglia infiltration. Sham-operated group had normal spinal cords. Treatment group showed significantly better morphological results than the control group. These results suggest that LA has also beneficial effects on preserving the normal spinal cord morphology by reducing the oxidative stress.
Severe oxidative DNA damage may trigger activation of the cysteine protease caspase-3 and consequently death by apoptosis. The onset of apoptosis in oligodendroglia, distant to the site of injury, appears to be unique in acute spinal cord ischemia and contributes to axonal demyelination and dysfunction with long-term neurological deficits [14]. However, caspase-3 immunohistochemical examination did not show any significant difference between the treatment and control group in the present study. LA has no significant anti-apoptotic effect on spinal cord ischemia–reperfusion injury model according to our study.
The compensatory activation of endogenous antioxidant molecules important for neutralizing ROS occurs at later time points. Impaired mitochondrial function and lipid peroxidation occurs within the first hour following spinal cord injury, with an increase in ROS occurring at fourth and 24th hours following injury. Oxidative stress persists for 5 days following SCI in rats [13]. These findings suggest that antioxidant mechanisms increase several hours after the generation of ROS. Antioxidant treatments should be initiated at early stages following injury [3]. That is why we administrated LA just after the injury. Although we administered only a single dose of LA in this study, the effectiveness can be increased by injecting secondary doses 3–5 days after the injury. On the other hand, the effectiveness of the agent should also be tested in traumatic spinal cord injury models such as weight drop or clip compression experiments. Different mechanisms of injury could reveal different results.
In the presented study, only biochemical, histopathological, and immunohistochemical parameters were analyzed. The lack of functional outcome assessment is the major pitfall of our study. Functional outcome assessment should be analyzed with different dose regimens and longer administration of LA in the further studies to achieve better outcome.
Conclusion
The results of the present study clearly demonstrated the favorable effects of LA in experimental spinal cord I/R injury. Although there may be other mechanisms involved, it seems that LA is successful in alleviating the effects of oxidative stress. Further experiments including a traumatic spinal cord injury model should be performed before any recommendations can be made for LA to be tested in humans.
References
Amar AP, Levy ML (1999) Pathogenesis and pharmacological strategies for mitigating secondary damage in acute spinal cord injury. Neurosurgery 44:1027–1039, discussion 1039–1040
Aykac G, Uysal M, Yalcin AS, Kocak-Toker N, Sivas A, Oz H (1985) The effect of chronic ethanol ingestion on hepatic lipid peroxide, glutathione, glutathione peroxidase and glutathione transferase in rats. Toxicology 36:71–76
Azbill RD, Mu X, Bruce-Keller AJ, Mattson MP, Springer JE (1997) Impaired mitochondrial function, oxidative stress and altered antioxidant enzyme activities following traumatic spinal cord injury. Brain Res 765:283–290
Baptiste DC, Fehlings MG (2006) Pharmacological approaches to repair the injured spinal cord. J Neurotrauma 23:318–334
Bilska A, Wlodek L (2005) Lipoic acid—the drug of the future? Pharmacol Rep 57:570–577
Bowes MP, Swanson S, Zivin JA (1996) The AMPA antagonist LY293558 improves functional neurological outcome following reversible spinal cord ischemia in rabbits. J Cereb Blood Flow Metab 16:967–972
Bracken MB, Shepard MJ, Holford TR, Leo-Summers L, Aldrich EF, Fazl M, Fehlings MG, Herr DL, Hitchon PW, Marshall LF, Nockels RP, Pascale V, Perot PL Jr, Piepmeier J, Sonntag VK, Wagner F, Wilberger JE, Winn HR, Young W (1998) Methylprednisolone or tirilazad mesylate administration after acute spinal cord injury: 1-year follow up. Results of the third National Acute Spinal Cord Injury randomized controlled trial. J Neurosurg 89:699–706
Budwit-Novotny DA, McCarty KS, Cox EB, Soper JT, Mutch DG, Creasman WT, Flowers JL, McCarty KS Jr (1986) Immunohistochemical analyses of estrogen receptor in endometrial adenocarcinoma using a monoclonal antibody. Cancer Res 46:5419–5425
Cameron NE, Cotter MA, Horrobin DH, Tritschler HJ (1998) Effects of alpha-lipoic acid on neurovascular function in diabetic rats: interaction with essential fatty acids. Diabetologia 41:390–399
Cao X, Phillis JW (1995) The free radical scavenger, alpha-lipoic acid, protects against cerebral ischemia–reperfusion injury in gerbils. Free Radic Res 23:365–370
Casini AF, Ferrali M, Pompella A, Maellaro E, Comporti M (1986) Lipid peroxidation and cellular damage in extrahepatic tissues of bromobenzene-intoxicated mice. Am J Pathol 123:520–531
Chaudhary P, Marracci GH, Bourdette DN (2006) Lipoic acid inhibits expression of ICAM-1 and VCAM-1 by CNS endothelial cells and T cell migration into the spinal cord in experimental autoimmune encephalomyelitis. J Neuroimmunol 175:87–96
Christie SD, Comeau B, Myers T, Sadi D, Purdy M, Mendez I (2008) Duration of lipid peroxidation after acute spinal cord injury in rats and the effect of methylprednisolone. Neurosurg Focus 25:E5
Chronidou F, Apostolakis E, Papapostolou I, Grintzalis K, Georgiou CD, Koletsis EN, Karanikolas M, Papathanasopoulos P, Dougenis D (2009) Beneficial effect of the oxygen free radical scavenger amifostine (WR-2721) on spinal cord ischemia/reperfusion injury in rabbits. J Cardiothorac Surg 4:50
Conti A, Miscusi M, Cardali S, Germano A, Suzuki H, Cuzzocrea S, Tomasello F (2007) Nitric oxide in the injured spinal cord: synthases cross-talk, oxidative stress and inflammation. Brain Res Rev 54:205–218
Diaz-Ruiz A, Rios C, Duarte I, Correa D, Guizar-Sahagun G, Grijalva I, Madrazo I, Ibarra A (2000) Lipid peroxidation inhibition in spinal cord injury: cyclosporin-A vs methylprednisolone. NeuroReport 11:1765–1767
Dumont RJ, Okonkwo DO, Verma S, Hurlbert RJ, Boulos PT, Ellegala DB, Dumont AS (2001) Acute spinal cord injury, part I: pathophysiologic mechanisms. Clin Neuropharmacol 24:254–264
Dumont RJ, Verma S, Okonkwo DO, Hurlbert RJ, Boulos PT, Ellegala DB, Dumont AS (2001) Acute spinal cord injury, part II: contemporary pharmacotherapy. Clin Neuropharmacol 24:265–279
Erten SF, Kocak A, Ozdemir I, Aydemir S, Colak A, Reeder BS (2003) Protective effect of melatonin on experimental spinal cord ischemia. Spinal Cord 41:533–538
Genovese T, Cuzzocrea S (2008) Role of free radicals and poly(ADP-ribose)polymerase-1 in the development of spinal cord injury: new potential therapeutic targets. Curr Med Chem 15:477–487
Gilgun-Sherki Y, Rosenbaum Z, Melamed E, Offen D (2002) Antioxidant therapy in acute central nervous system injury: current state. Pharmacol Rev 54:271–284
Gunasekar PG, Kanthasamy AG, Borowitz JL, Isom GE (1995) NMDA receptor activation produces concurrent generation of nitric oxide and reactive oxygen species: implication for cell death. J Neurochem 65:2016–2021
Hall ED (1993) The role of oxygen radicals in traumatic injury: clinical implications. J Emerg Med 11(Suppl 1):31–36
Hall ED, Springer JE (2004) Neuroprotection and acute spinal cord injury: a reappraisal. NeuroRx 1:80–100
Hall ED, Yonkers PA, Andrus PK, Cox JW, Anderson DK (1992) Biochemistry and pharmacology of lipid antioxidants in acute brain and spinal cord injury. J Neurotrauma 9(Suppl 2):S425–S442
Han D, Sen CK, Roy S, Kobayashi MS, Tritschler HJ, Packer L (1997) Protection against glutamate-induced cytotoxicity in C6 glial cells by thiol antioxidants. Am J Physiol 273:R1771–R1778
Hosaka N, Kimura S, Yamazaki A, Wang X, Denda H, Ito T, Hirano T, Endo N (2008) Significant correlation between cerebrospinal fluid nitric oxide concentrations and neurologic prognosis in incomplete cervical cord injury. Eur Spine J 17:281–286
Hulsebosch CE (2002) Recent advances in pathophysiology and treatment of spinal cord injury. Adv Physiol Educ 26:238–255
Ildan F, Polat S, Oner A, Isbir T, Gocer AI, Tap O, Kaya M, Karadayi A (1995) Effects of naloxone on sodium- and potassium-activated and magnesium-dependent adenosine-5'-triphosphatase activity and lipid peroxidation and early ultrastructural findings after experimental spinal cord injury. Neurosurgery 36:797–805
Kamencic H, Griebel RW, Lyon AW, Paterson PG, Juurlink BH (2001) Promoting glutathione synthesis after spinal cord trauma decreases secondary damage and promotes retention of function. FASEB J 15:243–250
Kaptanoglu E, Solaroglu I, Okutan O, Surucu HS, Akbiyik F, Beskonakli E (2004) Erythropoietin exerts neuroprotection after acute spinal cord injury in rats: effect on lipid peroxidation and early ultrastructural findings. Neurosurg Rev 27:113–120
Kaymaz M, Emmez H, Bukan N, Dursun A, Kurt G, Pasaoglu H, Pasaoglu A (2005) Effectiveness of FK506 on lipid peroxidation in the spinal cord following experimental traumatic injury. Spinal Cord 43:22–26
Kimura S, Hosaka N, Yuge I, Yamazaki A, Suda K, Taneichi H, Denda H, Endo N (2009) Cerebrospinal fluid concentrations of nitric oxide metabolites in spinal cord injury. Spine (Phila Pa 1976) 34:E645–E652
Kochhar A, Zivin JA, Lyden PD, Mazzarella V (1988) Glutamate antagonist therapy reduces neurologic deficits produced by focal central nervous system ischemia. Arch Neurol 45:148–153
Kowluru RA, Kern TS, Engerman RL (1997) Abnormalities of retinal metabolism in diabetes or experimental galactosemia. IV. Antioxidant defense system. Free Radic Biol Med 22:587–592
Kristian T, Siesjo BK (1998) Calcium in ischemic cell death. Stroke 29:705–718
Kurtel H, Granger DN, Tso P, Grisham MB (1992) Vulnerability of intestinal interstitial fluid to oxidant stress. Am J Physiol 263:G573–G578
Lang-Lazdunski L, Heurteaux C, Dupont H, Widmann C, Lazdunski M (2000) Prevention of ischemic spinal cord injury: comparative effects of magnesium sulfate and riluzole. J Vasc Surg 32:179–189
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lukacova N, Halat G, Chavko M, Marsala J (1996) Ischemia–reperfusion injury in the spinal cord of rabbits strongly enhances lipid peroxidation and modifies phospholipid profiles. Neurochem Res 21:869–873
McCarty KS Jr, Miller LS, Cox EB, Konrath J, McCarty KS Sr (1985) Estrogen receptor analyses. Correlation of biochemical and immunohistochemical methods using monoclonal antireceptor antibodies. Arch Pathol Lab Med 109:716–721
Miranda KM, Espey MG, Wink DA (2001) A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 5:62–71
Mitsui Y, Schmelzer JD, Zollman PJ, Mitsui M, Tritschler HJ, Low PA (1999) Alpha-lipoic acid provides neuroprotection from ischemia–reperfusion injury of peripheral nerve. J Neurol Sci 163:11–16
Mizuno Y, Ohta K (1986) Regional distributions of thiobarbituric acid-reactive products, activities of enzymes regulating the metabolism of oxygen free radicals, and some of the related enzymes in adult and aged rat brains. J Neurochem 46:1344–1352
Nagamatsu M, Nickander KK, Schmelzer JD, Raya A, Wittrock DA, Tritschler H, Low PA (1995) Lipoic acid improves nerve blood flow, reduces oxidative stress, and improves distal nerve conduction in experimental diabetic neuropathy. Diab Care 18:1160–1167
Oruckaptan HH, Ozisik P, Atilla P, Tuncel M, Kilinc K, Geyik PO, Basaran N, Yuksel E, Ozcan OE (2009) Systemic administration of interleukin-10 attenuates early ischemic response following spinal cord ischemia reperfusion injury in rats. J Surg Res 155:345–356
Packer L, Tritschler HJ, Wessel K (1997) Neuroprotection by the metabolic antioxidant alpha-lipoic acid. Free Radic Biol Med 22:359–378
Panigrahi M, Sadguna Y, Shivakumar BR, Kolluri SV, Roy S, Packer L, Ravindranath V (1996) alpha-Lipoic acid protects against reperfusion injury following cerebral ischemia in rats. Brain Res 717:184–188
Pekarkova I, Parara S, Holecek V, Stopka P, Trefil L, Racek J, Rokyta R (2001) Does exogenous melatonin influence the free radicals metabolism and pain sensation in rat? Physiol Res 50:595–602
Prehn JH, Karkoutly C, Nuglisch J, Peruche B, Krieglstein J (1992) Dihydrolipoate reduces neuronal injury after cerebral ischemia. J Cereb Blood Flow Metab 12:78–87
Qian H, Liu D (1997) The time course of malondialdehyde production following impact injury to rat spinal cord as measured by microdialysis and high pressure liquid chromatography. Neurochem Res 22:1231–1236
Rokkas CK, Helfrich LR Jr, Lobner DC, Choi DW, Kouchoukos NT (1994) Dextrorphan inhibits the release of excitatory amino acids during spinal cord ischemia. Ann Thorac Surg 58:312–319, discussion 319–320
Roy S, Packer L (1998) Redox regulation of cell functions by alpha-lipoate: biochemical and molecular aspects. Biofactors 8:17–21
Saunders RD, Dugan LL, Demediuk P, Means ED, Horrocks LA, Anderson DK (1987) Effects of methylprednisolone and the combination of alpha-tocopherol and selenium on arachidonic acid metabolism and lipid peroxidation in traumatized spinal cord tissue. J Neurochem 49:24–31
Schillace RV, Pisenti N, Pattamanuch N, Galligan S, Marracci GH, Bourdette DN, Carr DW (2007) Lipoic acid stimulates cAMP production in T lymphocytes and NK cells. Biochem Biophys Res Commun 354:259–264
Schmidley JW (1990) Free radicals in central nervous system ischemia. Stroke 21:1086–1090
Schonheit K, Gille L, Nohl H (1995) Effect of alpha-lipoic acid and dihydrolipoic acid on ischemia/reperfusion injury of the heart and heart mitochondria. Biochim Biophys Acta 1271:335–342
Serbinova E, Khwaja S, Reznick AZ, Packer L (1992) Thioctic acid protects against ischemia–reperfusion injury in the isolated perfused Langendorff heart. Free Radic Res Commun 17:49–58
Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM (2009) Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta 1790:1149–1160
Skvarilova M, Bulava A, Stejskal D, Adamovska S, Bartek J (2005) Increased level of advanced oxidation products (AOPP) as a marker of oxidative stress in patients with acute coronary syndrome. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 149:83–87
Smith AR, Shenvi SV, Widlansky M, Suh JH, Hagen TM (2004) Lipoic acid as a potential therapy for chronic diseases associated with oxidative stress. Curr Med Chem 11:1135–1146
Sugawara T, Lewen A, Gasche Y, Yu F, Chan PH (2002) Overexpression of SOD1 protects vulnerable motor neurons after spinal cord injury by attenuating mitochondrial cytochrome c release. FASEB J 16:1997–1999
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500
Taskiran D, Kutay FZ, Sozmen E, Pogun S (1997) Sex differences in nitrite/nitrate levels and antioxidant defense in rat brain. NeuroReport 8:881–884
Tsai S-K, Hung L-M, Fu Y-T, Cheng H, Nien M-W, Liu H-Y, Zhang FB-Y, Huang S-S (2007) Resveratrol neuroprotective effects during focal cerebral ischemia injury via nitric oxide mechanism in rats. J Vasc Surg 46:346–353
Varija D, Kumar KP, Reddy KP, Reddy VK (2009) Prolonged constriction of sciatic nerve affecting oxidative stressors & antioxidant enzymes in rat. Indian J Med Res 129:587–592
Witko-Sarsat V, Friedlander M, Capeillere-Blandin C, Nguyen-Khoa T, Nguyen AT, Zingraff J, Jungers P, Descamps-Latscha B (1996) Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int 49:1304–1313
Wolz P, Krieglstein J (1996) Neuroprotective effects of alpha-lipoic acid and its enantiomers demonstrated in rodent models of focal cerebral ischemia. Neuropharmacology 35:369–375
Xu M, Yip GW, Gan LT, Ng YK (2005) Distinct roles of oxidative stress and antioxidants in the nucleus dorsalis and red nucleus following spinal cord hemisection. Brain Res 1055:137–142
Yagi K (1982) Assay for serum lipid peroxide level and its clinical significance. In: Yagi K (ed) Lipid peroxides in biology and medicine. Academic, New York, pp 223–242
Yoshioka T, Kawada K, Shimada T, Mori M (1979) Lipid peroxidation in maternal and cord blood and protective mechanism against activated-oxygen toxicity in the blood. Am J Obstet Gynecol 135:372–376
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
Currently, there is a tremendous research focus in animals and humans to improve the devastating effects of spinal cord injury. Some of the main strategies include avoidance of prehospital hypoxia and hypotension, early surgical decompression and spinal fracture stabilization, therapeutic hypothermia, neuroprotectants such as alpha-lipoic acid in this study, neurotrophic agents, cell transplantation, blocking myelin-based protein inhibitors, using neural scaffolds, anti-inflammatory agents, reducing glial scar formation, and new rehabilitation strategies.
H. Emmez and colleagues have reported a carefully conducted study using the antioxidant alpha-lipoic acid in an experimental spinal cord ischemia and reperfusion injury model in 24 rabbits. The animals were sacrificed 48 h after the injury was induced, and it was found that tissue and spinal cord tissue levels of various markers of oxidative stress were significantly improved and there was less cellular degeneration and inflammatory change in the affected spinal cord after intraperitoneal alpha-lipoic acid was administered shortly after 30 min of ischemia.
This experiment is a long way from the use of this agent in human spinal cord injury but is an important step along the way. There is a component of ischemia–reperfusion injury in human spinal cord injury, but there is also the mechanical injury. The ischemia–reperfusion injury model the authors have used is not therefore a complete model of human spinal cord injury. This antioxidant should be further investigated experimentally with different dose scales, administered at different time intervals after the trauma, for longer observation periods, and with different models such as weight drop or clamping that more closely replicate mechanical injury to the spinal cord or parts of it before sacrifice of the animals. Clearly there are ethical challenges to keeping animals alive longer after partial or complete spinal cord injury. We encourage the authors to continue their excellent research on this promising therapeutic agent.
Jeffrey V Rosenfeld
Departments of Neurosurgery and Surgery, The Alfred Hospital and Monash University, Melbourne, Australia.
Rights and permissions
About this article
Cite this article
Emmez, H., Yildirim, Z., Kale, A. et al. Anti-apoptotic and neuroprotective effects of alpha-lipoic acid on spinal cord ischemia–reperfusion injury in rabbits. Acta Neurochir 152, 1591–1601 (2010). https://doi.org/10.1007/s00701-010-0703-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-010-0703-9