Abstract
In recent years, with the higher median life expectancy, the number of hip and knee replacements has increased. Clinical examination and morphological studies are essential to evaluate patients with a painful arthroplasty. Nuclear medicine examinations also play an important role, their main usefulness being the exclusion of prosthesis complications. Nevertheless, conventional examinations, namely bone scan and white blood cell scintigraphy, can also identify complications, such as loosening and infection. This study describes the normal and pathologic patterns of a bone scan and exemplifies ten common situations that can cause pain in patients with hip or knee arthroplasty, other than loosening and infection, which can be disclosed on a bone scintigraphy. The ten situations that should be considered and looked for when analysing a bone scan are: referred pain, patellofemoral pain syndrome, fractures, fissures, abscess/haematoma, bone insert behaviour, heterotopic ossification, greater trochanter pseudarthrosis, osteoarthritis extension in a knee with an unicompartmental prosthesis, and systemic disease with bone involvement.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
European context
Joint arthroplasty began in the nineteenth century [1, 2], and the modern surgical procedure was developed in 1950s for hip prosthesis and in 1970s for knee prosthesis [3].
According to the Organisation for Economic Co-operation and Development (OECD) data, the number of hip and knee replacements has increased in recent years in most European countries, with knee replacements generally growing more rapidly, although the volume of knee replacements still remains below that of hip replacements [4] (Figs. 1 and 2).
The majority of arthroplasties are successful in relieving pain and improving function. However, complications may occur in some patients, loosening and infection being the most common. Loosening can occur 10–20 years after joint replacement in 2–18% of prosthesis [5]. Infections occur in 2–3% of all primary joint replacements and in 5–8% of revision joint replacement [2, 6]. Because infection and loosening require different treatments, the differentiation between these two complications is very important.
Painful arthroplasty evaluation
When evaluating a patient with painful arthroplasty, the clinical history, history of the prosthesis (including the surgical procedure) and physical examination are crucial for diagnosis. Blood analysis (erythrocyte sedimentation rate, C-reactive protein and peripheral white blood cell count) and joint aspiration followed by culture are usually very important [1, 6]. Plain X-rays can help, but they often appear to be normal. Computed tomography (CT) and magnetic resonance imaging (MRI) are not very useful due to imaging artefacts caused by the metallic implants. In contrast to these techniques, nuclear medicine examinations are not conditioned by the metallic implants [6]. Some of the available conventional nuclear medicine examinations are [2, 6]:
-
1.
Triple-phase bone scan is frequently used because it is ubiquitously available, inexpensive, easily performed and has a high sensitivity and a low radiation dose. A normal examination excludes arthroplasty complication with high accuracy. It is sensitive for identifying the failed joint replacement, but cannot clearly differentiate between infection and loosening [1]. The radiotracer uptake is related to blood flow and the rate of new bone formation. In fact, any cause of accelerated new bone formation, including post-operative physiologic bone remodelling, as well as pathological conditions such as heterotopic ossification, fractures, neoplasms and arthritis, may show increased periprosthetic uptake, explaining the low specificity of this modality [7].
-
2.
Gallium scintigraphy has been used in combination with bone scintigraphy to increase diagnostic accuracy. A gallium scan can detect infection with an accuracy of 65–80% [1, 6]. However, gallium uptake is mainly related to inflammation and not to infection specifically. In addition, the suboptimal imaging characteristics of gallium radioisotope, the high radiation dose and the need for multiple imaging sessions over several days are disadvantages of this technique.
-
3.
White blood cell (WBC) imaging is also used for diagnosing complications of arthroplasty, and in combination with bone marrow scintigraphy with 99mTc-sulphur colloid, its accuracy can be improved, reaching sensitivity of about 90% [1, 2, 6]. Both WBC imaging and bone marrow imaging reflect radiotracer accumulation in the reticuloendothelial system of the bone marrow. Together these two radiotracers can reduce the interference of ectopic bone marrow induced by prosthetic surgery, which can lead to false-positive findings when interpreting the WBC scan alone. However, the distinction between delayed (chronic) infection and failure related to prosthetic wear debris can be very difficult. Other causes of decreased sensitivity are the formation of a biofilm by bacteria in bone infection and the influence of antibiotics. To overcome these aspects, and increase accuracy, it is important to acquire late images (day after injection) [8], as well as, to perform a single-photon emission computed tomography/computed tomography (SPECT/CT) [9].
Presently, bone scintigraphy is useful for screening purposes and the diagnosis of infection should be based on the combined interpretation of findings from bone scintigraphy and other radionuclide techniques like WBC imaging [1, 6]. Dual-phase WBC scintigraphy and WBC SPECT/CT combined with bone marrow scintigraphy seem to be the imaging modality of choice, showing the highest accuracy.
Since the beginning of the twenty-first century, another nuclear medicine examination has been increasingly used for studying prosthesis complication, the positron emission tomography with 18F-fluorodeoxyglucose ( 18 F-FDG PET). In infection and inflammation, the increased glycolytic activity in neutrophils and activated macrophages leads to FDG uptake. A meta-analysis stated that the sensitivity and specificity of 18F-FDG PET are 82 and 87%, respectively [10]. A PET without abnormal radiotracer uptake can rule out infection due to its high sensitivity, but when abnormal uptake foci are identified, it is not possible to distinguish infection from loosening based only on standardized uptake value (SUV) [11]. Therefore, it is essential to observe the uptake pattern (Reinartz et al. 2005 tried to develop a classification pattern using five uptake categories: patterns I, II and III are not associated with loosening, pattern IV should be associated with loosening, and pattern V should be associated with infection [12,13]) and, the most important, to correlate the findings from PET imaging with all the clinical information. Furthermore, the use of hybrid imaging introduced the possibility of performing 18F-FDG PET/CT, which has high sensitivity for detecting small particle disease. This aspect has significant clinical value as it can predict loosening in an early phase [6].
Recently, some studies have been developed to evaluate the usefulness of sodium fluoride( 18 F-FNa) PET. Most of the 18F-FNa delivered to the bone is retained after a single pass of blood, making it an excellent radiotracer to assess subtle changes in bone cortex hyperaemia and turnover, because bone marrow uptake is negligible. Its sensitivity is about 97%, and its specificity is about 88% [5]. It is very useful to evaluate bone metabolism at resurfacing arthroplasty, namely excluding aseptic femoral head necrosis, which is a major risk of total hip replacement, and analysing bone formation and mineralization patterns to study responses to prosthetic implants [2]. In 2011, Kobayashi et al. [14] published a prospective study presenting patterns to distinguish between normal, loosening and infected prostheses.
With hybrid imaging, we observed a metamorphosis of both bone scan and PET. The SPECT/CT and PET/CT have a fourfold advantage because it adds the following factors to the functional information: anatomical localization, increased spatial resolution, improved image quality due to attenuation correction and addition of extra diagnostic data. All these factors improve the hybrid examination specificity [1, 2, 6, 9, 15].
Aim
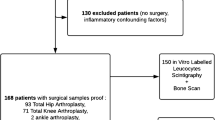
The aims of this study were: (1) to describe the patterns of a normal and a pathologic bone scan; and (2) to exemplify ten common situations that can cause painful hip or knee prosthesis, other than loosening and infection, and that can be found on bone scan.
Bone Scan and SPECT/CT patterns of knee and hip arthroplasty
The bone scan interpretation of a patient with a painful joint prosthesis is challenging because:
-
Pain is normal in the post-operative period (hip prosthesis for up to 2–3 months and knee prosthesis for up to 6–12 months) and is usually imprecisely described;
-
A joint prosthesis complication can be clinically and imagiologically similar to other problems or pathologies (i.e. soft tissue abscess, degenerative pathology with referred pain, fractures);
-
The periprosthetic radiotracer uptake can physiologically persist for several years;
-
There are several hundred types of prosthesis.
The normal bone scan
A bone scan without abnormal uptake foci rules out infection with high confidence. To avoid misinterpretation of a physiologic uptake and to reduce the false-positive examinations, it is essential to correlate all the clinical information with the morphologic and functional examinations. The normal aspect of a bone scan is [16, 17]:
-
hip prosthesis homogenous uptake that can persist for two years after the arthroplasty. There might be foci of enhanced uptake in the acetabulum, greater trochanter and prosthesis tip;
-
knee prosthesis heterogeneous uptake that can persist up to four years after arthroplasty. The uptake can be more evident at the tibia medial compartment (Table 1).
Table 1 Scintigraphic patterns of physiologic, infection and loosening uptake in hip and knee prosthesis. (Schemes based on J. Theriault images)
The knee prosthesis interpretation is usually more difficult than the hip prosthesis, mainly due to its variety, asymmetry and the long period necessary to be considered a pathological uptake (Table 1).
The pathologic bone scan
Differentiation between infection and loosening or inflammation is challenging, even when using specific uptake pattern classifications [6]. The examination is suspected of being pathological depending on the radiotracer uptake extent and intensity. The scintigraphic pattern of infection is a diffuse and intense radiotracer uptake around the prosthesis, in both the early (blood pool—soft tissue phase) and late images (bone phase). The scintigraphic pattern of loosening is a more focal uptake and more evident in the late images. In hip prosthesis, the uptake is usually enhanced at the acetabulum (main location), greater trochanter and in the prosthesis tip, while in knee prosthesis the uptake is located in the tibia tray and keel (main locations) and posterior region of the femoral condyles (both medial and lateral) [16, 17] (Table 1).
Concerning the variety of uptake patterns and the physiologic uptake period after a knee arthroplasty, bone scan has a limited value in the evaluation of a painful knee prosthesis. The identities in which the bone scan plays a true role are: femorotibial and/or patellofemoral osteoarthritis in a knee that already has a unicompartmental prosthesis, periprosthetic fractures/fissures and heterotopic ossifications [17].
The SPECT/CT
When analysing a hybrid examination, CT should also be considered. To decrease metal artefacts, especially in bilateral prostheses, it is advisable to optimize technical CT parameters [i.e. preferably use a high voltage (140 kV), high amperage (350–450 milliamperes (mAs) and up to 600 mAs if there are bilateral hip arthroplasties), narrow collimation and thin sections] and to correctly reconstruct the CT images [generate thick sections combining several acquired thin sections, by using low kernel values (standard reconstruction algorithm, using the soft filter B20) and by extending CT scale (possible up to 40,000 Hounsfield units in some scanners)] [18, 19]. It is also important to pay attention to the scout view and always observe the images in bone and soft tissues CT windows.
The radiologic signs of infection can include [6, 16, 19, 20, 21] widening of the periprosthetic lucency; presence of geodes (or subchondral cysts); osteolysis badly defined; presence of granulome, abscess or effusion; fistulous pathway; air bubbles; and intense periosteal reaction. The radiologic signs of loosening can include periprosthetic lucency more than 2 mm thick or progressive; osteosynthesis material or prosthesis migration or tilt; presence of granuloma; and cement fracture.
It is also very important to localize the radiotracer uptake. To facilitate communication between nuclear medicine physicians, radiologists, orthopaedists, rheumatologists and other physicians, it is recommended to use the DeLee & Charnley classification [22] for the acetabular region and Gruen classification [23] for the femoral region (Fig. 3). Concerning knee prosthesis, some classifications have been proposed, the more commonly referred are the Ewald classification and Hirschmann classification (Fig. 4), but there is no consensus yet.
Ten clinical situations that can cause pain, other than infection and loosening
When a patient has a painful hip or knee prosthesis and performs a bone scan on account of the clinician’s suspicions of prosthesis complication, the nuclear medicine physician should bear in mind that there are situations that can also cause arthroplasty pain, other than loosening and infection. The ten common situations that can be found on a bone scan and should be considered are: referred pain, patellofemoral pain syndrome, fractures, fissures, abscess/haematoma, bone insert behaviour, heterotopic ossification, greater trochanter pseudarthrosis, osteoarthritis extension in a knee with an unicompartmental prosthesis, and systemic disease with bone involvement.
-
1.
Referred pain
Whenever possible, it is advantageous to perform a SPECT/CT of the lumbar spine, to search for degenerative pathology (mainly osteophytes, listhesis and scoliosis) or fractures (e.g. vertebral bodies collapses or sacral ala fractures) that can cause referred pain and simulate a prosthesis complication. Ideally, the lumbar SPECT/CT should be performed systematically in every patient presenting a painful hip or knee prosthesis. It is also important to evaluate the contralateral articulation.
This is a common entity that can justify knee pain. Its diagnosis is mainly done by clinical and physical examinations. Usually, pain starts with movement (pain caused by squatting and descending stairs is a typical sign).
It can be caused by patellofemoral arthropathy (mainly in a bicompartmental knee prosthesis), patellar instability, patella fractures or patellar insertion mobilization. Although the diagnosis is usually made by clinical observation, on bone scan it appears as a “hot patella” and the radiotracer uptake is more intense at the patella posterior region, patella lateral region and trochlea union.
Fractures usually occur in the periprosthetic bone (mainly due to stress shielding that accelerates bone reabsorption and increases bone fragility), but they can also happen in the osteosynthese material (more common at the femoral stem). When analysing the images, besides analysing the scintigraphy, one should always examine the scout view and the bone window on CT, to increase the capacity to detect a fracture. Trojani et al. 1999 [25] classified fractures in 3 types, depending on its localization, stability and mobilization degree in extraprosthetic, intraprosthetic and prosthetic stem fractures.
Bone scan is very useful to identify fractures. It is important to report the location of the fracture and to search for loosening signs, especially in stem fractures.
-
4.
Fissures
The forceful procedure of trying to achieve a tight fit of the uncemented prosthesis, compared to cemented devices where a space is left for the cement to act as a grout, makes iatrogenic fissures and minor fractures a severe risk in the cementless prosthesis. Bone scan can be very helpful to diagnose these entities. Intense radiotracer uptake, with focal or linear pattern, without evident correspondence on CT is highly likely to be due to a fissure (Fig. 5).
-
5.
Abscess/haematoma
These lesions are usually a consequence of the surgical procedure or osteosynthesis material. They are evident in the soft tissue CT window, but sometimes are not evident in the metabolic study. Nevertheless, an abscess can show a peripheral tracer uptake in the late bone scan phase and a haematoma can be identified in the blood flow phase. This demonstrates the importance of changing the CT window from bone to soft tissues. Some common examples are the psoas muscle abscess (Fig. 6) and the pes anserinus (“goose foot”) haematoma (Fig. 7).
A 35-year male with right hip prosthesis, due to osteonecrosis, presented a sudden lameness. A right psoas abscess, caused by a traumatic contact with a prosthetic screw, was identified. a Anterior and posterior projections of the whole body bone scan. b SPECT/CT of the zone with enhanced uptake (transverse, coronal and sagittal projections). c Axial projection of the abscess in soft tissues CT window
These substances are synthetic bone substitutes, have different components (e.g. hydroxyapatite nanocrystals and tricalcium phosphate) and are increasingly used. They have osteoconduction, osteoinduction and osteoformation properties. The porous structure of these implants is analogous to the architecture of cancellous bone and allows fibrovascular and bone ingrowth, promoting the healing process. Planar scintigraphy and SPECT/CT can show an intense uptake within those implants in the early phase as well as in the late phase of the bone scan. This hypothesis should be considered in patients with such bone replacement materials, in order to avoid a false diagnosis of inflammation or infection (Fig. 8).
It is more common in hip prosthesis than in knee prosthesis. Usually, osteomas are identified during the 3 months following the arthroplasty and can progress for up to 2 years after the surgery. Some are similar to spurs, and they are more common in patients with vertebral ankylosing hyperostosis (e.g. Forestier disease). They can be located in tendons, ligaments, muscles and bones. Only 1% causes mobility limitation. Bone scan can be useful to determine the osteomas maturation degree before its surgical resection. The Brooker classification [28] of peri-hip prosthesis osteomas can also be used for knee prosthesis.
It is found when a trochanteric osteotomy, followed by a cerclage, is performed, and its incidence is about 6%. This shows the importance of knowing the surgical procedure when analysing images. Trochanteric non-union associated with total hip arthroplasty may result in isolated fracture of the greater trochanter, complication of trochanteric osteotomy, or fracture secondary to cystic formation or osteolysis from wearing debris post-arthroplasty. Bone scan shows a linear radiotracer uptake at the trochanter union, which corresponds to the region linked by the cerclage on CT images (Fig. 9). This pseudoarthrosis does not consolidate, and it can cause loosening due to the exacerbated macrophagic reaction in this region. Reattachment may be accomplished using various techniques, including abductor slide, trochanteric claw plate, grip system and various wiring techniques. Persistent non-union following reattachment ranges up to 39%.
-
9.
Osteoarthritis extension in a knee with an unicompartmental prosthesis [20]
The unicompartmental knee arthroplasty is an option applied when only half of the knee joint is affected (the most common is the medial femorotibial compartment). Bone scan shows a persistent periprosthetic uptake that can remain for up to three years after surgery. The femoral and tibial components uptake is usually asymmetric, as the intensity is higher at the tibia baseplate, because this region is submitted to more mechanical stress, and that can, therefore, lead to loosening. The value of bone scan, complementary to radiography, is in the evaluation of the remaining femorotibial and/or patellofemoral compartments, as it can detect degenerative changes suggesting the necessity to perform a total knee arthroplasty.
Many systemic diseases have bone involvement. Rheumatologic diseases often present articular manifestations. Several cancers can originate bone metastases, mainly breast and prostate cancer. This demonstrates the importance of integrating all clinical information.
Conclusion
Nuclear medicine plays an important role in the evaluation of a patient with a painful prosthesis. In spite of the advances of PET and the promising expectations that are being investigated, conventional examinations are widely accepted and performed. Bone scintigraphy is useful for screening. Labelled leucocytes scintigraphy, complemented with bone marrow scintigraphy, are established as the main examinations for prosthesis complication identification, namely loosening and infection.
There are several causes of painful arthroplasty, other than loosening and infection, which should be investigated when analysing a bone scan. Bone scan has lots of potential, and its diagnostic accuracy increases when complemented with SPECT/CT and correlated with the clinical information.
References
Love C, Marwin SE, Palestro CJ (2009) Nuclear Medicine and the infected joint replacement. Semin Nucl Med. doi:10.1053/j.semnuclmed.2008.08.007
Palestro CJ (2014) Nuclear medicine and the failed joint replacement: past, present, and future. World J Radiol. doi:10.4329/wjr.v6.i7.446
Palestro CJ, Love C, Marwin SE (2006) Nuclear medicine of the painful joint replacement. Appl Radiol. http://appliedradiology.com/articles/nuclear-medicine-of-the-painful-joint-replacement
OECD European Union (2014) Hip and knee replacement, health at a glance: Europe 2014. OECD Publishing, Paris. doi:10.1787/health_glance_eur-2014-34-en
Adesanya O, Sprowson A, Masters J, Hutchinson C (2015) Review of the role of dynamic 18F-NaF PET in diagnosing and distinguishing between septic and aseptic loosening in hip prosthesis. J Orthop Surg Res. doi:10.1186/s13018-014-0147-7
Jansen A, Smit F, Arias-Bouda LMP (2012) The role of Nuclear Medicine techniques in differentiation between septic and aseptic loosening of total hip and knee arthroplasty. JTijdschr Nucl Geneesk 34(4):988–994
Ouyang Z, Li H, Liu X, Zhai Z, Li X (2014) Prosthesis infection: diagnosis after total joint arthroplasty with three-phase bone scintigraphy. Ann Nucl Med. doi:10.1007/s12149-014-0899-5
Vries EFJ, Roca M, Jamar F, Israel O, Signore A (2010) Guidelines for the labelling of leucocytes with 99 mTc-HMPAO. Eur J Nucl Med Mol Imaging. doi:10.1007/s00259-010-1394-4
Erba PA, Israel O (2014) SPECT/CT in infection and inflammation. Clin Transl Imaging. doi:10.1007/s40336-014-0092-9
Kwee TC, Kwee RM, Alavi A (2008) FDG-PET for diagnosing prosthetic joint infection: systematic review and metaanalysis. Eur J Nucl Med Mol Imaging. doi:10.1007/s00259-008-0887-x
Zoccali C, Teori G, Salducca N (2009) The role of FDG-PET in distinguishing between septic and aseptic loosening in hip prosthesis: a review of literature. Int Orthop. doi:10.1007/s00264-008-0575-2
Reinartz P, Mumme T, Hermanns B, Cremerius U, Wirtz DC, Schaefer WM et al (2005) Radionuclide imaging of the painful hip arthroplasty: positron-emission tomography versus triple-phase bone scanning. J Bone Joint Surg Br. doi:10.1302/0301-620X.87B4.14954
Reinartz P (2009) FDG-PET in patients with painful hip and knee arthroplasty: technical breakthrough or just more of the same. Q J Nucl Med Mol Imaging 53(1):41–50
Kobayashi N, Inaba Y, Choe H, Ike H, Fujimaki H, Tezuka T et al (2011) Use of F-18 fluoride PET to differentiate septic from aseptic loosening in total hip arthroplasty patients. Clin Nucl Med. doi:10.1097/RLU.0b013e3182291ae7
Saha S, Burke C, Desai A, Vijayanathan S, Gnanasegaran G (2013) SPECT-CT: applications in musculoskeletal radiology. Br J Radiol. doi:10.1259/bjr.20120519
Girma A, Paycha F (2013) Place de la scintigraphie osseuse planaire et TEMP/TDM dans l’exploration des prothèses de hanche douloureuses. Méd Nucl. doi:10.1016/j.mednuc.2013.07.003
Girma A, Paycha F, Nizard R (2013) Rôle de la scintigraphie osseuse dans l’exploration des prothèses de genou douloureuses. In: Presented at the 69th Réunion Scientifique de l’ACOMEN « Prothèses orthopédiques et Médecine Nucléaire »
Cyteval C, Bourdon A (2012) Imaging orthopedic implant infections. Diagn Interv Imaging. doi:10.1016/j.diii.2012.03.004
Jacobson JA, Morag (2015) Hip replacement imaging. Medscape website. http://emedicine.medscape.com/article/398669-overview. Updated oct 27
Roth TD, Maertz NA, Parr JA, Buckwalter KA, Choplin RH (2012) CT of the hip prosthesis: appearance of components, fixation, and complications. RadioGraphics. doi:10.1148/rg.324115183
Cyteval C, Hamm V, Sarrabère MP, Lopez FM, Maury P, Taourel P (2002) Painful Infection at the site of hip prosthesis: CT imaging. Radiology. doi:10.1148/radiol.2242010989
DeLee JG, Charnley J (1976) Radiological demarcation of cemented sockets in total hip replacement. Clin Orthop 121:20–32
Gruen TA, McNeice GM, Amstutz HC (1979) "Modes of failure" of cemented stem-type femoral components: a radiographic analysis of loosening. Clin Orthop 141:17–27
Draper CE, Fredericson M, Gold GE, Besier TF, Delp SL, Beaupre GS, Quon A (2012) Patients with patellofemoral pain exhibit elevated bone metabolic activity at the patellofemoral joint. J Orthop Res. doi:10.1002/jor.21523
Fontaine C, Vannineuse A (2005) Fractures du genou. Springer, Paris, pp 317–330. http://eknygos.lsmuni.lt/springer/165/317-330.pdf
Tabouret-Viauda C, Maintaa I, Boudabbous S, Amzalaga G, Ratiba O, Ragera O, Paycha F (2014) High protracted 99mTc-HDP uptake in synthetic bone implants—a potentially misleading incidental finding on bone scintigraphy. Knee. doi:10.1016/j.knee.2014.08.005
Shehab D, Elgazzar AH, Collier BD (2002) Heterotopic ossification. J Nucl Med 43(3):346–353
Brooker AF, Bowerman JW, Robinson RA, Riley LH Jr (1973) Ectopic ossification following total hip replacement. Incidence and a method of classification. J Bone Joint Surg Am 55(8):1629–1632
Capello WN, Feinberg JR (2008) Trochanteric excision following persistent nonunion of the greater trochanter. Orthopedics 31(7):711
Salai M, Zippel D, Perelman M, Chechik A (1999) Revision hip arthroplasty in patients with a history of previous malignancy. J Surg Oncol 70:122–125
Schmidt AH, Walker G, Kyle RF, Thompson RC (1996) Periprosthetic metastatic carcinoma: pitfalls in the management of two cases initially diagnosed as osteolysis. J Arthroplasty 11(5):613–619
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have nothing to disclose.
Rights and permissions
About this article
Cite this article
Vaz, S., Ferreira, T.C., Salgado, L. et al. Bone scan usefulness in patients with painful hip or knee prosthesis: 10 situations that can cause pain, other than loosening and infection. Eur J Orthop Surg Traumatol 27, 147–156 (2017). https://doi.org/10.1007/s00590-016-1884-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-016-1884-6